The Effect of Fusarium Oxysporum and Macrophomina Phaseolina on the Proximate Composition of Jatropha Curcas Seed a Biofuel Plant
Article Information
Isalar Oluwatoyin F1,2*, Ataga Anthony E2
1Department of Biological Sciences, Edwin Clark University, Kiagbodo, Delta State, Nigeria
2Department of Plant Science and Biotechnology, University of Port Harcourt, Rivers State, Nigeria
*Corresponding Author: Isalar Oluwatoyin F, Department of Biological Sciences, Edwin Clark University, Kiagbodo, Delta State, Nigeria
Received: 21 December 2018; Accepted: 31 December 2018; Published: 10 January 2019
Citation:
Isalar Oluwatoyin F, Ataga Anthony E. The Effect of Fusarium Oxysporum and Macrophomina Phaseolina on the Proximate Composition of Jatropha Curcas Seed a Biofuel Plant. Journal of Biotechnology and Biomedicine 2 (2018): 001-008.
View / Download Pdf Share at FacebookAbstract
A study was carried out on the effects of Fusarium oxysporum and Macrophomina phaseolina on the proximate compositions of Jatropha curcas seeds. The proximate composition of the various nutrient component (Dry matter, Moisture, Lipids, Free fatty Acid, Carbohydrates, Ash, Fibre, Crude protein and Amino acid) in both fungus-inoculated and uninoculated seeds at the incubation period of 7 days were measured by the methodology proposed by the Association Official Analytical Chemists [1]. The results obtained from each food component were subjected to statistical analysis using the Analysis of Variance (ANOVA). In the results of the proximate analysis, Fusarium oxysporum and Macrophomina phaseolina caused significant increase (P=0.05) in moisture content 7.0% and 6.0%, crude protein 2.1% and 1.7%, and 0.2% and 0.7% ash, respectively in seeds inoculated at room temperature (28 ± 2°C) for 7days when compared with uninoculated controls. There was a remarkable reduction of 11% and 10.5% in dry matter, 0.8% and 0.2% in carbohydrate, 5.0% and 3.3% in lipids, 2.6% and 5.7% in fibre content of the seeds inoculated with Fusarium oxysporum and Macrophomina phaseolina, respectively when compared with their controls. The results are discussed in relation to the changes in the nutritional composition of the seeds due to the activities of these pathogenic fungi.
Keywords
Jatropha curcas, Proximate Composition, Pathogenic fungi
Jatropha curcas articles Jatropha curcas Research articles Jatropha curcas review articles Jatropha curcas PubMed articles Jatropha curcas PubMed Central articles Jatropha curcas 2023 articles Jatropha curcas 2024 articles Jatropha curcas Scopus articles Jatropha curcas impact factor journals Jatropha curcas Scopus journals Jatropha curcas PubMed journals Jatropha curcas medical journals Jatropha curcas free journals Jatropha curcas best journals Jatropha curcas top journals Jatropha curcas free medical journals Jatropha curcas famous journals Jatropha curcas Google Scholar indexed journals Proximate Composition articles Proximate Composition Research articles Proximate Composition review articles Proximate Composition PubMed articles Proximate Composition PubMed Central articles Proximate Composition 2023 articles Proximate Composition 2024 articles Proximate Composition Scopus articles Proximate Composition impact factor journals Proximate Composition Scopus journals Proximate Composition PubMed journals Proximate Composition medical journals Proximate Composition free journals Proximate Composition best journals Proximate Composition top journals Proximate Composition free medical journals Proximate Composition famous journals Proximate Composition Google Scholar indexed journals Pathogenic fungi articles Pathogenic fungi Research articles Pathogenic fungi review articles Pathogenic fungi PubMed articles Pathogenic fungi PubMed Central articles Pathogenic fungi 2023 articles Pathogenic fungi 2024 articles Pathogenic fungi Scopus articles Pathogenic fungi impact factor journals Pathogenic fungi Scopus journals Pathogenic fungi PubMed journals Pathogenic fungi medical journals Pathogenic fungi free journals Pathogenic fungi best journals Pathogenic fungi top journals Pathogenic fungi free medical journals Pathogenic fungi famous journals Pathogenic fungi Google Scholar indexed journals J.podagrica articles J.podagrica Research articles J.podagrica review articles J.podagrica PubMed articles J.podagrica PubMed Central articles J.podagrica 2023 articles J.podagrica 2024 articles J.podagrica Scopus articles J.podagrica impact factor journals J.podagrica Scopus journals J.podagrica PubMed journals J.podagrica medical journals J.podagrica free journals J.podagrica best journals J.podagrica top journals J.podagrica free medical journals J.podagrica famous journals J.podagrica Google Scholar indexed journals morphological characters articles morphological characters Research articles morphological characters review articles morphological characters PubMed articles morphological characters PubMed Central articles morphological characters 2023 articles morphological characters 2024 articles morphological characters Scopus articles morphological characters impact factor journals morphological characters Scopus journals morphological characters PubMed journals morphological characters medical journals morphological characters free journals morphological characters best journals morphological characters top journals morphological characters free medical journals morphological characters famous journals morphological characters Google Scholar indexed journals biodiesel production articles biodiesel production Research articles biodiesel production review articles biodiesel production PubMed articles biodiesel production PubMed Central articles biodiesel production 2023 articles biodiesel production 2024 articles biodiesel production Scopus articles biodiesel production impact factor journals biodiesel production Scopus journals biodiesel production PubMed journals biodiesel production medical journals biodiesel production free journals biodiesel production best journals biodiesel production top journals biodiesel production free medical journals biodiesel production famous journals biodiesel production Google Scholar indexed journals Fusarium oxysporum articles Fusarium oxysporum Research articles Fusarium oxysporum review articles Fusarium oxysporum PubMed articles Fusarium oxysporum PubMed Central articles Fusarium oxysporum 2023 articles Fusarium oxysporum 2024 articles Fusarium oxysporum Scopus articles Fusarium oxysporum impact factor journals Fusarium oxysporum Scopus journals Fusarium oxysporum PubMed journals Fusarium oxysporum medical journals Fusarium oxysporum free journals Fusarium oxysporum best journals Fusarium oxysporum top journals Fusarium oxysporum free medical journals Fusarium oxysporum famous journals Fusarium oxysporum Google Scholar indexed journals Macrophomina phaseolina articles Macrophomina phaseolina Research articles Macrophomina phaseolina review articles Macrophomina phaseolina PubMed articles Macrophomina phaseolina PubMed Central articles Macrophomina phaseolina 2023 articles Macrophomina phaseolina 2024 articles Macrophomina phaseolina Scopus articles Macrophomina phaseolina impact factor journals Macrophomina phaseolina Scopus journals Macrophomina phaseolina PubMed journals Macrophomina phaseolina medical journals Macrophomina phaseolina free journals Macrophomina phaseolina best journals Macrophomina phaseolina top journals Macrophomina phaseolina free medical journals Macrophomina phaseolina famous journals Macrophomina phaseolina Google Scholar indexed journals Mycology articles Mycology Research articles Mycology review articles Mycology PubMed articles Mycology PubMed Central articles Mycology 2023 articles Mycology 2024 articles Mycology Scopus articles Mycology impact factor journals Mycology Scopus journals Mycology PubMed journals Mycology medical journals Mycology free journals Mycology best journals Mycology top journals Mycology free medical journals Mycology famous journals Mycology Google Scholar indexed journals Plant Pathology articles Plant Pathology Research articles Plant Pathology review articles Plant Pathology PubMed articles Plant Pathology PubMed Central articles Plant Pathology 2023 articles Plant Pathology 2024 articles Plant Pathology Scopus articles Plant Pathology impact factor journals Plant Pathology Scopus journals Plant Pathology PubMed journals Plant Pathology medical journals Plant Pathology free journals Plant Pathology best journals Plant Pathology top journals Plant Pathology free medical journals Plant Pathology famous journals Plant Pathology Google Scholar indexed journals
Article Details
1. Introduction
Jatropha (Jatropha curcas L.) or physic nut is a drought resistant specie, which is widely cultivated in the tropics as a living fence. It is a deciduous large shrub or small tree 5 (-8) m tall, belongs to the genus Jatropha which consists of over 170 species. It belongs to the Euphorbiaceae family, producing oil containing seeds. Four of the eight species recorded in West Tropical Africa and described by Hutchison et al. [2] occur in the Niger Delta. These are J.curcas L., J.gossypifolia L., J.multifida L. and J.podagrica. Ratha et al. [3] described 12 species of Jatropha curcas in india using morphological characters. Nwokocha et al. [4] confirmed the relatedness of the 4 species present in the Niger Delta from the phytochemical studies carried out on the leaf, stem, root and seed of four Jatropha L. Species.
Jatropha curcas is a plant with high potential source of biodiesel production. The oil content and fatty acid (FA) composition of J.curcas seeds are of vital importance. The oil content of J. curcas seeds, together with the seed yield, remarkably influences its economic value, especially for large scale agricultural production. Furthermore, probably due to its toxic characteristics, the area of research on the nutritional importance of physic nut seed has not been fully harnessed. This present study is therefore embarked upon to evaluate the effect of Fusarium oxysporum and Macrophomina phaseolina on proximate compositions of J. curcas seeds.
2. Materials and Methods
2.1 Source of plant materialThe Physic nut seeds (Jatropha curcas L) used in this study were sourced from Federal College of Forestry, Jos, Plateau State. The research work was conducted at the Mycology/ Plant Pathology and Physiological Unit of the Department of Plant Science and Biotechnology, University of Port-Harcourt.
2.2 Inoculation of seeds with fungi for proximate analysis
Fifty grams each of healthy Jatropha curcas seeds were weighed into individual 250 ml Erlenmeyer conical flasks. The flasks containing seeds were sterilized at 121°C for 15 mins to eliminate any internal and external seed borne micro-organisms [5]. After sterilization, each flask was allowed to attain room temperature. 20 ml of sterile distilled water was added to individual flask and shaken gently to wet all the seeds, create humid and conducive environment for the test fungi growth. Each flask containing seeds was individually inoculated with a disc of 7 day old spores of each fungus of Fusarium oxysporum and Macrophomina phaseolina respectively obtained from the pure culture of isolated fungi from the infected seeds. This was carried out with a 1.5 cm diameter sterile cork borer. The inocula were spread properly by gently shaking the flask for about 15 minutes to obtain uniform distribution of the mycelium among the seeds. The control flask were maintained without fungi added to it. The entire flasks which include the fungi inoculated and uninoculated seeds were incubated at room temperature for 7 days. At the end of the incubation period, two flasks of each fungal treatment and one flask for control were collected for biochemical analysis. The seeds in each flask were transferred into a pre-weighed watch glass, dried at 45°C for 24 hours and the spores and mycelia of the fungi removed by sieving [5].
The proximate composition of the various nutrient component (Dry matter, Moisture, Lipids, Free fatty Acid, Carbohydrates, Ash, Fibre, Crude protein and Amino acid) in both fungus-inoculated and uninoculated seeds at the incubation period of 7 days were determined following procedures recommended by the Association Official Analytical Chemists [1]. The data obtained from each food component were analysed statistically using the Analysis of Variance (ANOVA).
3. Results
The results of the biochemical analysis of Physic nutseeds inoculated with Fusarium oxysporum, Macrophomina phaseolina and uninoculated controls incubated for 7 days are presented in Figure 1. The test fungi grew well on the Physic nut seed and caused appreciable changes in the nutrient component of the seed. There were significant increase (P=0.05) in the moisture, ash and protein, content of the seeds inoculated with the test organisms compared with the control seeds. (Figures 1, 5 and 7) There were significant decrease (P=0.05) in dry matter, lipid, carbohydrate and fibre content of the seeds inoculated with fungi when measured with the control. (Figures 2, 3, 4 and 6).
3.1 Changes in moisture content
There was significant (P=0.05) increase in moisture content of seeds inoculated with Fusarium oxysporum (37.1%) and Macrophomina phaseolina (36.1%)when compared to uninoculated seeds (Figure 1).
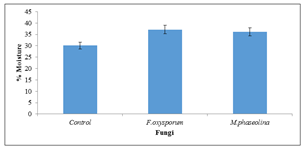
Figure 1: Moisture content of inoculated and uninoculated Jatropha curcas seeds.
3.2 Changes in dry matter
The dry matter was significantly (P=0.05) reduced by Fusarium oxysporum (58.9%) and Macrophomina phaseolina (59.4%) compared with the uninoculated control (Figure 2).
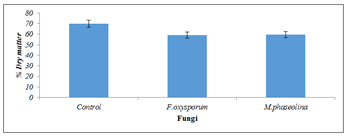
Figure 2: Dry matter of inoculated and uninoculated Jatropha curcas seeds.
3.3 Changes in lipid content
The lipid content increased significantly(P=0.05) in all the samples inoculated with Fusarium oxysporum and Macrophomina phaseolina in contrast with the uninoculated control (Figure 3).
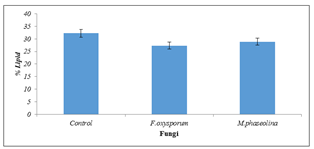
Figure 3: Lipid content of inoculated and uninoculated Jatropha curcas seeds.
3.4 Changes in carbohydrate content
The carbohydrate content of inoculated seeds were significantly (P=0.05) reduced by all the test fungi as compared to the uninoculated seeds (Figure 4).
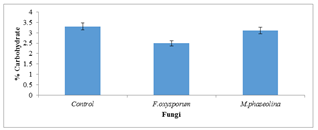
Figure 4: Carbohydrate content of inoculated and uninoculated Jatropha curcas seeds.
3.5 Changes in ash content
There was significant (P=0.05) increase in ash content of seeds inoculated with Fusarium oxysporum (6.2%) and Macrophomina phaseolina (7.3%) respectively (Figure 5).
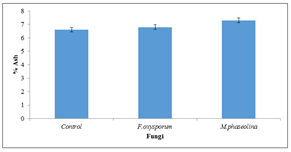
Figure 5: Ash content of inoculated and uninoculated Jatropha curcas seeds.
3.6 Changes in crude fibre content
A significant (P=0.05) reduction of crude fibre content was observed in samples inoculated with Fusarium oxysporum (7.51%) and Macrophomina phaseolina (4.39%) when compared to uninoculated samples (Figure 6).
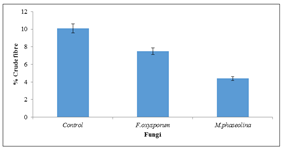
Figure 6: Crude fibre content of inoculated and uninoculated Jatropha curcas seeds.
3.7 Changes in crude protein content
There was significant (P=0.05) increase in protein content of seeds inoculated with Fusarium oxysporum by (19.6%) and Macrophomina phaseolina by (19.3%) over the uninoculated control (Figure 7).
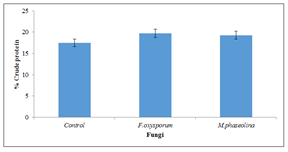
Figure 7: Crude protein content of inoculated and uninoculated Jatropha curcas seeds.
4. Discussion
The moisture content of fungus-inoculated seeds increased when compared to uninoculated control. There was a significant difference at P=0.05, Fusarium oxysporum had the highest moisture content (37.06%) followed by Macrophomina phaseolina (36.10%). The increase caused by fungi is due to the utilization of some of the components of the seed as food nutrient thereby converting them into water in the process. Similar results were obtained by [5, 6]. Also, Amienyo et al. [7] reported similar result in Sweet Potato inoculated with fungi. Deteriorative reactions occur more readily in seeds at higher moisture content and subsequently, this condition constitute hazard to the longevity of seed survival [8].
The inoculation of Jatropha curcas seeds with pure cultures of Fusarium oxysporum and Macrophomina phaseolina were incubated at ambient temperature for 7days, underwent different degrees of deterioration. Fungi secretes pectic enzymes which spread rapidly through the intercellular spaces of parenchyma down the middle lamella of the host cells tissue, a process which results in the maceration of tissues. The activities of these enzymes may be responsible for the decrease in Dry matter and increase in moisture content. From the biochemical analysis carried out it was observed that there was a decrease (P=0.05) in dry matter of fungus inoculated seeds compared with that obtained using uninoculated control. In this study, Fusarium oxysporum had the highest decrease compared to Macrophomina phaseolina, this agrees with earlier report of [9] that fungi produces extracellular cellulolytic and pectic enzyme and secondary metabolites which may be responsible for the drastic depletion of dry matter. The extracted lipid from the seeds inoculated with Fusarium oxysporum and Macrophomina phaseolina decreased from the uninoculated control (32.17%). Fusarium oxysporum (27.27%) and Macrophomina phaseolina (28.97%) respectively. This is in agreement with the report of Ward et al. [10] who reported that fungi caused a decrease in total oil of groundnut. In accordance with these, Ogundero [11] explained that the decrease in oil content could be due to the hydrolysis of oil to free fatty acid (FFA). This occurred at different rates for the individual micro-organisms.
The free fatty acid expressed as percent Oleic acid for both inoculated and uninoculated seeds increased. Macrophomina phaseolina induced the highest value of free fatty acid of (56.6%) while seeds inoculated with Fusarium oxysporum had the lowest free fatty acid value of (53.61%). The increase in free fatty acid seems to suggest that these fungi have high lipase activity. Similar to the result of this present study, the increase in the formation of free fatty acid was found to be associated with a decrease in total oil, in line with Ward and Diener, [10] who reported increase in free fatty acids of groundnut inoculated with Aspergillus sp. and noted that the fungus utilized the free fatty acid as carbon source. There was significant (P=0.05) decrease in the carbohydrate content of the inoculated seeds when compared to the control. The highest reduction was recorded in seeds inoculated with Fusarium oxysporum thereby showing the amylolytic activity of the fungus compared to Macrophomina phaseolina. Solomon [12] mentioned that carbohydrates are usually hydrolyzed to glucose and then used as a source for carbon and energy for microbial growth, which also explains the decrease in carbohydrate content observed. Raimbualt [13] has attributed the depletion in carbohydrate to transformation of starch and mineral salts by fungi (especially Aspergilli) into fungal protein. Embaby et al. [14] reported that, most reduction and loss percent were found with fat, carbohydrate due to Fusarium oxysporum in legume seeds. The result of the investigation revealed that in comparison with the uninoculated control, the presence of the individual microorganism in the flasks increased the ash content of the seeds, in agreement with Ataga et al. [5]. The crude fibre content decreased in all the seeds inoculated with the test fungi. This is in agreement with the report of Onifade et al. [15] about a decrease in crude fibre content, in the enrichment of sweet potato flour using A.niger and Saccharomyces cerevisiae. El-Tinay et al. [16] explained that crude fibre tends to decrease during fermentation, this he concluded was a result of being utilized by the fermentation microbes.
In this study, the fungi, caused increases in protein when compared to the control. The significant increase (P=0.05) in protein content by Fusarium oxysporum and Macrophomina phaseolina could have resulted from slight protein synthesis of enzyme proteins or other constituents [17]. Several studies have shown that mold form proteases during fermentation which may result in increased protein amino acid. This result agrees with the report of Nwaukwu and Ataga [18].
5. Conclusion
In conclusion, the diverse ways the fungi affected the nutritional value is of paramount importance. Due to the high value of the main plant, the need for ways to achieve disease free seeds, which result to healthy useful plant parts, is key.
References
- AOAC. Association Official Analytical Chemists. Official Method of Analysis (13th Edn) Washington DC (1995): 1018.
- Hutchison J, Dalziel JM. Floral of West Tropical Africa, Vol.1. Part II. Crown Agent for Oversea Governments and Administrations, London (1958).
- Ratha PK, Paramathma M. Potentials and Jatropha species. wealth of India Curr Sci 97 (2009): 1000-1004.
- Nwokocha AB, Agbagwa IO, Okoli BE. Comparative Phytochemical Screening of Jatropha L. Species on the Niger Delta. Research Journal of phytochemistry (2011).
- Ataga AE, Umechuruba CI. Biochemical changes in African Yam Bean seeds caused by Botryodiplodia theobromae, Fusarium palidoreseum and Penicillium oxalim. Global Journal of Pure and Applied Science 4 (1997): 381-384
- Nwaukwu IA, Ikechi-Nwogu C. Biochemical Changes induced by the Effect of Six Pathogenic Fungi on Dialium quineense: Black Edible Fruit. International Organization of Scientific Research Journal of Pharmacy and Biological Sciences 2 (2012): 20-24.
- Amienyo CA, Ataga AE. Biochemical changes induced by four rot fungi on Sweet Potato (Impomoea batatas (L) Lam) tubers. Nigerian Journal of Microboilogy 20 (2006): 1334-1338.
- Vashisth, Nagarajan. Deteriorative reactions occur more readily in seeds at higher moisture content and subsequently, this condition constitute hazard to the longevity of seed survival (2009).
- Okonkwo SN, Amund OO, Ogunsanya CO. Microbial rotting and preservation of banana fruit (Musa sapientium L) in Nigeria. Microbes. Letter 44 (1990): 175-176.
- Ward HS, Diener UL. Biochemical changes in shelled peanuts caused by storage fungi in Effect of Aspergillus tamari, Four species of A.glaucus group and Penicillium citrinum. Phytopathology 51 (1961): 244-250.
- Ogundero VW. Hydrolysis of vegetable oils and triglycerides by thermotolerant and zoopathogenic species of Aspergillus from Nigeria Palm Produce. Mycopathogia 77 (1992): 43-46.
- Solomon B. Microbial conversion of hydrolysed maize and sorghum dusts to high protein foods. Nigerian Food Journal 5 (1987): 1-11.
- Raimbualt M. Fermentation of milleu solid croissanceb de champing ons filamentoux sur substrats amylases edited by Orstom-Paris, Sorie Travicuet Document no (1981): 127-291.
- Embaby EM, Mona M, Abdel-Galil. Seed Borne Fungi and Mycotoxins Associated with Some Legume Seeds in Egypt. Journal Appl Sci Res 2 (2006): 1064-1071.
- Onifade AK, Atum HN, Adebolu TT. Nutrient enrichment of Sweet potato (Ipomoea batatas L). Global Journal of Pure and Applied Sciences 10 (2004): 31-36.
- El-Tinay AH, Abdel-gadir AM, EL-hidal. Sorghum fermented Kisra bread I: Nutritive value of Kisra. Journal of Science and Food Agriculture 30 (1979): 859-361.
- Kuo UH, Bau HM, Quemener B, et al. Solid State fermentation of Lathyrus sativus seeds using Aspergillus oryzae and Rhizopus oligosporus SP T-3 to eliminate the neurotoxin B-ODAP without loss of nutritional value. Journal of Science, Food and Agriculture 69 (1995): 81-89.
- Nwaukwu IA, Ataga AE. Biochemical Changes induced by five Pathological Fungi on seeds of Hibiscus sadariffa (YAKWA) Scientia Africana 12 (2012): 1-6.
- Christensen CM, Kaufman HH. Grain storage. The role of fungi in quality losses. Univ. Minnesota, Press Minneapolis (1969).
- Osborne Voogt. The analysis of nutrient in foods. Academic Press, London (1978): 159.


 Impact Factor: * 5.3
Impact Factor: * 5.3 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 