Evaluation of Stability against Oxidation in Edible Fats and Oils
Selin Sahin*
Istanbul University-CerrahpaSa, Engineering Faculty, Department of Chemical Engineering, Istanbul, Turkey
*Corresponding Author:Selin Sahin, Istanbul University-CerrahpaSa, Engineering Faculty, Department of Chemical Engineering, 34320 Avcilar, Istanbul, Turkey
Received: 06 September 2019; Accepted: 23 September 2019; Published: 30 September 2019
Article Information
Citation:
Selin Åžahin. Evaluation of Stability against Oxidation in Edible Fats and Oils. Journal of Food Science and Nutrition Research 2 (2019): 283-297.
DOI: 10.26502/jfsnr.2642-11000027
View / Download Pdf Share at FacebookAbstract
There is a lipid oxidation problem in edible oils and fats due to the fact that they are continuously exposed to various storage conditions or overheating. Therefore, investigation of the stabilization of edible oil products is an area worth researching due to consumer-related health concerns. Consequently, thermal oxidative deterioration might be prevented if the stability of the product is comprehended fairly. Thus, food formulations can be regulated depending on the findings of the stability measurements. In this review article, the mechanism and structure of lipid oxidation process have been explained. Furthermore, the significance of prediction of shelf-life has been mentioned as well as kinetic and thermodynamic comprehension in oxidation process. Additionally, all the mentioned stability measuring methods have been discussed, respectively.
Keywords
<p>Lipid oxidation, Thermal oxidative stability, Fat-containing food products, Fats and oils, Shelf-life prediction</p>
Article Details
1. Introduction
Consumption of foodstuffs keeps increasing as a result of the rise in human population of the world. So, consumption of vegetable oil, which is one of the basic components of foodstuffs has been rising rapidly. With the increase in the need for vegetable oil in the food industry, it has been emerged to carry out operations in this direction with the aim of preserving the existing quality without deterioration until the vegetable oil reaches the consumers from the production stage. The big quality problem in vegetable oil industry is Lipid Oxidation (LO), which gives rise to the existence of unsatisfactory and toxic compounds in the relevant products [1].
Some tests have been arisen for the determination of lipid oxidation in the selected oil at accelerated conditions [2, 3, 4]. A thermal analysis method known as Differential Scanning Calorimetry (DSC) is a more than fifty year-old method to assess the thermal oxidation process in lipid containing products [5, 6]. Active Oxygen Method (AOM) is another way for measuring the resistance of the fat-containing sample to oxidation [7, 8]. Oxygen uptake method (oxydograph) method has been also applied to anlayse the oil stability [9]. Thermogravimetric analysis (TGA) might be utilized to evaluate the oxidative stability in the fats and oils by detecting the related sample’s mass change through thermal degradation [10]. Fourier transform infrared spectra (FTIR) might also be evaluated for detection of the decomposition of the oxidative stability in fats and oils [11]. Schaal oven is also known as another accelerated assay for measuring the stability of oils [12]. Rancimat is an easy test method, including parameters such as amount of sample, air flow rate and temperature [13]. It requires no extra analyses such as titrations with too much time and chemical consumptions [14].
2. Lipid Oxidation
Deteriorative intermediates of lipid oxidation has adverse effect on shelf-life and characteristics (deterioration some physical properties such as of taste, color and odour) of lipid-containing food products [15]. Formation of this hazardous intermediate products in the concerning food is also inconvenient for the health of consumers. The relavant oxidation process is attributable to 2 structures of oxygen, which are singlet (1O2) and triplet (3O2) oxygens [16]. Those species are also known as Reactive Oxygen Species [17].
Figure 1 represent the 3 stages (initiation, propagation and termination) of LO process. RH represents the fatty acids/acylglycerols in fat-containing food product oil, while R is the lipid alkyl occured in the initiation stage depending on the food processing, transportation and preservation conditions [18]. Later, R· forms ROO· (lipid peroxy radical) by means of the reaction with triplet oxygen. This reactive species also reacts with the hydrohen of another RH, leading to generation of anaother R·. In the end of the related chain reactions, undesired and toxic compounds such as aldehydes and ketones, hydrocarbons, organic acids, volatile and polymeric compounds [3]. In the termination step, the reaction is terminated after lipid alkyl radicals react with each other [16].
3. Prediction of Shelf-life
It is well known that peroxide value (PV) is usually used as quality parameter for primary lipid oxidation, but if oil goes rancid further to secondary oxidation, PV is not necessary indicating oxidation status. Actually, PV is a measure of the extent of primary oxidation reactions but primary reactions do not prevent rancidity development but they are just the early reactions of lipids oxidation [19]. In order to avoid such inconsistencies, several oxidation tests have been emerged to comprehend the oxidation in the relevant products under several conditions. On the other hand, kinetics and thermodynamic factors are necessary for prediction of the lipid oxidation in oils under several conditions for composing better preparations regarding quality indicators [20]. The time for the resistance of the oil againt to oxidation is expressed as induction time (IT), which is utilized as an indicator for oxidative stability of fat-containing foods [21].
The following model describes the relationship between the induction time (stability of the oil against oxidation) and temperature:

t= Temperature (ºC)
a, b= Coefficients of the Eq.(1)
Arrhenius equation is used to define the robust relationship between the reaction rate constant (k) and the temperature in lipids:

k= Reaction rate constant (h-1)
Ea= Energy for activation (kJ mol-1)
A= Frequency factor
R= Universal gas constant (J mol-1 K-1)
T= Absolute temperature (K)
Actually, k is stated with the reciprocal of the induction time, since lipid oxidation in fats is assumed as first-order kinetic reaction [20]. Thermodynamics of a chemical process is a must to have knowledge about the nature of a system. Using Activated Complex approach, enthalpy (?Hº) and entropy (?Sº) were derived from the Eyring equation:
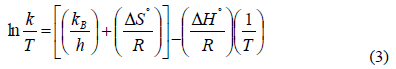
Where kB is Boltzmann (1.38065x10-23 J K-1) constant, and and h is known as Planck’s (6.62608x1034 J s) constant. One of the fundamental equations of thermodynamics is applied to calculate the change of Gibbs free energy (?Gº, kJ mol-1) through enthalpy change, the universal gas constant and absolute temperature:

Thermodynamic parameters obtained from Eqs.(3) and (4) are necessary in order to decide if the chemical reaction will be spontaneous or non-spontaneous, exergonic or endergonic, and exothermic or endothermic in the future.
3.1 Active oxygen method
Active oxygen method (AOM) is an option to identify the oil oxidation stability. A specific amount of sample is exposed to air at an elevated temperature. The sample is received regularly from the analyzed substance to measure the peroxide value. The time required to attain a certain amount of peroxide value is accepted as an indicator for the oxidative stability [9]. However, it has many disadvantageous such as being labor-intensive and costly due to the fact that AOM contains analyses such as the peroxide titration with too much time and chemical consumptions [8]. Therefore, the relevant method has not been applied into the fats and oils recently, although the literature has been reviewed thoroughly for the last 5 years.
3.2 Schaal oven test
Schaal oven test is accepted as the easiest method among the accelerated oxidation tests since certain amount of oil sample is heated in an oven at aroun 70 °C [9]. However, it requires extra analytical methods such as PV, p-anisidine value and iodine value determinations [22]. After treated with rosemary extract, cottonseed, soybean and rice bran oils were analysed with Schaal oven test at 62 °C by measuring their PV, total phenolic and tocopherol contents, antioxidant activity and fatty acid concentration [1]. Thermal oxidative stability of refined palm olein enrihed with natural antioxidants was also assessed by Schaal oven test by measuring its total oxidation, peroxide, iodine, p-anisidine and thiobarbituric acid values for 30 days [22, 23]. Schaal oven test was also performed by Souza et al. , where chia oil was monitored with respect to thermal oxidation according to its fatty acid and α-linolenic change [24]. Oxidation process of sunflower oil enriched by cold-pressed black cumin oil was investigated by variation in PV, conjugated dienes and trienes, tocopherols, tyhymoquinone and volatile compounds [25]. Moringa oil was monitored depending on its structure variation due to the thermal oxidative degradation through infrared spectra [26]. Several edible oils were compared depending on their thermal oxidative stability by measuring their peroxide and anisidine values under Schaal oven test conditions [27]. In order to see the effect of lycopene on the quality of walnut oil, Schaal ovent test was applied by measuring its antioxidant activity, total phenolic ingredient, PV, acid value and fatty acid [28]. Recently, Kiralan et al. used this method for the observation of thermal oxidative stability of grape, flax and black cumin seed oils by measuring the PV and conjugated dienes [29].
3.3 Fourier transform infrared spectroscopy
Fourier transform infrared (FTIR) spectroscopy has been proposed to be an alternative in deteriation studies of fat-containing food products with its advantegous as an easy, rapid and precise [30]. Some specific peaks can be examined during thermal oxidation process. Hu et al. used FTIR spectroscopy combined with infrared quartz cuvette to observe the oxidation in several edible oils (rapeseed, cottonseed, walnut, sesame, linseed, sunflower and soybean) by determination of acid value [31]. Shang et al. also used FTIR with infrared quartz cuvette to detect the oxidation process in several oil samples by identification of PV [32]. Canola oil was also investigated with FTIR regarding oxidative stability to comprehend the effect of heating and frying [33]. Heating (conventional and microwave) effect on the thermal deterioration of corn and soybean oils was monitored by peak changes infrared spectra of FTIR, proving the degradation of the products due to the secondary oxidation compounds [34]. Corn, sunflower, colza and a mixture of frying oils were studied by FTIR coupled with attenuated total reflectance (ATR) with the assistance of multivariate curve resolutionalternative least square (MCR-ALS) [35]. It was also proposed as a cost effective method as well as including no time-consuming sample preparation stage. FTIR technique together with a mesh cell was applied to determine the change of some functional groups during the lipid oxidation in some oils under ambient storage conditions [36]. Tena et al. also used similar technique to study the stability of virgin olive oil [37].
3.4 Thermogravimetric analysis
Thermal deterioration of the oils due to the lipid oxidation can be also examined by thermogravimetric analysis (TGA). Oxidation process in the product is monitored by means of weight change according to the oxygen taking and thermally deterioration [10, 38]. Gao and Birch suggested TGA as a relatively simple and time-saving method for the prediction of oxidation initiation in flax, hemp, and canola seed oils [39]. Shelf-life of hoki oil was estimated as 0.56 by using TGA through Arrhenius extrapolation, while it was predicted as 1.39 years by DSC [10]. Li et al. exploited TGA to compare the thermal oxidation stabilities of several vegetable oils such as palm, rapeseed, sunflower and linseed oils [40]. Alzate Arbeláez et al. studied the thermal stability of Lecythis tuyrana oil by means of TGA [41].
3.5 Differential scanning calorimetry
Differential scanning calorimetry (DSC) has been emerged to measure the stability of the fat-containing food against oxidation. It is a thermal analysis method with advantages such as efficient sensitivity, fastness and low sample requirements [21]. This accelerated method also allows to track the thermal activity in oxidation process continuously consideering the other methods [42].
It also required less stability measurement period comparing to Rancimat method. Similarly, Ramezan et al. compared the findings of Rancimat with that of DSC for 8 oil types such as sunflower, canola, palm, soybean, maize, peanut, sesame and coconut oils oxidized at 110, 120, 130 and 140 C [43]. They also recommended DSC as an alternative accelerated method with its advantegous (as mentioned above) over Rancimat. By using different heating rates (5, 7.5, 10, 12.5 and 15 C per minute), refined palm, olive, grapeseed, sunflower, corn, soybean, safflower and sesame oils were heated at 100-400 C by means of DSC [6]. Kinetics of the lipid oxidation occured in the selected oils were investigated by means of reaction rate constant and activation energy parameters. Activation energy changed depending on the compositon of the oils Tengku-Rozaina and Birch measured the stabilitiesof hoki and tuna oils against oxidation at 80 C [10]. Prediction of shelf-lifes of the products were conducted through Arhenius model. Srivastava et al. reported the thermodynamic structure of virgin coconut oil blended with different oils (refined soyabean and refined safflower oils) depending on the findings of DSC [44]. Belayneh et al. investigated Camelina seed oil to comprehend the effect of extraction method (cold press, soxhlet and supercritical CO2 extractions) on oxidative stability of the oil [45]. Symoniuk et al. monitored the thermal oxidation of some selected cold-pressed oils by DSC [46]. Recently, Echium oil has been studied to observe the effect of rosemary extract and hydroxytyrosol on the stability of the oil towards oxidation [47].
3.6 Rancimat test
Rancimat is an easy test method, including parameters such as amount of sample, air flow rate and temperature [13]. It requires no extra analyses such as titrations with too much time and chemical consumptions [14]. Sahin et al. performed Rancimat test to define the influences of olive leaf and lemon balm extracts on the shelf-life of corn oil [48]. Sahin et al. also reported the stability of virgin olive oil against oxidation after they enriched the oil with olive leaf extract through different methods [49, 50]. Similarly, they exposed the oil samples to air at a flow rate of 20 L/h for the Rancimat accelerated conditions (130°C). The same conditions were also applied to sunflower oil to assess the effect of olive leaf extract on the oil stability to oxidation [51].
Kinetics and thermodynamics factors are necessary for prediction of the lipid oxidation in oils under several conditions for composing better preparations regarding quality indicators [20]. However, the kinetics data obtained by rancimat method to assess the oxidative stability in vegetable oils are scarce. Farhoosh and Hoseini-Yazdi reported the olive oil oxidation process regarding kinetics studies achieved by accelerated rancimat conditions (100-130ºC) [4]. Upadhyay and Mishra enriched the sunflower oil with sage extract, and investigated the kinetic and thermodynamic parameters of the lipid oxidation to comprehend the nature of the process [52]. The fact of lipid oxidation occured in vegetable oils is distinctive for each system. Therefore, Arrhenius equation has to be reproduced for every oil system to discriminate the characteristics of the related products [20]. Elhussein et al. identified the kinetic and thermodynamic parameters of sesame oils of different origins (Turkey, Yemen and Sudan) by using Rancimat test under accelaretad conditions such as 110, 120, 130 and 140ºC [53]. KurtulbaS et al. also carried out similar study to evaluate the nature of the lipid oxidation process in cottonseed oil treated with phytonutrients (gallic acid, rutin and carotenoid) [54]. Recently, kinetics of the lipid oxidation in sunflower and sesame oils were calculated through Arrhenius model under Rancimat accelerated conditions [55].
4. Concluding Remarks
Table 1 summarizes the stability measurement tests of
several edible oils and fats against oxidation. Quality of the fat-containing food product might be monitored by the proposed methods produced from the relevant tests. However, it is not a good way to state which is the best method after mentioning the advantages and disadvantages of these measurement methods. Therefore, the results of multiple test methods should be given comparatively in an investigation of thermal oxidative stability. Kinetic and thermodynamic information should be calculated in the light of data produced from the selected methods. In this way, formulation of the fat-containing food product should be improved after having a provision on the nature of the complex lipid oxidation.

Figure 1: Stages of lipid oxidation process occured in fat-containin food products.
Table 1: Summary of the reported studies on oxidative stability of several edible oils and fats.
Acknowledgement
The authors thank the Research Fund of Istanbul University for financial support for this research project (Project No: BEK-2017-26410).
Disclosure Statement
No potential conflict of interest was reported by the author.
References
- Yang Y, Song X, Sui X, et al. Rosemary extract can be used as a synthetic antioxidant to improve vegetable oil oxidative stability. Ind Crops Prod 80 (2016): 141-147.
- Velasco J, Andersen ML, Skibsted LH. Evaluation of oxidative stability of vegetable oils by monitoring the tendency to radical formation. A comparison of electron spin resonance spectroscopy with the Rancimat method and differential scanning calorimetry. Food Chem 85 (2004): 623-632.
- Ostrowska-Ligeza E, Bekas W, Kowalska D, et al. Kinetics of commercial olive oil oxidation: Dynamic differential scanning calorimetry and Rancimat studies. Eur J Lipid Sci Technol 112 (2010): 268-274.
- Farhoosh R, Hoseini-Yazdi SZ. Evolution of Oxidative Values during Kinetic Studies on Olive Oil Oxidation in the Rancimat Test. J Am Oil Chem Soc 91 (2014): 281-293.
- Chiavaro E. Differential Scanning Calorimetry?: Applications in Fat and Oil Technology, n.d.
- Qi B, Zhang Q, Sui X, et al. Differential scanning calorimetry study-Assessing the influence of composition of vegetable oils on oxidation. Food Chem 194 (2016): 601-607.
- AktaS N, UzlaS?r T, Tunçil YE. Pre-roasting treatments significantly impact thermal and kinetic characteristics of pumpkin seed oil. Thermochim Acta 669 (2018): 109-115.
- Läubli MW, Bruttel PA. Determination of the oxidative stability of fats and oils: Comparison between the active oxygen method (AOCS Cd 12-57) and the rancimat method. J Am Oil Chem Soc 63 (1986): 792-795.
- Velasco J, Dobarganes C. Oxidative stability of virgin olive oil. Eur J Lipid Sci Technol 104 (2002): 661-676.
- Tengku-Rozaina TM, Birch EJ. Thermal oxidative stability analysis of hoki and tuna oils by Differential Scanning Calorimetry and Thermogravimetry. Eur J Lipid Sci Technol 118 (2016): 1053-1061.
- Guillén MD, Cabo N. Fourier transform infrared spectra data versus peroxide and anisidine values to determine oxidative stability of edible oils. Food Chem 77 (2002): 503-510.
- Liang C, Schwarzer K. Comparison of four accelerated stability methods for lard and tallow with and without antioxidants. J Am Oil Chem Soc 75 (1998): 1441-1443.
- Farhoosh R. The Effect of Operational Parameters of the Rancimat Method on the Determination of the Oxidative Stability Measures and Shelf-Life Prediction of Soybean Oil. J Am Oil Chem Soc 84 (2007): 205-209.
- Hasenhuettl GL, Wan PJ. Temperature effects on the determination of oxidative stability with the metrohm rancimat. J Am Oil Chem Soc 69 (1992): 525-527.
- Johnson DR, Decker EA. The Role of Oxygen in Lipid Oxidation Reactions: A Review. Annu Rev Food Sci Technol 6 (2015): 171-190.
- Choe E, Min DB. Mechanisms and Factors for Edible Oil Oxidation. Compr Rev Food Sci Food Saf 5 (2006): 169-186.
- Lee J, Koo N, Min DB. Reactive Oxygen Species, Aging, and Antioxidative Nutraceuticals. Compr Rev Food Sci Food Saf 3 (2004): 21-33.
- Van Dyck S. The impact of singlet oxygen on lipid oxidation. Lipid Technol 19 (2007): 278-280.
- Adhvaryu A, Erhan SZ, Liu ZS, et al. Oxidation kinetic studies of oils derived from unmodified and genetically modified vegetables using pressurized differential scanning calorimetry and nuclear magnetic resonance spectroscopy. Thermochim Acta 364 (2000): 87-97.
- Tan CP, Che Man YB, Selamat J, et al. Application of arrhenius kinetics to evaluate oxidative stability in vegetable oils by isothermal differential scanning calorimetry. J Am Oil Chem Soc 78 (2001): 1133.
- Pardauil JJR, Souza LKC, Molfetta FA, et al. Determination of the oxidative stability by DSC of vegetable oils from the Amazonian area. Bioresour Technol 102 (2011): 5873-5877.
- Djikeng FT, Hilaire M Womeni, Enti Anjaneyulu, et al. Effects of natural antioxidants extracted from Cameroonian ginger roots on the oxidative stability of refined palm olein. European Food Research and Technology 244 (2018): 1015-1025.
- Womeni HM, Tonfack Djikeng F, Iruku NSSP, et al. Valorization of soursop flowers (Annona muricata L.) as potent source of natural antioxidants for stabilization of palm olein during accelerated storage. Food Sci Nutr 4 (2016): 802-810.
- Souza AL, Martínez FP, Ferreira SB, et al. A complete evaluation of thermal and oxidative stability of chia oil. J Therm Anal Calorim 130 (2017): 1307-1315.
- Kiralan M, UlaS M, Özaydin A, et al. Blends of Cold Pressed Black Cumin Oil and Sunflower Oil with Improved Stability: A Study Based on Changes in the Levels of Volatiles, Tocopherols and Thymoquinone during Accelerated Oxidation Conditions. J Food Biochem 41 (2017): 12272.
- Duarte AM, Aquino JS, Queiroz N, et al. A comparative study of the thermal and oxidative stability of moringa oil with olive and canola oils. J Therm Anal Calorim 134 (2018): 1943-1952.
- Maszewska M, Florowska A, D?u?ewska E, et al. Oxidative Stability of Selected Edible Oils. Molecules 23 (2018): 1746.
- Xie C, Ma ZF, Li F, et al. Storage quality of walnut oil containing lycopene during accelerated oxidation. J Food Sci Technol 55 (2018): 1387-1395.
- Kiralan M, Çalik G, Kiralan S, et al. Stability and volatile oxidation compounds of grape seed, flax seed and black cumin seed cold-pressed oils as affected by thermal oxidation. Grasas y Aceites 70 (2019): 295.
- Rohman A, Man YBC. Fourier transform infrared (FTIR) spectroscopy for analysis of extra virgin olive oil adulterated with palm oil. Food Res Int 43 (2010): 886-892.
- Hu K, Huyan Z, Geng Q, et al. Rapid Determination of Acid Value of Edible Oils via FTIR Spectroscopy Using Infrared Quartz Cuvette. J Oleo Sci 68 (2019): 121-129.
- Shang J, Wu X, Hu K, et al. A simple and practical method to determine peroxide values in edible oils via infrared quartz cuvette-based Fourier transform infrared spectroscopy. Anal Methods 10 (2018): 3675-3679.
- Naz S, Saeed R. OXIDATIVE STABILITY OF CANOLA OIL BY PHYSICO-CHEMICAL ANALYSIS AND FT-IR SPECTROSCOPY. Asian J Pharm Res Dev 6 (2018): 9-15.
- Saeed R, Naz S, Naz S. Effect of heating on the oxidative stability of corn oil and soybean oil. Grasas y Aceites 70 (2019): 303.
- Mahboubifar M, Hemmateenejad B, Javidnia K, et al. Evaluation of long-heating kinetic process of edible oils using ATR–FTIR and chemometrics tools. J Food Sci Technol 54 (2017): 659-668.
- Xu L, Yu X, Liu L, et al. A rapid method for evaluating the edible oil oxidative stability during ambient storage by FTIR spectroscopy using a mesh cell. Anal Methods 8 (2016): 5117-5122.
- Tena N, Aparicio R, García-González DL. Virgin olive oil stability study by mesh cell-FTIR spectroscopy. Talanta 167 (2017): 453-461.
- Marleen van Aardt, Susan E Duncan, Timothy E Long, et al. Effect of Antioxidants on Oxidative Stability of Edible Fats and Oils: Thermogravimetric Analysis. J Agric Food Chem 52 (2004): 587-591.
- Gao F, Birch J. Oxidative stability, thermal decomposition, and oxidation onset prediction of carrot, flax, hemp, and canola seed oils in relation to oil composition and positional distribution of fatty acids. Eur J Lipid Sci Technol 118 (2016): 1042-1052.
- Li J, Liu J, Sun X, et al. The mathematical prediction model for the oxidative stability of vegetable oils by the main fatty acids composition and thermogravimetric analysis. LWT 96 (2018): 51-57.
- Alzate Arbeláez AF, Cogollo Pacheco A, Rojano B. Composition, antioxidant activity, thermal and oxidative stability of Lecythis tuyrana oil. J Food Nutr Res 57 (2018): 87-97.
- Grajzer M, Prescha A, Korzonek K, et al. Characteristics of rose hip (Rosa canina L.) cold-pressed oil and its oxidative stability studied by the differential scanning calorimetry method. Food Chem 188 (2015): 459-466.
- Ramezan Y, Bahmaei M, Givianrad MH. The Application of Differential Scanning Calorimetry As a Mean to Determine the Oxidative Stability of Vegetable Oils and its Comparison with Rancimat Natural Antioxidants View project Effect of Additives on Characterization and Photocatalytic Activity of Tio 2 /ZnO Nanocomposite Prepared via Sol-Gel Process View project. Artic Orient J Chem 31 (2015): 1389-1394.
- Srivastava Y, Semwal AD, Sajeevkumar VA, et al. Melting, crystallization and storage stability of virgin coconut oil and its blends by differential scanning calorimetry (DSC) and Fourier transform infrared spectroscopy (FTIR). J Food Sci Technol 54 (2017): 45-54.
- Belayneh HD, Wehling RL, Cahoon EB, et al. Effect of Extraction Method on the Oxidative Stability of Camelina Seed Oil Studied by Differential Scanning Calorimetry. J Food Sci 82 (2017): 632-637.
- Symoniuk E, Ratusz K, Ostrowska-Lig?za E, et al. Impact of Selected Chemical Characteristics of Cold-Pressed Oils on their Oxidative Stability Determined Using the Rancimat and Pressure Differential Scanning Calorimetry Method. Food Anal Methods 11 (2018): 1095-1104.
- Bañares C, Martin D, Reglero G, et al. Protective effect of hydroxytyrosol and rosemary extract in a comparative study of the oxidative stability of Echium oil. Food Chem 290 (2019): 316-323.
- Sahin S, Bilgin M, Say?m E, et al. Effects of natural antioxidants in the improvement of corn oil quality: olive leaf vs. lemon balm. Int J Food Sci Technol 52 (2017): 374-380.
- Sahin S, Say?m E, Bilgin M. Effect of olive leaf extract rich in oleuropein on the quality of virgin olive oil. J Food Sci Technol 54 (2017): 1721-1728.
- Sahin S, Sevgen S, Samli R. Estimation of quality parameters in virgin olive oil treated with olive leaf extract: application of artificial neural networks. Chem Pap 73 (2019): 1189-1197.
- Sahin S, Sayim E, Samli R. Comparative study of modeling the stability improvement of sunflower oil with olive leaf extract. Korean J Chem Eng 34 (2017): 2284-2292.
- Upadhyay R, Mishra HN. Multivariate Analysis for Kinetic Modeling of Oxidative Stability and Shelf Life Estimation of Sunflower Oil Blended with Sage (Salvia officinalis) Extract Under Rancimat Conditions. Food Bioprocess Technol 8 (2015): 801-810.
- Elhussein E, Bilgin M, Sahin S. Oxidative stability of sesame oil extracted from the seeds with different origins: Kinetic and thermodynamic studies under accelerated conditions. J Food Process Eng 41 (2018): e12878.
- KurtulbaS E, Bilgin M, Sahin S. Assessment of lipid oxidation in cottonseed oil treated with phytonutrients: Kinetic and thermodynamic studie. Ind Crops Prod 124 (2018): 593-599.
- Ghosh M, Upadhyay R, Mahato DK, et al. Kinetics of lipid oxidation in omega fatty acids rich blends of sunflower and sesame oils using Rancimat. Food Chem 272 (2019): 471-477.


 Impact Factor: * 3.8
Impact Factor: * 3.8 Acceptance Rate: 77.96%
Acceptance Rate: 77.96%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 