Environmental and public health impacts of plastic wastes due to healthcare and food products packages: A Review
Ugoeze KC1,*, Amogu EO1, Oluigbo KE2, Nwachukwu N1
1Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmaceutical Sciences, University of Port Harcourt, Port Harcourt, Nigeria
2Department of Clinical Pharmacy and Biopharmaceutics, Faculty of Pharmaceutical Sciences, Enugu State University of Science and Technology, Agbani City, Enugu, Nigeria
*Corresponding Author: Ugoeze, Kenneth Chinedu, Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmaceutical Sciences, University of Port Harcourt, Port Harcourt, Nigeria
Received: 11 January 2021; Accepted: 16 January 2021; Published: 27 January 2021
Article Information
Citation: Ugoeze KC, Amogu EO, Oluigbo KE, Nwachukwu N. Environmental and public health impacts of plastic wastes due to healthcare and food products packages: A Review. Journal of Environmental Science and Public Health 5 (2021): 1-31.
View / Download Pdf Share at FacebookAbstract
Plastic products have gained contemporary extensive utility, became indispensable and the commonest material used in product packaging in healthcare, food and other articles essential to humanity. These packaging materials, produced from plastic bases like the bio-degradable, epoxy resins, polystyrenes, expanded polystyrenes, polyolefin, polyvinyl chloride and polycarbonates also contain several unsafe substances incorporated into them to aid their production and product aesthetics. The innovative exploitation of plastic-based packaging materials in healthcare, food and related products has evolved to the extent of harnessing single-use and discard after-use packages which have merely given rise to the prevalence of menacing plastic wastes generally, creating heaps of plastic wastes in several spots, defacing the cities, contaminating the environments and upsetting the ecosystem deleteriously with its public health outcomes. The atmosphere, animals and humanity are confronted with threats of extinction due to the discharges originating from the harmful constituents of these plastic wastes which saturate and contaminate the ecosystem. These jeopardies could be abridged if the bulk of plastic junks due to the healthcare, food and allied packages are properly managed or recycled at the end of their useful life. The emerging economies that is mostly affected by these challenges should adopt plastic waste management approaches, especially recycling, reuse and establishment of enabling policies on the manufacture, use and disposal of plastic-based packaging products. Effective majors will ensue if the government and policymakers would enhance enlightenment efforts at various levels of each society coupled with strict regulations over the management of plastic-based packaging materials.
Keywords
<p>Environmental; Public health; Plastic wastes; Healthcare; Food; Packages</p>
Article Details
1. Introduction
Plastic products have gained wider usage and became consistently essential on daily basis [1]. Due to their extensive application in packaging, etc. heaps of plastic wastes are generated in quite a lot of spots which have exceedingly affected the environs, permitting visual pollutions with impending dangers as well as defacing of the living cities [2, 3, 4]. Plastic wastes constitute between 60-80 % of marine debris and are one of the world’s most pervasive pollution problems impacting our oceans and waterways, according to the U.N. [5]. Over six decades, manufacture of plastic products and their resultant wastes have extremely increased due to rapid urbanization, increasing consumption in both high and low-income countries and increased production of “use and discard” products, coupled with the fact that the bulk of plastic packages are not recycled at the end of their useful life. From raw material extraction through to plastic contaminating the ocean, plastics exemplify the failure of a predominantly fossil fuel-based, linear economic system [5]. Several nations, particularly the developing economies are faced with the challenge of developing plastic waste-management approaches from options that include reduction of waste generated, incineration, landfilling, recycling and reuse [4].
2. What are Plastics?
Plastics are a wide range of polymeric components available synthetically, semi-synthetically or organically which could be shaped into a required form when they are hot and retain that shape when they are cold or hardened. The term “plastic” is derived from the Greek word “plastikos”, meaning, fit for moulding, pointing to their malleability during processing or manufacture, which enables them to be cast, pressed or extruded into a variety of shapes such as films, fibres, plates, tubes, bottles, boxes, etc. They are useful in making water bottles, clothing, medical and food packaging, electronics, construction materials, etc. Polymers (classified as natural or synthetic), the sources of plastics are macromolecules comprising of large numbers of repeated single units (monomers) bonded through a chemical procedure described as polymerization. The natural polymers (biopolymers) are obtainable from plants or animals and include polysaccharides, nucleic acids and proteins, etc. The synthetic polymers are man-made products obtained through chemical manipulations of other polymers, even the natural polymers. They are the structural materials exhibited generally in plastics, synthetic fibres, paints, building materials, adhesives, etc. The synthetic polymers may be divided into thermoplastic polymers and thermoset plastics which is centred on their response when they are exposed to heat. Thermoplastics are the plastics that do not undergo a chemical change in their composition when heated and can be remoulded. They include polyethylene (employed in plastic bags), polypropylene, polystyrene (used in plastic cups), polyvinyl chloride (PVC) (used for food wraps, bottles and drain pipes) and polytetrafluoroethylene (PTFE) or Teflon (used for non-sticking surfaces). Thermosets can be melted, formed into the desired shape which remains permanent as the material gets cold and solidified. They include vulcanized rubber, bakelite, polyepoxide, etc. [6-14].
Plastics was devised by the use of natural materials that had intrinsic plastic properties such as shellac and chewing gum. It was followed by the chemical treatment of natural materials such as rubber, nitrocellulose, collagen or galalite which culminated in the widespread series of entirely synthetic materials that could be acknowledged as modern plastics. One of the initial instances was invented by Alexander Parkes in 1855, who named his invention Parkesine. It is recognized presently as celluloid. Polyvinyl chloride (PVC) was initially polymerized between1838-1872. A key advance came in 1907, when Belgian-American chemist, Leo Baekeland created Bakelite, the first real synthetic, mass-produced plastic [15].
The basic raw materials that are crucial in making plastics are sourced from petroleum, natural gas or other organic materials like cellulose, coal, salt, etc. The manufacture of plastics is initiated with the distillation of crude oil by refining it to lighter fractions, each being a mixture of compounds of hydrocarbon chains differing in terms of the size and molecular structures. Naphtha is one of these fractions and is the key composite for the manufacture of plastics. Plastics are got by two techniques known as polymerization and polycondensation, each requiring a specific catalyst. In a polymerization reactor, monomers such as ethylene and propylene are joined to constitute stretched polymer chains, each polymer taking its specific characteristics, structure and size subject to the different types of basic monomers employed. Plastics can be assembled into two major polymer groups such as thermoplastics (the types that soften under heat, then toughen on cooling) and thermosets (the types that never soften once they have been moulded) [15].
3. Chemical Constituents of Plastic Products
During polymerization, some additives like accelerators, initiators, solvents and catalysts, etc. are incorporated in their essential extents to support the procedures. While producing the plastic products also, various further additives are added to aid in their production and enhance their properties, such as prolonging of their shelf life and aesthetic values. Such additives include plasticizers (e.g. short, medium and long chain chlorinated paraffins (SCCP/MCCP/LCCP), Diisoheptylphthalate (DIHP), Benzyl butyl phthalate (BBP), Bis (2-ethylhexyl)phthalate (DEHP): Bis(2-methoxyethyl) phthalate (DMEP), etc.) [16], flame retardants (Short, medium, long chain chlorinated paraffins), boric acid; Brominated flame retardants with antimony (Sb) as synergist (e.g. Polybrominated diphenyl ethers (PBDEs); Decabromodiphenylethane; tetrabromobisphenol A (TBBPA)); Phosphorous flame retardant (e.g. Tris (2-chloroethyl)phosphate (TCEP) Tris (2-chlorisopropyl)phosphate (TCPP) [16, 17], acid scavengers (synthetic hydrotalcites, metallic stearates and zinc oxides), light and heat stabilizers (Bisphenol A (BPA); Cadmium and Lead compounds, metallic salts, organometallic compounds, non-metallic organic stabilizers, organophosphites and epoxies) [18-21], lubricants (silicones) [22], pigments (titanium dioxide),(antioxidants(phenolics and phosphites) [23], colourants (azo dyes, anthraquinones) [24], antistatic agents (rubbers, polyesters) [25], slip compounds ( Fatty acid amides (primary erucamide and oleamide), fatty acid esters, metallic stearates (for example, zinc stearate), and waxes ) [26] and thermal stabilizers (Cadmium and Lead compounds; Nonylphenol (barium and calcium salts), lead salts, metal soaps and organo-tin compounds which are extremely lethal and ecological contaminants) [27-32].
4. Types of Plastics used in Packaging of Pharmaceutical and Food Products
Packaging is essential in pharmaceutical and food industries to conserve the value of products through their shelf life from external elements such as light and moisture which are capable of affecting their integrity. The selection of the packaging material is based on the type of product in question, their physical and chemical properties as well as the properties of the packaging material in relationship to the product. The following are varieties of some plastic materials that could be used in the design of packages for food or pharmaceutical products.
4.1 Bio-based and biodegradable plastics
These are sourced from renewable biological resources such as sugar cane, starch, etc. Sugar cane is processed to produce ethylene, which could be used to produce, for instance, polyethylene. Starch can be processed to produce lactic acid and later polylactic acid (PLA) which are elastic, long-lasting, clear, heat resistant, etc. They could be starch, cellulose or protein-based [33, 34]. These class of plastics could be degraded by micro-organisms into the water, carbon dioxide (or methane) and biomass under specified conditions. They are often manufactured with renewable raw materials, micro-organisms, petrochemicals or mixture of the three. They are used as disposable items employed in packaging and catering items, bags, trays, containers for fruits, vegetables, eggs and meat, bottles for carbonated drinks and dairy products as well as blister foils for fruit and vegetables [33-37].
4.2 Epoxy resins
The epoxy resins are also known as polyepoxides. They are a class of reactive prepolymers and polymers containing epoxide groups. A chemical reaction amongst the polyepoxides or with polyfunctional hardeners yields a thermosetting polymer, often with enhanced mechanical properties and high thermal and chemical resistance. They are used in the production of cans for soft-drinks, closures and caps for medical products, in cover linings aimed at protecting the contents and retaining flavor [33, 37, 38].
4.3 Polystyrene
Polystyrene (PS) is a thermostatic synthetic aromatic polymer formed from styrene, monomers and a liquid petrochemical found naturally in strawberries, cinnamon, coffee and beef [33]. It is often used in products that require clarity and often used to protect and package food and other consumer products as in meat/poultry trays and egg cartons for protection against damage or spoilage. They are employed as food service items, such as cups, plates, bowls, cutlery, hinged takeout containers (clamshells), meat and poultry trays and rigid food containers for yoghurt [33, 39, 40].
4.4 Expanded polystyrene
Expanded polystyrene (EPS) is a thermoplastic material manufactured from styrene monomer using a polymerization method to obtain translucent spherical beads of polystyrene, a solid thermoplastic at room temperature which could melt at a higher temperature and re-solidify to emerge into a material that could fit in for anticipated use [38]. Considering its shock-absorbing, thermal insulation and moisture resistant capacities, it is employed in the storage and conveyance of breakable items. It is very useful in the packing of cooked food to retain its warmth or coolness and avert wastage. It is also useful in the packaging perishable items such as seafood, fruit, and vegetables to preserve their integrity [38, 39].
4.5 Polyolefins
Polyolefins are of the category of polyethylene and polypropylene thermoplastics, being the combined term for the varieties of plastics that include polyethylene, specifically low-density polyethylene (LDPE), linear low-density polyethylene (LLDPE), high-density polyethylene (HDPE) and polypropylene (PP). They are made essentially from oil and natural gas by a process of polymerization of ethylene and propylene respectively. Polyolefins are extremely suitable and safe for packaging because they are 'chemically inert' materials. It is believed that the foodstuffs and other items packed in them cannot absorb harmful chemicals, additives or by-products from the container, hence, they have great advantages employing them in various packaging materials. The LDPE is used in cling film, carrier bags, agricultural film, milk carton coatings, hot and cold beverage cups, squeezable bottles for honey and mustards, etc. The HDPE is useful in crates and boxes, bottles (for food products, detergents and cosmetics), food containers, etc. The PP is utilized in food packaging, including yoghurt, margarine pots, sweet and snack wrappers, microwave-proof containers, medical packaging and appliances, etc. The polyethylene terephthalate (PET, PETE) is clear, tough, and has good gas and moisture barrier properties. This resin is commonly used in beverage bottles and many injections- moulded consumer product containers. They are utilized as plastic bottles for soft drinks, water, juice, sports drinks, beer, mouthwash, catsup and salad dressing. They are useful as food jars for peanut butter, jelly, jam and pickles. The polypropylene (PP) are employed for yoghurt, margarine, takeout meals and deli food, medicine bottles, bottle caps and closure [33, 40].
4.6 Polyvinyl chloride
Polyvinyl chloride (PVC) was one of the first plastics discovered and is also one of the most extensively used. The essential raw materials for PVC are derived from salt (57 %) and oil (43 %). There are two forms of PVC, the rigid type that is often abbreviated as RPVC and the flexible type [33, 41]. They are useful in the packaging of healthcare and medical devices (blood and plasma transfusion storage bags/sets, catheters and cannulas, surgical and examination gloves, intravenous solution giving sets, urine bags, endotracheal tubing, blister packs, shrink wrap, tamper resistance and dosage packs for pharmaceutical products) [41].
4.7 Polycarbonates
These are employed in the packaging of consumer goods like reusable bottles. However, it has bisphenol-A (BPA). In case it is exposed to high temperature, BPA could leach from the polycarbonated bottle into the content. Due to the health implication of BPA as reported in various researches, the utilization of polycarbonated plastics has become decreased [26, 33].
5. Application of Plastics in Pharmaceutical and Food Packaging
Packaging plays a crucial role in Pharmaceutical products as it helps to retain their original form and preserve the quality of the content until it is consumed by the end-user. Suitable packaging protects the product from external elements such as light and moisture which could affect its integrity. The selection of the packaging material is based on the type of product being manufactured. Therefore, all the physical and chemical properties of the product as well as the packaging materials are of utmost importance. Plastics are generally considered for the packaging of pharmaceutical preparations because plastic containers are not heavy, not fragile and do not easily develop leakage. They are cheap to manufacture since they are easily moulded or remoulded with aesthetic and excellent finishing. Their lids or closures are resistant to rust or corrosion [42, 43].
Currently, plastics are produced and employed in the packaging of pharmaceutical products as follows [44-48]:
- Rigid bottles as packaging materials for solid dosage forms like tablets, capsules or powders.
- Sterile plastic packaging materials for human blood samples and their components.
- Plastic packaging materials for aqueous solutions.
- Plastic bags for parenteral sterile solutions and their components.
- Containers for dry powders and metered-dose inhalers.
- Squeezable bottles for eye, ear drops and nasal sprays.
- Jars for powders, creams, ointments and other multi-dose topical applications.
- Pre fillable syringes.
- Pill boxes
- Flexible tubes
- Sachets, blister and strip packs, cartridges, nebulizers, vials, etc.
6. Resin Identification Coding of Plastic Packaging Containers
On the bottom of any plastic packaging container is a symbol designated as a resin identification code (RIC). This is a symbol with a number in its middle. Each symbol is unique and refers to the type of plastic employed in the manufacture of the container in question. Each symbolic code is designated to the types of products that it could be used to package. It then serves as a guide to producers to ensure that products are not packaged in the wrong containers. The Society of the plastics industry (SPI) established this classification system in 1988 in an effort to evolve standardization in the production of plastics and reprocessing of recycled plastic materials. The coding system also enables producers of consumer goods, consumers and recyclers to harness and appropriately identify different types of plastics [49]. With the knowledge of this, the consumers can ascertain that the particular product they are procuring is packaged in the right container, especially, the internally consumed products ranging from pharmaceuticals to foods and beverages [50]. The codes are as follows:
6.1 PET: The Polyethylene terephthalate
Polyethylene terephthalate, commonly abbreviated as PET or PETE is a general-purpose thermoplastic polymer belonging to the polyesters. It is one of the most recycled thermoplastics and has its SPI resin identification code (RIC) as “1”. It is an excellent water and moisture barrier material, hence, plastic containers made from PET are widely used for mineral water and carbonated soft drinks, foods, cooking oils, packaging trays and pharmaceutical packages and medical applications [13, 51, 52]. It prevents oxygen from getting in and spoiling the product packaged in them and equally helps to keep the carbon dioxide in a carbonated drink from escaping. Though this material absorbs odours and flavours from foods and drinks that are stored in them, it does not contain harmful bisphenol-A (BPA), phthalates, dioxins (a group of compounds sometimes formed with high-temperature combustion above 750 o F and certain types of industrial processes involving chlorine) lead, cadmium or endocrine disruptors [52].
6.2 HDPE: High density polyethylene
High-density polyethylene (HDPE) is used to make many types of bottles. It has a good barrier and is suitable for packaging products with a short shelf life and has good chemical resistance. It is commonly recycled and has its SPI resin identification code as “2”. This could be opaque, translucent (and often white). It is used as containers for milk, yoghurt, fruit juices, pharmaceutical products, caps for food packaging and plastic grocery bags [13]. The HDPE is the most commonly used plastic in the production of plastic bottles and does not contain bisphenol-A (BPA) or phthalates [53].
6.3 PVC: Polyvinyl chloride
Polyvinyl chloride (PVC) was one of the first plastics discovered and is also one of the most extensively used. It is made softer and flexible by adding phthalates and bisphenol-A. It has its SPI resin identification code as “3”. It is used in toys, blister wrap, blood bags, detergent bottles, food packages, lunch-boxes, etc. PVC is harmful for use in storing anything to be ingested. It is sometimes recycled but is rarely accepted by recycling programmes as it releases dioxins, phthalates, vinyl chloride, ethylene dichloride, lead, cadmium and other toxic chemicals such as bisphenol-A (BPA) and is considered the most hazardous plastic for health and the environment [54, 55].
6.4 LDPE: Low-density polyethylene
This type of plastic has its SPI RIC as “4” and is used in grocery stores, bread and sandwich bags, squeezable bottles, drug dispensing bottles, juice and milk cartons, etc. It has resistance to moderate levels of heat, recyclable, durable and flexible [56-58].
6.5 PP: Polypropylene
This has its SPI resin identification code as “5” and could occasionally be recycled. It is strong and can withstand high temperatures and is safe for food and beverages, however, caution needs to be exercised with microwaving them with edible items to avoid leakage of certain chemicals [59]. The plastics are useful in medicine bottles, straws, bottle caps, syrup bottles, and some yoghurt containers as well as in flexible packaging of food, confectionaries and tobacco [60]. Polypropylene (PP) has a high heat tolerance and as such, does not seem to leach many of the chemicals other plastics do [61].
6.6 PS: Polystyrene
The polystyrene has its SPI resin identification code as “6”. It is also known as Styrofoam and is used for cups, plates, take-out containers, supermarket meat trays, and packing peanuts. Their materials can leach styrene, a suspected carcinogen, especially in the presence of heat [61]. In safety matters, there was some confusion over the safety of polystyrene with the inclusion of "styrene" in the National Toxicology Program's (NTP) 12th Report on Carcinogens in 2011. Styrene is naturally present in foods such as strawberries, peaches, cinnamon, beef and coffee and is produced in the processing of foods such as beer, wine and cheese. It is also used to make polystyrene, a material used to make some foodservice packaging. In 2013, the American Chemistry Council's Plastics Foodservice Packaging Group provided updated styrene migration data to FDA. The data show that current exposures to styrene from the use of polystyrene food contact products remain extremely low, with the estimated daily intake calculated at 6.6µg per person per day, a value that is more than 10,000 times below the safety limit set by FDA. The FDA’s acceptable daily intake value of styrene is calculated to be 90,000 micrograms per person per day [62-64].
6.7 Others
Any plastic item not made from the other six plastics as highlighted earlier is lumped together and designated with the SPI resin identification code of “7”. They may likely leach bisphenol-A (BPA) and/or bisphenol-S (BPS) (a chemical analogue of BPA), both potent endocrine disruptors linked to interfering with proper mood, growth, development, sexual function, reproductive function, and puberty, among other essential human developmental processes. They are also suspected of increasing the risk of adult reproductive cancers, obesity, heart disease, and type 2 diabetes [61].
The various types of polymers engaged in various packaging materials and their respective RIC are summarized in Table 1 [65, 66].
|
Symbol |
Type of plastic |
Common uses |
|
|
Polyethylene terephthalates |
soft drink containers, water bottles, containers, salad dressing, biscuit trays and salad domes. |
|
|
High density polyethylene (HDPE) |
Shopping bags, freezer bags, buckets, shampoo, milk bottles, ice cream containers, juice bottles, chemical and detergent bottles, rigid agricultural pipe, crates. |
|
|
Polyvinyl Chloride (PVC) Plasticized Polyvinyl chloride PVC-P. |
Cosmetic container, plumbing pipes and fittings, electrical conduct, blister packs, wall cladding, roof sheeting, bottles, garden hose, Shoe soles, cable sheathing, blood bags and tubing. |
|
|
Low density polyethylene (LDPE) |
Refuse bags, Irrigation tubings, mulch film, cling wrap, garbage bags, squeeze bottles. |
|
|
Polypropylene (PP) |
Microwave dishes, lunch boxes, packaging tape, garden furniture, kettles, bottles and ice cream tubs, potato chip bags, straws |
|
|
Polystyrene (PS) Expanded polystyrene (PS-E) |
CD cases, plastic cutlery, imitation glassware, low cost brittle toys, video cases/foamed polystyrene cups, protective packaging, building and food insulation |
|
|
Other |
Automotive and appliance components, computers, electronics, cooler bottles, packaging |
Table 1: Types of plastics, their properties, common uses and their SPI/RIC symbol/number.
7. Environmental and Public Health Impacts of Plastic Wastes
7.1 Impact of plastic wastes on the environment
In Europe, around 2006, plastic materials were prevalent in the packaging (38 %), followed by building and construction (21%), automotive (7%), electrical and electronic (6 %), and other sectors like medical and leisure (28 %) [44]. This shows that packaging constituted the largest market for plastic products due to a global shift from reusable to single-use containers. It led to the increase in the share of plastics in municipal solid waste (by mass) from less than 1 % in 1960 to more than 10 % by 2005 in middle and high-income nations [67]. At the same time, global solid waste generation, which is strongly correlated with the gross national income per capita, has grown steadily over the past five decades [68, 69].
Plastics represent 15-25 % of all hospital waste in the United States. Some newer plastics are biodegradable, but the rest is incinerated, disposed of in landfills or recycled. These techniques are bound with ecological risk. Biodegradable plastics may break down in the atmosphere into smaller polymer components, which may still pose a risk to the surroundings [70]. The enormous majority of monomers used to make plastics, such as ethylene and propylene, are derived from fossil hydrocarbons and the plastics made from these are not biodegradable. Instead of decomposing, they accumulate in landfills or the natural environs [71].
Incineration liberates greenhouse gases associated with climate change. Landfilling of plastics, particularly in the enormous volumes, maybe an impractical use of land resources and a danger exists of plastic constituents which are toxic chemical additives like phthalates, heavy metals, bisphenol-A, brominated flame retardants, nonylphenol, polychlorinated biphenyl ethers, phenanthrene, dichlorodiphenyldichloroethylene, antimony trioxide sipping into the land and entering the groundwater. With flooding, these toxic chemicals also enter the rivers, seas, oceans, etc. The recycling of plastics requires careful sorting of plastic material, which is difficult. Recycled plastics tend to be of lower quality and may not be practical for health care and other applications [70].
The leading source of plastics-related environmental destruction stems from the overuse of items whose long-term damage outweighs their short-term profit. Such challenges often come through consumer convenience items which are most of the time hurriedly discarded after a short use-life. They include plastic water bottles, grocery bags, packaging, Styrofoam cups, Teflon-coated dental floss and other products [70]. Plastic debris has been found in all major ocean basins [71], with an estimated 4-12 million metric tons (Mt) of plastic waste generated on land entering the marine environment in 2010 alone [67]. Pollution of freshwater systems and terrestrial habitats is also increasingly more documented [72-74], as is environmental contamination with synthetic fibres [74, 75]. Plastic waste is now so ubiquitous in the environment that it has been suggested as a geological indicator of the proposed Anthropocene era [76].
There are three possible environmental problems to be considered. Firstly, plastics are mostly made from oil, natural gas or coal, and these are all limited natural resources that must be conserved for other utilization [77].
Secondly, the manufacture of plastics produces a lot of harmful pollutants which manufacturing companies need to deal with properly. Plastics being versatile, pliable, durable, cheap to produce, and pervasive, are also both a life-saving miracle product and the scourge of the earth. In 2012, about 8 % of the world’s oil production was used to make plastic and power its production. That figure is projected to rise to 20 % by 2050 [78]. Considering the global distribution of plastic production, half the world’s plastics are made in Asia. The lion’s share of that, 29 % is made in China, home to 18 % of the world’s population. The rest of Asia produces 21 %, Europe (19 %), the North America Free Trade Agreement (NAFTA) countries (18 %), Middle/East Africa (7 %), Latin America (4 %) and others (2 %) [78]. Less than a fifth of all the plastics made are recycled. Plastic effluence is most visible in emerging Asian and African nations, where trash gathering systems are often disorganized or non-existent. The rates of plastic recycling are higher in Europe (about 30 %), China (25 %), the United States recycle just 9 % of her plastic trash [78].
Thirdly, old and undesirable plastics are not always easy to dispose of. When they are left in landfill sites they take a very long time to decay. Some 18 billion pounds of plastic waste flows into the oceans every year from coastal regions leading to degradation of marine habitat which eventually affects aquatic organisms. That is the equivalent of five grocery bags of plastic trash sitting on every foot of the coast globally [67]. Diversities of the plastics used in the manufacture of many consumable products including medical devices, food packaging and water bottles, in addition to being synthetic organic polymers, also contain toxic chemicals additives like phthalates, heavy metals, bisphenol-A, brominated flame retardants, nonylphenol, polychlorinated biphenyl ethers, phenloballyanthrene, dichlorodiphenyldichloroethylene, antimony trioxide, etc. which can leach out to have adverse effects on environmental and public health. Long term usage and exposure of plastics and plastic products to high temperature can lead to leaching of these toxic chemical additives into food, drinks and water. Chaotic disposal of plastics on land and open- air burning can lead to the release of toxic chemicals into the air causing public health hazards [79-82].
By 2015, humans had produced 6.3 billion tons of plastic waste. Of that total, only 9 % was recycled; 12% was incinerated and 79% accumulated in landfills or the natural environment. With the continuation of such a trend, roughly 12 billion metric tons of plastic waste will be in landfills or the natural environment by 2050 [83]. As at 2017, the global plastic production by industry (in millions of tons) were: building construction (72 million tons), packaging (161 million tons), textiles (65 million tons), consumer products (46 million tons), transportation (30 million tons), electrical (19 million tons), industrial machinery (3 million tons), other (52 million tons). About 40 % of plastics produced is packaging, used just once and then discarded [78, 84].
8. Impacts of Plastic Wastes on Animals
A considerable aspect of the planet is bathing in rejected plastics which though harms animals, exerts an indirect effect on human health. Food materials for human intake can be undesirably affected if animals serving as meat to man are poisoned by toxic elements from plastic wastes. Microplastics have been established in more than 100 marine species, comprising fish, shrimp, and mussels that are edible [85]. Millions of animals are exterminated by plastics yearly, ranging from birds to fish to other marine organisms through eating and entanglement of plastics or starvation, even though ingestion is more common than entanglement and starvation. Their stomachs when packed with plastics decrease their appetite. Animals like the seals, whales, turtles, etc. are strangled by abandoned fishing gear or cast-off six-pack rings [79, 85, 86]. Aquatic trash constituting plastics mainly affects sea turtles, etc. that feed on jellyfishes since they regularly mistake these wastes for jellyfish. Similarly, sea birds mistake microplastics for cuttlefish (a swimming marine mollusc). Fishes also mistake plastic wastes for their natural prey [87]. These microplastics which enter the digestive systems of these animals have often been found to block their digestive tracts or pierced the organs of these animals leading to death. Plastics have also been consumed by land-based animals like elephants, hyenas, zebras, tigers, camels, cattle and other large mammals, often also leading to death [88]. Tests have also confirmed liver and cell damage and disruptions to reproductive systems, prompting some species, such as oysters, to produce fewer eggs [2, 85].
9. Public Health Impacts of Plastic Wastes
Prolonged storage of liquid in PET-based containers leads to the leaching of antimony trioxide (a compound considered as a carcinogen) due to the catalyst used in making PET which is chemically bound into the polymer at very low levels of 200 -300 parts per million. Prolonged contact to a hot environment coupled with extended storage of liquids in them might cause trace amounts of antimony to migrate into the liquid content or beverages left in them. Irrespective of the trace amount of antimony - averaging less than 1 part per billion getting leached into liquids, long storage of liquids in PET containers as well as leaving such in warm temperatures inside cars, garages and enclosed storage could be risky [52]. Equally, exposure to the harmful effect of the PVC could take place by the consumption of food or water polluted with PVC, or food enclosed in PVC packaging or containers, inhaling emissions from manufacturing or incinerating PVC or landfills or by skin contact with materials made from the PVC [89].
Varieties of the plastics employed in the manufacture of medical devices, food packaging and water bottles are made from synthetic organic polymers with toxic additives like phthalates, heavy metals, bisphenol-A (BPA), brominated flame retardants, nonylphenol, polychlorinated biphenyl ethers, phenanthrene, dichlorodiphenyldichloroethylene, antimony trioxide, etc. At various environs, these could permeate into the contents of the packages, land, air and ground waters as they are disposed from their entry point to the ecosystem and gain access to the food chain with their multiple adverse effects on ecological and public health. With continual use of plastic-based products, contact to their adverse effects are expected to thrive into new areas of the atmosphere and food chain, especially as these plastic products break up into micro-particles and their toxic chemical constituents get saturated in the environment with their unpleasant human health impacts arising at every stage of the plastic lifecycle: from its extraction, mining or transportation, production site to store shelves for consumer use and to waste management (by incineration, recycling, etc.) to their influences as air, water and soil impurities. In the course of these cycles, there are major inhalations, ingestion or skin contacts through the air, water and soil of emissions of micro-plastics and myriads of toxic effluences [79].
Up to 99 % of plastics originates from fossil fuels. The mining of oil and gas, mainly hydraulic fracturing for natural gas, discharges a range of toxic discharges like benzene and volatile organic compounds (VOCs) into the air and water in considerable amounts. Exposure to these occurs by inhalation and ingestion of contaminated air and water. Their health effects upset the immune system, sensory organs, liver and kidney and could lead to cancers, neuro-reproductive and age-related toxicity [2].
Transforming fossil fuel into plastic resins and additives discharge carcinogenic and other extremely toxic constituents like benzene, polycyclic aromatic hydrocarbons (PAHs) and styrene into the air. Exposure to these is by inhalation, ingestion and skin contact of contaminated air, water and soil, possibly leading to cancers, neurotoxicity, reproductive toxicity, low birth weight, eye and skin irritation. Human activities in patronizing consumer goods, as well as those packaged with plastics, bring about exposure to certain emissions such as heavy metals, persistent organic pollutants (POPs), carcinogens, ethylene dichloride (EDCs) and micro-plastics. Contact to these is by inhalation, ingestion and skin contact as several of these chemicals migrate from the plastic packaging to the foods they contain. They affect the renal, cardiovascular, gastrointestinal, neurological, reproductive, and respiratory systems and possibly causing cancers, diabetes and developmental toxicity [2].
10. Public Health and Environmental Influences due to Plastic Waste Management Techniques
Some of the plastic waste management approaches like incineration, co-incineration, gasification and pyrolysis release toxic metals like lead and mercury, organic substances (dioxins and furans), polycyclic aromatic hydrocarbons (PAHs), acid gases and other toxic substances to the air, water, and soils. These plastic waste management approaches expose workers in such sites and the near-by communities directly or indirectly to toxic constituents through inhalation of polluted air, direct contact with filthy soil or water and consumption of foods that were developed in surroundings made unhygienic due to these elements. Pollutants from emissions, fly ash and slag in a burn pile can extend to far places depositing in soil and water and at the end entering human bodies after being accumulated in the tissues of plants and animals. Their health effects include cancers, neurological damages and damages to immune, reproductive, nervous and endocrine system [90].
11. Public Health and Environmental Impacts of Plastic Additives
During the processing and production of plastic products, various additives are incorporated to boost their production, enhance their aesthetic values and durability [16-32]. Irrespective of the reasons for the utilization of these additives, they are associated with undesirable ecological and human health effects as follows [91]:
- Direct toxicity, as in the cases of lead, cadmium, and mercury.
- Carcinogens, as in the case of diethylhexyl phthalate (DEHP).
- Endocrine disruption, which can lead to cancers, birth defects, immune system suppression and developmental problems in children.
The public is exposed to these chemicals not only during manufacturing but also by using plastic packages since some chemicals migrate from the plastic packaging to the foods they contain. Instances of plastic-based contamination of food have been documented as emanating from several plastic types, including styrene from polystyrene, plasticizers from PVC, antioxidants from polyethylene and acetaldehyde from PET [91]. Other plastic additives with threatening influences on public health include bisphenol-A (BPA), phthalates, flame retardants, etc.
11.1 Bisphenol-A
The BPA is vastly employed in the production of polycarbonate plastics and epoxy resins. Polycarbonate plastics are engaged in the packaging of medical devices, food and drink while epoxy resins are used as lacquers to coat metal products such as food cans, bottle tops and water supply pipes. These serve as key possible sources of exposure to BPA among several other sources as BPA can leach into food from the protective internal epoxy resin coatings of any of the packages of the above items. The extent of its leaching from polycarbonate bottles into liquid may depend more on the temperature to which the liquid or bottle is exposed to, than the age of the container [92]. BPA is metabolized in the liver to form bisphenol-A glucuronide and is excreted in this form with the urine. Due to its phenolic structure, BPA has been shown to interact with estrogen receptors and acts as agonist or antagonist via estrogen receptor (ER) dependent signalling pathways and has played a role in the pathogenesis of several endocrine disorders including female and male infertility, precocious puberty, hormone-dependent tumours such as breast and prostate cancer and several metabolic disorders including polycystic ovary syndrome (PCOS), etc. [93-97].
11.2 The phthalates
Exposure to phthalates could be through inhalation, ingestion and dermal absorption [98]. Phthalates are semi-volatile organic compounds which are widely applied as plasticizers into polyvinyl chloride (PVC) to impart flexibility of plastic in consumer products such as toys, flooring materials, wallpaper, furniture, building materials, food containers or medical devices [99]. Since phthalates are not covalently bound to the polymers they can easily migrate into the environment with time and exposure [100]. Phthalates and their metabolites could have adverse effects on the human body and are associated with endometriosis [101], reduced sperm count and quality [102-103], decreased testosterone levels [104], metabolic diseases such as diabetes, obesity and breast cancer [105-107], as well as allergy and asthma [108, 109].
11.3 Flame retardants
The flame retardants commonly used as additives in plastics and other consumer products include the brominated flame retardant (BFRs), polychlorinated biphenyls (PCBs), etc. to prevent the start or slow the growth of a fire. Flame retardants are grouped based on whether they contain bromine, chlorine, phosphorus, nitrogen, metals or boron [110].
11.4 Brominated flame retardant (BFR)
The most commonly used BFR are tetrabromo-bisphenol A (TBBPA) and polybrominated diphenyl ethers (PBDEs). They have been linked to endocrine disruption among other effects [111, 112]. They are not chemically bound to the polymer making it possible for PBDEs to leach out and contaminate surrounding environment [113]. Fish and fish products contain the highest levels of BFRs and dominate the dietary intake of frequent fish eaters in Europe, while meat, followed by seafood and dairy products accounted for the highest US dietary intake [111, 114, 115]. PBDEs and TBBPA are hormone disruptors, altering the activities of thyroid hormones and oestrogen, thereby causing impaired development of both the nervous and reproductive systems [111].
11.5 Polychlorinated biphenyls (PCBs)
Marine food web has always been polluted with polychlorinated biphenyls (PCBs) for the last 70 years, particularly in seabirds through plastic bits [116]. Ingestion of PCBs may cause reproductive disorders, enhance disease proliferation, alters hormone levels and death [116-118]. PCBs have been shown to cause cancer and non-cancer related effects in animals including effects on the immune, reproductive, nervous, endocrine systems and other health disorders like endocrine changes, immunological alterations, neurodevelopment and reproductive changes [119-121]. The ban on PCBs under the Toxic Substances Control Act (TSCA) in the United States is principally due to a rising scientific indication that PCBs accumulate in the environment and can adversely impact humans and other biotas. The PCBs do not degrade easily in the environment and since they are lipophilic, they tare bio-accumulate in adipose tissue of higher predators, including humans. Thus, even though they are no longer manufactured or used by industry, they continue to be encountered as contaminants of concern at hazardous waste sites and in the environment [122].
12. Curbing the Public Health and Environmental Impacts of Plastic Wastes
About 50 % of the annual plastic production are engaged in packaging, especially in the food and healthcare sector with its resultant high plastic wastes. There should be a second uprising of plastics in which life-cycle reflections are incorporated in the manufacture and utilization resolutions to curb the ample present-day flow of plastic wastes, most of which are intended for disposal after a single-use [123]. To reduce the myriads of problems posed by plastic products, they must be designed and produced, for example as biodegradable, used and disposed thoughtfully to prevent or minimize plastic waste from entering the land, rivers and seas, etc. [55]. According to the National Geographic Society, this could be accomplished with improved waste management systems and recycling, better product design that takes into account the short life of disposable packaging and reduction in the manufacturing of unnecessary single-use plastics [2]. Contemporary research offers substantial anticipation for enhancements to human and environmental health. Better biodegradable plastics are now being developed using carbon dioxide and carbon monoxide compounds and applying metal complexes as catalysts [123]. To curb the public health and environmental impacts arising from plastic wastes, there will be a very essential need to clear up the marine systems. It will be necessary to create an adjustment in the mindset of the general public regarding the intrinsic predicaments from plastic product wastes regardless of their indispensability. The general public needs to reduce the use of plastic materials and consider alternatives where there are, especially, the disposable packaging items which could be reused where it becomes necessary instead of outrightly always picking up new ones and throwing them away after use without contemplation of the proper way it could be disposed of. While re-orientating and educating the citizens of every nation of the need to imbibe the right attitude in handling plastic products, especially with its disposal, the need for recycling of such products cannot be overemphasized [70, 124, 125]. Government policy such as imposing a very high tariff or value added-tax on the prices of all plastic packaging materials especially on the types that could be reused. This will cause the large population of the nation where such tariffs are applicable to become thoughtful and considerate in attempt to preserve such reusable packaging materials for their next use instead of outright discarding them and picking a new one in the next shopping exercises.
13. Curbing the Public Health and Environmental Impacts of Plastic Wastes Through the Management of Plastic Wastes
The packaging materials which are often hurriedly discarded after a short use-life include plastic water bottles, grocery bags, packaging, Styrofoam cups and several other pharmaceutical and packaging products. These are the largest source of plastics-related environmental damage due to the overuse of items whose long-term damage outweighs their short-term profit [70]. The practice of waste management provides services, specifically to remove waste from the human habitat to ensure hygienic living conditions. This very basic task, which was the main aim of waste management in the developed world until the end of the 19th century and still is in many developing countries, was attained in Europe with the introduction of modern sanitation practices. In parallel with the population explosion leading to increased production and consumption, the growing need and role for waste management as a “filter” between human activities and the environment became apparent, resulting in the development of safe and reliable technologies such as modern collection systems, incinerators, and sanitary landfills. Also, recycling was introduced and soon became recognized as a means to reduce the exploitation of primary resources and thus to reduce pollution created by mining and ore processing [126].
The first step in the management of plastic waste is measuring the degree of waste and this is very tasking due to of lack of details about their real sources and where they are likely ending up [125]. This statistics is very essential since material flow breakdown helps to put a figure on the flow of products and wastes [127]. This process was developed in the late 1990s by industrial ecologists for waste management [128]. It integrates diverse types of data, tracking imported or newly factory-made products inward bound economies to their use and reuse, comprising recycling, export or disposal in landfills [125]. A review of the application of material flow analysis [127] in Trinidad and Tobago, for instance, disclosed that most of the plastic arriving the nation’s landfills – a total of 49,000 tons per year – was not produced or imported, rather, they entered the nation as packaging materials around imported goods. In other words, the leading aggregate of landfilled plastic emanated from other products [125]. Further origins of plastic wastes that pollute the environment, apart from those originating as packaging around the imported goods in several countries, include food wrappers and containers (31.14 %), bottle and container caps (15.5 %), plastic bags (11.8 %), straws and stirrers (8.13 %), beverages bottles (7.27 %) and take-away (6.27 %) [129]. Plastic wastes that started as packaging materials congests tropical landfills and countries around the world throw away millions of tons of plastic junk every year. Finding methods to manage plastic wastes is overwhelming even for wealthy nations, but for smaller and less-developed nations it can be devastating [125]. Geyer et al, 2017 piloted a comprehensive plastic waste by disposal, 1980 to 2015 applying the estimated share of global plastic waste by disposal method [130]. They projected the global fate of all the plastic waste within the year under study by categorizing the waste plastics as recycled, incinerated or discarded. The data they generated showed that before 1980, recycling and incineration of plastic were taken into thought and indicates that 100 % was discarded. From 1980 for incineration and 1990 for recycling, rates increased on average by about 0.7% per annum [130]. In 2015, an estimated 55 % of global plastic waste was discarded, 25% was incinerated, and 20 % recycled. Geyer et al [130] postulated that an extrapolation of historic developments through to 2050, that incineration rates would increase to 50 %; recycling to 44 % and discarded waste would fall to 6 %, though, they stated that it should be noted that the postulation was established on the simplistic extrapolation of historical pattern and does not characterize actual estimates [130]. The entire plastic wastes that are discarded end up in the landfill and some in the ocean with their multiple environmental and human impact as have earlier been enumerated.
Globally, plastic production was estimated to be 380 million tonnes in 2018. Since 1950 to 2018, plastics of about 6.3 billion tonnes have been manufactured universally, 9 % and 12 % of which have been recycled and incinerated, respectively [130]. Plastics of about 5 million tonnes are yearly expended in the UK alone, with only about one-quarter recycled, and the rest landfilled. It has been suggested by investigators that by 2050, oceans might hold more plastics than fish in terms of weight [131]. Annually, approximately 500 billion plastic bags are used out of which a projected 13 million tonnes ends up in the ocean, destroying about 100,000 marine lives [130]. Several ways of managing plastic wastes include landfilling, plastic incineration and plastic recycling [79].
14. Landfilling
The total generation of municipal solid waste (MSW) in the USA in 2017 was 267.8 million tons or 4.51 pounds per person per day out of which, nearly 67 million tons were recycled and 27 million tons were composted. In all, more than 94 million tons of MSW were recycled and composted, corresponding to a 35.2 % recycling and composting rate. Further, more than 34 million tons of MSW (12.7 %) were combusted with energy recovery and more than 139 million tons of MSW (52.1 %) were landfilled [132]. As of 2018, within the EU Member States, 40 % of the total plastic manufacture was for packaging and out of the plastic waste control, 31 % went for landfilling [133]. It has been estimated that globally, 55 % of plastic wastes go straight to landfill or were discarded [130, 134].
In as much as landfilling is the conservative communal waste control technique in several countries, though, deficiency of space for landfills is defying the approach [135, 136]. Historically, landfilling was pretty in the UK since it is comparatively inexpensive and simple without necessarily requiring treatment, cleaning or separation. In 1999, 65% (8.4 million tonnes per annum) of the overall household waste recoverable plastics were sent to landfill in Western Europe [137], but at present in the UK, plastic waste landfilling is the least favoured waste management option [138, 139]. There is a rising ecological and public health anxiety around the probable effects of landfills due to the categories and extents of toxic chemicals and their possibilities for leaching at landfill sites [139, 140, 141]. It is currently a government policy in the UK to decrease the extent of wastes landfilled (e.g. Landfill Directive European Commission 1999/31/EC) which has been challenging to arrive as an estimated 60 % of England's municipal wastes is still sent to the landfills in comparison to a projection of 20 % and 37 % in Germany and France, separately [138, 139].
Environmental contamination and its associated dangers to public health can be curtailed if the landfills are properly controlled. The main hazards with landfilling are the chances of soil and groundwater contamination by broken plastic by-products and additives which persists in the surroundings [142, 143]. Landfill locations are not attractive to sight and there are numerous objectionable consequences for its adoption in as much as it permits less economic impact [79, 137, 144]. The major unwanted implications of the landfill are toxins, leachates and greenhouse gases being the degraded yields of plastic constituents as well as the additives with which they were manufactured [18-32, 61, 70, 71]. These seep into the soil and saturate in the groundwater or aquifers and establish environmental dangers for years [1, 4, 145]. Plants that thrive around contaminated soil absorb unhealthy ingredients. Human beings or animals may ingest these plants and get affected and may equally inhale soil filths through a dust-free in the air or absorb harmful pollutants through their skin. Water from aquifers is exploited in the cities and irrigation of the farms. It has been projected that about 97 % of the planet’s liquid fresh water is stored in aquifers and about 1.5 billion people worldwide depend on ground water as portable water [135, 136, 146-156].
Gases like methane that is released from landfill is a greenhouse gas that is 25 times more toxic than carbon dioxide with its high degree of implications in global warming and climate alteration. Methane is as well an inflammable gas which could be unsafe if permitted to accumulate in concentrations. It is certain that plastic wastes left in landfill degrade at a slower rate and constitute challenges for many years [157, 158].
However, landfilling could be adopted with minimal damage and contamination to the environment, especially the soil from where the contaminated soil water could escape into the body of rivers or oceans. Modern well-designed engineering techniques could be employed to manage the disposal of plastic wastes in landfills. Such a landfill is constructed considering varying compartmental planes, with the lowest bearing the least capacity while the volume is increased upwards. The variation in dimensions is to avoid collapsing of land. The lowest part is the linear arrangement designed with condensed clay and high-density plastics to hinder the pollution of underground water by the penetrating liquids coming from the waste materials. The other phase of the design is the drainage which takes the accumulated fluid to a treatment plant. Still another part of the strategy is the gas pool portion in which volatile and toxic gases like methane are managed and harnessed to generate electricity. In the contemporary system, the filled trash is shielded with artificial plastic or clay-like the one used below. The layers of discarded materials and soils are alternated to aid the reduction of odour and enhance fast breakdown [159-161].
15. Incineration of Plastics
Incineration of plastic wastes is a substitute to landfilling of plastic waste [162, 163]. Burning of plastic trashes in an open place is a key cause of air contamination even though there is increasing apprehensions concerning the imminent atmospheric release of hazardous chemicals in the course of incineration [134]. Often, the Municipal Solid Waste (MSW) comprising about 12 % of plastics is burnt, discharging toxic gases like dioxins, furans, mercury and polychlorinated biphenyls, hydrochloric acid, sulphur dioxide and several particulates into the air which pollute the air and modify the climate. They also aggravate respiratory conditions and stress human immunity [164]. The release of harmful fumes into the atmosphere has been the major drawback of incineration of plastic wastes [165]. The dioxins are classified under the persistent organic pollutants (POPs) which bio-accumulate in the food chain, mostly in the fatty tissues of an animal or human body. Dioxins are highly toxic and can cause reproductive and developmental problems, damage the immune system, interfere with hormones and also cause cancer [166]. Short-term exposure of humans to high levels of dioxins may result in skin lesions, such as chloracne and patchy darkening of the skin, and altered liver function. Long-term exposure is linked to impairment of the immune system, the developing nervous system, the endocrine system and reproductive functions [167].
The incineration heater of the flue systems is permanently damaged by plastics during plastic burning and the products of this plastic ignition are injurious to both humans and the surroundings [168]. Compounds of low molecular weight can vapourize directly into the air thereby poisoning the air and depending on their diversities, some may form a combustible mixture, while others may oxidize in solid form [169]. During the ignition of plastic wastes, soot, ashes and diverse powders are formed which settle on plants and soil and probably migrate to the aquatic environment. Being toxic substance, rainfall can make them sip into the soil, pollute the groundwater or absorbed by plants growing on this soil, and getting assimilated into the food chain [162, 170]. Some of these plastic incineration yields can chemically react with water and the subsequent composites can modify the pH and alter the effectiveness of the aquatic ecologies [168, 147]. Computational models for the environmental transportation and outcome of pollutants over air, soil, water and food can offer valuable details for assessing significant contact paths for humans. The prototypes recommend that ingestion of fish constitutes the main pathway of human contact to mercury and that meats, dairy products, and fish are probably the vital means for dioxins and furans [162]. Due to the potential pollution impact on the environment, plastic incineration is less employed for waste management in comparison to recycling and landfilling [79, 171].
16. Recycling of Plastics
Nearly all of the plastic that served one useful purpose or the other turns into waste and a huge amount of them eventually end up in the oceans, rivers and terrestrial territories [172]. The solution to avert such dangerous outcomes is to stop with such treatments and undertake to the recycling of the plastics that were previously used. Though reducing the use of plastic is the best, but recycling them at any time possible is the next-best key [173]. Since 1950, only 9 % of plastic used has been sufficiently recycled [84] and close to half of the plastics utilized ended up in landfill or dumped in the wild. Each year about 8 million metric tons of plastic wastes are destined to the oceans [67, 83], swept along by the world’s rivers. In addition to being a critical environmental problem, lack of recycling represents a tremendous amount of value that local economies fail to capture [175]. The goal of plastic recycling is to reduce high rates of plastic contamination while putting less pressure on virgin materials to produce brand new plastic products. This approach helps to save resources and averts plastics from landfills, incineration or accidental endpoints such as the oceans.
Currently, only PET, HDPE and PVC plastic products are recycled under curbside recycling programs. PS, PP and LDPE typically are not recycled because these plastic materials get stuck in the sorting equipment in recycling facilities causing it to break or stop. Lids and bottle tops cannot be recycled as well. “To recycle or not to recycle” is a big question when it comes to plastic recycling. Some plastic types are not recycled because it is not economically feasible to do so. Recycling offers prospects to lessen oil usage, discharges of carbon dioxide and the extents of waste necessitating discarding [176]. It takes 88 % less energy than producing plastics from new raw materials and would still remain a better way of managing plastic wastes, to minimize the tonnes of plastic wastes that surge into the oceans or landfill across the world [177]. Recycling of plastic wastes is significant to real end-of-life plastic control. Enhancing recycling rates will result in the more public consciousness and the better efficiency of recycling processes. Working proficiency will be improved if the investment is increased in research and development of advanced ways to curb wastes accumulating from plastic packaging materials. Recycling of a larger collection of post-consumer plastic products and packaging will additionally increase the idea of plastic recycling and divert more end-of-life plastic wastes from landfills and incineration. Industries and policymakers can also help stimulate recycling activity by requiring or incentivizing the use of recycled resin versus virgin plastics [177].
17. Conclusion
The twentieth century was a period of enormous progress, achieved in part by ignoring the possible consequences of the ways in which that progress was being made to happen. The conjunction of inadequately thought-out technological approaches with rapidly rising populations and an increasing culture of consumption led to the trouble besetting mankind due to the menace of plastic wastes accruing from the packaging of healthcare and food products among others. There are roles for many players in addressing the need to transform the technology–society–environment relationship. Social scientists need to understand consumption and how it may evolve and be modified. Environmental scientists and material science experts need to understand the limits imposed by a planet with limited resources and limited assimilative capacity for industrial emissions. Technologists need to develop design and manufacturing approaches that are more environmentally sound. Industrialists need to understand all these frameworks for action and develop ways to integrate the concepts within today’s corporate structures. Policymakers need to provide the proper mix of regulations and incentives to promote the long-term health of the planet rather than short-term fixes [128]. However, effort should be geared by the government agencies and policymakers of various nations, especially the developing economies on educating the populace of the dangers inherent in careless disposals of plastic wastes.
References
- Rodrigues MO, Abrantes N, Gonçalves FJM, et al. Impacts of plastic products used in daily life on the environment and human health: What is known? Environ. Toxicol. Pharmacol 72 (2019): 103239.
- Parker L. The world's plastic pollution crisis explained. Environment Explainer (2019).
- Parker L. We depend on plastic. Now we are drowning in it. National Geographic (2018).
- Ferronato N, Torretta V. Waste mismanagement in developing countries: A Review of global issues. Int J Environ Res Public Health 16 (2019): 1060.
- Kumar S, Stephen R. Smith GF, et al. Challenges and opportunities associated with waste management in India. R. Soc. Open Sci 4 (2017): 160764.
- Harsulkar AA, Sreenivas SA, Mandade RJ, et al. Polymers in mucoadhesive drug delivery system- A review. Intl. J. Drug Formulation Res 2 (2018): 61-67.
- Roiter I, Minko S. AFM single molecule experiments at the solid-liquid interface: In situ conformation of adsorbed flexible polyelectrolyte chains. J. Amer. Chem. Soc 127 (2005): 15688-15689.
- Ugoeze KC, Nwachukwu N, Nwodo CC. Excipient functionality of a novel hydrophilic biopolymer derived from Ipomoea batatas tubers. Indo Am. J. Pharm. Res 7 (2017): 360-368.
- Chukwu A, Ofoefule SI, Ugoeze K. Studies of the pharmaceutical application of a polysaccharide derived from Treculia africana fruit. Bolletino Chimico Farmaceutico 136 (1997): 539-544.
- Painter PC, Coleman MM. Fundamentals of Polymer Science: An Introductory Text, 2nd ed.; Technomic Publishing Co: Lancaster PA (1997).
- Ugoeze KC. Bioadhesive polymers for drug delivery applications. In: Bioadhesives in Drug Delivery, 1st ed.; Mittal KL, Bakshi IS, Narang JK (Editors). John Wiley and Sons: NJ, USA and Scrivener Publishing LLC: MA, USA. 29-56.
- American Chemistry Council. History of Polymers and Plastics. Available from: http://plastics.americanchemistry.com/Education-Resources/Hands-on-Plastics [accessed 17.06.20].
- Katz DA. Polymers. Available from: http://www.chymist.com/Polymer%20lab.pdf [accessed 17.06.20].
- Plastics Europe. What are plastics? Available from: https://www.plasticseurope.org/en/about-plastics/what-are-plastics [accessed 17.06.20].
- Plastics Europe. How plastics are made. Available from: https://www.plasticseurope.org/en/about-plastics/what-are-plastics/how-plastics-are-made [accessed 18.06.20].
- Chlorinated Paraffins Industry Association. Letter to Darryl Arfsten (NRC), from Robert J. Fensterheim, Executive Director, CPIA, regarding chlorinated paraffins for use in furniture flame retardants. Chlorinated Paraffins Industry Association, Washington, DC (1999).
- S. Environmental Protection Agency. Environmental Profiles of Chemical Flame-Retardant Alternatives for Low-Density Polyurethane Foam (Report). EPA 742-R-05-002A (2005).
- Thürmer A. Acid scavengers for polyolefins. In: Pritchard G. (ed). Plastics Additives. Polymer Science and Technology Series, Springer: Dordrecht 1 (1998).
- Crawford J. 2(2-hydroxyphenyl) 2H-benzotriazole ultraviolet stabilizers. Progress in Polymer Science 24 (1999): 7-43.
- Geuskens G, Bastin P, Lu V, et al. Photo-oxidation of polymers: Part IV-Influence of the processing conditions on the photo-oxidative stability of polystyrene. Polymer Degradation and Stability 3 (1981): 295-306.
- Allsopp MW, Vianello G. Poly (vinyl chloride). In: Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH: Weinheim (2012).
- Titow WV. Lubricants. In: PVC Plastics. Springer: Dordrecht (1990): 294-312.
- Christie RM. Pigments for plastics. In: Pritchard G. (ed.), Plastics Additives. Polymer Science and Technology Series, Springer: Dordrecht 1 (1998): 485-498.
- Colorants for Plastic Industry. Dyes for Plastics Industry, Pigments for Plastics Industry. Dyes and Pigments, Dyes Manufacturers, Dyes Intermediates, Pigments Suppliers, Wholesale Dyes Intermediates.
- Pritchard G. Antistatic agents. In: Pritchard G. (ed.) Plastics Additives. Polymer Science and Technology Series, Springer: Dordrecht 1 (1998): 108-114.
- Patel P, Savargaonkar N. A Review of Additives for Plastics: Slips and Anti-blocks, Plastics Engineering-Connecticut 63 (2007): 48-51.
- Hahladakisa JN, Velisa CA, Weber R, et al. An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J. Hazard. Mater 344 (2018): 179-199.
- Advani SG, Sozer EM. Process Modeling in Composites Manufacturing, 2nd ed. Taylor and Francis Group: LLC (2010).
- Dean DA, Evans ER, Hall IH. Pharmaceutical Packaging Technology. Canada: Taylor and Francis: Canada (2005).
- Pareek V, Khunteta A. Pharmaceutical Packaging: Current Trends and Future. Int J Pharm Pharm Sci 6 (2014): 480-485.
- Shivsharan US, Raut ES, Shaikh ZM. Packaging of Cosmetics: A Review. J. pharm. sci. innov. 3 (2014): 286-293.
- World Health Organization. Annex 9, Guidelines on packaging for pharmaceutical products, WHO Technical Report Series, No. 902 (2002).
- Plastics Europe. Types of plastics. Available from: https://www.plasticseurope.org/en/about-plastics/what-are-plastics/large-family [accessed 18.05.20].
- Chua H, Yu PHF, Ma CK. Accumulation of biopolymers in activated sludge biomass. Appl Biochem Biotech 78 (1999): 389-399.
- Ammala A. An overview of degradable and biodegradable polyolefins. Progress in Polymer Science 36 (2011): 1015-1043.
- Harris W. How long does it take for plastics to biodegrade? How Stuff Works (2010).
- Association of Plastics Distributors (APD) Education Committee. Amorphous and Semi-Crystalline Engineering Thermoplastics, Module 4. Basic Plastics Education tutorials. International Association of Plastics Distributors.
- May CA (ed.). Epoxy Resins-Chemistry and Technology, 2nd ed.; Marcel Deccker: New York (1988).
- Scheirs J, Priddy D (eds.). Modern Styrenic Polymers: Polystyrenes and Styrenic Copolymers, John Wiley and Sons: NJ (2003).
- Cossee P. Ziegler-Natta catalysis I. Mechanism of polymerization of α-olefins with Ziegler-Natta catalysts. J.Catal 3 (1964): 80-88.
- British Plastic Federation. Polyvinyl Chloride (PVC). Available from https://www.bpf.co.uk/plastipedia/polymers/pvc.aspx [accessed 19.05.20].
- S Department of Health and Human Services, FDA. FDA Guidance on Container Closure Systems for Packaging Human Drugs and Biologicals; May (1999).
- Rawlins EA (ed.). Bentley's Text Book of Pharmaceutics 8th ed. Bailliere Tindall:London (2002).
- Plastics Europe. The compelling facts about plastics. An analysis of plastics production, demand and recovery for 2006 in Europe.
- FDA Guidance on Container Closure Systems for Packaging Human Drugs and Biologicals, U.S Department of Health and Human Services, FDA, May (1999).
- Engelhart A. The Fiber Year 2009/10. A World Survey on Textile and Nonwovens Industry, Oerlikon Textile GmbH and Co. KG, Remscheid (2010).
- Andrady AL, Neal, MA Applications and societal benefits of plastics, Philos. Trans. R. Soc. Lond. B: Biol. Sci 364 (2009): 1977-1984.
- Miller B. 17 Biggest Advantages and Disadvantages of Plastics. Green garage (2002).
- Toloken S. SPI's new name and look to evolve the way people think about plastics - Plastics News (2016).
- International Trade Centre (ITC). Packaging for Organic Foods, Geneva: ITC, 2012. xi, 68 pages (Technical paper) Doc. No. SC-12-223.E. (Sustainability Market Guides) (2012).
- Omnexus: The material selection platform. Polyethylene Terephthalate (PET): A Comprehensive Review. Available from: https://omnexus.specialchem.com/selection-guide/polyethylene-terephthalate-pet-plastic [accessed 28.05.20].
- PET Resin Association (PETRA). The science behind PET. Available from: http://www.petresin.org/science_behindpet.asp [accessed 28.05.20].
- Precision Plastic Ball Company. Benefits of using HDPE. Available from: https://www.precisionplasticball.com/blog/post/benefits-of-using-hdpe [accessed 28.05.20].
- Ehrlich B. The PVC Debate: A Fresh Look (2014). Available from: https://www.buildinggreen.com/feature/pvc-debate-fresh-look [accessed 29.05.20].
- Thompson RC, Moore CJ, vom Saal FS, et al. Plastics, the environment and human health: current consensus and future trends. Philos Trans R Soc Lond B Biol Sci 364 (2009): 2153-2166.
- Malpass DB. Introduction to Industrial Polyethylene: Properties, Catalysts, and Processes. John Wiley and Sons: NJ (2010).
- United States Environmental Protection Agency (EPA). Municipal Solid Waste Generation, Recycling, and Disposal in the United States. Available from: https://www.epa.gov/facts-and-figures-about-materials-waste-and-recycling/advancing-sustainable-materials-management-0 [accessed 28.05.20].
- British Plastics Federation (BPF). Polyethylene (Low Density) LDPE, LLDPE. Available from: https://www.bpf.co.uk/plastipedia/polymers/LDPE.aspx [accessed 28.05.20].
- Whiteley KS, Heggs TG, Koch H, et al Polyolefins. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH (2005).
- British Plastics Federation (BPF). Polypropylene. Available from: https://www.bpf.co.uk/plastipedia/polymers/pp.aspx [accessed 28.05.20].
- Schwartz L. Ecowatch: Environmental News for a Healthier Planet and Life;. 7 Types of Plastic Wreaking Havoc on Our Health (2016).
- ILSI Europe Report Series: Packaging material 2: Material: Polystyrene for Food Packaging Applications. Available from: https://ilsi.eu/wp-content/uploads/sites/3/2018/06/PS-ILSI-Europe-Report-Update-2017_Interactif_FIN-June-2018.pdf [accessed 17.05.20].
- Plastics foodservice packaging group (PFPG). Styrene. Available from: https://www.fpi.org/Foodservice-Packaging-and-Styrene [accessed 19.05.20].
- FDA: Safety of Polystyrene Foodservice Packaging. Available from: https://www.plasticfoodservicefacts.com/foodservice-safety/fda-safety-of-polystyrene-foodservice-products/ [accessed 19.05.20].
- AAA Polymer. Recyclable Polymer Materials. Available from: https://www.aaapolymer.com/recyclable-plastic-materials/ [accessed 08.06.20].
- American Chemistry Council. Plastic Packaging Resins. https://plastics.americanchemistry.com/Plastic-Resin-Codes-PDF/ [accessed 04.01.21].
- Jambeck JR, Geyer R, Wilcox C, et al. Plastic waste inputs from land into the ocean. Science 347 (2015): 768-771.
- Hoornweg D, Bhada-Tata P, Kennedy C. Environment: Waste production must peak this century. Nature 502 (2013): 615-617.
- Wilson DC. Global Waste Management Outlook (International Solid Waste Association and United National Environment Programme (2015).
- Harth R. Health and environment: A closer look at plastics. Medical Express (2013).
- Barnes DKA, Galgani F, Thompson RC, et al. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B 364 (2009): 1985-1998.
- Wagner M, Scherer C, Alvarez-Muñoz D, et al. Microplastics in freshwater ecosystems: What we know and what we need to know. Environ. Sci. Eur 26 (2014): 12.
- Rillig MC. Microplastic in terrestrial ecosystems and the soil? Environ. Sci. Technol 46 (2012): 6453-6454.
- Zubris KAV, Richards BK. Synthetic fibers as an indicator of land application of sludge. Environ. Pollut 138 (2005): 201-211.
- Dris R, Gasperi J, Mirande C, et al. A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ. Pollut 221 (2016): 453-458.
- Zalasiewicz J, Waters CN, Sul JID, et al. The geological cycle of plastics and their use as a stratigraphic indicator of the Anthropocene. Anthropocene 13 (2016): 4-17.
- The Library of Congress. History of the Oil and Gas Industry. Business and Economics Research Advisor (5/6) (2006).
- Parker L. Planet or plastic: Fast facts about plastic pollution. National Geographic (2018).
- Alabi OA, Ologbonjaye KI, Awosolu O, et al. Public and Environmental Health Effects of Plastic Wastes Disposal: A Review. J Toxicol Risk Assess 5 (2019): 021.
- Sepulveda A, Schluep M, Renaud FG, et al. A review of the environmental fate and effects of hazardous substances released from electrical and electronic equipment during recycling: Examples from China and India. Environ Impact Assess Rev 30 (2010): 28-41.
- Zhang XL, Luo XJ, Liu HY, et al. Bioaccumulation of several brominated flame retardants and dechlorane plus in water birds from an e-waste recycling region in South China: Associated with trophic level and diet sources. Environ Sci Technol 45 (2011): 400-405.
- Alabi OA, Bakare AA, Xu X, et al. Comparative evaluation of environmental contamination and DNA damage induced by electronic-waste in Nigeria and China. Sci Total Environ 423 (2012): 62-72.
- UN Environment. Our planet is drowning in plastic pollution. Available from: https://www.unenvironment.org/interactive/beat-plastic-pollution/ [accessed 05.01.21].
- Geyer R (2017). A Plastic Planet. University of California, Santa Barbara. Available from: https://www.news.ucsb.edu/2017/018137/plastic-planet [accessed 24.05.20].
- Daniel DC. Creating a sustainable future. Jones and Bartlett Learning. Environ Sci 8 (2004): 517-518.
- Karleskint G, Small J, Turner R. Introduction to marine biology. Ceng Learning 536 (2009): 71.
- Gregory MR. Environmental implications of plastic debris in marine settings-entanglement, ingestion, smothering, hangers-on, hitch-hiking and alien invasions. Philos. Trans. R Soc. Lond B Biol Sci 364 (2009): 2013-2025.
- Hammer J, Kraak MH, Parsons R. Plastics in the marine environment: The dark side of a modern gift. Rev Environ Contam Toxicol 220 (2012): 144.
- Town T. National Institute of Health (NIH): U.S. National Library of Medicine. Poly vinyl chloride (2017).
- Center for International Environmental Law (CIEL). Plastic and Health: The Hidden Costs of a Plastic Planet. Available from: https://www.ciel.org/plasticandhealth/ [accessed 30.05.20].
- Ecology Centre. Adverse Health Effects of Plastics. Available from: https://ecologycenter.org/factsheets/adverse-health-effects-of-plastics/ [accessed 31.05.20].
- Fenichel P, Chevalier N, Brucker-Davis F. Bisphenol A: An Endocrine and Metabolic Disruptor. Ann Endocrinol (Paris) 74 (2013): 211-220.
- Konieczna A, Rutkowska Q, Rachon D. Health Risk of Exposure to Bisphenol A (BPA). Rocz Panstw Zakl Hig 66 (2015): 5-11.
- Rochester JR. Bisphenol A and human health: a review of the literature. Reprod Toxicol 42 (2013): 132-155.
- Legeay S, Faure S. Is bisphenol A an environmental obesogen? Fundam Clin Pharmacol 31 (2017): 594-609.
- Chevalier N, Fénichel P. Bisphenol A: Targeting metabolic tissues. Rev Endocr Metab Disord 16 (2015): 299-309.
- Welshons WV, Nagel SC, vom Saal FS. Large effects from small exposures. III. Endocrine mechanisms mediating effects of bisphenol A at levels of human exposure. Endocrinology 147 (2006): S56-S69.
- Kamrin MA. Phthalate risks, phthalate regulation, and public health: A review. J. Toxicol. Environ. Health B 12 (2009): 157-174.
- Wang Y, Zhu H, Kannan K. A review of biomonitoring of phthalate exposures. Toxics 7 (2019): 21.
- Bornehag C, Lundgren B, Weschler CJ, et al. Phthalates in Indoor Dust and Their Association with Building Characteristics. Environ. Health Perspect 113 (2005): 1399-1404.
- Reddy BS, Rozati R, Reddy BV, et al. General gynaecology: Association of phthalate esters with endometriosis in Indian women. Int. J. Gynaecol. Obstet 113 (2006): 515-520.
- Högberg J, Hanberg A, Berglund M, et al. Phthalate diesters and their metabolites in human breast milk, blood or serum, and urine as biomarkers of exposure in vulnerable populations. Environ. Health Perspect. 116 (2008): 334-339.
- Guo Y, Alomirah H, Cho HS, et al. Occurrence of phthalate metabolites in human urine from several Asian countries. Environ. Sci. Technol 45 (2011): 3138-3144.
- Joensen UN, Frederiksen H, Jesen MB. Et al. Phthalate excretion pattern and testicular function: A study of 881 healthy Danish men. Environ. Health Perspect 120 (2012): 1397-1403.
- James-Todd T, Stahlhut R, Meeker JD, et al. Urinary phthalate metabolite concentrations and diabetes among women in the national health and nutrition examination survey (NHANES) 2001–2008. Environ. Health Perspect 120 (2012): 1307-1313.
- Yaghjyan L, Sites S, Ruan Y, et al. Associations of urinary phthalates with body mass index, waist circumference and serum lipids among females: National health and nutrient examination survey 1999-2004. In. J. Obes 39 (2015): 994-1000.
- López-Carrillo L, Hernández-Ramírez RU, Calafat AM, et al. Exposure to phthalates and breast cancer risk in Northern Mexico. Environ. Health Perspect 118 (2010): 539-544.
- Hoppin JA, Jaramillo R, London SJ, et al. Phthalate exposure and allergy in the U.S. population: Results from NHANES 2005–2006. Environ. Health Perspect 121 (2013): 1129-1134.
- Braun JM, Sathyanarayana S, Hauser R. Phthalate exposure and children’s health. Curr. Opin. Pediatr 25 (2013): 247-254.
- National Institute of environmental Health Science (NIH). Flame Retardants. Available from: https://www.niehs.nih.gov/health/topics/agents/flame_retardants/index.cfm [accessed 01.06.20].
- Agency for Toxic Substances and Disease Registry (ATSDR). A toxicological Profile for Copper. Agency for Toxic Substances and Disease Registry, Atlanta, USA (2004).
- Lyche JL, Rosseland C, Berge G, et al. Human Health Risk Associated With Brominated Flame-Retardants (BFRs), Environ Int 74 (2015): 170-180.
- European Union Diphenyl ether, pentabromo derivative (pentabromodiphenyl ether). European Union Risk Assessment Report. Office for Official Publications of the European Committees; Luxembourg, Europe (2001): 1-124.
- Xu F, Giovanoulis G, van Waes S, et al. Comprehensive Study of Human External Exposure to Organophosphate Flame Retardants via Air, Dust, and Hand Wipes: The Importance of Sampling and Assessment Strategy. Environ Sci Technol 50 (2016): 7752-7760.
- Zheng X, Qiao L, Covaci A, et al. Brominated and phosphate flame retardants (FRs) in indoor dust from different microenvironments: Implications for human exposure via dust ingestion and dermal contact. Chemosphere 184 (2017): 185-191.
- Ryan PG, Connell AD, Gardner BD. Plastic ingestion and PCBs in seabirds: Is there a relationship? Mar Pollut Bull 19 (1988): 174-176.
- Lee K, Tanabe S, Koh C. Contamination of polychlorinated biphenyls (PCBs) in sediments from Kyeonggi Bay and nearby areas, Korea. Mar Pollut Bull 42 (2001): 273-279.
- Zitko V, Hanlon M. Another source of pollution by plastics skin cleaners with plastic scrubbers. Mar Pollut Bull 22 (1991): 41-42.
- Rochman CM, Hoh E, Kurobe T, et al. Ingested plastic transfers hazardous chemicals to fish and induces hepatic stress. Sci Rep 3 (2013b): 3263.
- Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Polychlorinated Biphenyls. Public Health Service, U.S. Department of Health and Human Services, Atlanta, GA (1997).
- Faroon O, Ruiz P. Polychlorinated biphenyls: New evidence from the last decade. Toxicology and Industrial Health 32 (2016): 1825-1847.
- Johnson GW, Quensen III JF, Chiarenzelli JR, et al. Polychlorinated Biphenyls (PCBs). In: Environmental Forensic: Contaminant specific guide. Robert D. Morisin and Brian L. Murphy (Eds.), Academic Press:Philadelphia (2005): 187-225.
- Souhrada L. Reusables revisited as medical waste adds up. Hospitals 62 (1988): 82.
- Solar Impulse Foundation. How to reduce plastic waste. Available from https://solarimpulse.com/plastic-pollution-solutions [accessed 04.06.20].
- Millette S. The first step in managing plastic waste is measuring it. The Conversion (2020).
- Mastellone ML, Brunner PH, Arena U. Scenarios of Waste Management for a Waste Emergency Area: A Substance Flow Analysis. J Ind Ecol 13 (2009): 735-757.
- Laner D, Rechberger H. Material Flow Analysis. In: Finkbeiner M. (ed). Special Types of Life Cycle Assessment. LCA Compendium – The Complete World of Life Cycle Assessment. Springer: Dordrecht (2016): 293-332.
- Allenby BR, Graedel T . Humanity and Technology. In: Industrial Ecology and Sustainable Engineering. Prentice Hall: New Jersey (2010): 2-11.
- Martinko K. 6 most common sources of plastic pollution. Treehugger (2017).
- Geyer R, Jambeck JR, Law KL. Production, use and fate of all plastics ever made. Science Advances 3 (2017): e1700782.
- Sutter JD. How to stop the sixth mass extinction. CNN (2016).
- United States Environmental Protection Agency (EPA). National Overview: Facts and figures about material waste and recycling. Available from: https://www.epa.gov/facts-and-figures-about-materials-waste-and-recycling/national-overview-facts-and-figures-materials#Landfilling [accessed 06.06.20].
- News: European Parliament. Plastic waste and recycling in the EU: facts and figures. Available from: https://www.europarl.europa.eu/news/en/headlines/society/20181212STO21610/plastic-waste-and-recycling-in-the-eu-facts-and-figures [accessed 06.06.20].
- Ritchie H, Roser M. (2018). Plastic Pollution. Published online at OurWorldInData.org. Available from: https://ourworldindata.org/plastic-pollution [accessed 06.06.20].
- Joshi R, Ahmed S. Status and challenges of municipal solid waste management in India: A review. Cogent Environ. Sci 2 (2016): 1139434.
- Idris A, Inane B, Hassan MN. Overview of waste disposal and landfills/dumps in Asian countries. J. Mater Cycles and Waste Manag 16 (2004): 104-110.
- Association of Plastics Manufacturers in Europe (APME)). Plastics consumption data by sector; Brussels, Belgium (2002).
- Waste and Resources Action Programme (WRAP). Understanding plastic packaging and the language we use to describeit. Available from: http://www.wrap.org.uk/sites/files/wrap/Understanding%20plastic%20packaging%20FINAL.pdf [accessed 06.06.20].
- House of Commons Library (2020). Plastic waste. Available from: https://commonslibrary.parliament.uk/research-briefings/cbp-8515/ [accessed 06.06.20].
- Miller R. The landscape for biopolymers in packaging. Miller-Klein Associates report. Summary and Full Report available from the National Non-Food Crops Centre (NNFCC); 2005, Heslington, York, UK. Available from: http://miller-klein.com/wp-content/uploads/2016/12/2007LandscapeforBiopolymers.pdf [accessed 06.06.20].
- Song JH, Murphy RJ, Narayan R, et al. Biodegradable and compostable alternatives to conventional plastics. Philos Trans R Soc Lond B Biol Sci 364 (2009): 2127-2139.
- Oehlmann J, Schlte-Oehlmann U, Kloas W, et al. A critical analysis of the biological impacts of plasticizers on wildlife. Philos Trans R Soc Lond B Biol Sci 364 (2009): 2047-2062.
- Teuten EL, Saquin JM, Knappe DR, et al. Transport and release of chemicals from plastics to the environment and to wildlife. Philos Trans R Soc Lond B Biol Sci 364 (2009): 2027-2045.
- Environment Victoria. The problem with landfill; 2013. Available from https://environmentvictoria.org.au/resource/problem-landfill/ [accessed 07.06.20].
- Metropolitan Transfer Station. Modern waste management techniques; 2018. Available from: https://www.metropolitantransferstation.com.au/blog/modern-waste-management-techniques [accessed 07.06.20].
- Papadopoulou MP, Karatzas GP, Bougioukou GC. Numerical modelling of the environmental impact of landfill leachate leakage on groundwater quality-a field application. Environ Model Assess 12 (2007): 43-54.
- Nesaratnam S. Effects of pollutants on the aquatic environment. Adapted extract from the Open University course T868 Environmental monitoring and protection. Available from https://www.open.edu/openlearn/ocw/mod/oucontent/view.php?id=18241&printable=1 [accessed 08.06.20].
- Ramos JAL, Barrón LER, Sandoval IM. Combined use of aquifer contamination risk maps and contamination indexes in the design of water quality monitoring networks in Mexico. Geofís. Int 43 (2004): 641-650.
- Taylor R, Allen A. Waste disposal and landfill: Potential hazards and information needs. In: Schmoll O, Howard G, Chilton J, Chorus I (eds). Protecting Groundwater for Health: Managing the Quality of Drinking-water Sources. IWA Publishing: London (2006): 643-646.
- Soil Science Society of America (SSSA). Soil contaminants; 2020. Available from: https://www.soils.org/about-soils/contaminants [accessed 07.06.20].
- University of Massachusetts Amherst Centre for Agriculture, Food and the Environment. Water: Supply and sources. Available from: https://ag.umass.edu/greenhouse-floriculture/greenhouse-best-management-practices-bmp-manual/water-supply-sources [accessed 07.06.20].
- Food and Agriculture organization of the United Nations. Water for Sustainable Food and Agriculture: A report produced for the G 20 Presidency of Germany; 2017. Available from: http://www.fao.org/3/a-i7959e.pdf [accessed 07.06.20].
- Shammas MI, Jack G. Management of the Salalah plain aquifer, Oman. In: Bhattacharya P, Ramanathan AL, Mukherjee AB, Bundschuh J, Chandrasekharam D, Keshari AK (eds). Groundwater for Sustainable Development: Problems, Perspectives and Challenges. CRC Press: Florida (2008): 410-420.
- Gulf of Maine Aquarium. Aquifer: Source of Pure Water; 1998. Available from: https://www.safewater.org/fact-sheets-1/2017/1/21/aquifers [accessed 07.06.20].
- American Groundwater Trust. The ABC of Aquifers. Available from: http://www.tafths.org/ourpages/auto/2012/1/19/46927603/abcsofaquifers.pdf [accessed 07.06.20].
- S. Geological Survey. Aquifers and Groundwater. Available from: https://www.usgs.gov/special-topic/water-science-school/science/aquifers-and-groundwater?qt-science_center_objects=0#qt-science_center_objects [accessed 07.06.20].
- European commission. Climate Action: Causes of climate change. Available from: https://ec.europa.eu/clima/change/causes_en [accessed 07. 06.20].
- United States Environmental Protection Agency. Overview of Greenhouse Gases. Available from: https://www.epa.gov/ghgemissions/overview-greenhouse-gases [accessed 05.01.21].
- Cossu R, Stegmann R. Solid Waste Landfilling: Concepts, Processes, Technology. Elsevier Inc: Amsterdam (2019).
- Cointreau S. Sanitary landfill design and siting criteria (English). Infrastructure notes; 2004. Washington, DC: World Bank. Available from: http://documents.worldbank.org/curated/en/461871468139209227/Sanitary-landfill-design-and-siting-criteria [accessed 07.06.20].
- Interstate Technology and Regulatory Council. Characterization, Design, Construction, and Monitoring of Bioreactor Landfills. Technical/Regulatory Guideline (2006).
- National Research Council. Waste Incineration and Public Health. Washington, DC: The National Academies Press: Washington, DC (2000).
- Buekens A. Incineration Technologies. In: Meyers RA. (eds). Encyclopedia of Sustainability Science and Technology. Springer: New York (2012).
- Verma R, Vinoda KS, Papireddy M, et al. Toxic Pollutants from Plastic Waste- A Review. Procedia Environ Sci 35 (2016): 701-708.
- National Research Council (US) Committee on Health Effects of Waste Incineration. Waste Incineration & Public Health. Washington (DC): National Academies Press: Washington, DC (2000): 34-70.
- National Institute of Environmental Health Sciences. Dioxins. Available from: https://www.niehs.nih.gov/health/topics/agents/dioxins/index.cfm [accessed 09.06.20].
- World Health organization (WHO). Dioxins and their effects on human health (2016).
- Nagy A, Kuti R. The Environmental Impact of Plastic Waste Incineration. AARMS 15 (2016): 231-237.
- Carson P, Mumford C. Hazardous Chemicals Handbook, 2nd ed. Butterworth-Heinemann: Oxford (2002).
- Shemwell BE, Levendis YA. Particulates Generated from Combustion of Polymers (Plastics). J Air Waste Manag Assoc 50 (2000): 94-102.
- Abdel-Shafy HI, Mansour MSM. Solid waste issue: Sources, composition, disposal, recycling, and valorization. Egypt. Pet 27 (2018): 1275-1290.
- Cho R. Earth Institute, Columbia University; 2012. What happens to all that plastics? Available from: https://blogs.ei.columbia.edu/2012/01/31/what-happens-to-all-that-plastic/ [accessed 14.06.20].
- How is Plastic Recycled: Step by Step. Available from: https://greentumble.com/how-is-plastic-recycled-step-by-step/ [accessed 14.06.20].
- Rhodes CJ. Plastic pollution and potential solutions. Sci Prog 101 (2018): 207-260.
- d’Ambrières W. (2019). Plastics recycling worldwide: current overview and desirable changes. The Journal of Field Action, Field Actions Science Report, Special Issue 19 (2019): 12-21.
- Mwanza BG, Mbohwa C. Drivers to Sustainable Plastic Solid Waste Recycling: A Review. Procedia Manufacturing 8 (2017): 649-656.
- Leblanc R. An overview of plastic recycling. The Balance Small Business; 2019. Available from: https://www.thebalancesmb.com/an-overview-of-plastic-recycling-4018761 [accessed 15.06.20].



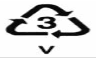


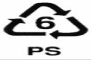


 Impact Factor:
* 3.6
Impact Factor:
* 3.6
 Acceptance Rate:
76.49%
Acceptance Rate:
76.49%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
