Exposure Factors to Toxic Trace Elements among Post-Delivery Mothers Living in the Vicinity of Mining Operating Plants in Lubumbashi
Cham LC1*, Chuy KD2, Mwembo TA1,2, Chenge MF1,2,3, Tamubango H4, Kaniki A5, Kalenga MK1, 2
1Department of Gynecology and Obstetrics, University of Lubumbashi, Democratic Republic of Congo
2Public Health School, University of Lubumbashi, Democratic Republic of Congo
3Health Knowledge Center (HKC), Democratic Republic of Congo
4High School of Medical Technics of Likasi, Democratic Republic of Congo
5Engineering School, University of Lubumbashi, Democratic Republic of Congo
*Corresponding Author: Dr. Cham Chamy Lubamba, Department of Gynecology and Obstetrics, University of Lubumbashi, Democratic Republic of Congo
Received: 02 March 2020; Accepted: 09 March 2020; Published: 12 March 2021
Article Information
Citation: Cham LC, Chuy KD, Mwembo TA, Chenge MF, Tamubango H, Kaniki A, Kalenga MK. Exposure Factors to Toxic Trace Elements among Post-Delivery Mothers Living in the Vicinity of Mining Operating Plants in Lubumbashi. Journal of Environmental Science and Public Health 5 (2021): 155-168.
View / Download Pdf Share at FacebookAbstract
Background: The environmental pollution contributes to human exposure to TE through direct skin contact with soil, inhalation of wind-blown dust, direct deliberate or unintentional ingestion of soil and ingestion of foods grown on TE-contaminated soils and waters.
Method: A prospective cohort study of mothers and newborns exposed to TE was conducted. Informations on exposure conditions were collected by a questionnaire-based interviews of 378 post-delivery mothers from whom TE (Al, As, Cd, Cr, Ni, and Pb) were tested at the laboratory of the Congolese Control Office of Lubumbashi by ICP-OES in total blood samples.
Results: The pregnant women working in mining plants had four times the risk of having high concentration of Aluminum: RR 3.84(1.697-8.709), and two times for Lead: 2.08 (1.237-3.50) ; The post-delivery mothers who consumed clay (geophagia) had two fold the risk of having high concentration of Nickel: RR2.14 (1.37-3.34) and Chromium :RR 1.57 (1.04-2.39).Those living in the immediate vicinity of welding workshops had four times the risk of having high concentration of Arsenic: RR 3.97 (1.74-9.05) , when the immediate proximity to an artisanal or industrial mining plants increased two fold the risk of having high concentration of Cadmium: RR 1.86 (1.19-2.91).
Conclusion: Among potential exposure factors to trace elements studied in Lubumbashi: geophagia, immediate proximity to mining plants, use of well water for drinking or housework were the most predominant and associated to high concentration of toxic trace element in total blood of the post-delivery mothers. Implementation of preventive measures should consider these factors.
Keywords
<p>Exposure; Toxic trace elements; Post-delivery mothers; Newborns; Lubumbashi</p>
Article Details
1. Introduction
Trace elements (TE) have been widely found in various environmental media of the city of Lubumbashi (soil, water, air, and food). Specifically, in the areas immediately surrounding the mining plants. These areas are characterized by bare and sparsely vegetated land and soils that are severely eroded, acidic, and contaminated with elevated levels of TE (Cu, Zn, Pb, Cd, As…). TE such as Pb, Zn, Cd, Hg, As and Cr are emitted into the environment in large quantities through wastewater irrigation, solid waste disposal, sludge application, vehicular exhaust, and atmospheric emission. The lack of preventive measures and strict regulations rules in Lubumbashi and worldwide favor the environmental pollution [1-7].
The above-mentioned environmental conditions may contribute to human exposure to TE through direct skin contact with soil (dermal exposure), inhalation of wind-blown dust (inhalation route), direct deliberate or unintentional ingestion of soil and ingestion of foods grown on TE-contaminated soils and waters (oral exposure) [8-10]. Blood levels of TE are reliable biomarkers of recent exposure, its concentrations come down to a minimum level within a few hours or days of exposure [11]. TE are persistent in the human body, their excretion half-lives last for decades and they can lead to a wide range of toxic effects (mutagenic, teratogenic, and carcinogenic effects [12-15]. High concentrations of TE in the body can affect the central nervous system and act as co-factors, initiators, or promoters of some diseases [16]. As, Cr, and Pb are among the TE of greatest concern due to their high degree of toxicity (17].
Excessive concentration of toxic TE (Al, As, Cd et Pb) with bioaccumulation factor >1 were observed in the total blood of post-delivery mothers and their newborns residing in the city of Lubumbashi < of 3km and >3km from the mining processing plants [18]. Another study on the same population noticed low essential TE concentration (Cu, Se and Zn) with bioaccumulation factor <1 and excessive geometric mean for Cr [19]. These two studies unlighted the need to analyze the determinants of TE bioaccumulation in these vulnerable groups. Identifying the potential exposure routes to TE in Lubumbashi can help implement preventive measures.
It is difficult to identify the key exposure route of humans to TE because of lack of multipathway risk analysis in studies; media or pathway-specific approach may also fail to ensure public safety. Therefore, it is necessary to assess the aggregate exposure factors and pathways of different environmental contaminants [20].
In a mining city like Lubumbashi, where populations are exposed to TE, up to now no study has focused on contamination routes. To address this knowledge gap, the aim of this study is to identify main exposure routes of post-delivery mothers to toxic TE in Lubumbashi.
2. Materials and Methods
2.1 Study design
A prospective cohort study of mothers and newborns exposed to TE was conducted from November 30, 2018 to May 30, 2019.
2.2 Study area
The study was done in Lubumbashi, a city born from the exploitation by the Union Minière du Haut Katanga (Gécamines) of copper deposits (1906-1910). Since the 2000s, minerals are also exploited by private mining companies including: STL, Ruashi mining, Chemaf, Somika, CDM, Anvil Mining, etc.
2.3 Study population
This study included 378 post-delivery mothers consecutively enlisted in ten medical institutions in Lubumbashi (Lubumbashi University Clinics, Jason Sendwe Provincial General Hospital, CMDC, Imani Polyclinic, Tshamilemba Learning and Researcher Health Center, la garde Medical Center, Ruashi Military Hospital, Eben Ezer Health Center, Crisem Medical Center, and Luna Medical Center).
The post-delivery mother’s home were < of 3km and >3km from the mining processing plants. Subjects were grouped in accordance with TE concentration in total blood. The groups were constituted whether the TE concentration was higher or lower than the TE reference value.
2.4 Data collection
Informations on exposure conditions were collected by a questionnaire-based interviews and the following data were recorded: age, place of residence < of 3km and >3km from the mining processing plants, occupation of the post-delivery mothers whether she worked in a mining processing plant or not, occupation of the partner whether he worked or not in a mining processing plant, source of water for drinking and domestic use, type of energy used for cooking, geophagia behaviors, the presence of welding or recycling batteries workshop near the home.
2.5 Metal trace elements analysis
The dosage of TE in total blood was done using Perkin Elmer brand ICP- OES Optima 8300 (Optical Emission inductively coupled plasma spectrometry system) at the laboratory of the Congolese Control Office (ISO 9150 quality certification since 2010). The following TE (Al, As, Cd, Cr, Ni, and Pb) were measured.
2.6 Statistical analysis
Our data was encoded using the EPI info 7 software, SPSS 23 allowed us to set the two groups according to TE concentration, to calculate the chi square and relative risk for each exposure factor. We have correlated the two groups of each TE concentrations (higher or lower than the reference value) to the potential exposure factors by calculating a chi square. If the Chi square value was statistically significant, we calculated the relative risk for each TE to have higher TE concentration than the reference value according to each exposure factors. Only significant results are reported in this study.
For toxic TE there were a health risk if the RR value were above 1 with a significant confidence interval.
2.7 Ethical considerations
This study was approved by the Medical Ethics Committee of the School of Public Health of the University of Lubumbashi (UNILU/CEM/117/2018 delivered the 10/10/2018).
All participants gave their informed consent prior to answer to the questionnaire and collection of blood samples.
3. Results
3.1 Main sociodemographic characteristics of our study population
In this table we present the main sociodemographic characteristics of our study population.
|
Sociodemographic characteristics |
Number (n=378) |
Pourcentage (%) |
|
Age (years) <18 18-35 >35 |
2 321 55 |
0,50 84,90 14,6 |
|
Residential district < 3km from mining plants > 3km from mining plants |
152 226 |
40.2 59.8 |
|
Level of education Analphabete Primary school Secondary school University Post-graduate |
19 30 184 138 7 |
5,0 7,9 48,7 36,5 1,9 |
|
Occupation of the pregnant woman Unemployed State official Informal Artisanal miner |
243 68 65 2 |
64,3 18,0 17,2 0,5 |
|
Profession of the partner/spouse State official Informal Artisanal miner Unemployed |
216 143 14 5 |
57,1 37,8 3,7 1,3 |
|
Parity Nulliparous(0) Primiparous (1) Pauciparous (2-3) Multiparous (4-5) Large multiparous (≥6) |
4 86 140 91 57 |
1,1 22,8 37,0 24,1 15,1 |
The mean age of the post-delivery mothers was 29 ± 6 years; the majority (59.8%, n=226) resided in neighbourhoods located >3 km from the mines; 48.70% (n= 226) had a secondary education level and 36.5% (n=138) university level;64.30% (n=243) were unemployed;57.10% (n=216) had a spouse state official;37% (n=140) were pauciparous, 22.8% (n=86) of primiparous and 15.1% (n=57) of large multiparous.
Table 1: Sociodemographic characteristics of the post-delivery mothers.
3.2 Potentially exposure factors of our study population to TE
All the potentially exposure factors of our study population to TE are listed in the following table based on the declarative information from the post-delivery mothers.
In this table we report all the potential exposure factors of the 378 post-delivery mothers to toxic trace elements, dichotomized as presence or absence of the factor.
|
Potential exposure factors |
n |
Present |
Absent |
|
Immediate proximity to an artisanal or industrial mining plant |
378 |
152 (40.2) |
226 (59.8) |
|
Geophagia |
378 |
192 (50.8) |
186 (49.2) |
|
Pregnant woman working in mining plants |
378 |
73 (19.3) |
305 (80.7) |
|
Partner working in mining plants |
378 |
112 (29.62) |
266 (69.38) |
|
Painting workshop in the immediate vicinity of the home |
378 |
22 (5.82) |
356 (94.18) |
|
Welding workshop in the immediate vicinity of the home |
377 |
50 (13.26) |
327 (86.73) |
|
Battery recycling workshop in the immediate vicinity of the home |
374 |
49 (13.10) |
325 (86.89) |
|
Use of REGIDESO water for drinking |
378 |
169 (44.7) |
209 (55.3) |
|
Use of well water for drinking |
378 |
56 (14.81) |
322 (85.19) |
|
Use of mineral water for drinking |
378 |
95 (25.13) |
283 (74.87) |
|
Mixing of different waters for drinking |
378 |
58 (15.34) |
320 (84.66) |
|
Use of REGIDESO water for housework |
378 |
217 (57.4) |
161 (42.6) |
|
Use of well water for housework |
378 |
86 (22.75) |
292 (77.25) |
|
Mixing of different waters for housework |
378 |
75 (19.84) |
303 (80.16) |
|
Electricity used for cooking food |
378 |
48 (22.7) |
330 (87.3) |
|
Biomass used for cooking food |
378 |
191 (50.52) |
187 (49.48) |
|
Mixture for cooking food |
378 |
139 (36.77) |
239 (73.33) |
Table 2: Potential exposure factors of post-delivery mothers to TE.
In this table among the potential exposure factors, geophagia was present in 50.8% of the study population, biomass were used for cooking food in 50.52% of cases, while electricity were used only by 22.7% of the population study. Well water were used for drinking by 14.81% and 27.51% of post-delivery mothers lived in direct proximity to artisanal or industrial mining plants.
|
Exposure factors |
ETM |
Chi 2(p) |
RR (CI) |
Comments |
|
Immediate proximity to an artisanal or industrial mining plant |
Lead Cadmium Aluminium |
0.000 0.007 0.007 |
2.32 (1.98-5.63) 1.86 (1.19-2.91) 2.15 (1.23-3.76) |
High Risk |
|
Pregnant woman working in mining plants |
Aluminum Lead |
0.000 0.006 |
3.84 (1.69-8.70) 2.08 (1.23-3.50) |
- |
|
Geophagia |
Aluminum Lead Nickel Chromium |
0.002 0.000 0.001 0.035 |
2.23 (1.34-3.70) 2.94 (1.89-4.57) 2.14 (1.37-3.34) 1.57 (1.04-2.39) |
- |
|
Welding workshop in the immediate vicinity of the home |
Arsenic Nickel |
0.000 0.003 |
3.97 (1.74-9.05) 3.09 (1.41-6.77) |
- |
|
Biomass used for cooking food |
Arsenic Lead Nickel |
0.035 0.002 0.003 |
2.01 (1.08-3.75) 3.51 (1.53-8.09) 2.59 (1.34-4.86) |
- |
|
Use of well water for household work |
Lead Cadmium Nickel |
0.000 0.048 0.002 |
3.05 (2.20-5.96) 1.54 (1.02-2.32) 2.04 (1.29-3.21) |
- |
|
Use of well water for drinking |
Lead Cadmium Nickel |
0.000 0.003 0.031 |
3.10 (1.97-4.90) 1.97 (1.26-3.08) 1.73 (1.05-2.86) |
- |
Table 3: Association between exposure factors and high concentration of TE in the total blood of post-delivery mothers.
In this table we report among all exposure factors those that showed a significant association with high TE concentration in the total blood of post-delivery mothers through their Chi square and relative risk values.
We can notice in this table that the following exposure factors were associated to the risk of having high toxic TE concentration in the total blood of post-delivery mothers:
- The post-delivery mothers whose houses were within immediate proximity to artisanal of industrial mining plants had two times the risk of having high concentration of Lead: RR 2.32 (1.98-5.63); Cadmium: RR 86 (1.19-2.91) and Aluminum: RR 2.15 (1.23-3.76)
- The pregnant women working in mining plants had four times the risk of having high concentration of Aluminum: RR84 (1.697-8.709) but two times the risk for Lead: RR 2.08 (1.237-3.50)
- The post-delivery mothers with geophagia behavior had two times the risk of having high concentration of Aluminum: RR2.23 (1.34-3.70); Nickel: RR2.14 (1.37-3.34); Chromium: RR1.57 (1.04-2.39) and three times for Lead: RR2.94 (1.89-4.57)
- The post-delivery mothers living in the immediate vicinity of welding workshops had four-fold the risk of having high concentration of Arsenic: RR 97 (1.74-9.05) and almost three-fold of having high concentration of Nickel: RR 3.09 (1.41-6.77)
- The post-delivery mothers who used biomass for cooking food had two times the risk of having high concentration of Arsenic: RR2.01 (1.08-3.75) and Nickel: RR 2.59 (1.34-4.86) but all most four times risk of having high level of Lead: RR 3.51 (1.53-8.09)
- The post-delivery mothers who used well water for household work had two times the risk of having high concentration of Cadmium RR 1.54 (1.02-2.32); Nickel: RR 2.04 (1.29-3.21) and three times for Lead: RR 3.05 (2.20-5.96)
- The post-delivery mothers who used well water for drinking had two times the risk of having high concentration of Cadmium RR 1.97 (1.26-3.08); Nickel: RR 1.73 (1.05-2.86) and three times for Lead: RR 3.10 (1.97-4.90)
3.3 Cumulative risk assessment
To assess a complete exposure pathway to TE, it is important to consider all exposure factors. They should not be taken separately but in global manner. Therefore, it is advised to analyse the presence of all exposure components such as source of contamination, environmental medium and transport mechanism, point of exposure, route of exposure and the exposed population.
In the figure 1 we cumulate for each TE the exposure factors to be considered in our settings.
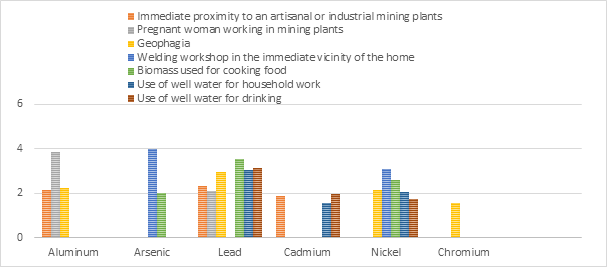
Figure 1: Cumulative toxic TE risk assessment.
From this figure we can notice that among exposure factors to TE, the most prominent are immediate proximity to mining plants, geophagia and use of well water for drinking and housework. Lead and Nickel were the most present TE in all potential exposure media.
4. Discussion
High concentration of TE in the total blood of post-delivery mothers and their newborns as reported in Lubumbashi [18, 19] is a public concern regarding the harmful effects of excessive toxic TE concentration and unbalanced essential TE concentration in pregnant women and their newborns [21]. Therefore, it is important to identify the main exposure sources of post-delivery-mothers to TE in order to implement protective measures that will help reduce harmful health effects on mothers and their newborns. Despite our methodological approach that allowed us to have declarative information from post-delivery mothers on the exposure factors to TE in the city of Lubumbashi, it clearly showed the potentially main exposure factors of our study population to TE. For the mining city of Lubumbashi, it is the first study focused on exposure factors of post-delivery mothers and their newborns to TE.
4.1 Immediate proximity to an artisanal or industrial mining plant and pregnant woman working in mining plants
The direct contact with TE is among the principal exposure route through direct contact with naked skin and inhalation [8-10]. Depending on the exploitation and treatment procedure used in the mining plant whether artisanal or industrial; dust, smoke and other rejections containing high concentration of TE can get directly in contact with humans [22].
In our study we noticed that through immediate proximity to mining plants, the post-delivery mothers were exposed to high concentrations of Aluminum, Cadmium and Lead. Aluminum is ubiquitous in the environment, being one of the most abundant elements in the earth’s crust. Aside from its natural presence, it is an environmental contaminant, due to anthropogenic releases associated with industrial processes. Humans are exposed by the inhalation of ambient air and the ingestion of food and drinking water. Additional sources are food additives and the migration of Aluminum from food contact materials such as cooking utensils and packaging materials. At this point in our setting especially in the suburb of Lubumbashi cooking utensils are artisanally made from recycled aluminum.
Certain consumer products (personal care products and cleaning agents) and pharmaceuticals (antacids and vaccines) are further sources of exposure for humans. In humans, the oral bioavailability from food and drinking water is low (0.1-0.8%), from antiacids is also low (<0.01-0.2%). Dermal absorption from antiperspirants is very low (0.00052%). Absorption following inhalation is estimated at 1.5-3% for the respirable fraction. This bioavailability is approximately 10-fold higher on average than the gastrointestinal resorption [22]. Therefore, in our study the high concentration of Aluminum observed in post -delivery mothers having immediate proximity to mining plants and working in it can be related mainly to inhalation of dust containing Aluminum. These dust and smoke come from direct emission from the mining exploitation (removal of earth crust and combustion) but also by the dust generated by trucks carrying mineral products. The environment is usually polluted by cadmium through the discharge of polluted water from plating works, by dust from mining and especially from lead and zinc melting and plating work [10, 23]. Increase of traffic volume, abuse of fertilizers and pesticides causing heavy metal concentration of soils, dusts, and roadside vegetation contribute also to the pollution of the environment [23].
Cadmium is absorbed by the body through the lungs and the digestive tract. In the work environment, absorption through the lungs is important [24]. In our study, the post-delivery mothers who were in immediate proximity to mining plants had high blood level of cadmium supporting that this trace element is emitted in the environment as a result of human activities. Organic forms of heavy metals (monomethyl mercury and dimethyl cadmium) are very toxic and adversely affect water qualities by seepage and pollute underground water sources. Lead is emitted during its mining and smelting activities, from automobile exhausts and from old lead paints It’s emissions also occur from non-related industries activities such as other metals works, incinerators, and in small amounts from power station. Humans are exposed to through inhalation of contaminated air and dust, ingestion of contaminated soil and water but through wine bottle wraps, mirror coatings, batteries, tiles and linolein [10].
4.2 Geophagia
Geophagia involves deliberate eating of earth such as clay. It is a widespread practice in many parts of the world such as in Asia, Africa, South America, North America, and parts of Europe [25]. Most of the eaten clay contain a mixture of toxics elements (As, Cd, and Pb) [26]. Pregnant women are of particular concern in this context since As, Pb, Cd, and Mn can transfer from the mother to the foetus, placing the health of the unborn baby at risk [27, 28]. In our study the post-delivery mothers who consumed clay during pregnancy were at high risk of having high concentration of Aluminum, Lead, Nickel, and Chromium. This finding shows that the soil is polluted by toxic TE which principally originate from mineral exploitation. Thus, all the women consuming clay are at risk of having high concentration of toxic TE even if they do not live near or work in mining plants [18]. We are also aware that due to the stigma associated with this practice, many women are ashamed to admit that they consume clay, then the number of post-delivery mothers consuming clay may be minored in our study. Even though this practice is harmful to the pregnant women and their newborns by exposing them to high blood concentration of toxic TE. Chromium is widely distributed in the earth's crust. Soils and rocks may contain small amounts of chromium, almost in the trivalent state. In soil, trivalent chromium predominates, in water trivalent chromium forms hydroxides and complexes that are adsorbed at relatively high pH values. In surface waters, the ratio of trivalent chromium to hexavalent chromium varies widely, and relatively high concentrations of the latter can be found locally. In general, hexavalent chromium salts are more soluble than those of trivalent chromium, making hexavalent chromium relatively mobile. In air, chromium is present in the form of aerosols. It can be removed from the atmosphere by wet and dry deposition. Both trivalent and hexavalent chromium are released into the air [29].
4.3 Welding workshop in the immediate vicinity of the home
Welding involves the process of heating the base and filler metals to a high temperature that leads to the formation of fumes. Welding emissions consist of gaseous pollutants, micron and sub-micron particles of different heavy metals (Aluminum, Antimony, Arsenic, Beryllium, Cadmium, Chromium, Cobalt, Copper, Iron, Lead, Manganese, Molybdenum, Nickel, Silver, Tin, Titanium, Vanadium, Zinc) [30]. Arsenic in its inorganic form, can be found in soil, air, and water. Human exposure can occur through many different pathways: occupations, the environment, and food. Arsenic contamination of drinking water from geologic and manmade sources has been found globally. Human activity, including mining, smelting, pesticide use, and coal ash disposal, has been linked to water and soil contamination. Use of arsenic contaminated water for agricultural irrigation extends the exposure pathway to soil and food crops. Other sources of soil contamination with arsenic include release of arsenates in pressure-treated lumber (chromated copper arsenate) and localized industrial activities [31]. In our study the proximity to mining plants can lead to water and soil contamination and explain the high concentration of Arsenic found in post-delivery mothers. Previous studies in the same city found high level of toxic TE in the soil, vegetables, and water among them Arsenic [5-7].
Nickel is a micronutrient for most organisms, but excessive quantities have toxic effects by gaseous discharge and solid waste from metallurgical industries [23]. Human are therefore exposed to inhalation of dust and fumes containing Nickel.
In our study the post-delivery mothers living in the immediate vicinity of welding workshops were at risk of having high concentration of Arsenic and Nickel. They are exposed through inhalation, ingestion, and dermal contact with the weld fume. This finding show that welding workshop near homes of pregnant women is a source of environmental pollution (soil, air, and water). Thus, expose the pregnant women to high concentration of this toxic TE in the total blood.
4.4 Biomass fuel used for cooking food
The post-delivery mothers who used biomass for cooking food were at risk of having high concentration of Arsenic, Nickel and Lead. The main biomass fuel used in the city of Lubumbashi is charcoal. Charcoal burning is a source of gaseous and particulate pollutants, including CO, aromatic volatile organic chemicals, polycyclic aromatic hydrocarbons, carbonyl compounds and trace elements [32]. Human exposure may arise through the incidental inhalation of gases and ashed, respirable particulates, and via the ingestion of chemicals absorbed by or deposited on food. A Sharp and A Turner showed in their study high bio accessible concentrations of TE (Al, As, Cd, Cu, Fe, Hg, Mn, Ni, Pb and Zn) in the combusted residues of in six products containing lump wood charcoals and nine products containing briquette charcoals [32]. Though we have not realized such measures we can assume that for our population study the use of biomass for cooking food had contributed to the high concentration of toxic TE observed in the total blood of post-delivery mothers among them Arsenic and Nickel.
4.5 Use of well water for household work and drinking
Well water comes from the water stored in the earth and rocks below ground. Substances on the surface of the ground such as gas, animal and industrial waste can seep down and pollute it. Some natural substances stored in rocks and soil can also affect the smell, taste, color, and safety of well water. Lead is not naturally found in water. Most lead in well water results from corrosion of metal in old lead pipes, lead-based solder, or brass fittings. In our study, the post-delivery mothers who used well water for household work were at risk of having high concentration of Lead, Cadmium and Nickel in their total blood. Therefore, these findings can be related to the environmental pollution from mining and other human activities.
4.Conclusion
Immediate proximity to an artisanal or industrial mining plant, pregnant woman working in mining plants, geophagia, welding workshops in the immediate vicinity of the home and biomass fuel used for cooking food were amongst the exposure factors positively associated to high concentration of toxic TE. To assess a complete exposure pathway of these TE, we should consider the presence of all exposure components such as a source of contamination, an environmental medium and transport mechanism, a point of exposure, a route of exposure and the exposed population.
References
- Banza Lubaba Nkulu C, Casas L, Haufroid V, et al. Sustainability of artisanal mining of cobalt in DR Congo. Nat Sustain 1 (2018): 495-504.
- Nkulu cbl. (sd). Rapport de l’enquete sur la pollution chimique dans les quartiers Tshamilemba et kabecha de la ville de lubumbashi. rapport du Centre Carter intitulé les investissements miniers en RDC : Développement ou appauvrissement des communautés locales Octobre (2012).
- Musa Obadia P, Kayembe-Kitenge T, Haufroid V, et al. Preeclampsia and blood lead (and other metals) in Lubumbashi, DR Congo. Environ Res 167 (2018): 468-471.
- Godart et Bogaert. Pollution et contamination des sols aux métaux lourds dues à l’industrie métallurgique à Lubumbashi : Empreinte écologique, impact paysager,pistes de gestion,ULB mémoires) (2010).
- Kashimbo Kalala S, Mbikayi E, Ngoy Shutcha M, et al. Evaluation du risque de contamination de la chaine Alimentaire en éléments traces métalliques de trois espèces maraichères cultivées au bord de la rivière Lubumbashi, International Journal of Innovation and Applied Studies10 (2015): 1125-1133.
- Mpundu Mubemba Mulambi Michel, Useni Sikuzani Yannick, Ntumba Ndaye François, et al. Évaluation des teneurs en éléments traces métalliques dans les légumes feuilles vendus dans les différents marchés de la zone minière de Lubumbashi, Appl. Biosci (2013).
- Mylor Ngoy Shutcha, Robert-Prince Mukobo, Donato Kaya Muyumba, Michel Mpundu Mubemba, Michel-Pierre Faucon, Tolérant Luba lega Kimbamba, Ludovic Andres, et al. Fond pédogéochimique et cartographie des pollutions des sols à Lubumbashi, Bogaert J, Colinet G, Mahy G. Anthropisation des paysages katangais. Gembloux,Belgique : Presses Universitaires de Liège – Agronomie-Gembloux (2018).
- Pierzynski GM, Strategies for remediating trace-element contaminated sites, in: Iskandar IK, Adriano DC (Eds.). Remediation of Soils Contaminated with Metals,Science Reviews, Northwood, UK (1997).
- Qu C-S, Ma Z-W, Yang J, et al. Human exposure pathways of heavy metals in a lead-zinc Mining Area, Jiangsu Province, China. PLoS One 7 (2012): e46793.
- Duruibe JO1, Ogwuegbu MOC, Egwurugwu JN. Heavy metal pollution and human biotoxic effects, International Journal of Physical Sciences 2 (2007): 112-118.
- Mandal BK, Ogra Y, Suzuki KT. Speciation of arsenic in human nail and hair from arsenic-affected area by HPLC-inductively coupled argon plasma mass spectrometry, Toxicol. Appl. Pharmacol 189 (2003): 73-83.
- Tong S, von Schirnding YE, Prapamontol T. Environmental lead exposure: a public health problem of global dimensions. B World Health Organ 78 (2000): 1068-1077.
- Ja¨rup L, Hellstro¨m L, Alfve´n T, et al. Low level exposure to cadmium and early kidney damage: the OSCAR study. Occup Environ Med 57 (2000): 668.
- Thomas LDK, Hodgson S, Nieuwenhuijsen M, et al. Early kidney damage in a population exposed to cadmium and other heavy metals. Environ Health Persp 117 (2009): 181.
- Putila JJ, Guo NL. Association of arsenic exposure with lung cancer incidence rates in the United States. PLoS ONE 6 (2011): e25886.
- Faiz Y, Tufail M, Javed MT, et al. Road dust pollution of Cd, Cu, Ni, Pb and Zn along Islamabad Expressway Pakistan. Microchem. J 92 (2009): 186-192.
- Tchounwou PB, Yedjou CG, Patlolla AK, et al. Heavy metals toxicity and the environment. Molecular, Clinical and Environmental Toxicology 101 (2012); 133-164.
- Cham LC, Chuy KD, Mwembo TA, et al. Toxic metallic trace elements in post-delivery mothers and their newborns residing near and far from mining operating plants in Lubumbashi, J Environ Sci Public Health 4 (2020): 367-379.
- Cham LC, Chuy KD, Mwembo TA, et al. Essential metallic trace elements in post-delivery mothers and their newborns residing near and far from mining operating plants in Lubumbashi, J Environ Sci Public Health 4 (2020): 442-454.
- Falah S Al-Fartusie, Saja N. Mohssan, Essential Trace Elements and Their Vital Roles in Human Body, Indian Journal of Advances in Chemical Science 5 (2017): 127-136.
- Hu J, Dong Z. Development of Lead source-specific exposure standards based on aggregate exposure assessment: Bayesian inversion from biomonitoring information to multipathway exposure. Environ Sci Technol (2011).
- Affourtit F, Bakker MI, Pronk MEJ. Human health risk assessment of aluminium, National Institute for Public Health and the Environment, RIVM report (2020): 0001.
- Sumia Ahmed Mohamed Nimir, Environmental impact of some trace elements emission from the work environment of atbara cement factory and atbara railway foundry, Thesis Department of Chemistry Faculty of Education University of Khartoum.
- Wojciech Wasowicz, Jolanta Gromadzinska, Konrad Rydzynski, Blood concentration of essential trace elements and heavy metals in workers exposed to lead and cadmium. International Journal of Occupational Medicine and Environmental Health 14 (2001): 223-229.
- Shaban W Al-Rmalli, Richard O Jenkins, Michael J Watts, et al. Risk of human exposure to arsenic and other toxic elements from geophagy: trace element analysis of baked clay using inductively coupled plasma mass spectrometry, Environmental Health 9 (2010): 79.
- Chowdhury MAI, Ahmed MF, Ali MA. In Fate of Arsenic in the Environment. ITN Centre, BUET and United Nations University, ISBN 984-32-0507-3 (2003): 21-37.
- Rudge CV, Rollin HB, Nogueira CM, et al. The placenta as a barrier for toxic and essential elements in paired maternal and cord blood samples of South African delivering women. J Environ Monit 11 (2009): 1322-1330.
- Sorkun HC, Bir F, Akbulut M, et al. The effects of air pollution and smoking on placental cadmium, zinc concentration and metallothionein expression. Toxicology 238 (2007): 15-22.
- Guidelines for drinking-water quality, 2nd ed. Health criteria and other supporting information. World Health Organization, Geneva 2 (1996).
- Jilla, Abhinay. Evaluation of total fume and heavy metal emission factors applicable to gas metal arc welding . University of New Orleans Theses and Dissertations (2019): 2612.
- Beth A Baker, Victoria A Cassano, Carolyn Murray. Arsenic Exposure, Assessment, Toxicity, Diagnosis, and Management. Guidance for Occupational and Environmental Physicians, American College of Occupational and Environmental Medicine (2018).
- Annabel Sharp, Andrew Turner. Concentrations, and bio accessibilities of trace elements in barbecue charcoals, Journal of Hazardous Materials 262 (2013) 620-626.


 Impact Factor: * 3.6
Impact Factor: * 3.6 Acceptance Rate: 76.49%
Acceptance Rate: 76.49%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 