Spectrum of Bleeding Disorders In Pakistan: A Cross-Sectional Study
Muhammad Sohaib Asghar1 *, Ayesha Asghar2, Fatima Asghar3, Mohammed Akram4
1Resident Physician of Internal Medicine, Dow University Hospital, Dow University of Health Sciences, Karachi, Pakistan
2Graduate, Masters of Science in Biochemistry, Karachi University, Karachi, Pakistan
3Graduate, BDS (Dentistry), Dow International Dental College, Karachi, Pakistan
4Intern of Internal Medicine at Liaquat National Hospital & Medical College, Karachi, Pakistan
*Corresponding Author: Muhammad Sohaib Asghar, Resident Physician of Internal Medicine, Dow University Hospital, Dow University of Health Sciences, Karachi, Pakistan
Received: 13 July 2020; Accepted: 23 July 2020; Published: 31 July 2020
Article Information
Citation: Muhammad Sohaib Asghar, Ayesha Asghar, Fatima Asghar, Mohammed Akram. Spectrum of Bleeding Disorders In Pakistan: A Cross-Sectional Study. Fortune Journal of Health Sciences 3 (2020): 110-121.
View / Download Pdf Share at FacebookAbstract
Abstract
This is a cross-sectional study conducted to determine the frequency and spectrum of bleeding disorders in different regions of Pakistan and to improve facilities to cure them. Samples were collected from different regions of Pakistan. There bleeding presentation and history of the family were recorded. 435 patients were selected from the different regions of Pakistan; 318 male (73%) and 117 female (26%). Laboratory analysis included Prothrombin time, Activated partial thromboplastin time, Bleeding time, platelet counts and fibrinogen assay were performed. According to their results, vWF:Ag, vWF:RCo, inhibitor screening, Bethesda test, urea clot solubility test, and platelet aggregation test were performed to diagnosed bleeding disorders in patients. Out of 435 patients, 283 constitute CBD, 80 diagnosed with RBD, and 72 remain undiagnosed. Patients diagnosed with 52% hemophilia A, 1.4% with its inhibitor, 8.4% with hemophilia B, 0.35% with its inhibitor, 3.88% with VWD, 33.5%with its type III. A rare bleeding disorder is diagnosed in 80 subjects with 20% fibrinogen, 16.25% FXIII, 15% FVII, 11.25% FV, 10% vitamin k dependent factors, 8.25% GT, 5% FXII and epinephrine, 2.5% FII and FX and 1.25% FV + FVIII, BSS and FXI deficiencies. Clinical bleeding episodes varied in patients with respect to its deficiencies include gum bleeding, menorrhagia, hematoma, bruises, umbilical cord bleeding, hemorrhagia, and hemarthrosis. Study shows that hemophilia A is more frequent than hemophilia B. vWD type III has a higher prevalence. In RBD, fibrinogen deficiency is the most common, followed by FXIII, FVII, and FV respectively.
Keywords
<p>Bleeding; Disorders; Hemophilia</p>
Article Details
Introduction
The clinical presentation of bleeding disorder prevalence is worldwide. Haemophilia and von Willebrand disease is the most frequent disorder among all the bleeding disorders. They both constitute about 95 to 97% of all inherited bleeding disorders [1]. Prevalence of other inherited recessive disorders, transmitted in both sexes was rare and uncommon among them [1]. But their prevalence is comparatively high in the areas where marriages among blood-relations are common [2]. Pakistan has high rates of consanguineous marriages [3]. The disorders which are found rarely are deficiencies of factors that involve in the coagulation process and abnormal functioning of platelets. These disorders constitute 3 to 5% of all bleeding disorders [1].
The rare bleeding disorders include deficiencies of clotting factors I, II, V, VII, X, XI, XIII, vitamin K dependent clotting factors [II, VII, IX, and X], V & VIII, and von Willebrand disease type 3, Glanzmann thrombasthenia (GT) and Bernard-Soulier syndrome (BSS). The presentation and bleeding pattern and episodes in these patients vary according to the etiology of each disorder. Rarely, Life-threatening bleeding episodes also occurred, e.g. CNS or musculoskeletal bleeding.
Haemophilia is a quantitative and qualitative X-linked inherited bleeding disorder. It is characterized as hemophilia A and hemophilia B with deficiency and defects of factor VIII and IX respectively with the frequency of about 1 in 5,000 and 1 in 25,000 [4]. Depending upon its factor level it categorized into severe (factor level <1 IU/dl), moderate (1-5 IU/dl), and mild forms (5-40 IU/dl) [5]. VWD is an inherited disorder of autosomal dominant or recessive pattern. It is characterized as a defect in von Willebrand factor (large multimeric glycoprotein) gene on chromosome 12 [6]. VWD is characterized as a quantitative defect of VWF as type I and III and type II as a qualitative defect of factor. VWD type III is the rarest but severe type and it is associated with the total absence of VW factor [7].
Bernard-Soulier syndrome is an autosomal recessive disorder that has a prevalence of 1/1,000,000 [8]. Platelets from BSS patients lack the major surface membrane glycoprotein complex, glycoprotein (GP) Ib-IX-V [9]. Glanzmann thrombasthenia is the most frequently diagnosed inherited disorder of platelet function with prevalence 1 per 10, 00,000 [10]. Patients with GT lack or have nonfunctional αIIbβ3 integrin [11]. Fibrinogen deficiency has a prevalence of 1 in 1,000,000. It is divided into types I (afibrinogenemia) and II (hypofibrinogenemia) on the basis of quantitative defect and dysfibrinogenemia and hypodysfibrinogenemia based on qualitative defect [12]. Prothrombin deficiency has a prevalence of approximately 1 in 2,000,000 [13]. Factor V [FV] deficiency has Prevalence of about 1 per 1,000,000 [14]. Combined deficiency of factor V and VIII are associated with mutations in the LMAN1 and MCFD2 genes [15]. Factor VII deficiency presents as a hemophilia-like bleeding disorder with an estimated prevalence is 1 in 300,000 - 500,000 [16]. Factor X has an estimated prevalence of 1 in 1,000,000 individuals [17]. Occurrence of factor XI is approximately 1 per 1,000,000 [18]. Factor XI, XII, XIII is also an inherited disorder and prevalence among the population. Vitamin K is required in the synthesis of factors like factor II, VII, XI, X, prothrombin and protein C, and S, which are the part of the coagulation pathway, its deficiency also serves as a bleeding disorder [19, 20] Approx. 1 in 2 million of the population is suffered from FII and FVIII deficiencies and 1 in 50,000 patients were recorded for FVII deficiency in a general population [21].
The aim of the study is to determine the frequency and spectrum of bleeding disorders in different regions of Pakistan and to improve facilities to cure them.
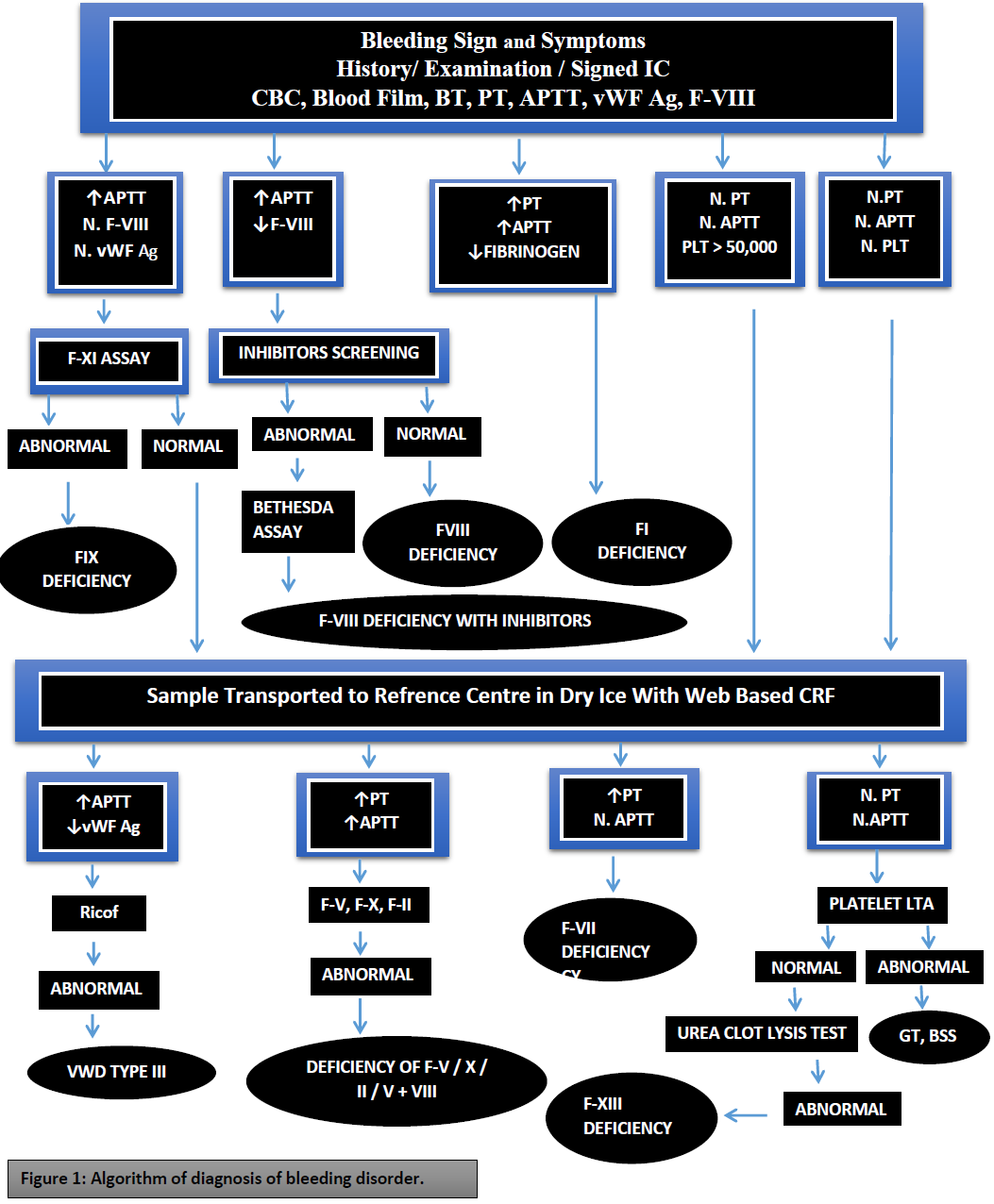
Figure 1: Algorithm of diagnosis of bleeding disorder.
Methods
The study was carried out in the department of biochemistry, University of Karachi, Pakistan, and department of hematology, National Institute of Blood Disease and Bone Marrow Transplantation, Karachi, Pakistan. This cross-sectional multi-center study was coordinated from NIBD (hematology tertiary care center) in Karachi. Samples were collected from patients residing in urban and rural areas of the province of Sindh (NIBD and Fatimid foundation Karachi), Punjab [Chughtai’s laboratory and the Children’s Hospital Lahore (CHL)], Islamabad [Pakistan Atomic Energy Commission hospital (PAEC)] and Khyber Pakhtunkhwa (KPK) [Hayatabad Medical College (HMC), Lady Reading Hospital (LRH), Peshawar]. Some patients may either misdiagnosed or remain undiagnosed and labeled as bleeding disorder patients because there was a severe lack of diagnostic facilities in Pakistan. It is a cross-sectional study in which patients with a history of bleeding tendencies were selected and their history, symptomal bleeding episodes, and clinical findings were recorded. Data collected included a detailed history of bleeding, family history, and demographic data. The bleeding score was assessed using the Tosetto bleeding score method [22]. Samples were collected into trisodium citrate 0.109 M 3.2%. Initially, the samples were collected and processed at local reference centers and platelet-poor plasma was collected in labeled aliquots and then transported to study coordinating center (NIBD, Karachi) in different phases via maintaining cold chain aliquots that centrifuged at 1200-1500 x G for 15 minutes under a suitable temperature of about 18-25°C then samples were frozen at -40°C or -80°C for required time without loss of most hemostatic activity. Laboratory analysis included prothrombin time (PT), activated partial thromboplastin time (APTT), bleeding time (BT), platelet counts, and fibrinogen assay were performed. First-line coagulation profile including manual prothrombin time (PT) [23] and activated partial thromboplastin time (APTT) [24]. Bleeding time was determined using the Dukes methodology and fibrinogen levels by the Clauss method [25]. Patients with prolonged APTT were tested for factors VIII (FVIII) and IX (FIX). This was done using the one-stage APTT based factor assay [26]. If FVIII was low, von Willebrand factor: antigen (vWF:Ag) and von Willebrand factor: ristocetin cofactor (vWF:RCo) were performed. Inhibitor screening and Bethesda test are performed when FVII is low. When PT and APTT both were prolonged and fibrinogen is low, FV, FX, and FII were tested. Factors II, V, and X were tested using an assay based on PT. Factors II, V, and X were tested using an assay based on PT.
In patients with normal PT and APTT, we suspected platelet disorder like Glanzmann thrombasthenia and Bernard Soulier Syndrome. Peripheral blood samples were analyzed to identify any platelet morphological abnormalities. Platelet aggregation studies were done when there was prolonged BT on a standard aggregometer (Helena Aggram, Beaumont, Texas) using standard aggregation reagents (ADP: 2.25µM; Adrenaline: 5µM; Collagen 4µg/ml; Ristocetin 0.5mg/ml; Arachidonic acid: 500µg/ml). Urea clot solubility test was done when all coagulation tests were normal using commercially available thrombin, on those patients whose tests turned to be negative for other coagulation tests.
Results
Out of 435 patients diagnosed with bleeding disorders, 283 subjects had CBDs (65%) and 80 subjects diagnosed with RBDs (18%). Consanguinity is present in 89% of cases.
Among 283 subjects with CBDs, 236 (83%) were male and 47 (16%) were female. 148 subjects suffering from hemophilia A (52.2%) while only 4 subjects with its inhibitors (1.4%). 24 subjects diagnosed with hemophilia B (8.4%) while 1 patient with its inhibitor (0.35%). VWD is found in 11 patients (3.88%) while type III is occurring in 95 patients (33.5%). Haemophilia A is more frequent as compared to hemophilia B. VWD type 3 is present in a high proportion in the population (Figure 2).
Rare bleeding disorder found in 80 patients (18.39%) 46 males (57.5%) and 34 females (42.5%). The most common deficiency was fibrinogen deficiency with 16 patients (20%), followed by F-XIII deficiency with 13 patients (16.25%), 12 with F-VII deficiency (15%), 9 with F-V deficiency (11.25%), Vitamin K dependent factor deficiency was found in 8 patients (10%), platelets function defects found in 7 with GT (8.75%), 4 with F-XII deficiency (5%), epinephrine receptor defect found in 4 patients (5%) 2 suffer with F-II deficiency (2.5%), 2 with F-X deficiency (2.5%), 1 with F-V + VIII deficiency (1.25%), 1 with F-XI deficiency (1.25%), and 1 with BSS (1.25%). (Figure 3) 70 patients were remain undiagnosed (16.09%).
Comparative patient’s data and hemostatic status diagnosed by performing different tests on several patients samples taken from different labs of provinces of Pakistan (Figure 4).
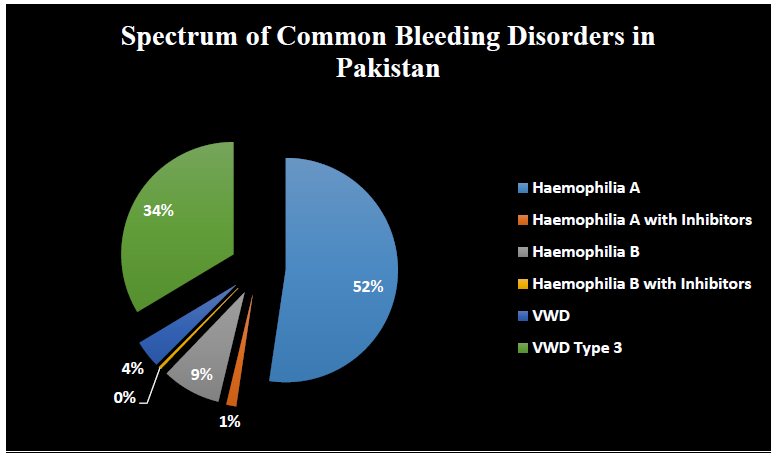
Figure 2:
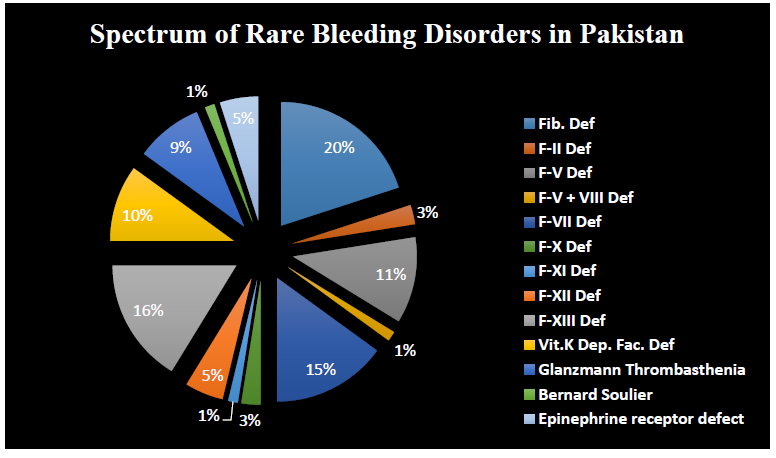
Figure 3:
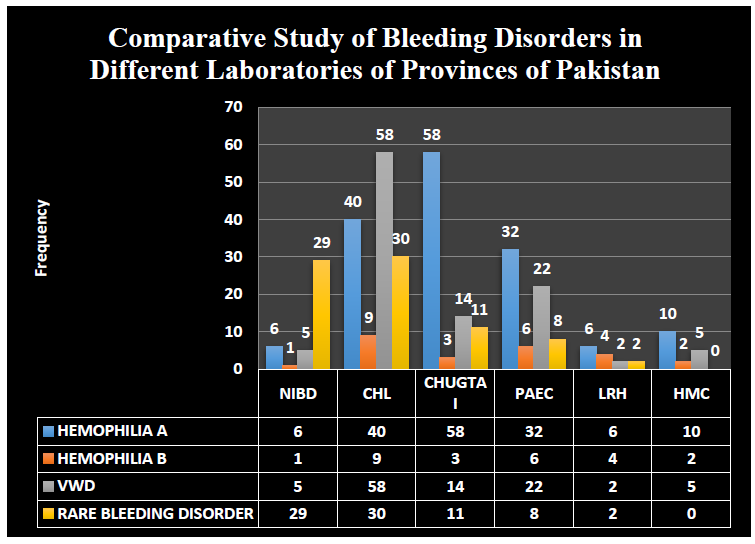
Figure 4:
Clinical bleeding episodes are reported as gum bleeding (52-36%), easy bruising (37%), Spontaneous bleeding (6%), Menorrhagia (6%), hemorrhage (4%) Gum bleeding was more prominent in patients with Glanzmann thrombasthenia, Bernard-Soulier syndrome, hemarthrosis was most common in patients suffering from factor VII deficiency, and hematoma is prominent with patients with factor XIII, factor V and vitamin K dependent clotting factor deficiency. Patients with factor XIII deficiency had the highest incidence of prolonged umbilical cord bleeding. Prolonged bleeding after trauma was associated with factor VII and vitamin K dependant clotting factor deficiency, GT, and BSS. Easy bruising is a prominent feature of GT, BSS, and factor XI deficiency according to our study cohort (Table 1).
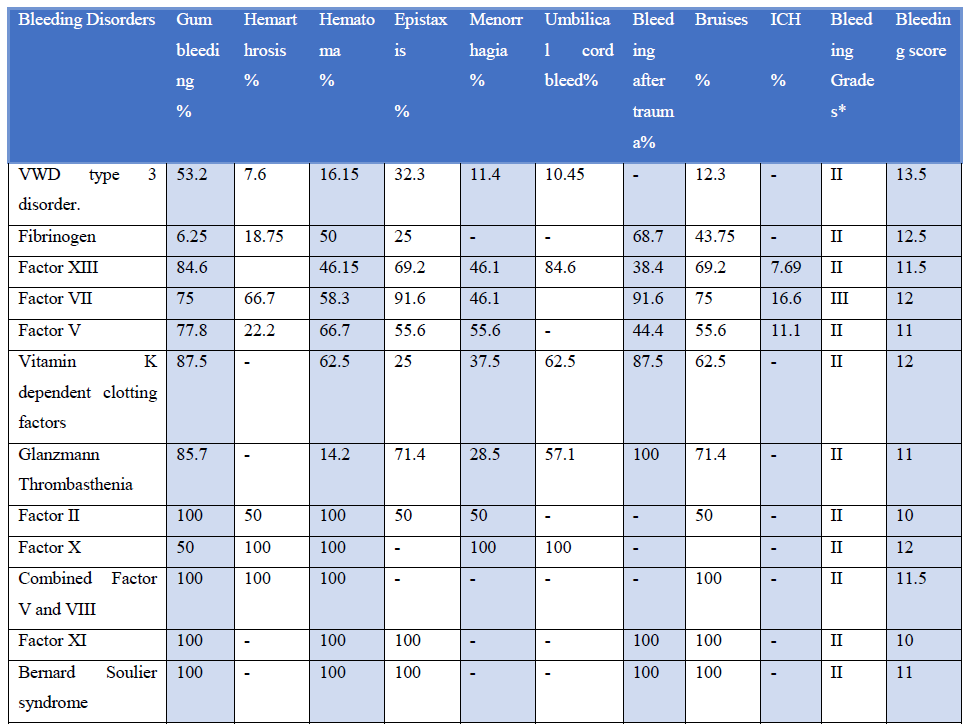
Table 1:
Discussion
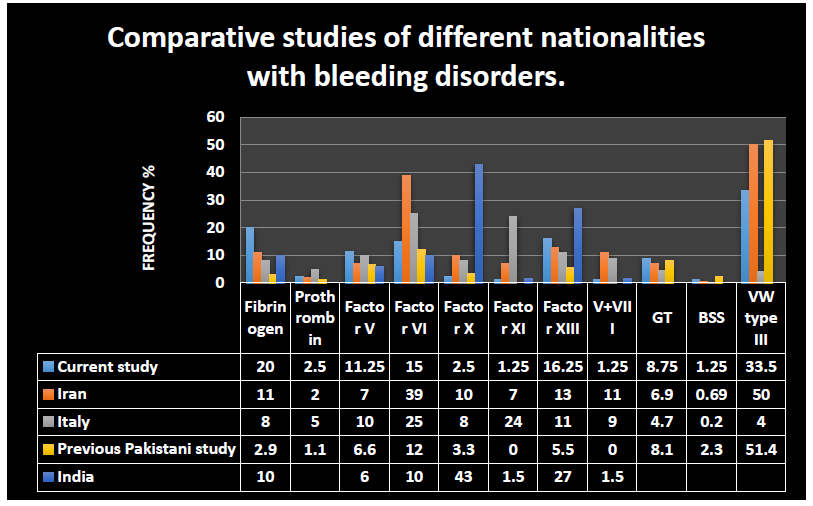
Figure 5:
In Figure 5, we compared our results with a study conducted to investigate patients with recessively inherited coagulation disorders from Italy, Iran [1], and India [27] countries with high rates of bleeding disorders due to consanguineous marriages. In the present study out of 435 patients suspected of having a bleeding disorder, 80 were diagnosed as having rare bleeding disorders. After hemophilia A with 148 patients, vWD type 3 was the most common with 95 patients (33.5%), although, in a similar, local study, the percentage was 51.4% [28], in Iran, it was 50% and in Italy just 4% [1]. The second most common deficiency found in this study was fibrinogen deficiency in 16 patients (20%). In an earlier, local study, it occurred at a rate of 2.9% [28], 11% in the Iranian study, 8% in the Italian study [1], and 10% in the Indian study [27]. This bleeding disorder is a clinically important diagnosis in this geographical location because if diagnosed early it can prevent fatal bleeding episodes. GT, a relatively well-understood platelet disorder, was diagnosed in 7 (8.75%) of patients, the rate of diagnosis was 6.9% in the Iranian study, 4.7% in the Italian study, and 8.1% in an earlier Pakistani study.
Conclusion
It can be concluded from this data that hemophilia A is considered to be highly prevalent among all bleeding disorders. Hemophilia A is more frequent than hemophilia B. VWD type 3 has the highest incidence amongst the ARBDs in our study cohort followed by fibrinogen deficiency. Glanzmann thrombasthenia was found to be the third most common disorder among ARBDs. The incidence of autosomal recessive bleeding disorders in this region is higher than previously thought.
References
- Mannucci PM, Duga S, Peyvandi F. Recessively inherited coagulation disorders. Blood 104 (2004): 1243-1252.
- Bhopal RS, Petherick ES, Wright J, et al. Potential social, economic and general health benefits of consanguineous marriage: results from the Born in Bradford cohort study. European Journal of Public Health (2013).
- Hussain R, Bittles AH. The prevalence and demographic characteristics of consanguineous marriages in Pakistan. Journal of Biosocial Science 30 (1998): 261-275.
- Sajid R, Khalid S, Mazari N, et al. Clinical audit of inherited bleeding disorders in a developing country. Indian J Pathol Microbiol 53 (2010): 50-53
- Sajid R, Khalid S, Mazari N, et al. Clinical audit of inherited bleeding disorders in a developing country. Indian Journal of Pathology and Microbiology 53 (2010): 50.
- Castaman G, Federici AB, Rodeghiero F, et al. Von Willebrand's disease in the year 2003: towards the complete identification of gene defects for correct diagnosis and treatment. Haematologica. 88 (2003): 94-108.
- Borhany M, Shamsi T, Naz A, et al. Congenital bleeding disorders in Karachi, Pakistan. Clinical and Applied Thrombosis/Hemostasis. 17 (2011): E131-E137.
- Lanza F. Bernard-Soulier syndrome (hemorrhagiparous thrombocytic dystrophy). Orphanet Journal of Rare Diseases 1 (2006): 1-6.
- Andrews RK, Berndt MC. Bernard-Soulier syndrome: an update. Seminars in Thrombosis and Hemostasis 39 (2013): pp. 656-662.
- Nurden AT. Glanzmann thrombasthenia. Orphanet Journal of Rare Diseases 1 (2006): 10.
- Nurden AT, Pillois X, Nurden P. Understanding the genetic basis of Glanzmann thrombasthenia: implications for treatment. Expert Review of Hematology 5 (2012): 487-503.
- Peyvandi F, Cattaneo M, Inbal A, et al. Rare Bleeding Disorders. Haemophilia. 14 (2008): 202-210.
- Lancellotti S, Basso M, De Cristofaro R. Congenital prothrombin deficiency: an update. Seminars in Thrombosis and Hemostasis 39 (2013): pp. 596-606.
- Huang JN, Koerper MA. Factor V deficiency: a concise review. Haemophilia : the official journal of the World Federation of Hemophilia 14 (2008): pp. 1164-1169.
- Zhang B, McGee B, Yamaoka JS, et al. Combined deficiency of factor V and factor VIII is due to mutations in either LMAN1 or MCFD2. Blood 107 (2006): 1903-1907.
- Mariani G, Bernardi F, editors. Factor VII deficiency. Semin Thromb Hemost 35 (2009): pp. 400-406.
- Spreafico M, Peyvandi F. Combined Factor V and Factor VIII Deficiency. Seminars in Thrombosis and Hemostasis 35 (2009): pp. 390-399
- Gomez K, Bolton?Maggs P. Factor XI deficiency. Haemophilia 14 (2008): 1183-1189.
- Oldenburg J, Von Brederlow B, Fregin A, et al. Congenital deficiency of vitamin K dependent coagulation factors in two families presents as a genetic defect of the vitamin K-epoxide-reductase-complex. Thrombosis and Haemostasis 84 (2000): 937-941.
- Pechlaner C, Vogel W, Erhart R, et al. A new case of combined deficiency of vitamin K dependent coagulation factors. Thrombosis and Haemostasis 68 (1992): 617.
- Palla R, Peyvandi F, Shapiro AD. Rare bleeding disorders: diagnosis and treatment. Blood, The Journal of the American Society of Hematology 125 (2015): 2052-2061.
- Tosetto A, Castaman G, Plug I, et al. Prospective evaluation of the clinical utility of quantitative bleeding severity assessment in patients referred for hemostatic evaluation. Journal of Thrombosis and Haemostasis 9 (2011): 1143-1148.
- Caen J, Larrieu MJ, Samama M. L'hémostase. Méthodes d'Exploration et Diagnostic Pratique 1 (1975): 315.
- Langidell RD, Wagner RH, Brinkhous KM. Effect of antihemophilic factor on onestage clotting tests; a presumptive test for hemophilia and a simple one-stage antihemophilic factor assy procedure. J Lab Clin Med 41 (1953): pp. 637– 647.
- Acharya SS, Dimichele DM. Rare inherited disorders of fibrinogen. Haemophilia: the official journal of the World Federation of Hemophilia 14 (2008): 1151-1158.
- Kirkwood TB, Snape TJ. Biometric principles in clotting and clot lysis assays. Clin Lab Haematol 2 (1980): 155-167.
- Sharma SK, Kumar S, Seth T, et al. Clinical profilem of patients with rare inherited coagulation disorders: a retrospective analysis of 67 patients from northern India. Mediterr J Hematol Infect Dis 4 (2012): pp. 57.
- Borhany M, Shamsi T, Fatima N, et al. Rare Bleeding Disorders are not so Rare in Pakistan. J Hematol Thromb Dis 2 (2013): pp.122.


 Impact Factor: * 6.2
Impact Factor: * 6.2 Acceptance Rate: 76.33%
Acceptance Rate: 76.33%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 