Nutritional and functional properties of flour and protein isolates from germinated Solojo Cowpea Vigna unguiculata (l.) Walp.
Olubamike Adetutu Adeyoju1, Kayode Oyebode Adebowale2, Bamidele Iromidayo olu-owolabi3, Henry Okwudili Chibudike4*, and Eunice Chinedum Chibudike5
1Production, Analytical and Laboratory Management, Federal Institute of Industrial Research, Oshodi, F.I.I.R.O., Lagos-Nigeria
2Department of Chemistry, Industrial Unit, University of Ibadan, Ibadan-Nigeria
3Department of Chemistry, Analytical Unit, University of Ibadan, Ibadan-Nigeria
4Chemical, Fiber and Environmental Technology Department, Federal Institute of Industrial Research, Oshodi, Lagos-Nigeria
5Planning, Technology Transfer and Information Management, Federal Institute of Industrial Research, Oshodi., F.I.I.R.O., Lagos-Nigeria
*Corresponding Author: Henry Okwudili Chibudike, Chemical, Fiber and Environmental Technology Department, Federal Institute of Industrial Research, Oshodi, Lagos-Nigeria
Received: 28 May 2021; Accepted: 05 June 2021; Published: 29 June 2021
Article Information
Citation: Olubamike Adetutu Adeyoju, Kayode Oyebode Adebowale, Bamidele Iromidayo olu-owolabi, Henry Okwudili Chibudike, Eunice Chinedum Chibudike. Nutritional and functional properties of flour and protein isolates from germinated Solojo Cowpea Vigna unguiculata (l.) Walp. Journal of Food Science and Nutrition Research 4 (2021): 161-174.
DOI: 10.26502/jfsnr.2642-11000069
View / Download Pdf Share at FacebookAbstract
Nutritional and functional properties of flours derived from two (2) varieties of cowpea, Dark-Ash Solojo (DAS) and Brown Solojo (BS) were studied before and after dehulling of the germinated seeds. Ungerminated seeds were used as the control. Protein isolates from dehulled defatted solojo cowpea seeds were prepared using isoelectric (CPIA) procedure. Dehulled samples had a higher protein solubility compared with germinated and control samples. Both varieties of cowpea (DAS and BS) investigated were soaked in distilled water and germinated at varying periods i.e. 0, 6, 24, 36, 48 and 72hrs. Protein isolates were obtained from the treated and processed samples by isoelectric precipitation method which was subsequently followed by proximate and anti-nutritional analyses. Functional properties were also analysed which include Water Absorption Capacity (WAC) and Oil Absorption Capacity (OAC) of the protein isolates using standard methods. Amino acids and molecular weight of the protein isolates were determined by amino acid analyser and sodium-dodecyl-sulphate-polyacrylamide-gel-electrophoresis. Surface morphology, functional group and thermal properties were determined for protein isolates by scanning electron microscopy, Fourier Transform Infrared (FTIR) spectrometry and differential scanning calorimetry, respectively. Data were analysed by descriptive statistics and ANOVA at α0.05. The moisture content, crude protein, crude fat, crude fibre and total ash of DAS ranged from 9.00-11.40, 24.82-31.00, 1.56-2.66, 1.43-1.67, and 3.20-4.14%, respectively; while those of BS flours ranged from 7.10-9.50, 24.90-30.14, 1.17-2.37, 1.06-1.52 and 3.05-3.93%, respectively. The protein contents for DAS were 81.57±0.53, 86.44±0.84, 89.39±1.51, 90.23±0.53, 91.81±0.77 and 94.85±0.86, while for BS were 84.39±0.39, 85.44±0.56, 90.05±0.10, 90.47±0.89, 92.78±0.28 and 95.81&plusm
Keywords
<p>Solojo Cowpea, Underutilised legumes, Protein isolate, Antinutrients properties, DAS, BS</p>
Article Details
1. Introduction
Protein malnutrition is one of the major problems in developing parts of the world in addition to the increase and escalating demand for protein due to world population explosion and growth of the food industry, cumulating in increasing demand for animal protein thereby putting pressure on the conventional animal sources. Some health issues like kwashiorkor is prevalent in children owing to protein deficiency, whereas in adults, results in reduced work capacity. Bridging the gap between increased food consumption and production is one of the most challenging tasks in the developing parts of the world. Most folks in the developing countries rely upon grain legumes as major sources of dietary protein, because, animal proteins are expensive. Various research efforts are now on going on the application of non-animal proteins for the evolution of innovative nutritional produce or substitute against high- priced animal proteins. Collaborative efforts towards exploiting the capacity of legumes to curtail the complication of malnutrition (protein) in Africa and to reduce the pressure on the commonly consumed legumes is on-going. Legumes not only possess significant protein content but also essential protein character, research has also shown their capacity to oppose the action of malnutrition especially in emerging nations by including them in the everyday regime. Seed proteins should in addition to providing essential amino acid have the useful properties required by industries producing food [1-5]. The drive to discover the physical, chemical and functional characteristics of these legume proteins can therefore not be compromised. The inherent characteristics of the protein which induces their usefulness in the food industry are functional properties which also control their behaviour in food structure during preparation, production, depository and management. The capability of legume protein to meet the recommended standard of functional properties as required by the food industry like, swelling, pasting, water absorption, emulsifying, gelation and foaming properties are vital in novel food product evolution. For legume protein to be effectively utilised, the varied functional and nutritional characteristics must be well channelled to the required demand of manufactured edible materials. This has been found to be challenging because many native proteins possess limited functionality, as a result of their stability which is affected by the application of heat, carbon-based solvents and proteolytic degradation, thus makes industrial applications of food proteins to be limited. Therefore, for a wide application of legume proteins, they must be converted to more stable forms [6-10]. The need for multiple functional food products or ingredient has compelled the food industries and researchers to develop means of modifying plant protein and endow them with desirable functional characteristics to meet the various needs. Functional properties are the physical and chemical characteristics of the specific protein influencing its behavior in food system during processing, storage, cooking and consumption. Examples of functional properties include bulk density, protein solubility, water and oil absorption capacity, emulsifying and foaming properties. The factors that affect the functional behavior of proteins in foods are their size, shape, amino acid composition and sequence, net charge, hydrophobicity, structure, molecular rigidity in response to external environment (pH, temperature, salt concentration) or interaction with other food constituents. This work therefore is designed to evaluate the ability of biochemical modification in enhancing the functional properties, and nutritive quality of Solojo protein. Solojo an underutilized legume commonly grown in the South-West region of Nigeria, will be biochemically modified for its possible industrial application through its functional properties [11,12].
2. Experimental Materials and Methods
2.1 Materials
The raw material investigated in this research study is Solojo Cowpea (Vigna unguiculata L.) which occur in two varieties i.e. Dark-ash solojo (DAS) and Brown Solojo (BS). These two underutilized varieties found in South-West region of Nigeria where they are called ‘Solojo’ were obtained from Bodija market in Ibadan, Western Nigeria. They were stored in polyethylene bags at room temperature (25-26oC).
2.2 Methods
The dehulled cowpea seeds were cleaned and screened to get rid of every irrelevant materials and unwholesome seeds. The Solojo seeds (DAS and BS) for germination were sterilized by soaking in 0.07% Sodium hypochlorite (Rumiyati et al., 2012) for 30 min, then rinsed thoroughly. The Solojo seeds were then immersed for 6h in distilled water at ambient temperature (1:10 w/v) (~25oC), then placed in a colander and germinated under subdued light in an open laboratory (Rusydi, 2011) for 0, 6, 24, 36, 48 and 72hrs. Other treated portions of the Solojo seeds (DAS and BS) were dehusked, dried, milled into flour and defatted. Protein was isolated by isoelectric precipitation method. Proximate, antinutritional analysis and functional properties [Water Absorption Capacity (WAC), Oil Absorption Capacity (OAC)] of the flours and protein isolates were determined by standard methods. Amino acids and molecular weight of the protein isolates were determined by amino acid analyzer and sodium-dodecylsulphate-polyacrylamide-gel-electrophoresis. Surface morphology, functional group and thermal properties were determined for protein isolates by scanning electron microscopy, Fourier Transform Infrared (FTIR) spectrometry and differential scanning calorimetry, respectively. Data were analysed by descriptive statistics and ANOVA at α0.05
2.3 Preparation of Flours
2.3.1. Raw flour: The grains were segregated to remove the spoilt ones; then dry dehulled with a mechanical dry dehuller (Fabricated in FIIRO), dried at 40oC and later milled dry to powder then sifted using 80 μm mesh. The flour was stored in flexible bags and preserved at 4oC preceding utilization in a refrigerator freezer. 6hour Soaked flour: The seeds were segregated to remove the unwholesome ones, then immersed for 6h in the ratio (1:10 w/v) (seed/water). The grains were then frozen to prevent germination from setting in, then the hull was removed manually, dried for 48 h at 40oC later milled dry to smooth powder prior to sieving using 80 μm mesh screen. The resulting flour was packaged in plastic pack and preserved in a fridge-freezer at 4oC pending utilization [13-17].
2.3.2 Germination of seed: This was implemented by the method of Mubarak with minor adjustment. The seeds for germination were disinfected by soaking in 0.07% Sodium hypochlorite for 30 mins, then, it was rinsed painstakingly. The Solojo seeds were then immersed for 6 h at ambient temperature in water in the ratio (1:10 w/v) (seed/water) (~25oC), then placed in a colander and germinated under subdued light in an open laboratory for various h, 24, 36, 48 and 72h. The process of germination was terminated by freezing, the seeds were manually dehulled, dried in a draught oven (Schutzart DIN EN 60529-IP 20. Memmert, Germany) at 40oC for 48h, cooled, milled and packaged in an air tight plastic bag in the refrigerator pending analysis.

Figure 1: Dark- ash Solojo Cowpea
Figure 2: Brown Solojo Cowpea

Figure 3: Germinated Dark- ash Solojo Cowpea Figure 4: Germinated Brown Solojo Cowpea
3. Results and Discussion
Results of the proximate composition of the seed flour and the protein isolates are presented in Table 1. The whole (WCF) and dehulled defatted (DDCF) cowpea seed flour contained 22.30%-26.73% protein, 2.10%-2.30% fat, 4.10%-1.02% fibre, 3.77%-3.87% ash and 60%-59% carbohydrates respectively (on dry weight basis) as major components.The data obtained is comparable to that reported by Sosulski et al., Abdalla et al. and Ragab et al. Protein isolates (CPIA and CPIB) showed 75% and 76% protein content and a decrease in carbohydrate content from 59.78% to 13%. The nutritional chemical analysis of both raw and germinated seed flours of full fat and defatted dark-ash and brown solojo cowpea [18-21].
Figure 5: Preparation of Beans Flour/Schematic representation
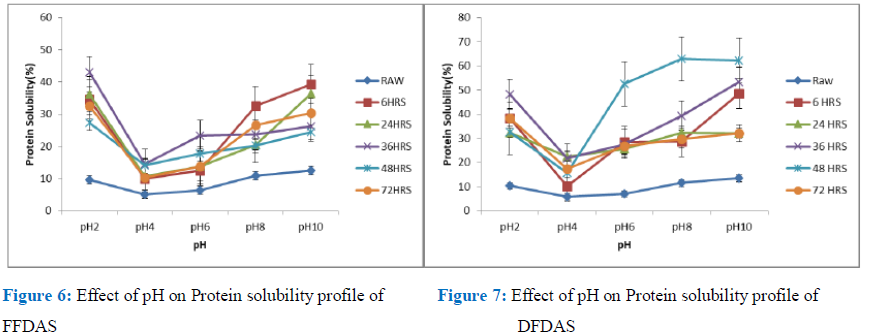
Figure 6: Effect of pH on Protein solubility profile of Figure 7: Effect of pH on Protein solubility profile of
FFDAS DFDAS
Protein solubility of flours and isolates at different pH are presented in figures 6-11. Results indicated the lowest solubility for all the flours and isolates germinated and control for both the DAS and BS, full fat, defatted and isolates were around pH 4. The maximum pH of solubility was pH 2 and pH10 for different treatments but on the whole, solubility is inversely proportional to the pH. Decrease in pH was observed from pH 2 till minimum value of the isoelectric point was reached, which is around pH 4 and pH 6. Solubility progressively went up as the pH increased till pH 10. Solubility also increased with germination. This rise in protein solubility was ascribed to change in structure and configuration of protein caused by the unfolding of the protein molecule [22-25].
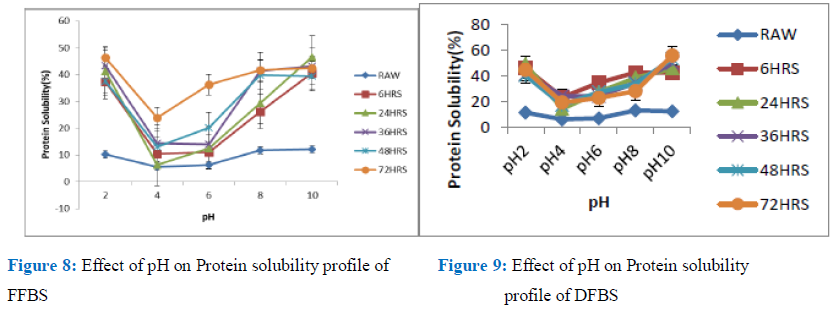
Figure 8: Effect of pH on Protein solubility profile of Figure 9: Effect of pH on Protein solubility
FFBS profile of DFBS
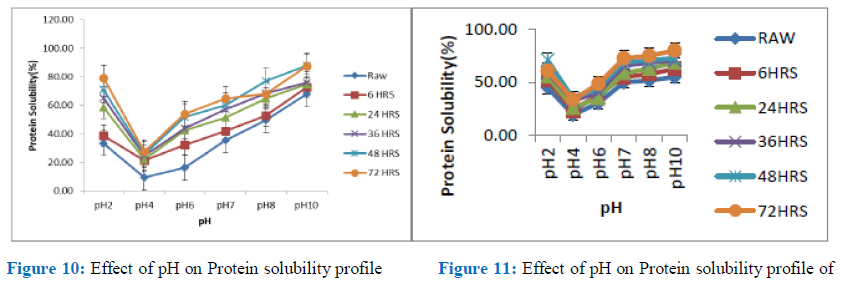
Figure 10: Effect of pH on Protein solubility profile Figure 11: Effect of pH on Protein solubility profile of BS Protein of DAS Protein isolate isolate
FFDAS- Full Fat Dark Ash Solojo Cowpea for germinated and non germinated at different times
DFDAS- Defatted Dark Ash Solojo Cowpea for germinated and non germinated at different times
FFBS- Full Fat Brown Solojo Cowpea for germinated and non germinated at different times
DFBS- Defatted Brown Solojo Cowpea for germinated and non germinated at different times
DAS- Dark-Ash Solojo Cowpea protein isolate for germinated and non germinated at different times
BS- Dark-Ash Solojo Cowpea protein isolate for germinated and non germinated at different times
The solubility profile of the cowpea flours and isolates, gave a curve with U-shape in the region pH 2- pH 10 which is close to what was obtained for many oil seeds and most vegetable legume protein, according to the observation of Lawal et al; Khalid and Elharadallou, for seeds such as, Mucuna bean, Jack bean, Bambara groundnut flour, African locust bean (albumin and globulin, full fat and defatted flours), Cowpea and lupin flour and isolate respectively. Solubility of protein at different pHs serves as a useful index of its behaviour in the food system, both for flour and the isolates; and also, the extent to which protein has been altered (denatured) as a result of heat or chemical treatment [26,27].
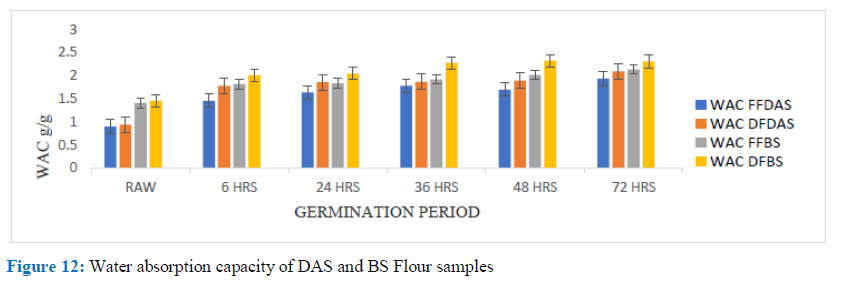
Figure 12: Water absorption capacity of DAS and BS Flour samples
The Water (H2O) Absorption Capacity (WAC) can be defined as the amount of water taken in by every gram of protein material. WAC represents the capability of a produce to interact with H2O in a situation where water is limited. WAC of flours (Full fat and Defatted) of DAS and BS Cowpea as well as the isolates is as shown in figures 12 and 13. Data showed that flours (full fat and defatted) had their WAC increasing with time of germination, the same was observed for the isolates of DAS and BS, with the germinated flour having better WAC than the non-germinated flour (0.95 to 1.94g/g for FFDAS and 0.94 to 2.10g/g for DFDAS; 1.41±0.02 to 2.14±0.02g/g for FFBS; 1.46±0.02 to 2.33±0.01g/g for DFBS; 2.27±0.06 to 3.41±0.03g/g for DAS isolate and 2.21±0.09 to 4.15±4.15g/g for BS isolate). This observation agreed with the findings of Akaerue and Onwuka, Murugkar et al. Desalegn, and others; with WAC ranging for various legumes between values of 0.89 to 8.10g/g, such as chickpea flour, pigeon pea, green gram, mung bean, lentils, African oil bean and so on [28].
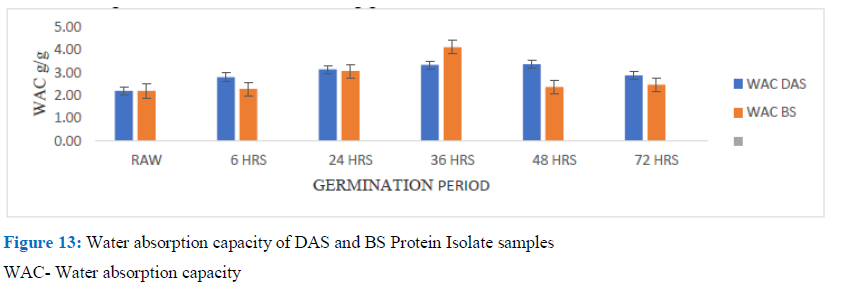
Figure 13: Water absorption capacity of DAS and BS Protein Isolate samples
WAC- Water absorption capacity
The results obtained for the germinated Solojo cowpea were found to fall within the reported range in literature. Yellavila et al. obtained a range 0.89 to 1.2g/g for five lima flour samples, these values were smaller than those obtained in this study for the germinated flour samples. Wisaniyasa et al. had an increase of WAC from 216.65±1.64 to 250.73±1.09% after 72h of germination, while Ojo et al. recorded a value of 1.84g/g and 1.31g/g for two varieties of Vigna subterranean. Similar values were also reported for sprout of amaranth flour and mucuna flour. The WAC of flours of protein seeds may be affected by shape and size of protein, steric factors, equilibrum of the water-loving-oil-loving amino acid of protein molecule [29].
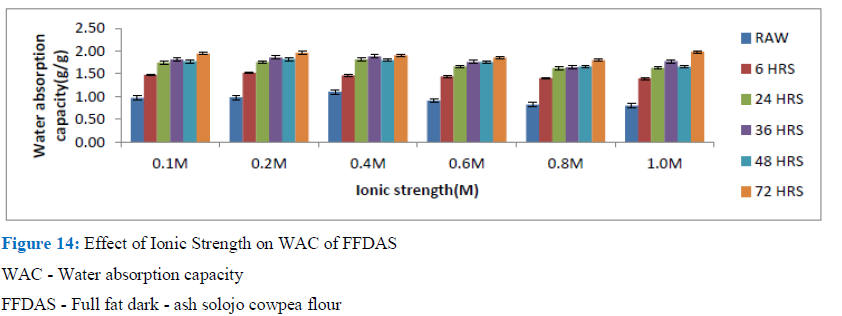
Figure 14: Effect of Ionic Strength on WAC of FFDAS
WAC - Water absorption capacity
FFDAS - Full fat dark - ash solojo cowpea flour
A commonly used component for enhancement of palatability, preservation of food, and improvement of handling in food industry is the common salt; but it brings about the alteration of protein functionality strongly. Figures 14-19, show the Ionic strength effect on WAC of both the full and defatted Dark -ash and Brown Solojo cowpea. The effect varied in the two flour varieties both with the full fat and defatted flours. Rise in NaCl concentration brought about corresponding increase in WAC generally for the FFDAS, as well as DFDAS samples for 6, 48 and 72h of the FFDAS and 6 and 36 h of DFDAS showing maximum increase in WAC as ionic strength increased to 0.2M.

Figure 15: Effect of Ionic Strength on WAC of DFDAS
WAC - Water absorption capacity
DFDAS - Defatted dark - ash solojo cowpea flour
Whereas, the raw, 24h, 36h of FFDAS and the raw, 24 h and 48h of DFDAS needed up to 0.4M concentration of the salt to achieve the highest WAC, beyond which any more rise in ionic strength brought about a lowering. The value ranged between 0.80 ±0.01 and 1.98 ± 0.02 g/g for FFDAS, while that of DFDAS ranged between 0.80 ± 0.02 and 2.10 ± 0.01 g/g. The 24, 36 and 72h germinated samples of FFBS only required 0.1M ionic strength to cause an increase in WAC of the sample, whereas the raw and 6h germinated samples as well as the DFBS samples experienced decrease in WAC as ionic strength increased to various concentration ranging between 0.1 M to 0.6M. Berhanu and Amare observed increase in WAC with salt of minimal ionic strength for the defatted flour of Brebra (Millettia ferruginea) seed [30]. This observed inclination may be due to charge masking as concentration increased, causing reduction in electrostatic interaction and hydration, but bringing about increasing hydrophobic interaction. Berhanu and Amare observed increase to 0.5% and 1% respectively for defatted brebra and defatted soya bean flour.
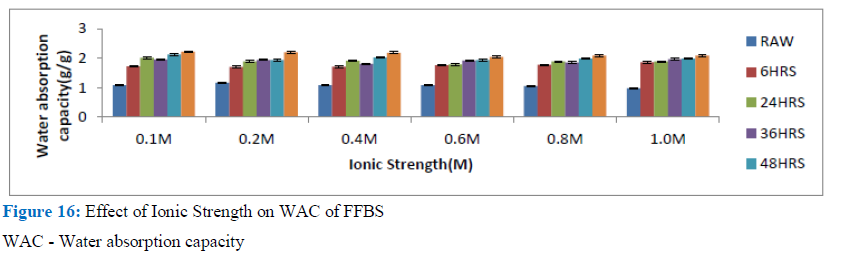
Figure 16: Effect of Ionic Strength on WAC of FFBS
WAC - Water absorption capacity
FFBS - Full fat brown solojo cowpea
This observation is also comparable to the findings of Lawal et al. for the albumin protein fraction, where they reported that, raising the salt concentration up to 0.4M caused an improvement in the water imbibing capability when compared with the control, but any more increment in ionic strength brought about reduction in the water absorption capacity ever more. In the case of the globulin fraction and flours, rise in ionic strength up to 0.2M brought about increase in WAC following which no more increase was observed [31].
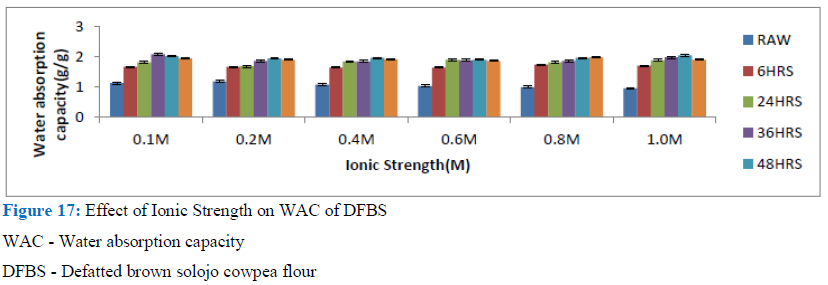
Figure 17: Effect of Ionic Strength on WAC of DFBS
WAC - Water absorption capacity
DFBS - Defatted brown solojo cowpea flour
A significant functionality trait in foods is its WAC, especially for dough forming foods like sausages and custards. The result suggested that DAS flour samples could be utilised as ingredient in the development of pulverized foods such as sausages. Likewise, it enables the addition to dough of more water in order to enhance the handling peculiarities by bakers and enable freshness to be maintained in bread. The presence of sodium chloride in trace level is required for some protein to be soluble, little salt has also been found to improve the solubility of protein, hence the observed improvement in the WAC but as concentration increases, it brings about a decrease in water holding capacity of protein. This happens by preventing the hydrophilic amino acids of protein from relating with water by competing with it for the available water. This is termed as charge screening.
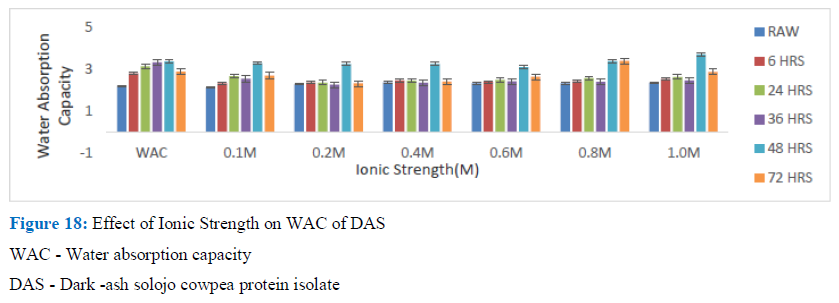
Figure 18: Effect of Ionic Strength on WAC of DAS
WAC - Water absorption capacity
DAS - Dark -ash solojo cowpea protein isolate
In the case of the isolates, generally it was observed that when NaCl was added, it brought about a reduction in the WAC of germinated DAS and BS cowpea when compared with that of the control. For the DAS cowpea after the initial drop usually there was a gradual increase in the observed WAC. The drop was between 0.1 M and 0.2M. The BS did not give a regular pattern. This observed effect of NaCl leading to decrease of WAC was also observed by Ivanova et al. for sun flower meal protein isolated by isoelectric point method where it was reported that addition of 0.25M of NaCl led to over threefold reduction of WAC when equated with that of the control. A related observation was made by Aremu, et al. for WAC of Kersting’s groundnut protein, with decrease up to a certain concentration and increased and decreased again. Ogungbenle likewise reported about decrease in WAC with increase in NaCl up to 10%, which was attributed to the nature of anion and cation present. Andualem and Gessesse suggested the decrease in WAC with increase in concentration of NaCl to be probably due to charge screening as concentration increased, which reduced electrostatic inter-reaction and hydration but improved hydrophobic interaction.
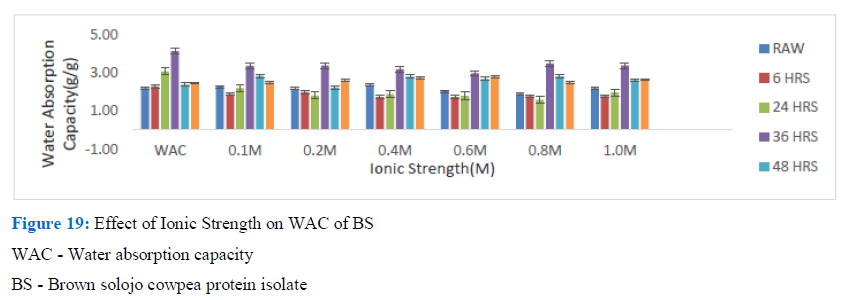
Figure 19: Effect of Ionic Strength on WAC of BS
WAC - Water absorption capacity
BS - Brown solojo cowpea protein isolate
The packed density for FFBS was 0.59 to 0.88 g/cm3 and that of DFBS was between 0.69 and 0.91 g/cm3.There was a general initial increase in BD with germination for the samples, both full fat flours, defatted flours and the protein isolates. Except for FFDAS samples, which showed decrease at 24hrs and then an increase occurred from 36 h. 24 h, 48 h LBD of FFDAS; 24 hBD of both the DAS and BS protein isolates all showed lower or same values as the raw. The reduction observed goes along with previous literature report by Chinma et al. for two varieties of Tigernut flour; and Desalegn, for chickpea flour. This could be as a result of decrease in heaviness. Factors affecting BD are, particle size, intensity of attractive interparticle forces and number of contact points. High bulk density also indicates greater compactness ability of the particles, this is because, particle size and bulk density have an inverse relationship. The high BD shows the flour will be very useful in food formulation for easier dispersion and reduced thickness of paste, which is very significant in the production of infant foods. Flours with low bulk density are said to be desirable for the preparation of weaning foods because they give reduced/ low paste thickness and viscosity on reconstitution. However, flours that have high bulk density, have been said to enhance fat absorption, even though this is not very good for weaning food, it is a very good property for flours used for baked and pastry products. The isolates had the bulk density varying between 0.36 to 0.50g/cm3 for the LBD of DAS and 0.47 to 0.59g/cm3 for PBD; while the BS isolate had for LBD 0.35 to 0.47g/cm3 and 0.45 to 0.59g/cm3 for PBD. Butt and Batool, had values of 0.71, 0.68, 0.55 and 0.53g/cm3 for cowpea, pea, mungbean and pigeon pea protein isolates respectively. Sibt-e-Abbas et al. obtained for two varieties of peanut, Golden 0.51±0.01g/cm3 and Bari 0.54±0.01g/cm3 which are comparable to our result. When compared with enzymatic hydrolysis, Wani et al. observed a decreased in BD with enzymatic hydrolysis using Indian black gram cultivars (Phaseolus mungo L.) with value range between 0.21- 0.17g/mL. Rajesh and Prakash also reported that bulk density of albumin fraction of lentil went down from 0.94g/mL to 0.88g/mL on hydrolysis. Their results were higher than that reported in this study.
5. Conclusion
This research work shows that biochemical modification (Germination/Malting/ Sprouting) had an enormous impact on the nutritional composition, functional properties, mineral bioavailability, anti-nutrient content and amino assay of Solojo bean, thus, it could be used as protein supplement in infant, young children and geriatric foods. Efforts should be increased to promote the cultivation, encourage the consumption and industrial application of this under-utilized legume by the Government, especially in the south-western region where it can survive the rain fall level. Large scale production of this legume which is gradually going into extinction should be encouraged in order to fight the menace of malnutrition in developing countries where animal protein price is exorbitant; This will ensure food security and also creation of jobs, because people can engage in different aspects of the production process and thereby reducing the rate of unemployment.
References
- American Association of Cereal Chemists (8th edtn), (2019).
- Abdalla MH, Elkhalifa AO, Eltinay AH (2001) Factors affecting protein extraction from cowpea. J Food Sci Technol 38: 532-533.
- Aghajanpour M, Reza -Nazer M, Obeidavi Z, et al. Functional foods and their role in cancer prevention and health promotion: a comprehensive review American journal for cancer Research 7 (2017): 740-769.
- Ahmed SH, Ahmed IAM, Eltayeb MM, et al. Functional Properties of Selected legumes flour as Influenced by pH. Journal of Agricultural Technology 7 (2011): 2091-2102
- Aiking H. Future protein supply. Trends in Food Science and Technology 22 (2011): 112-120.
- Ali NA, Ahmed SH, Mohamed EA, et al. Changes in functional properties by transglutaminase cross linking as a function of pH of legumes protein isolate. Innovative Romanian Food Biotechnology 7 (2010): 12-20
- Alashi A, Blanchard C, Mailer R, et al. Improving the emulsifying properties of canola meal protein isolate by enzymatic modification 17th Australian Research Assembly on Brassicas (ARAB) Wagga Wagga (2011).
- Barac M, Cabrilo S, Pesic M, et al. Functional Properties of Pea (Pisum sativum, L.) Protein Isolates Modified with Chyosin. International Journal of Molecular sciences 12 (2011): 8372-8387.
- Beuchat LR. Functional and electrophoretic characteristics of succinylated peanut flour proteins. J Agric Food Chem 25 (1977): 258-261.
- Aletor O, Ojelabi A. Comparative evaluation of the nutritive and functional attributes of some traditional nigerian snacks and oil seed cakes. Parkistan Journal of Nutrition 6 (2007): 99-103.
- Coffmann CW, Garciaj VV. Functional properties and amino acid content of a protein isolate from mung bean flour. Int J Food Sci Technol 12 (1977): 473-484.
- D’Astolfo DS, Pagliero RJ, Pras A, et al. Efficient Intracellular Delivery of Native Proteins. Cell 161 (2015): 674-690.
- Gaspar ALC, de Góes-Favoni SP. Action of microbial transglutaminase (MTGase) in the modification of food proteins: A Review. Food Chemistry 171 (2015) 315-322.
- Jahreis G, Brese M, Leiterer M. Legume flours: Nutritionally important sources of protein and dietary fiber. Ernahrungs Umschau 63 (2016): 36-42.
- Klupšaite D, Juodeikiene G. Legume: Composition, Protein Extraction and Functional Properties. A review. Chemine Technologija. Nutrition 1 (2015).
- Lawal OS, Adebowale KO. The acylated protein derivatives of Canavalis ensiformis (Jack bean). A study of functional characteristics. Food Technology 39 (2006): 918-929.
- MacDonald JI, Munch HK, Moore T, et al. One-step site-specific modification of native proteins with 2-pyridinecarboxyaldehydes. Nature Chemical Biology 11 (2015): 326-331.
- Marco-moles R, Hernando I, Perez- Munuera I. Influence of high-pressure homogenization (HPH) on the structural stability of an egg/dairy emulsion. Journal of Food Engineering 109 (2021): 652-658.
- Mirmoghtadaie L, Shojaee S, Seyede A, et al. Recent approaches in physical modification of protein functionality. Food Chemistry 199 (2016): 619-627.
- Mudryj AN, Yu N, Aukema HM. Nutritional and health benefits of pulses. Applied Physiology Nutrition and Metabolism 39 (2014): 1197-1204.
- Mune MAM, Minka SR, Mbome IL. Chemical composition and nutritional evaluation of a cowpea protein concentrate Global Advanced Research. Journal of Food Science and Technology 2 (2013): 035-043.
- Nahid AA, Salma A, ElShazali AM. Transglutaminase cross-link of legumes protein isolate: changes in functional properties as a function of pH. International Food Research Journal 17 (2010): 1011-1018.
- Ragab DDM, Babiker EE, Eltinay AH. Fractionation, solubility and functional properties of cowpea (Vigna unguiculata) proteins as affected by pH and/or salt concentration. J Food Chem 84 (2004): 207-212.
- Salma HA, Nahid AA, ElShazali AM, et al. Changes in the functional properties as a function of NaCl concentration of legumes protein isolate by transglutaminase cross linking. International Food Research Journal 17 (2010): 817-824.
- Shimelis EA, Rakshit SK. Effect of processing on antinutrients and in vitro protein digestibility of kidney bean (Phaseolus vulgaris L.) varieties grown in East Africa. Food Chemistry 103 (2007): 161-172.
- Sibt-e-Abbas M, Butt MS, Sultan MT, et al. Nutritional and functional properties of protein isolates extracted from defatted peanut flour. International Food Research Journal 22 (2015): 1533-1537.
- Singhal A, Can Karaca A, Tyler R, et al. Pulse Proteins: From processing to structure-function relationships (2016).
- Skylas DJ, Molloy MP, Willows RD, et al. Characterisation of Protein Isolates Prepared from Processed Mungbean (Vigna radiata) Flours. Journal of Agricultural Science 9 (2017): 12.
- Sosulski FW, Kasirye-Alemu EN, Sumner AK. Microscopic, nutritional and functional properties of cowpea flours and protein concentrates during storage. J Food Sci 52 (1987): 700-706.
- Wouters AGB, Rombouts I, Fierens E, et al. Relevance of the Functional Properties of Enzymatic Plant Protein Hydrolysates in Food Systems. Comprehensive Reviews in Food Science and Food Safety 15 (2016): 786-800.
- Zang X, Yue C, Wang Y, et al. Effect of limited enzymatic hydrolysis on the structure and emulsifying properties of rice bran protein. Journal of Cereal Science 12(2018).


 Impact Factor: * 3.8
Impact Factor: * 3.8 Acceptance Rate: 77.96%
Acceptance Rate: 77.96%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 