Ex vivo Nitrite Exposure and Response of Peripheral Blood Mononuclear Cells from Osteoarthritis Cases and Controls: Proliferation and Nitric Oxide Generation
Junu Shrestha1*, Lisa Beltz2, Catherine Zeman3
1Department of Public Health, University of Illinois at Springfield, Springfield, IL, USA
2Professor (1995-2008) Department of Biology, University of Northern Iowa, Cedar Falls, IA, USA
3Public Health, Department of Health, Recreation, and Community Services, University of Northern Iowa, Cedar Falls, IA, USA
*Corresponding Author: Junu Shrestha, Department of Public Health, University of Illinois at Springfield, Springfield, IL, USA
Received: 22 March 2021; Accepted: 29 March 2021; Published: 06 April 2021
Article Information
Citation: Junu Shrestha, Lisa Beltz, Catherine Zeman. Ex vivo Nitrite Exposure and Response of Peripheral Blood Mononuclear Cells from Osteoarthritis Cases and Controls: Proliferation and Nitric Oxide Generation. Journal of Environmental Science and Public Health 5 (2021): 200-225.
View / Download Pdf Share at FacebookAbstract
This ex vivo pilot study examined the effect of low dose nitrite exposure on peripheral blood mononuclear cells (PBMC) proliferation and nitric oxide (NO) generation among people with osteoarthritis (cases) and without osteoarthritis (controls). The hypothesis was that nitrite exposure produced different effects on proliferation and NO generation between the cases and controls. Human PBMC, composed of lymphocytes and monocytes, were isolated from the whole blood of 31 participants (15 cases and 16 controls) using density gradient centrifugation, then exposed to five different nitrite concentrations: 0 (negative controls), 1 mg/l (0.02 mM), 5 mg/l (0.10 mM), 10 mg/l (0.21 mM), and 15 mg/l (0.32 mM). PBMC proliferation and NO generation were determined after 96-hours and 48-hours, respectively, using a Muse Unit (Cell Analyzer/Flow Cytometer). Increasing nitrite exposure significantly suppressed the non-stimulated PBMC proliferation (F = 7.07, p<.0001). The study also showed PBMC proliferation was significantly suppressed among all participants when exposed to 5 mg/l nitrites, but that the degree of suppression was higher among participants with osteoarthritis (F=4.23, p<0.05). Increasing nitrite exposure together with stressful life events, use of multiple drugs (prescribed and supplements), and chronic disease also significantly impacted the NO generating cells and PBMC proliferation.
Keywords
<p>Nitrate; Nitrite; Nitric oxide; PBMC proliferation; Osteoarthritis; Chronic disease</p>
Article Details
1. Introduction
Nitrate is a widespread groundwater pollutant from both natural sources and agricultural application of artificial fertilizers and animal manure [1-3]. The use of fertilizers and intensive animal farming led to increased nitrate concentrations in groundwater in some agricultural regions [4]. Approximately 90% of the rural population of the United States obtain drinking water from private wells, mainly in agricultural regions that are not regulated at the federal level [5]. About 80% of nitrate in the human body enters through consuming vegetables, fruits, and processed food products [6]. The nitrate concentration in saliva is rapidly converted to nitrite by facultative anaerobic bacteria [3, 7]. Nitrite formed in this process is then converted to nitric oxide (NO) in the stomach under acidic conditions and aids in the host defense mechanism against pathogens [2]. Very low levels of nitrate (less than 2 mg/l nitrate-nitrogen) are relatively non-toxic and, via NO production, are important for cardiovascular health, including vascular relaxation and decreased platelet aggregation and blood pressure [8]. If high nitrate levels are present in drinking water in the absence of proper levels of antioxidants, such as those present in complex foodstuffs, their metabolites (nitrite, NO, and nitrosamine) are suspected of contributing to chronic disease and cancer [9]. The Environmental Protection Agency (EPA) calculated the oral exposure reference dose (RfD) of 0.1mg nitrite nitrogen/kg of body weight per day which is equivalent to 0.33 mg nitrite ion/kg of body weight per day [10, 11]. The mean weight of US adults between the age of 40 to 59 is 91.1 kg [12]. Therefore, the reference dose of nitrite for a person weighing 91.1 kg is 30.06 mg per day for acute health effects and considers both food and water sources [10-12]. The effects of nitrate dose on the chronic disease osteoarthritis (OA), either alone or in combination with other factors, have not yet been determined. In this study, sodium nitrite was used as the exposure agent. The biologically active form of nitrogen after nitrate ingestion, however, is approximately 13% of an ingested dose of nitrate and is found as nitrite in the oral cavity (conversion in saliva via microbes present in the mouth) and the plasma (absorption through the upper third of the digestive tract after action by gut microbiome) [13]. Thus, the sodium nitrite levels of 1, 5, 10, and 15 mg/l are comparable to nitrate ingestion levels of 7.69, 38.46, 76.92, and 115.38 mg/l nitrates. Converting the nitrate levels to the relevant, EPA-regulated form of nitrate-nitrogen would equate to drinking water exposures of 1.37, 8.69, 17.38, 26.06 mg/l nitrate-nitrogen. The lower end of this exposure is under the maximum contaminant level (MCL) for nitrate-nitrogen of 10 mg/l and the higher end is within the realistic realm of exposure from private well waters around the world [13, 14].
In the human body, NO acts as an inflammatory agent and has been found in excess in the synovial fluid of OA patients, playing a role in the disease progression by altering cartilage and bone metabolism [15]. Pro-inflammatory cytokines, such as interleukin-1 (IL-1), tumor necrosis factor-α (TNF-α), IL-6, IL-8, IL-15, IL-17, IL-18, and IL-21 have either been linked to OA or found in OA synovial fluid [16, 17]. Excessive levels of NO, IL-1, TNF-α, and pro-inflammatory prostaglandins are produced by chondrocytes from OA patients’ joints [18-20]. Serum nitrite levels are higher in patients with OA [21]. Older women using nitrates for angina were found to be at increased risk of OA of the hips, with noted increases in NO in articular cartilage and greater catabolism of the cartilage matrix, contributing to OA progression [15]. Further, general chemical metabolism studies following nitrate ingestion have established that increases in nitrate consumption led to corresponding increases of nitrite and its metabolites such as NO in plasma and saliva [22, 23]. Thus, it would be of interest to determine the impact of nitrite exposure on human lymphocytes and to determine if differing responses are noted between OA cases and controls.
Previous research conducted by the authors pointed to possible connections between complaints of OA associated pain and higher levels of nitrate ingestion in well water. A combination retrospective cohort study and ex vivo lymphocyte study performed with private well users demonstrated a positive association between nitrate exposure and self-reported health problems, including high body mass index, low recreational activity, poorer general health, susceptibility to illness, higher methemoglobin levels, intestinal difficulties, OA, and production of cytokines associated with bone and joint disorders, such as tumor necrosis factor-β (TNF-β) and the T regulatory cell cytokine IL-10 [24]. Additionally, ex vivo exposure of lymphocytes to nitrate or nitrite alters cytokine production in normal, healthy people [25].
OA is a degenerative disease of synovial joints and articular cartilage which exhibits thickening of subchondral bone and synovial inflammation, particularly in diarthrodial joints [26]. OA is the most prevalent of arthritic diseases, affecting 10-15% of the world’s population and 30 million American adults. OA frequency increases with age and its incidence is higher than 60% among those older than 65 years [27].
Elevated levels of nitrite and the proinflammatory factor NO are present in the serum and synovial fluid samples from OA and rheumatoid arthritis patients [28]. NO and its redox derivatives may help to protect joints initially following injury by stimulating a protective immune response, but when the local production or accumulation of NO is excessive or chronic, bone destruction and inflammatory arthropathies may occur [18]. Long-term exposure to high levels of nitrate is a risk factor for developing osteoarthritis by triggering chondrocyte apoptosis and matrix destruction [29]. Also, high concentrations of NO suppress T lymphocyte proliferation and reduce levels of the T helper 1 (Th1) cytokines IL-2, interferon-γ (IFN-γ), and TNF-β in some, but not all, subjects. This variation in lymphocyte proliferation and cytokine production among normal, healthy subjects suggests that environmental or internal factors exist that, alone or in combination, affect lymphocyte proliferation and functioning [24]. Determining the proportion of cells generating NO is important due to its antitumor and anti-microbial action [30]. With the authors’ previous work in mind, this case-control study was designed to determine whether exposure to nitrite in-vitro resulted in differences in PBMC proliferation and NO generation between those people with or without OA with the hypothesis that differences would be found between the controls and cases of OA (H0). Other potentially contributing factors were also tested for their effects on proliferation and NO generation alone or in combination with nitrite exposure in case and control populations. These factors include stressful life events, the use of multiple drugs (prescribed and supplements), and chronic disease conditions.
2. Materials and Methods
2.1 Selection of participants, sample collection, and survey
Following approval by the Institutional Review Board (IRB) at the University of Northern Iowa, (Protocol Number 14-0018) experimental subjects were recruited among individuals having a diagnosed case of OA and healthy controls ranging in age from 40 to 65 years in 2016. Venous blood was collected from 15 subjects with osteoarthritis and 16 normal, healthy controls, following full-informed consent. At that time, participants also completed a brief questionnaire that included information on demographic, general health (comorbidities, stressful life events in the last 6 months, smoking history, recent immunizations) and medication use including the number and names of prescribed medicines, herbs, and supplements. Participants with reported autoimmune diseases, including rheumatoid arthritis and psoriasis, were excluded from the study.
2.2 PBMC proliferation
PBMC were isolated from whole blood by Ficoll-paque density gradient centrifugation as used in multiple previous studies [31]. Using the MUSE Unit (Cell/Analyzer/Flow Cytometer) from Luminex, Model #7200121194, PBMC counts, and viability were determined to standardize the initial concentration of all cells to 4x106 cells/ml, which were then resuspended in RPMI 1640 medium (Gibco, #61870036) containing 10% fetal bovine serum (RMBIO, Lot #20140319FS) and 100µl/ml penicillin/streptomycin (MP Biomedical, SKU# 0916704). The case and control subjects’ PBMCs were then placed into 96-well microliter tissue culture plates in triplicate, according to an experimental exposure design of control (not exposed to nitrite solution) and treatment (exposed to four concentrations of nitrite solution at 1 mg/l (0.02 mM), 5 mg/l (0.10 mM), 10 mg/l (0.21 mM), and 15 mg/l (0.32 mM) under stimulated and non-stimulated conditions. Stimulated cells were treated with 4 mg/ml of the stimulant phytohemagglutinin (Sigma-Aldrich, Product # L1668) sourced from crude extracts of the red kidney bean Phaseolus vulgaris, while non-stimulated cells were incubated with the same volume of culture medium [25]. The non-stimulated condition refers to resting cells that mimic natural cell growth phenomenon in vivo whereas the stimulated condition represents the addition of stimulants such as phytohemagglutinin that activate immune responses of PBMC.
2.3 Nitrite exposure, plating, and incubation
Sodium nitrite (Fisher Chemical, Catalog # M-12680) was prepared in a culture medium and filtered through a 0.2-micron pore size syringe filter. The stimulated and non-stimulated cell cultures were then exposed to the five different nitrite concentrations, 0 (negative or controls), 1 mg/l (0.02mM), 5 mg/l (0.10mM), 10 mg/l (0.21 mM), and 15 mg/l (0.32 mM) and incubated at 35 °C in the presence of 5% CO2 acting as a buffer. The plate configurations are shown in Figure 1. NO generation as a percentage of live and dead cell numbers as well as PBMC counts and viability were determined after 48-hours and 96-hours incubation, respectively, according to the Muse Unit manufacturer’s Kit User Guide [32, 33].
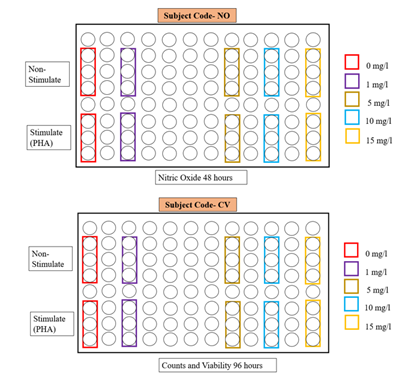
Figure 1: Cultured PBMC were tested for proliferation and NO generation under the various experimental conditions (non-stimulated, stimulated, and nitrite exposure concentration).
2.4 Determining NO generation from live and dead cells
The Muse Unit was used to detect NO generation by PBMCs using stock reagent kits. The Muse Nitric Oxide assay simultaneously monitors cell counts and the percentage of cells generating NO and cell death, as per the manufacturer’s instructions. The Muse Unit determines the percentage and concentration of live cells, live cells generating NO, dead cells generating NO, dead cells, and total NO-generating cells consisting of live and dead cells using the Nitric Oxide Reagent and the Dead Cell dye 7-Aminoactinomycin D (7-AAD) diluted in the Muse Assay Buffer. Nitric Oxide Reagent is a non-fluorescent, cell-permeable reagent that can measure free NO and nitric oxide synthase (NOS) activity in living cells under physiological conditions. Both the nitric oxide and 7-AAD working solutions were prepared by diluting the stock solution. The reagent does not depend on the action of cellular esterase for reactivity and is photostable. The dead cell dye 7-AAD measures cell membrane structural integrity and cell death. NO generation was determined following the MUSE Unit manufacturer’s procedure. After 48 hours of incubation, the cell samples were stained with the Nitric Oxide Reagent and then incubated at 37 °C for 30 minutes. After incubation, 7-AAD was added, and the samples were then incubated at room temperature for 5 minutes protected from light and then run on the Muse Unit using the gating of 98%. The Nitric Oxide profile was recorded for cell populations and cells that were living without NO generation, live and generating NO, dead and generating NO, and dead and not generating NO. The detailed methodology and test principle is mentioned in the Muse Nitric Oxide Kit User’s Guide [32].
2.5 Determining PBMC proliferation
The Muse Counts and Viability reagent allows the quantitative analysis of cell count and viability on the Muse Unit. The assay provides reliable results with cell concentrations in the range of 1x105 to 1x107 cells/ml. The cell samples for this research were used at a concentration of 4x106 cells/ml. The Muse Unit provides three results: viable cell count, total cell count, and percentage viability of the sample. The reagent consists of a DNA binding dye (sodium azide) that stains cells which have lost their membrane integrity, allowing the dye to stain the nucleus of dead and dying cells.
The Muse unit scores viable cells as live cells that do not stain and non-viable dead or dying cells that do stain. After 96 hours of incubation, PMBC was mixed with the Counts and Viability Reagent and then incubated for 5 minutes at room temperature. After incubation, the cells were run through the Muse Unit with the gating set at 98% during the cell analysis by setting the threshold level to exclude debris. Finally, the counts and viability results were recorded and analyzed. The detailed methodology and test principle is mentioned in the Muse Count and Viability Kit User’s Guide [33].
2.6 Quality control
To ensure that the Muse Unit provided reliable and accurate results, the system check procedure was conducted prior to performing cell counts, viability, and NO generation assays. All reagents for NO generation and PBMC proliferation tests were stored at appropriate temperatures according to the protocol provided by the manufacturer and were used within the expiration dates for quality control.
2.7 Data analysis
Data were analyzed using univariate, bivariate, and multivariate statistical methods in SAS Institute, JMP 15. Specific tests included basic descriptive statistics, (i.e., age, sex, health status, and lifestyle habits) as well as bivariate and multivariate analysis including one-way ANOVA, t-test, correlational analysis, and whole model testing with significance at p≤ 0.05*, p<0.001**, and p<0.0001***.
3. Results
3.1 Demographic Information
The mean ages of the case and control groups were 54.1 and 52.6 years, respectively, and included 24 females and 7 males. The bivariate fit for age by PBMC proliferation and NO generation indicated that age did not impact overall PBMC proliferation, but significant NO generation was observed among live cells at 1mg/l and dead cells in the presence of 1 and 10 mg/l of nitrate when correlated with age (Table 1 and 10). The T-test did not show any significant difference between PBMC proliferation and NO generation when correlated by sex. Additional variables measured include life-changing events, comorbidities such as fibromyalgia, medication, and dietary supplement use.
|
Demographics |
Results |
|
Age |
Case: Mean (54.1 + 5.77) and Control: Mean (52.6 + 8.30) |
|
Sex |
Female (77%); Male (23%) |
|
Life-Changing Events |
Yes (13%); No (87%) |
|
Comorbidities |
Yes (6%); No (94%) |
|
Medications |
Yes (55%); No (29%); No Answer (16%); Mean (1.88 + 2); Range (0-6) |
|
Supplement Use |
Yes (52%); No (26%); No Answer (22%); Mean (1.8 + 1.68); Range (0-4) |
Table 1: Distributional analysis of demographic variables life-changing events, comorbidities, medications, and supplement use.
3.2 PBMC proliferation in non-stimulated and stimulated cells
PBMC counts and viability were determined to standardize the initial concentration of all cells to 4x106 cells/ml. After 96 hours of incubation and exposure to experimental nitrite conditions, the viable PBMC cell numbers and magnitude of response (r=stimulated/non-stimulated) were determined.
The analysis showed that there is a decreasing trend in mean PBMC proliferation with increasing nitrite exposures in both non-stimulated and stimulated conditions (Table 2 and Figure 2).
|
Nitrite Exposure |
NS Cells* Controls |
NS Cells* Cases |
NS Cells* Combined |
ST Cells** Controls |
ST Cells** Cases |
ST Cells** Combined |
|
0 mg/l |
3.8 + 0.51 |
4.17 + 0.62 |
3.98 + 0.39 |
4.24 + 1.14 |
4.22 + 0.64 |
4.23 + 0.65 |
|
1 mg/l |
3.34 + 0.52 |
3.54 + 0.39 |
3.44 + 0.32 |
4.04 + 1.04 |
3.63 + 0.53 |
3.84 + 0.59 |
|
5 mg/l |
2.96 + 0.47 |
3.26 + 0.41 |
3.11 + 0.31 |
3.99 + 1.05 |
2.74 + 0.39 |
3.38 + 0.58 |
|
10 mg/l |
2.48 + 0.34 |
2.47 + 0.26 |
2.47 + 0.21 |
3.20 + 0.75 |
2.47 + 0.37 |
2.85 + 0.42 |
|
15 mg/l |
2.09 + 0.30 |
1.78 + 0.28 |
1.95 + 1.20 |
3.29 + 0.70 |
2.28 + 0.29 |
2.80 + 0.39 |
Notes: *NS cells= 105 non-stimulated cells/ml, **ST=105 stimulated cells/ml; Mean + SE
Table 2: Effect of Nitrite on Mean Numbers of Non-stimulated (NS) and Stimulated (ST) PBMC among cases (with OA), controls (without OA), and Cases and Control Combined at 105 cells.
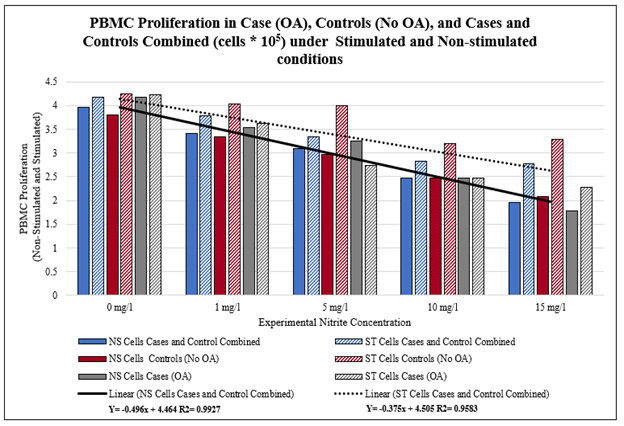
Figure 2: Effect of Nitrite Exposure on Numbers of Viable PBMC (Case, Control, and Combined Cells) under Stimulated and Non-stimulated conditions.
A bivariate fit to determine dose-response relationships between PBMC proliferation and control (0 mg/l) and the experimental conditions (nitrite exposure of 1, 5, 10, and 15 mg/l) indicated that in all situations: cases, controls, cases, and controls combined under both stimulated and non-stimulated conditions showed a significant dose response trend.
There were significant reductions in PBMC proliferation across all experimental exposure levels (1, 5, 10, 15 mg/l) indicating a dose response that was highly significant which encompassed all case, control subjects and most experimental exposures with measures of significance ranging from a p-value of p= 0.012 to p< 0.0001, except for one experimental situation, non-stimulated cases at 15 mg/l experimental exposure (Table 3).
|
No Exposure |
Experimental Nitrite Exposure |
||||
|
1 mg/l |
5 mg/l |
10 mg/l |
15 mg/l |
||
|
Cases (with OA) with Non-Stimulated Condition |
0 mg/l |
0.87, p<0.0001*** |
0.75, p<0.0001*** |
0.48, p=0.0041** |
0.19 |
|
Controls (No OA) with Non-Stimulated Condition |
0 mg/l |
0.84, p<0.0001*** |
0.79, p<0.0001*** |
0.53, p=0.0013** |
0.47, p=0.0031** |
|
Cases and Controls Combined with Non-Stimulated Conditions |
0 mg/l |
0.80, p<0.0001*** |
0.75, p<0.0001*** |
0.48, p<0.0001*** |
0.29, p=0.0015*** |
|
Controls (No OA) with Non-Stimulated Condition |
0 mg/l |
0.95, p<0.0001*** |
0.58, p=0.001** |
0.39, p=0.012* |
0.60, p=0.0006** |
|
Controls (No OA) with Stimulated Condition |
0 mg/l |
0.97, p<0.0001*** |
0.85, p<0.0001*** |
0.70, p<0.0001*** |
0.41, p=0.0075** |
|
Cases and Controls Combined with Stimulated Conditions |
0 mg/l |
0.96, p<0.0001*** |
0.76, p<0.0001*** |
0.62, p<0.0001*** |
0.41, p<0.0001*** |
|
Magnitude of Response for Cases (with OA) |
0 mg/l |
0.39, p=0.012* |
0.007 |
0.1 |
0.04 |
|
Magnitude of Response for Controls (No OA) |
0 mg/l |
0.1 |
0.02 |
0.09 |
0.08 |
|
Magnitude of Response for Cases and Control Combined |
0 mg/l |
0.14, p=0.03* |
0.027 |
0.10 |
0.07 |
Note: Magnitude(r)= stimulated/non-stimulated; R-Square, p<0.05*, p<0.001**, p<0.0001***
Table 3: Dose Response Relationship of PBMC between no exposure (0 mg/l) and treatments (Nitrite exposure of 1, 5, 10, and 15 mg/l).
ANOVA was used to analyze cases and controls across all experimental exposures which showed a significant reduction in mean number of viable PBMC in both non-stimulated (4.98, p=0.0014*) and stimulated (3.12, p= 0.019*) conditions. Although the mean viable PBMC count decreased among controls in non-stimulated conditions (2.35, p=0.06), this did not hold true for the stimulated controls, illustrating that under stimulation the subjects’ PBMCs without OA proliferated more effectively than the OA cases PBMC (Table 4).
Additional analysis using one-way ANOVA detected statistically significant differences in mean PBMC numbers among various nitrite concentrations (0, 1, 5, 10, and 15 mg/l) in non-stimulated cases and controls combined (F = 7.07, p<0.0001***), but not in stimulated cells (F=1.32, p=0.26), (Table 4). This highlights the more robust response of non-OA controls vs. cases following PHA stimulation (mimicking immune system challenge).
|
Nitrite Exposure |
Mean |
F Ratio, P-value |
Nitrite Exposure |
Mean |
F-Ratio, P-value |
||
|
Non- Stimulated Cases |
0 mg/l |
4.17 + 0.41 |
4.98, 0.0014* |
Stimulated Cases |
0 mg/l |
4.22 + 0.46 |
3.12, 0.019* |
|
1 mg/l |
3.54 + 0.41 |
1 mg/l |
3.63 + 0.46 |
||||
|
5 mg/l |
3.26 + 0.41 |
5 mg/l |
2.74 + 0.46 |
||||
|
10 mg/l |
2.47 + 0.41 |
10 mg/l |
2.47 + 0.46 |
||||
|
15 mg/l |
1.78 + 0.41 |
15 mg/l |
2.28 + 0.46 |
||||
|
Non- Stimulated Controls |
0 mg/l |
3.80 + 0.44 |
2.35, 0.061 |
Stimulated Controls |
0 mg/l |
4.24 + 0.95 |
0.24, 0.913 |
|
1 mg/l |
3.34 + 0.44 |
1 mg/l |
4.04 + 0.95 |
||||
|
5 mg/l |
2.96 + 0.44 |
5 mg/l |
3.99 + 0.95 |
||||
|
10 mg/l |
2.48 + 0.44 |
10 mg/l |
3.20 + 0.95 |
||||
|
15 mg/l |
2.09 + 0.44 |
15 mg/l |
3.29 + 0.95 |
||||
|
Non- Stimulated Combined |
0 mg/l |
3.98 + 0.30 |
7.07, <0.0001*** |
Stimulated Combined |
0 mg/l |
4.23 + 0.54 |
1.3299, 0.2613 |
|
1 mg/l |
3.44 + 0.30 |
1 mg/l |
3.84 + 0.54 |
||||
|
5 mg/l |
3.11 + 0.30 |
5 mg/l |
3.38 + 0.54 |
||||
|
10 mg/l |
2.47 + 0.30 |
10 mg/l |
2.85 + 0.54 |
||||
|
15 mg/l |
1.95 + 0.30 |
15 mg/l |
2.80 + 0.54 |
Note: Cases: Both NS and ST (F ratio, *p<0.05, significant); Controls: Both NS and ST (F ratio, p>0.05, Non-significant); Combined: NS (F ratio, ***p<0.0001, significant) ST (F ratio, p>0.05, Non-significant); Mean + SE.
Table 4: ANOVA: Effects of Nitrite on the numbers of viable, Non-stimulated (NS) and Stimulated (ST) PBMC at 105 cells for Case, Control, and Combined (Cases + Controls).
3.3 Magnitude of response exposure to 0, 1, 5, 10, and 15 mg/l nitrite
One-way ANOVA analysis of the magnitude of response (r = stimulated/non stimulated) detected a statistically significant difference (F = 2.47, p = 0.046*) in the mean PBMC proliferation magnitude at 15 mg/l nitrite exposure (Table 5, Figure 3) indicating that there is significant disruption of PBMC response with increased nitrite exposure. Also, the bivariate fit between magnitude of response for cases and controls between 0mg/l and experimental exposure (1, 5, 10, and 15 mg/l) indicated a significant effectin two situations; cases and combined (cases and control) both at 1mg/l of nitrite exposure (Table 3).
The mean magnitude of response among increase nitrite exposure significantly high (F = 3.01, p=0.023*) among cases, but not among the controls (Table 5). Lastly, when compared among cases and controls,the mean PBMC proliferation magnitude of response was significantly lower in cases than in controls groups at 5mg/l nitrite (Table 6 and Figure 6) as indicated by the student’s T-test (F=4.23, p=0.048*).
|
Nitrite Exposure |
Mean Magnitude of Response, Cases |
Mean Magnitude of Response, Controls |
Mean Magnitude of Response, Combined |
|||
|
Mean |
F Ratio, P-value |
Mean |
F Ratio, P-value |
Mean |
F Ratio, P-value |
|
|
0 mg/l |
0.97 + 0.13 |
3.01, 0.023* |
1.06 + 0.19 |
0.90, 0.46 |
1.06 + 0.12 |
2.47, 0.046* |
|
1 mg/l |
0.99 + 0.13 |
1.22 + 0.19 |
1.11 + 0.12 |
|||
|
5 mg/l |
0.84 + 0.13 |
1.33 + 0.19 |
1.09 + 0.12 |
|||
|
10 mg/l |
1.03 + 0.13 |
1.20 + 0.19 |
1.12 + 0.12 |
|||
|
15 mg/l |
1.46 + 0.13 |
1.57 + 0.19 |
1.52 + 0.12 |
|||
Note: Cases (F ratio, *p<0.05, significant); Controls ((F ratio, p>0.05, Non-significant); Combined (F ratio, *p<0.05, significant); Mean + SE
Table 5: ANOVA: Mean Magnitude (r=stimulated/non-stimulated) of Response by Various Nitrite Exposures among Case, Control, and Combined (Cases + Controls).
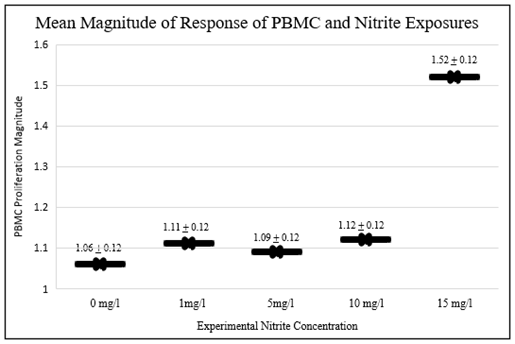
Figure 3: Mean Magnitude of Stimulated to Non-stimulated (r=stimulated/non-stimulated) PBMC by Nitrite Exposure (Mean ± SE).
|
Level |
Mean |
Lower 95% |
Upper 95% |
T Ratio |
R square |
DF |
F Ratio |
P Value |
|
Control |
1.33 + 0.16 |
0.99 |
1.67 |
-2.16 |
0.13 |
1 |
4.23 |
0.048* |
|
Case |
0.84 + 0.17 |
0.49 |
1.19 |
Note: F Ratio, * p<0.05, Mean + SE
Table 6: Mean PBMC Proliferation Magnitude (r=stimulated/non-stimulated) between Case and Controls Exposed to 5 mg/l Nitrite.
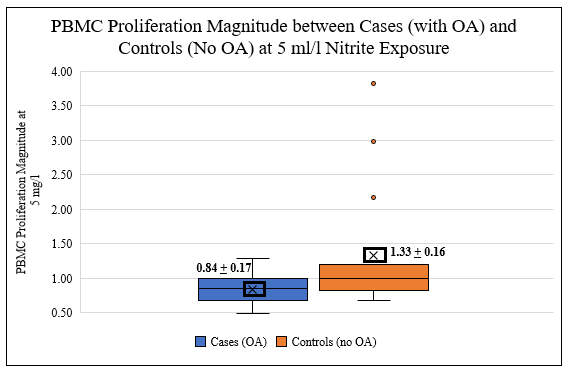
Figure 6: PBMC Proliferation Magnitude (r= stimulated/non-stimulated) between Cases (OA) and Controls (no OA) at 5 mg/l nitrite exposure, student’s T-test (F=4.23; p=0.048*).
3.4 Magnitude of response of PBMC proliferation as pooled nitrite concentrations
To explore breakpoints in the dose-response relationships, nitrite proliferation magnitudes were pooled into two different sets of breakpoints and compared using the student’s T-test.
The results indicate significant differences between responses at 0, 1, 5 mg/l, and 10, 15 mg/l with a clearly elevated response magnitude between the higher and lower pooled breakpoints (F=5.20, p = 0.023*). Significant elevations were detected between response magnitudes of 0, 1, 5, 10, and 15 mg/l (F=10.73, p = 0.001**) (Table 7, Figure 4 and 5).
|
Nitrite Exposure |
Mean |
Lower 95% |
Upper 95% |
T Ratio |
R square |
DF |
F Ratio |
P Value |
|
0,1,5 mg/l |
1.09 + 0.07 |
0.93 |
1.21 |
-2.28 |
0.02 |
1 |
5.20 |
0.023* |
|
10,15 mg/l |
1.32 + 0.08 |
1.15 |
1.49 |
|||||
|
0,1,5,10 mg/l |
1.10 + 0.06 |
0.96 |
1.21 |
-3.27 |
0.06 |
1 |
10.73 |
0.001** |
|
15 mg/l |
1.52 + 0.12 |
1.28 |
1.76 |
Note: F Ratio, * p<0.05, **p<0.01; Mean + SE
Table 7: Effects of Pooled Nitrite Exposure (0, 1, 5 mg/l and 10, 15 mg/l) and (0, 1, 5, 10 mg/l and 15 mg/l) on Mean PBMC Proliferation Magnitude (r=stimulated/non-stimulated).
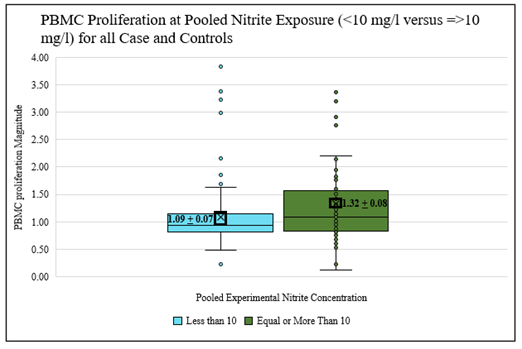
Figure 4: Effects of Pooled Nitrite Exposure all Cases and Controls (grouped concentration breakpoints, <10 mg/l versus ≥10 mg/l) on PBMC Proliferation Magnitude (r=stimulated/non-stimulated) (F=5.20; p= 0.023*).
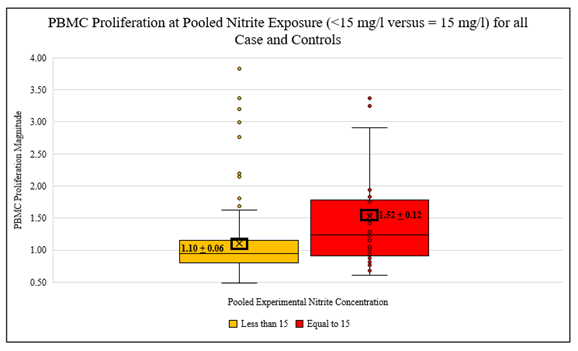
Figure 5: Effects of Pooled Nitrite Exposure (<15 mg/l versus = 15 mg/l) on PBMC Proliferation Magnitude (r= stimulated/non-stimulated) all Cases and Controls (F=10.73, p=0.001**).
3.5 Percentage of NO generating cells at various nitrite concentrations
The percentage of NO generating cells was measured among the treatment groups and evaluated as a percentage of live, dead, and total (live and dead) numbers of cells generating NO. Using one-way ANOVA analysis, dead cells showed a near significant trend increase in NO generation with increasing nitrite concentration, regardless of case-control status (Table 8) that, given the small sample size, should be considered.
Live and total cells also exhibited an increased percentage of cells producing NO with increasing nitrite concentration, but the result was not statistically significant. A whole model test indicated that life-changing events at 0, 5, 10, and 15mg/l nitrate and chronic disease comorbidity at 0 mg/l were correlated with NO generation at p< 0.05 (Table 12). Thus, illustrating the importance of these additional factors to NO generation in these PBMC cells.
|
Nitrite Exposure |
Live Cells |
Dead Cells |
Total Cells |
|||
|
Mean |
F Ratio, P-value |
Mean |
F Ratio, P-value |
Mean |
F Ratio, P-value |
|
|
0 mg/l |
0.48 + 0.23 |
1.24, 0.29 |
1.58 + 0.92 |
2.33, 0.058 |
0.80 + 0.31 |
1.38, 0.24 |
|
1 mg/l |
0.65 + 0.23 |
1.83 + 0.92 |
0.89 + 0.31 |
|||
|
5 mg/l |
0.44 + 0.23 |
2.09 + 0.92 |
0.56 + 0.31 |
|||
|
10 mg/l |
0.79 + 0.23 |
2.81 + 0.92 |
1.08 + 0.31 |
|||
|
15 mg/l |
1.09 + 0.23 |
5.07 + 0.92 |
1.54 + 0.31 |
|||
Table 8: ANOVA: PBMC Mean NO Generation (percentage) OA Cases and Controls Combined, Stimulated at Various Nitrite Exposures.
|
0mg/l |
1mg/l |
5mg/l |
10mg/l |
15mg/l |
|
|
Increasing Number of Dietary Supplements (range 0-4) |
1.12 |
1.19 |
1.12 |
1.02 |
1.52 |
|
Increasing Number of Medications (range 0-6) |
1.03 |
1.07 |
1.01 |
0.99 |
1.39,5.89, p = 0.023* |
|
Life Changing Events (Yes/No) |
1.06 |
1.18 |
1.09 |
1.26 |
1.52 |
|
Chronic Disease (Yes/No) |
1.06 |
1.11 |
1.09 |
1.12 |
1.52 |
Note: Nonsignificant (Mean of Response); Significant (Mean of Response, F-ratio, *p<0.05) Supplements, Medications, Life Changing Events, and Chronic Disease (N = 31).
Table 9: ANOVA: Analysis of PBMC Proliferation Magnitude by Medications/Supplements, Life Changing Events, and Chronic Disease at Various Experimental Nitrite Exposures.
|
% NO Generation |
Nitrite Exposure |
|||||
|
0mg/l |
1mg/l |
5mg/l |
10mg/l |
15mg/l |
||
|
Age |
Live |
0.49 |
0.65, 5.59, p=0.024* |
0.44 |
0.79 |
1.10 |
|
Dead |
0.31 |
0.14, 18.26, p=0.0002** |
0.11 |
0.28, 7.86, p=0.008** |
0.45 |
|
|
Total (Live + Dead) |
0.80 |
0.89, 5.96, p=0.029* |
0.56 |
1.08, 5.72*, p=0.023* |
1.54 |
|
|
Life Changing Events |
Live |
0.49 |
0.65, 6.04, p=0.02* |
0.44 |
0.79, 4.94, p=0.034* |
1.10, 5.45, p=0.02* |
|
Dead |
0.31, 6.48, p=0.016* |
0.14 |
0.11 |
0.28 |
0.45 |
|
|
Total (Live + Dead) |
0.80, 5.25, p=0.029* |
0.89, 5.23, p=0.029* |
0.56, 4.43, p=0.043* |
1.08, 6.05, p=0.02* |
1.54, 5.93, p=0.02* |
|
|
Osteoarthritis |
Live |
0.49 |
0.65 |
0.44 |
0.79 |
1.10 |
|
Dead |
0.31 |
0.14 |
0.11 |
0.28 |
0.45 |
|
|
Total (Live + Dead) |
0.80 |
0.89 |
0.56 |
1.08 |
1.54 |
|
|
Chronic Disease |
Live |
0.49 |
0.65 |
0.44 |
0.79 |
1.10 |
|
Dead |
0.31 |
0.14 |
0.11 |
0.28 |
0.45, 7.96, p=0.008* |
|
|
Total (Live + Dead) |
0.80 |
0.89 |
0.56 |
1.08 |
1.54 |
|
Note: Nonsignificant (Mean of Response); Significant (Mean of Response, F-ratio, *p<0.05, **p<0.01, ***p<0.0001)
Table 10: ANOVA: Analysis of Percentage of Stimulated NO Generating Cells by Age, Life Changing Events, Osteoarthritis, and Chronic Disease at Various Experimental Nitrite Exposures (N = 31).
|
% NO Generation |
Nitrite Exposure |
|||||
|
0mg/l |
1mg/l |
5mg/l |
10mg/l |
15mg/l |
||
|
Age |
Live |
0.58 |
0.62 |
0.60 |
0.59 |
0.85 |
|
Dead |
1.69 |
1.30 |
1.80 |
2.59 |
3.92 |
|
|
Total (Live + Dead) |
0.74 |
0.82 |
0.73, 5.25, p=0.03* |
0.81 |
2.59 |
|
|
Life Changing Events |
Live |
0.58 |
0.62, 4.48, p=0.042* |
0.60 |
0.59 |
0.85 |
|
Dead |
1.69 |
1.30 |
1.80 |
2.59 |
3.92 |
|
|
Total (Live + Dead) |
0.74 |
0.82 |
0.73 |
0.81 |
2.59, 8.61, p=0.0065** |
|
|
Osteoarthritis |
Live |
0.58 |
0.62 |
0.60 |
0.59 |
0.85 |
|
Dead |
1.69 |
1.30 |
1.80 |
2.59 |
3.92 |
|
|
Total (Live + Dead) |
0.74 |
0.82 |
0.73 |
0.81 |
2.59 |
|
|
Chronic Disease |
Live |
0.58 |
0.62 |
0.60 |
0.59 |
0.85 |
|
Dead |
1.69 |
1.30 |
1.80 |
2.59 |
3.92 |
|
|
Total (Live + Dead) |
0.74 |
0.82 |
0.73 |
0.81 |
2.59, 22.7, p=<0.0001*** |
|
Note: Nonsignificant (Mean of Response); Significant (Mean of Response, F-ratio, *p<0.05, **p<0.01, ***p<0.0001)
Table 11: ANOVA Analysis of Percentage of Non-Stimulated, NO Generating Cells by Age, Life Changing Events, Osteoarthritis, and Chronic Disease at Various Experimental Nitrite Exposures (N = 31).
|
Demographics |
Cell Population |
Nitrite Exposure |
||||
|
0mg/l |
1mg/l |
5mg/l |
10mg/l |
15mg/l |
||
|
Age |
Total (Live + Dead) |
0.04 |
0.671 |
0.005 |
1.93 |
1.65 |
|
Life Changing Events |
Total (Live + Dead) |
13.92, p=0.002** |
3.38 |
9.64, p=0.007** |
24.71, p=0.0002** |
34.95, p<0.001** |
|
Osteoarthritis |
Total (Live + Dead) |
0.001 |
0.46 |
2.14 |
1.56 |
1.96 |
|
Chronic Disease |
Total (Live + Dead) |
4.78, p=0.045* |
0.008 |
4.19 |
0.29 |
0.33 |
Note: F Ratio, *p <0.05; F Ratio, **p<0.01.
Table 12: Whole Model Test of Percentage of Stimulated Cells Producing NO at Various Nitrite Exposure by Age, Life Changing Events, Osteoarthritis, and Chronic Disease (N=31).
3.6Demographic variables and PBMC proliferation and NO generationParticipant responses to questions concerning numbers and types of dietary supplements, prescribed medication, age, life-changing events (i.e., known stressors or life changing events within the last six months prior to enrollment), and comorbidities outside of immunological disease such as fibromyalgia were analyzed using one way ANOVA regarding PBMC proliferation magnitude and NO generation under both stimulated and non-stimulated treatments (Table 9, 10, and 11). A whole model test was used to look for interacting relationships based on ANOVA findings. These are discussed in detail below.
3.6.1 Dietary supplement and medication usage: Of 25 participants responding to questions about supplement use, 52% took some form of dietary supplement and 26% did not (Table 1). In total, the participants used 28 different types of vitamins and supplements. Vitamins most often used included the following: Vitamin D (n=9), multivitamins (n=5), Vitamin B12 (n=2), and Vitamin C (n=1).
Other nutraceutical substances used included melatonin (n=2), fish oil (n=2), lysine (n=2), magnesium (n=2), and calcium (n=2). A correlational analysis of supplement usage (Range 0 to 4) did not show any significant relationship between supplement use and PBMC proliferation or NO generation.
Of 27 participants responding to questions about medication use, 55% took prescribed medications and 29% did not (Table 1). Reported medication usage was divided into the following broad categories: antidepressants (n=8), blood pressure (beta blockers) and LDL lowering medications (no oral nitrates were being used) (n=8), NSAIDs (n=3), hormone supplements (n= 3), and histamine blockers (n=3). Bivariate (ANOVA) analysis indicated that with increasing numbers of medications consumed (Range 0 to 6), PBMC proliferation decreased and became statistically significant at 15 mg/l nitrite concentration (Table 9).
There was no statistically significant relationship observed between the number of medications and percentage of NO generating PBMC at any of the experimental exposure levels.
3.6.2 Major life-changing events: Having experienced a major life-changing event within the past 6 months did not impact the PBMC proliferation magnitude but there was a positive correlation between increasing NO generation among live, dead, and total cells (live and dead) in the absence or presence of 1, 5,10, and 15 mg/l nitrites for both stimulated and non-stimulated cells (Table 10 and 11).
Additionally, a Whole Model test indicated that of total cells (live and dead), there was a significant and proportional impact on NO generation from life-changing events, exhibiting a dose-response trend with significantly more NO generated as the percentage of total live and dead cells (Table 12).
3.6.3 Chronic disease comorbidity: While there was no significant correlation between PBMC number and chronic disease comorbidity in the presence or absence of exogenous nitrite, the one-way ANOVA indicated a positive association between participants reporting fibromyalgia and increasing percentage of NO generating, dead PBMC cells at 15 mg/l experimental nitrite exposure, (Table 9) although it should be noted this was a very small number of subjects (n=2).
A whole model test indicated an increasing percentage of PBMC cells generating NO when correlated with chronic disease comorbidity for the control only and not at any of the nitrite experimental concentrations of 1,5,10, and 15 mg/l (Table 12).
4. Discussion
The results of this study illustrate that low-level nitrite exposure impacts PBMC viability, proliferation (Table 2 and 4). More importantly, differences exist in how the PMBC of osteoarthritis cases and control populations respond to nitrite exposures and consequent PBMC stimulation compared between no exposure (0 mg/l) versus increasing nitrite concentration (1, 5, 10, and 15 mg/l) (Table 3). When considered as a group (cases and controls), PBMC proliferation decreased across the exposure ranges for both the case and control participants in the stimulated and non-stimulated state when the nitrite exposure increased (1, 5, 10, and 15 mg/l nitrites), with the most significant increases in proliferation magnitude(r=stimulated/non-stimulated) observed at 15 mg/l nitrite exposure (Table 4, Table 5, and Figure 3).
The ex vivo studies also showed that increased sodium nitrite suppressed the growth of lymphocytes in a dose-dependent manner [25]. When comparing cases and controls, proliferation decreased significantly more among osteoarthritis participants at 5 mg/l nitrite exposure (Table 6, Figure 6, F=4.23, p<0.048*).
There was also a statistically significant association in the percentage of NO generating cells following PBMC stimulation (both live and dead) with increasing nitrite concentrations when considered together with age (1 and 10 mg/l), life-changing events (0, 1, 5, 10, and 15 mg/l), and chronic disease comorbidity (15 mg/l) (Table 10). Further, whole model tests indicated significant interactions between NO generation life-changing events (0, 5, 10, and 15 mg/l) and chronic disease comorbidities (0 mg/l) (Table 12). Life-changing events such as excessive stress are associated with the generation of NO and the formation of active nitrogen radicals. NO involvement in stress-related diseases may recommend the use of stress management and relaxation response techniques [34, 35]. NO activity was also found to be significantly higher in patients with fibromyalgia, a chronic musculoskeletal disease, and other health conditions such as chest pain, history of migraine, and morning stiffness [24, 36]. Even though the correlational analysis of supplement usage did not show any significant relationship between PBMC proliferation and NO generation, a large case-control treatment study found that the NOS activity increased significantly when patients with major depressive episodes were treated with antidepressants [37].
Other findings from this study are as follows:
- A clear statistically significant trend is present in decreasing PBMC proliferation for both OA cases and controls as nitrite exposure increases across the experimental conditions, and this trend is maintained in non-stimulated cells, and while stimulated cells still illustrate a decrease in proliferation across the experimental conditions, it is not statistically significant (Figure 2, Table 3, and Table 4, ANOVA, F= 7.07, p<0.0001). The mean PBMC proliferation was significantly suppressed among cases in both non-stimulated (F= 4.98, p= 0.0014) and stimulated (F= 3.12, p= 0.019) conditions but not among controls, indicating that participants suffering from OA might experience a significant impact on their PBMC count with increased nitrite exposure (Table 4).
- Additionally, the PBMC proliferation, magnitude of response (r = stimulated/ non-stimulated) also exhibited a significant difference among OA cases (Table 5, ANOVA, F= 3.01, p=0.023) and combined cases, controls (ANOVA, F= 2.47, p=0.046), but not among controls only. When comparing the mean between cases and controls, the analysis found that the magnitude of response was lower for OA cases vs. controls at 5 mg/l nitrite treatment (Table 6; Figure 6, t-test, F=4.23, p=0.048). It was also noted that the magnitude (r=stimulated/non-stimulated) of stimulated responses for both cases and controls combined increased significantly at 15 mg/l nitrite exposure (Table 5, Figure 3, ANOVA, F=2.47, p=0.046).
- Significant differences were seen when the mean magnitude of response was pooled into the breakpoints of 0-5 mg/l vs. 10-15 mg/l nitrite treatment (t-test; F=5.20, p = 0.023) as well as at 0-10 mg/l vs. 15 mg/l nitrite treatment (t-test; F=10.73, p = 0.001) (Table 7; Figures 4 and 5).
PBMC increased NO generation with increasing experimental exposures in all OA cases, controls, dead and live cells and dead and live cells combined under both stimulated and non-stimulated conditions. While the trend in increased NO generation was visible in most instances, it was not statistically significant, except for an instance in the OA cases and controls combined, stimulated dead cells where the trend approached significance (F= 2.33, p= 0.058) Given the small sample size and the instance that approached significance this is noted (Table 8). A whole model test showed possible effect modification with NO generation across nitrite treatment groups and life-changing events or chronic disease comorbidity (fibromyalgia, Table 12).
5. Conclusions
This pilot study is limited by small sample size but reveals important information that should be considered regarding chronic impacts of nitrate exposure, which are currently not reflected in the regulatory guidelines under the Safe Drinking Water Act (SDWA) since only the effects of acute exposure are covered by this statute. These observations are important and contribute to an increasing body of evidence that nitrite and nitrogen-derived NO may be acting on individuals with OA and other forms of arthritis in ways that could increase or prolong damaging cycles of inflammation. In the environmental health sense, this becomes important to ponder because:
- There is a directly proportional relationship between the intake of nitrate, increase in nitrite, and increase in nitric oxide in the body [22],
- Additional studies have indicated a relationship between high nitrate intake via nitrogen-containing medications and increase in the risk of OA of the hip with poor outcome [15],
- Studies of RA arthritis have indicated that higher serum levels of NO are associated with more inflammation, joint damage, and reports of greater morning stiffness and pain [38],
- Previous cohort study work by the researchers indicated that individuals consuming higher amounts of nitrate in their drinking water also complained of more musculoskeletal and joint pain [24].
- The exposure equivalent levels of sodium nitrite in this study were significant both below and above the current regulatory threshold (MCL) of 10 mg/l nitrate-nitrogen under the SDWA, and this regulatory threshold was established for acute, not chronic exposures [14].
While most demographic variables did not show significant interactions with the magnitude of PBMC proliferation or NO generation, life-changing events stood out as significant across most of the treatment categories. (Table 12, Whole Model Test) with significant findings at each treatment level above 1 mg/l.
Given these findings, additional work should be performed using larger sample sizes and in-vivo exposure samples. Additionally, research examining cytokine generation by PBMC should be conducted to determine the role of inflammatory cytokines relative to nitrite exposure levels of both cases and controls. This would aid our understanding of the relationships between the intake of nitrate/nitrite in the presence and absence of dietary or supplemental antioxidants and their implications for the health and well-being of individuals with OA. This would aid in a further understanding of the role that environmental exposures to various compounds play in chronic disease pathologies, especially at levels of exposure below that recommended by the Environmental Protection Agency.
Author Contributions
Dr. J. Shrestha performed the experimental research, sample analysis, data analysis, and writing of the initial manuscript. Dr. L. Beltz provided technical consultation for methods and the development and editing of the manuscript. Dr. C. Zeman served as a project chair, oversaw the development and performance of research in her laboratory at UNI, provided interpretation and analysis of data, and the writing of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the University of Northern Iowa (protocol number 14-0018).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare the lack of competing interests.
References
- World Health Organization. Nitrate and Nitrite in Drinking Water. WHO Guidelines for Drinking-water Quality (2011).
- Lundberg JO, Weitzberg E, Benjamin N. Nitrate, Bacteria, and Human Health. Nature Review Microbiology 2 (2004): 593-602.
- Santamaria P. Nitrate in Vegetables: Toxicity, Content, Intake and EC Regulation. Journal of the Science of Food and Agriculture 86 (2006): 10-17.
- Weyer PJ, Cerhan JR, Kross BC, et al. Municipal Drinking Water Nitrate Level and Cancer Risk in Older Women: The Iowa Women’s Health Study. Epidemiology 11 (2001): 327-338.
- Ward MH. Too Much of a Good Thing? Nitrate from Nitrogen Fertilizers and Cancer. Reviews on Environment Health 24 (2008): 357-363.
- Hord NG, Tang Y, Bryan NS. Food Sources of Nitrates and Nitrites: The Physiologic Context for Potential Health Benefits. The American Journal of Clinical Nutrition 90 (2009): 1-10.
- Lundberg JO, Weitzberg E, Gladwin MT. The Nitrate–Nitrite–Nitric Oxide Pathway in Physiology and Therapeutics. Nature Reviews 7 (2008): 156-167.
- Machha A, Schechter AN. Dietary Nitrite and Nitrate: A Review of Potential Mechanisms of Cardiovascular Benefits. European Journal of Nutrition 50 (2011): 293-303.
- Zeman C. Anthropologically Disrupted Biogeochemical Cycles and the Effect on Sustainable Human Health and Well-Being. In Encyclopedia of the UN Sustainable Development Goals; Filho L W, Wall T, Brandli L, Özuyar P, Eds.; Springer, Cham (2019).
- Mensinga TT, Speijers GJ, Meulenbelt J. Health Implications of Exposure to Environmental Nitrogenous Compounds. Toxicological Reviews 24 (2003): 41-51.
- Integrated Risk Information System. Nitrite (1987).
- Fryal CD, Kruszon-Morgan D, Gu Q, et al. Mean Body Weight, Height, Waist Circumference, and Body Mass Index Among Adults: The United States, 1999-2000 Through 2015-2016. National Health Statistics Reports 122 (2011): 1-15.
- Cortas NK, Wakid MW. Pharmacokinetic Aspects of Inorganic Nitrate Ingestion in Man. Pharmacology and Toxicology 98 (1991):192-195.
- New York State Human Health Fact Sheet. Ambient Water Quality Value for Protection of Sources of Potable Water (1998).
- Lane NE, Williams EN, Hung YY, et al. Association of Nitrate Use with Risk of New Radiographic Features of Hip Osteoarthritis in Elderly White Women: The Study of Osteoporotic Fractures. Arthritis and Rheumatism 49 (2003): 752-758.
- Hoff P, Buttgereit F, Burmester GR, et al. Osteoarthritis Synovial Fluid Activates P-inflammatory Cytokines in Primary Human Chondrocytes. International Orthopedics 37 (2013): 145-151.
- Kapoor M, Pelletier JM, Lajeunesse D, et al. Role of Proinflammatory Cytokines in the Pathophysiology of Osteoarthritis. Nature Review Rheumatology 7 (2011): 33-42.
- Abramson SB. Nitric Oxide in Inflammation and Pain associated with Osteoarthritis. Arthritis Research & Therapy 10 (2008): 1-7.
- Lin Z, Willers C, Xu J, et al. The Chondrocyte: Biology and Clinical Application. Tissue Engineering 12 (2006): 1971-1984.
- Pelletier JP, Pelletier J M, Abramson SB. Osteoarthritis, an Inflammatory Disease: Potential Implication for the Selection of New Therapeutic Targets. Arthritis and Rheumatism 44 (2001): 1237-1247.
- Ersoy Y, Özerol E, Baysal Ö, et al. Serum Nitrate and Nitrite Levels in Patients with Rheumatoid Arthritis, Ankylosing Spondylitis, and Osteoarthritis. Annals of the Rheumatic Diseases 61 (2002): 76-78.
- Kortboyer JM, Colbers EEPH, Vaessen HMAG, et al. A Pilot Study to Investigate Nitrate and Nitrite Kinetics in Healthy Volunteers with Normal and Artificially Increased Gastric pH After Sodium Nitrate Ingestion. In Health Aspects of Nitrate and its Metabolites (Particularly Nitrite), Proceedings of the International Workshop, Bilthoven, Netherlands (1995).
- Kleinbongard P, Dejam D, Rassaf T, et al. Plasma Nitrite Reflects Constitutive Nitric Oxide Synthase Activity in Mammals. Free Radical Biology and Medicine 35 (2003): 790-796.
- Zeman C, Beltz L, Linda M, et al. New Questions and Insights into Nitrate/Nitrite and Human Health Effects: A Retrospective Cohort Study of Private Well Users’ Immunological and Wellness Status. Journal of Environmental Health 74 (2011): 8-18.
- Ustyugova IV, Zeman C, Dhanwada K, et al. Nitrate/Nitrite Alters Human PBMC Proliferation and Cytokine Production. Environmental Contamination and Toxicology 43 (2002): 270-276.
- Loeser RF, Goldring SR, Scanzello CR, et al. Osteoarthritis: A Disease of the Joint as an Organ. Arthritis and Rheumatism 64 (2012): 1697-1707.
- Pelletier JM, Pelletier JP. Inflammatory Factors Involved in Osteoarthritis. In Osteoarthritis; Inflammation; and Degradation: A Continuum, Buckwater J., Eds.; Amsterdam: IOS Press (2007): 3-13.
- Misko TP, Radabaugh MR, Highkin M, et al. Characterization of Nnitro Tyrosine as a Biomarker for Arthritis and Joint Injury. Osteoarthritis and Cartilage 21 (2013): 151-156.
- Wimalawansa SJ. Nitric Oxide and Bone. Annals of the New York Academy of Sciences 1192 (2010): 391-403.
- Omer N, Rohilla A, Rohilla S, et al. Nitric Oxide: Role in Human Biology. International Journal of Pharmaceutical Sciences and Drug Research 4 (2012): 105-109.
- Kanof ME, Smith PD, Zola H. Isolation of Whole Mononuclear Cells from Peripheral Blood and Cord Blood in Immunologic Studies in Humans. Current Protocols in Immunology 19 (1996): 7.1.1-7.1.7.
- Luminex Corporation. The Muse Nitric Oxide Kit User’s Guide, Catalog No. MCH100112 (2020).
- Luminex Corporation. The Muse Count and Viability Kit, Catalog No. MCH100102 (2020).
- Kumar A, Chanana P. Role of Nitric Oxide in Stress-Induced Anxiety: From Pathophysiology to Therapeutic Target. Vitamins and Hormones 13 (2017): 147-167.
- Esch T, Stefano GB, Fricchione GL, et al. Stress-related diseases – a potential role for nitric oxide. Medical Science Monitor 8 (2002): 103-118.
- Bölgen Ö, Burak MY, Yapici T, et al. Arginase, NOS Activities, and Clinical Features in Fibromyalgia Patients. American Academy of Pain Medicine 5 (2009): 813-818.
- Loeb E, El Asmar K, Trabado S, et al. Nitric Oxide Synthase Activity in Major Depressive Episodes Before and After Antidepressant Treatment: Results of a Large Case-Control Treatment Study. Psychological Medicine First View (2020): 1-10.
- Ali AM, Habee, RA, et al. Higher Nitric Oxide Levels are Associated with Disease Activity in Egyptian Rheumatoid Arthritis Patients. Revista Brasileira de Reumatologia 54 (2014): 446-451.


 Impact Factor: * 3.6
Impact Factor: * 3.6 Acceptance Rate: 76.49%
Acceptance Rate: 76.49%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 