Coexistence of Axillary Tuberculosis Lymphadenitis and Primary Sternal Tuberculosis
Suphi Aydin1, Ahmet Dumanli1, Gürhan Öz1, Aydin Balci2, Adem Gencer3
1Department of Thoracic Surgery, Faculty of Medicine, Afyonkarahisar Health Science University-TURKEY
2Department of Pulmonology, Faculty of Medicine, Afyonkarahisar Health Science University-TURKEY
3Afyonkarahisar Public Hospital, Department of Thoracic Surgery, Afyonkarahisar, TURKEY
*Corresponding author: Suphi Aydin, Department of Thoracic Surgery, Faculty of Medicine, Afyonkarahisar Health Science University-TURKEY.
Received: 20 October 2021; Accepted: 28 October 2021; Published: 30 October 2021
Article Information
Citation: Suphi Aydın, Ahmet Dumanlı, Gürhan Öz, Aydın Balcı, Adem Gencer. Coexistence of axillary tuberculous lymphadenitis and primary sternal tuberculosis. Archives of Microbiology and Immunology 5 (2021): 384-391.
View / Download Pdf Share at FacebookAbstract
The appearance of tuberculosis primarily in the sternum or lymph nodes is a condition that causes difficulties and delays in diagnosis. Few cases have been reported in the literature. A 56-year-old male patient with diabetes presented with a complaint of swelling that had been growing for 6 months in the right axillary region and on the sternum. Various antibiotic treatments were applied for these swellings but no response was received. It was learned that the patient complained of fever from time to time, and had no complaints of night sweats and weight loss. On physical examination, fever was determined at 37.5 ° C, 10x6 cm, hard consistency, mobile, painless mass located on the right axillary fossa, and a hard mass, fixe, painful mass on the manibrium stern. There was no hyperemia, temperature increase and fluctuation on the masses. In the computed thorax tomographic examination of the patient, necrotic soft tissue was observed in the mediastinum and bilateral hilar region, causing lymphadenopathy and destruction of bone structure. Since fine needle aspiration biopsies from the right axillary region and over the sternum were not diagnostic, the right axillary mass was completely removed and an excisional biopsy was performed on the sternum. It was compatible with tuberculosis as a result of histopathological examinations of the pieces sent.
Keywords
<p>Tuberculosis, Lymphadenopathy, Biopsy, Bone, Diagnosis</p>
Article Details
1. Introduction
Tuberculosis is highly controlled in developed countries. In developing countries, it has become an important health problem due to the increase in the incidence of human immunodeficiency virus (HIV) and migration to developed countries [1]. According to the report published by the World Health Organization (WHO) in 2019, 10 million new tuberculosis patients appeared in 2018 and 1.5 million people died due to tuberculosis [2].
Tuberculosis bacillus, M. Tuberculosis, is located in the lungs 80%, and 20% extrapulmonary. Non-pulmonary tuberculosis may occur with an acute clinical picture, and may occur many years after primary infection. HIV infection is more common in conditions that weaken the immune system, such as diabetes, kidney failure, cirrhosis [3].
In the pathogenesis of non-pulmonary tuberculosis, infectious secretion causes infection in the respiratory tract and gastrointestinal tract, and the spread of tuberculosis bacillus through lympho-hematogenous and close relationship plays a role [4].
Among the extrapulmonary involvement of tuberculosis; pleural, lymphatic system, central nervous system, bone, skin, joint, miliary involvement, cardiovascular and genito-urinary system involvements can be seen [5].
After lung and pleural involvement, the most common involvement is the lymphatic system, often the cervical and supraclavicular lymph nodes are involved [6]. Clinical and radiological findings vary depending on the organs involved. Especially in patients with suppressed immune system, different clinical findings and radiological appearance of tuberculosis may complicate and delay diagnosis [7].
We aimed to present our 56-year-old male patient with diabetes who was diagnosed as bone tuberculosis and tuberculosis lymphadenitis as a result of the examinations, which suggests primarily chest wall malignancy and infection clinically and radiologically.
2. Case Report
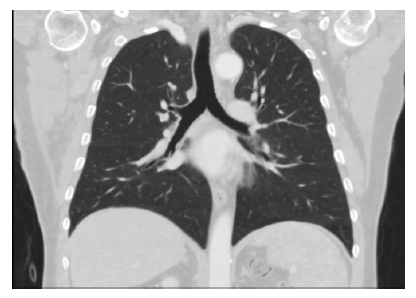
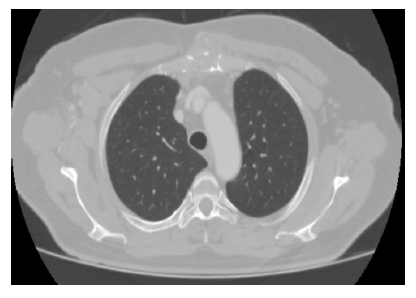
A 56-year-old diabetic male patient presented with a complaint of swelling in the right axillary region and over the sternum. It was learned that the swelling that started in the right axillary region and on the sternum occurred approximately 6 months before the history, various antibiotic treatments were applied for these swelling but no response was obtained. During this period, it was learned that he had complaints of fever from time to time, and that he had no complaints of night sweats and weight loss. In his physical examination, fever was determined at 37.5 ° C, approximately 80x60 mm, hard, mobile, painless mass located on the right axillary fossa, and a hard consistency, fixe, painful mass on the manibrium stern. There was no hyperemia, temperature increase and fluctuation on the masses. In laboratory examination; White blood cells 7.90x103 / UL (28.2% lymphocyte, 10.4% monocyte, eosinophil 4.4%), hemoglobin 12.5 g / dL, sedimentation rate 51 mm / hour, C-reactive protein (CRP) 2.40 mg / L. Blood biochemistry was within normal limits. Parenchyma was evaluated normally in the radiological images of the patient (figure 1,2).
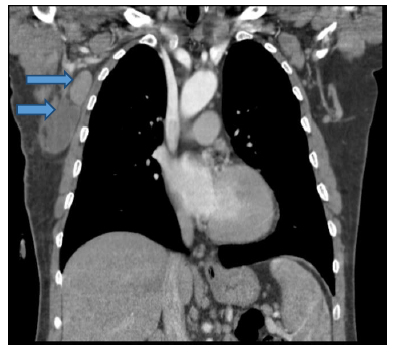
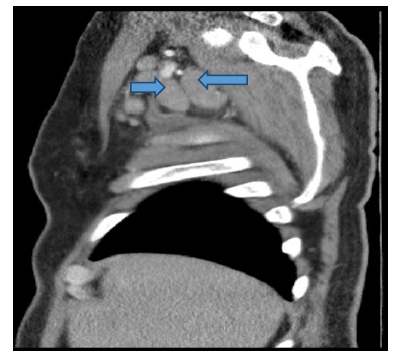
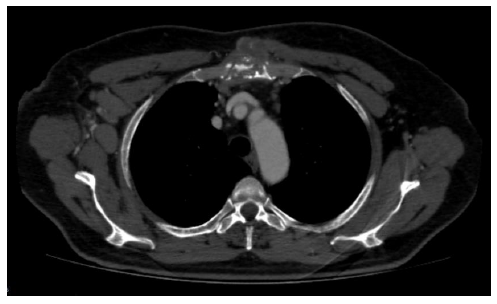
In thorax computed tomography (CT) examination; lymph nodes with a short diameter of 1 cm in the mediastinum and bilateral hilar region, the size of which is 28x19 mm in the right axillary fossa, the hilar fat content of which is not selected, multiple, conglomerate lymph nodes (figure 3,4).
Fine needle aspiration biopsy (FNAB) from the mass located in the right axillary fossa was not diagnostic. The mass was then excised totally. The pathology of the extracted mass was reported as caseified granulomatous lymphadenitis containing CD68 (+) and cytokeratin (+) conglomerated lymph nodes in immunohistochemical assay. In the immunohistochemical examination of the FNAB result from the mass on the sternum, TTF-1 (-) came as P40 (-). In the serial sections of the microscopic examination of the sample taken, calcification and necrosis in the middle. In the vicinity, granuloma formations containing epiteloid histiocytes and multinuclear Langhans type giant cells were observed. According to these findings, it was reported to be compatible with tuberculosis.
According to the result of the pathology, antituberculosis treatment with isoniazid, rifampin, pyrazinamide and ethambutol 4 was started. M. Tuberculosis also reproduced in the cultures of the samples. Treatment and follow-up of the patient continues.
3. Discussion
Tuberculosis is one of the oldest known diseases in human history. It is a preventable and treatable disease. Being late in the diagnosis of tuberculosis not only makes the treatment of this disease difficult, but also increases its spread in the society. Diagnosing tuberculosis is not always easy. In the absence of specific findings, both radiologically and clinically, it becomes difficult to diagnose when acid-resistant bacilli (ARB) cannot be produced in textiles and culture [8]. Various studies have shown that tuberculin skin test positivity varies between 20-80% in cases of extrapulmonary tuberculosis, chest radiography is above 50%, and other reasons that make the diagnosis difficult. It should be kept in mind that the negative tuberculin test and normal chest x-ray will not eliminate the possibility of tuberculosis. Routine biochemical and hematological examinations are generally not specific, do not help diagnosis. There may be abnormal findings such as high erythrocyte sedimentation rate, anemia, leukopenia or leukocytosis, thrombocytopenia [9].
Aydogdu K. et al. A 33-year-old woman with increasing swelling and pain complaints in the right chest posterolateral for the last 3 months examined the patient. On the chest X-ray, they detected bilateral hilar fullness and minimal pleural effusion on the right. In Magnetic Resonance Imaging (MRI), they detected a soft tissue mass approximately 5x6 cm in size extending from the 8th intercostal space to the diaphragm in the posterior, and diagnosed tuberculosis by performing an excisional biopsy [10].
Our case was a 56-year-old diabetic male patient. A growing mass was detected 6 months ago on the right axillary fossa and sternum. Lung parenchyma was normal in the imaging. There were no radiological findings to suggest infection, malignancy and tuberculosis. In thorax CT, multiple lymph nodes with a short diameter of 1 cm were detected in the bilateral hilar region and mediastinum. Multiple, conglomerated lymph nodes measured 28x19 mm in the right axillary fossa and at the level of corpus sterni manibrium stern junction, a 35x25 mm measured necrotic soft tissue that caused marked destruction in the bone structure was observed. These findings primarily suggested malignancy and metastasis. No pathology was observed on abdominal ultrasonography. FNA was performed from lesions in the axillary region and on the sternum. Since the biopsy results were not diagnostic, the mass in the right axillary region was totally removed. The result of histopathological examination of the mass was reported as chronic granulomatosis lymphadenitis compatible with tuberculosis.
Eren and Avci examined the patient who had chest pain that lasted for 6 months and a mass complaint growing in the anterior chest wall. On physical examination; They detected a hard, painful mass on the upper part of the sternum. In the laboratory examination, they found the sedimentation value of 70 mm / hour. In thorax CT, a 4x3 cm mass lesion was detected in the sternum, and they did not find any radiological pathology in the lung parenchyma. The patient underwent FNAB from the mass, and after the pathology was normal, the mass was removed by surgery and diagnosed primary sternum tuberculosis [11].
In our patient, the mass in the right axillary fossa was rigid, mobile and painless. The mass on the sternum was stiff, fixe, painful. There was no hyperemia, temperature increase and fluctuation on the masses. There were no symptoms such as cough, fever, weight loss, weakness, anorexia, and there was no evidence to suggest tuberculosis. In laboratory examination, hemogram and biochemistry values were observed within normal limits. The sedimentation rate was 51 mm / hour, C-reactive protein (CRP) 2.40 mg / L. The conglomerated LAP in the right axillary fossa and the mass on the sternum primarily suggested malignancy, metastasis and infectious diseases. The FNAB result from the mass was not diagnostic. Excisional biopsy was applied to the lesion on the sternum. The taken pieces were sent for specific culture and pathological examination. After the excisional biopsy pathology result obtained here was compatible with tuberculosis, the patient was diagnosed with Non-pulmonary tuberculosis and 4 antituberculosis treatment was started.
In tuberculosis radiologically, in 95% of cases, the lesion is seen in the superior segments of the upper lobes [12]. Atypical location is observed more frequently in cases such as diabetes mellitus (DM), pregnancy, kidney failure, liver failure, and corticosteroid use [13]. 1/3 of the lesions may appear in the form of cavity, bronchopneumonic miliary appearance, fibrocalcified lesions and tuberculoma [14].
Our patient had no lung involvement and had an unusual location of tuberculosis. The diabetic of our patient may have facilitated the extraordinary involvement of tuberculosis. The most common form of extrapulmonary tuberculosis is tuberculosis lymphadenitis. In the study conducted by Ugtur et al., the rate of tuberculosis lymphadenitis was found to be 24% [15]. Lymph node involvement occurs as a result of hematogenous or direct spread. Involvement is most common in cervical lymph nodes. In tuberculosis with severe systemic manifestations, lymphadenopathy is observed outside the cervical and supraclavicular area. These lymph nodes are often in the form of numerous, painless, hard, and fistulized masses (sucrofuloderma). PPD is often positive [16]. The rate of association with pulmonary tuberculosis is reported to be 60% [17]. In cases with skin involvement, erythema induratum and papulonecrotic tuberculids are seen in the skin. It can be seen in findings such as osteomyelitis and epididymitis as a result of bacillus spreading via hematogenous or adjacent [18]. Extrapulmonary findings did not accompany pulmonary tuberculosis in our case. Soft tissue tuberculosis is the least common type of tuberculosis and should be considered in diagnosis. There are publications indicating that underlying diseases such as trauma, organ transplantation history, immunosuppressive therapy and collagen vascular diseases are present in tuberculosis cases with soft tissue involvement [19].
Our case had a history of diabetes mellitus disease that would cause immunosuppression. If there is no shrinkage in lymph nodes despite the treatment, drainage of abscessed and fluctuating lesions, and total surgical excision can be planned in fistulized lesions [20].
In our case, a diagnosis was not made with FNAB performed from lesions on the right axillary fossa and on the sternum, and a total excision was made on the mass in the right axillary fossa, and the lesion on the sternum was diagnosed by performing excisional biopsy.
The exact diagnosis of tuberculosis is made by showing bacillus or in culture. With the microscopic examination of the sample taken, 50-90% of pulmonary tuberculosis and 25% of extrapulmonary tuberculosis can be detected [21].
In our case, the diagnosis was made by defining the result of histopathological examination of the samples taken from the mass on the sternum and right axillary lymph nodes as compatible with tuberculosis, as chronic granulomatosis lymphadenitis and the growth of M. tuberculosis in the specific culture.
In our country, the case of multidrug resistant tuberculosis is reported at 5.8%. Therefore, it is recommended that patients start treatment consisting of 4 drugs and prefer ethambutol over streptomycin [22]. Antituberculosis treatment, consisting of 4 drugs, was started in our patient and treatment and follow-up continues.
4. Conclusion
Tuberculosis is still a common disease in developing countries. For this reason, extrapulmonary tuberculosis should be kept in mind in the differential diagnosis of abscesses and in the differential diagnosis of lymphadenopathies, the etiology of which cannot be determined.
Conflict of interest:
None
Disclosure
No competing financial interests exist.
Informed consent
Informed consent was obtained from all individual participants included in this study.
It was presented as an e-poster at the 42nd congress of TÜSAD on October 2-8, 2020.
Ethical approval
All procedures performed in study involving patient with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding:
None
References
- Treatment of tuberculosis: Guidelines for national programmes, 3rd ed. Geneva, World Health Organization (2003).
- org.tr 73. Verem Egitim ve Propaganda Haftasi Bilgi Notu (2020): 05-11.
- Hass DW, Des Prez RG. Mycobacterium tuberculosis In: Mandell GL, Bennett JE, Dolin R, eds. Principles and Practice of Infectious Diseases. 5th ed. New York: Churchill Livingstone (2000): 2576-607.
- Aygen B. Ekstrapulmoner tüberküloz. In: Topçu A, Söyletir G, Doganay M, eds. Infeksiyon hastaliklari ve mikrobiolojisi. Cilt 1. 2. Baski. Istanbul: Nobel Tip Kitabevleri (2002): 591-9.
- Alverez S, Mc Cabe WR. Extrapulmonary tuberculosis reviseted; a review of experience at Boston City and other hospitals. Medicine (Baltimore) 63 (1984): 25-55.
- Euro TB (InVS/KNCV) and the national coordinators for tuberculosis surveillance in the WHO Europian Region. Surveillance of tuberculosis in Europe. Report on tuberculosis cases notified in 1998, February (2001).
- Yenel F. Immün sistemi yetersiz kiSilerde tüberküloz. KocabaS A (ed). Tüberküloz Klinigi ve Kontrolü. Adana: Çukurova Üniversitesi Matbaasi (1991): 137-9.
- Uçar N., Berberoglu B., Ögretensoy M., Metastatik Akciger Kanseri ile KariSan Bir Akciger Tüberkülozu (Tbc) ve Tbc Lenfadenit Olgusu. Tüberküloz ve Toraks Dergisi 49 (2001): 147-150.
- Tavusbay N, Aksel N, Çakan A, Gülerçe G, Dereli S, Özsöz A, Cases With Extrapulmonary Tuberculosis.
- Aydogdu K, Aydin S, Findik G, Acar L.N, Demirag F, Kaya S, Toraks Duvari Tümörünü Taklit Eden YumuSak Doku Tüberkülozu, Turkiye Klinikleri Arch Lung 13 (2012).
- Eren S, Avci A, Primary sternal tuberculosis: a case report., Türk Gögüs Kalp Damar Cer Derg 18 (2010): 235-237.
- Linnigton GA. Diffüse intertisial pattern. A Diagnostic approach to Chest Diseases. 3rd ed. Baltimore: Williams & Wilkins (1987): 306-31.
- Hadlock FP, Park SK, Awe RJ, Rivera M. Unusual radiographic findings in adult pulmonary tuberculosis. AJR (1980): 1015-8.
- Rossaman MD, Mayack RL. Akciger Tüberkülozu. Schlossberg D (ed). Tetikkurt C (Çeviri ed). Istanbul: Bilimsel ve Teknik Kurullari Çeviri Vakfi (1995): 75-83.
- Ugtur YS, Öztop A, Oguz V, Çakmak R. 50 ekstrapulmoner tüberküloz olgusu. Solunum Hastaliklari 10 (1999): 362-6.
- Haas DW. Mycobacterial diseases: Mycobacterium tuberculosis. In: Mandell GL, Bennett JE, Dolin R (eds). Principles and Practice of Infectious Diseases. 5th ed. Pennsylvania: Churchill Livingstone (2000): 2576-607.
- Kundakçi N, TaSpinar A. Lepra ve diger mikobakteriyel deri infeksiyonlari: Deri tüberkülozlari. Topçu AW, Söyletir G, Doganay M (editörler). Infeksiyon Hastaliklari. Nobel Tip Kitabevleri (1996): 803-17.
- Ertürk A, Arikan S, Ögretensoy M. Deri tüberkülozu: Iki farkli görünüm. Tüberküloz ve Toraks 47 (1999): 352-6.
- Puttick MP, Stein HB, Chan RM, et al. Soft tissue tuberculosis: A series of 11 cases. J Rheumatol 22 (1995): 1321-5.
- Karagöz T, Senol T, Bekçi T.T, Tüberküloz Lenfadenit, Toraks Dergisi 2 (2001): 74-79.
- Gupta A, Sharma SK, Pande JN. Diagnostic methods for tuberculosis. Indian J Chest Dis Allied Sci 35 (1993): 63-84.
- Tahaoglu K, Torun T, Sevim T, et al. The treatment of multidrug-resistant tuberculosis in Turkey. N Engl J Med 345 (2001): 170-4.


 Impact Factor: * 3.5
Impact Factor: * 3.5 Acceptance Rate: 71.36%
Acceptance Rate: 71.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
