Promoter’s Choice Drive CAR-T Efficiency
François Anna1,2#, Elodie Bole-Richard3#, Maxime Fredon3, Jean-Marie Certoux3, Philipe Souque2, Francine Garnache-Ottou3, Pierre Charneau2, Olivier Adotevi3, Pierre Langlade-Demoyen1, Julien Caumartin1 & Maria Loustau1*
1Invectys, Paris Biopark 12 rue Jean Antoine de Baïf ,75013 Paris, France.
2Molecular Virology and Vaccinology Unit, Virology Department, Institut Pasteur & CNRS URA 3015, 25-28 rue du Dr. Roux, 75015 Paris, France.
3Université. Bourgogne Franche-Comté, INSERM, EFS BFC, UMR1098, RIGHT Interactions Greffon-Hôte-Tumeur/Ingénierie Cellulaire et Génique, F-25000 Besançon, France.
#These authors contributed equally to this work.
*Corresponding Author: Maria Loustau, Invectys, Paris Biopark 12 rue Jean Antoine de Baïf ,75013 Paris, France
Received: 01 June 2021; Accepted: 10 June 2021; Published: 06 July 2021
Article Information
Citation:
François Anna, Elodie Bole-Richard, Maxime Fredon, Jean-Marie Certoux, Philipe Souque, Francine Garnache-Ottou, Pierre Charneau, Olivier Adotevi, Pierre Langlade-Demoyen, Julien Caumartin, Maria Loustau, Promoter’s Choice Drive CAR-T Efficiency. Journal of Biotechnology and Biomedicine 4 (2021): 63-83.
DOI: 10.26502/jbb.2642-91280036
View / Download Pdf Share at FacebookAbstract
The development and the functionality of Chimeric Antigen Receptor (CAR) therapy is highly linked to the stability and expression level of the receptor, for which the choice of the promoter is key. Although EF1α is a reference promoter to induce strong antigen binding and long-term CAR expression, we investigated the impact of this one and other constitutive promoters (MSCV, PGK, β2m and CMV) on a CAR123 construct in the Blastic Plasmocytoid Dendritic Cell Neoplasm (BPDCN). Our results indicate that lentiviral vector titer is affected by transgene nature and promoter activity. Interestingly, by moderating the expression level of CAR123 at the cell surface, its recognition capacity and selectivity were improved. We observed that in early stages after transduction, CAR123 expression level did not affect lymphocyte’s phenotype and its cytotoxic capacity against BPDCN cell line. However, following long-term culture and decrease of cell-surface CAR expression, a hallmark of transition from activation to memory state of T-cells was observed, and only T-cells transduced with CAR-EF1α or -β2m promoters were still functional. We identified β2m as a promising promoter. Though it shows lower antigen-binding CAR-T cell and antigen recognition rates compared to EF1α, β2m promoter still present interesting CAR expression levels, stability, and binding capacity
Article Details
1. Introduction
Along the last two decades several immunotherapeutic strategies have been developed to generate an efficient treatment against cancers. Development of chimeric antigen receptors (CAR) [1] has been an important breakthrough since CARs allow the redirection of autologous T cells to target tumor cells. Several CARs targeting a broad range of tumor antigens are currently under development and are in clinical phases, especially for hematological malignancies [2-5].
We have developed a novel cellular immunotherapy against CD123 protein, which is an Interleukin-3 Receptor α chain expressed at the cell-surface, demonstrated to be involved in hematopoietic differentiation [6, 7]. It was shown that CD123 is overexpressed in numerous hematologic malignancies, particularly in Blastic Plasmocytoid Dendritic Cell Neoplasm (BPDCN), acute myeloid leukemia [8] and acute lymphoblastic leukemia (ALL), whereas its expression is limited on normal hematopoietic stem cells, monocytes and endothelial cells [9, 10]. Consequently, CD123 was identified as membrane biomarker and a therapeutic target [11-14].
The present study focuses on the CAR123 construct based on the B4D5 monoclonal antibody, which was recently demonstrated to eliminate BPDCN in vitro and in vivo [15].
This CAR123-chain was firstly vectorized by a Moloney murine leukemia virus vector. Since transgene introduction and regulation are crucial for CAR-T cell functionality and antigen recognition, this vector was replaced by the HIV-1 lentiviral vector, which is more adequate for this use in human T cells [15]. Also, in the context of gene therapy, Moloney derived vector presented safety issues [16-18], showing a propensity to integrate near gene transcription initiation sites inducing proto-oncogenes [19]. On the contrary, HIV genome was shown to integrate in distal regions of transcription initiation sites [20, 21]. Thus, a lentiviral vector was selected for ongoing pre-clinical development because the risk of interference with flanking genes HIV-derived vectors is limited. More importantly, the type of promoter used to regulate the transgene expression is critical to ensure a long-term and stable expression of CAR-chain on human T cells [22]. Many constitutive promoters are described and can be used in lentiviral vectors, but few are able to sustain expression in human T cells.
In the present study, we investigated how different promoters influenced on our CAR123-chain expression, stability, and functionality, enabling to make a rational choice on the most appropriate one. Five different promoters were selected and assessed in primary human T cells: (i) human immediate early cytomegalovirus (CMV) promoter, a strong ubiquitous classically used viral promoter [23-25]; (ii) Murine stem cell virus (MSCV) retroviral LTR promoter, previously described in T cells to mimic transcription regulation of γ-retroviral vector [23, 26, 27]; (iii) Human phosphoglycerate kinase (PGK), an endogenous housekeeping promoter showed to sustain a moderate and stable expression level [23, 25]; (iv) Beta-2-Microglobulin (β2m) promoter, an ubiquitous and constitutive promoter especially strong in immune cells [28-30]; and (v) Human elongation factor 1 alpha (EF1α), a strong ubiquitous constitutive promoter classically used for stable gene transfer.
Here we report new CAR promoter optimizations to aim a stable and long-term cell surface expression on human T cells. We demonstrate that antigen binding is directly correlated to promoter intensity. Thus, by selecting a low intensity CAR123-T cell promoter we might attain a fine tuning anti-TAA engagement and reduce the “on-target, off-tumor” effect.
2. Material and Methods
2.1 Plasmids construction
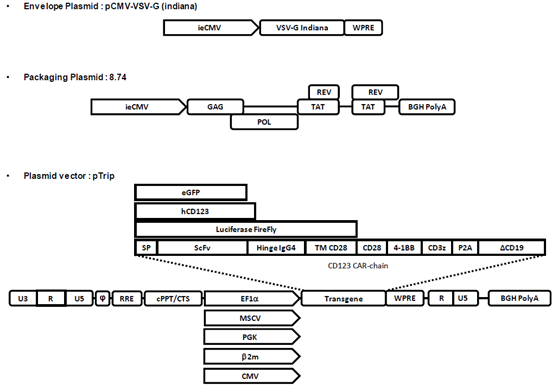
All vectors presented in this study derived from a pTrip-GFP [31] carrying a Woodchuck Hepatitis Virus (WH) Posttranscriptional Regulatory Element (WPRE) in 5’ of the transgene. GFP gene was replaced by B4D5 CD123 CAR-2A-ΔCD19 gene [15] by restriction/ligation. CMV promoter was replaced with different promoters by restriction/ligation. All promoters were extracted from their original plasmid by PCR with adaptive primers. β2m promoter was described in former studies [32] (Ref: LRG_1215; from 4556 to 5070). hPGK promoter comes from a pTrip hPGK-GFP plasmid generated in our laboratory (Ref: NG_008862.1; from 4649 to 5159). EF1α promoter comes from pEF-GFP plasmid, kindly donated by Connie Cepko (from 36 to 1217). MSCV promoter correspond to the MSCV 5’LTR from pMSCVpuro (Clonetech; nucleotide 1 to 515). All constructs were controlled by sequencing using the following primers for qPCR: U5R-M667 (5'-GGCTAACTAGGGAACCCACTG-3'), U5R-AASM (5'-GCTAGAGATTTTCCACACTGACTAA-3'), CD3 in 5' (5'-GGCTATCATTCTTCTTCAAGGTA-3') and CD3 in 3' (5'-CCTCTCTTCAGCCATTTAAGTA-3').
2.2 Lentiviral vector production
Production and titration of lentiviral vector were formerly described [33]. Lentiviral vectors were produced by transient calcium phosphate co-transfection of HEK-293T cells (ATCC) with the vector plasmid pTrip encoding the vector RNA, an envelope expression plasmid encoding the glycoprotein from Vesicular Stomatis Virus serotype Indiana (IND) and the p8.74 packaging plasmid for the production of integrative lentiviral vector particles (see Figure 1 for plasmid description). 48h post-transfection supernatant was harvested and centrifuged 5 min at 500g to remove cellular debris and then ultra-centrifuged during 1h at 22 000g. Pellets were resuspended in a storage buffer composed of PIPES pH 7.2, 2.5% sucrose 75mM NaCl. All productions correspond to 30 ml of supernatant pelleted and resuspended in 30 μl of buffer.
2.3 Lentiviral vector titration by qPCR and Luciferase assay
For titration, 4.105 HEK-293T cells were plated in 6 well-plates and transduced with vector suspension. 72h post transductions, cells were harvested and lysed. SYBR green qPCR was performed on 1:10 diluted cell lysates with a primer against U5 fragment of lentiviral vector Long Terminal Repeat [34] and a fragment of endogenous cell CD3ζ gene. Ratio U5/CD3 was calculated, and the “Transduction Units” per ml (TU/ml) were determined.
During titration, a part of non-treated cell lysate was used to quantify luciferase activity (Luciferase Assay System, Promega) and total protein for normalization (BCA protein assay Interchim, Clichy, France). Values were reported to the copy number measured by qPCR to determine Relative Luciferase Activity per Viral Copy Number (RLU/VCN).
2.4 Human T cell purification, stimulation, and transduction
Peripheral blood cell samples of healthy donors were collected at the French Blood Center (EFS BFC Besançon, France) after obtaining written informed consent. Blood samples collection was approved by the French Ministry of Higher Education and Research (agreement number #AC-2015-2408 of May 22nd, 2015).
T cells were purified by bead-sorting (Human Pan T Cell Isolation Kit, Miltenyi Biotech) and activated with CD3/CD28 beads (Human T Cell Activation/Expansion Kit, Miltenyi Biotech) for 2 days prior to transduction and cultured in complete RPMI medium: 10% heat-inactivated endotoxin-free Fetal Calf Serum (Eurobio, Courtaboeuf, France), 1% penicillin/streptomycin (Eurobio, Courtaboeuf, France); with 50μM Beta-mercaptoethanol (Gibco), 1mM Sodium Pyruvate (Gibco), 10 mM Hepes (Gibco), 1x Non-Essential Amino Acids (Eurobio, France) and 50 U/ml of Recombinant human IL-2 (Preprotech, France). Activated CD3+ T cells were either transduced with lentiviral vectors or not (activated but “non-transduced” T cells). For transduction, medium was renewed, and lentiviral vector was added at MOI 20 or 100. The day after transduction and every 3-4 days, half of the medium was changed, and cells were maintained at 1.106/ml.
2.5 CAR expression stability assay
To assess CAR cell surface expression, a truncated CD19 reporter (ΔCD19) was used as a selection marker [15]. Transduction efficiency was determined on the basis of ΔCD19 cell surface expression on transduced CD3+ T cells by flow cytometry. Activated T cells were transduced at MOI 20. From day 5 post-transduction and every 3-4 days, transduction efficiency was determined by flow cytometry analysis using CD8-PE-Cy7 (clone SK1, Biolegend), CD4-PerCP (clone SK3, Biosciences) and CD19-PE (clone LT19, Miltenyi Biotec, Germany) antibodies. Labelling was detected on an Attune flow cytometer (Thermo-Fisher Scientific) and analyzed with FlowJo software.
2.6 Evaluation of the binding capacity of the CAR in transduced cells
The expression of the CD123 CAR on cell surface of transduced T-cells was examined by flow cytometry (FACS LSR Fortessa). We evaluated the fixation of CD123 on the CD8+ ΔCD19+ CAR cells in comparison to non-transduced (NT) cells. T-cells (1.105 cells) were labeled with 0.25 μg per well of biotinylated CD123 protein for 1h at room temperature, followed by PE-streptavidin (BD Biosciences, New Jersey, USA) incubation (dilution 1:1000).
2.7 Evaluation of the expression of the CAR in transduced cells by Western blotting
Non-transduced (NT) and CD123-CAR-T cells were lysed by sonication in RIPA buffer supplemented with a protease inhibitor cocktail (complete Mini EDTA-free; Roche, France). Lysate’s protein content was quantified with a BCA protein assay (Interchim, Clichy, France). Then, equivalent amounts of cell protein (20 μg proteins) were separated by SDS-PAGE, and electro-transferred onto PVDF membranes (Bio-Rad, California, USA). Membranes were probed overnight with primary antibodies (diluted at 1:1000) specific for human CD3ζ chain (51-6527GR, BD Biosciences, New Jersey, USA). Membranes were also probed with specific anti-β-actin antibodies (clone AC15, #A5441, Sigma-Aldrich, Missouri, USA; dilution 1:1000) as an internal loading control. For immunodetection, membranes were probed with a secondary antibody: sheep anti-mouse IgG (#515-035-062, Jackson, Pennsylvania, USA; dilution 1:6000). Detection was carried out using chemiluminescence reagents (Clarity™ Western ECL Blotting Substrates, Bio-Rad, Cressier, Switzerland), using a camera and Bio-1D software (Wilber-Lourmat, Collégien, France).
2.8 In vitro Cytotoxicity assay
The CD123+ BPDCN cell line CAL-1 was kindly provided by Dr. Maeda, Nagasaki University, Japan. CAL-1 cells were cultured in complete RPMI 1640 glutamax medium supplemented with 10% heat-inactivated, endotoxin-free fetal calf serum, 1% penicillin/streptomycin (Eurobio, Courtaboeuf, France). CAL-1 cells (1.106 cells/ml) were labeled with CellTrace CFSE Cell Proliferation Kit (ThermoFisher, eBioscience, France) at 1μM during 25min. CAR-T cells were then cultured at 10:1 effector to target (E:T) ratio with CFSE-labelled CAL-1 at 37°C for 24h. After co-culture, cells were labeled with CD19-APC (clone LT19, Miltenyi), anti-CD8-VioBlue (clone SK1, Biolegend) antibodies and Fixable Viability Dye eFluor 780 (dilution 1:1000) (ThermoFisher, eBioscience, France) in order to evaluate cell survival (Viability Dye) of target cells (CFSE+).
2.9 Flow cytometry
Transduction efficiency was determined by flow cytometry analysis (FACS LSR Fortessa) using CD4PerCP, CD8PC7 (BD Bioscience, New Jersey, USA) and CD19-PE (Miltenyi Biotec, Bergisch Gladbach, Germany) antibodies to identify ΔCD19 reporter on transduced T cells. Non-transduced T cells (NT) were used as control. Cell surface phenotype was investigated with the following monoclonal antibodies: anti-CD19-APC and anti-CD3-VioBlue (Miltenyi Biotec, Bergisch Gladbach, Germany) for T cells, or anti-CD123-PC7 (Sony Biotechnology, California, USA) for target cells. Antibody binding was detected on an Attune flow cytometer (ThermoFisher) and analyzed with Flow Jo software.
A cytotoxic assay was performed as described above. Day prior to the assay and 12 days post CAL-1 co-incubation, surface phenotype was investigated with the following antibodies: CD8-PE (clone SK1, Biolegend), CD19-FITC (clone LT19, Miltenyi), CD62L-BV421 (clone DREG-56, BD Horizon) CD45RA-BV711 (clone H100, Invitrogen).
2.10 Statistical analysis
Independent experiments were originally performed 3 times and then repeated until a significant variation was reported for demonstration key points. The data were analyzed using GraphPad Prism.
3. Results
3.1 CAR expression level affects lentiviral vector production
CARs immunotherapy is highly dependent on lentivirus vector. CAR-T cells are mostly generated by transduction with lentiviral vectors, frequently obtained by the three-plasmid transfection method (Figure 1). To achieve high production yields of lentiviral vector, the whole machinery needs to be finely tuned and interference must be avoided. During production, the transcriptional unit encoding the transgene is functional and if the transgene is toxic or competes with any other component, the production will be dramatically affected.
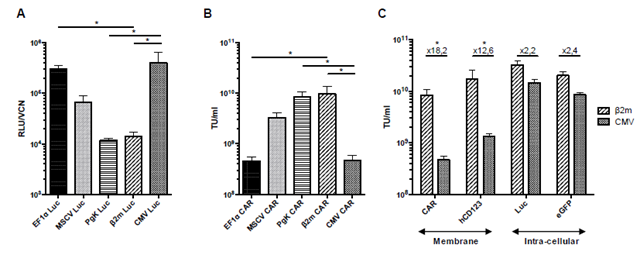
To investigate the effect of the CAR expression during vector production, we evaluated and compared CMV, MSCV, PGK, β2m and EF1α promoters. To do so, a 2nd generation lentiviral vector was used with transgenes under the control of each promoter (Figure 1). The HEK-293T cell line was firstly transduced with vectors expressing the luciferase firefly reporter to compare promoters’ activities (Figure 2A). We identified a group of strong promoters composed of CMV and EF1α that induced the highest protein expression level with a 50-fold increase compared to the group of weak promoters encompassing PGK and β2m. The MSCV promoter was considered as an intermediary expression promoter.
To assess the impact of the promoters’ expression level on the titer of the CAR vector, we compared and measured the functional titer of the CAR123 vector production by qPCR (Figure 2B). Strong promoters, CMV and EF1α induced weak vector titers (2.83 ± 0.82 .108 and 2.88 ± 1.7. 108 TU/ml, n=4). On the contrary, vector titers from the weak group (PGK and β2m) were determined to be about 30 times higher (7.90 ± 2.71 .109 and 8.44 ± 1.74.109 TU/ml, n=4). Vector titer with MSCV promoter remained intermediate (3.20 ± 0.86 .109 TU/ml, n=4). We observed that the CAR vector functional titer was inversely proportional to the promoter activity.
In order to determine if this difference of functional titer was related to the transgene nature, we replaced the CAR-chain with either transgenes encoding a type I membrane-bound (human CD123) or intracellular proteins (GFP and Luciferase Firefly). The different transgenes expression was regulated by a weak (β2m) or a strong (CMV) promoter. Vectors expressing intracellular proteins were poorly influenced by promoters’ nature with only a 2.2- and 2.4-fold variation between CMV and β2m (Figure 2C). Yet, for the membrane protein, the titer was higher with β2m promoter than with CMV promoter (18.2-fold increase for CAR-chain and 16.8-fold increase for hCD123).
Taken together, these results evidenced that cell-surface protein expression, particularly membrane CAR-chain expression, was clearly deleterious for vector production. As a result, CAR vectors with weak promoters in HEK-293T like β2m and PGK ensure better production than strong promoters like EF1α and CMV promoters. This observation is crucial for CAR development and manufacturing, but improving CAR
vector production should not be done at the expense of CAR functionality in primary human T-cells.
3.2 Expression stability of CAR123-chain is related to the promoter
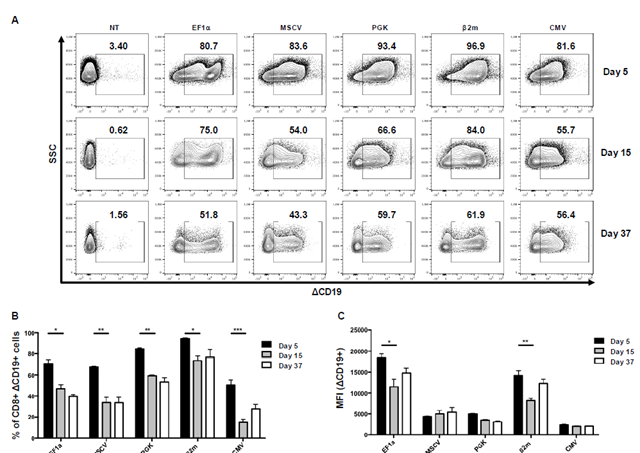
Expression level and stability are two critical parameters to generate functional CAR-T cells and we sought to study whether promoters could affect them. Since no direct immunolabeling of CAR protein could be performed, we used the 3rd generation CAR123 construct encompassing a CD19 truncated protein (ΔCD19) as a reporter protein to determine the promoter’s activity and stability. CD3+ T cells were purified from healthy PBMCs and activated using CD3/CD28/CD2 beads and transduced with the corresponding CAR123-promoter constructs. Non-transduced (NT) activated T cells were used as control. These CD3+ T cells were maintained in culture 37 days post-transduction to investigate the promoter’s influence on CAR expression and stability overtime.
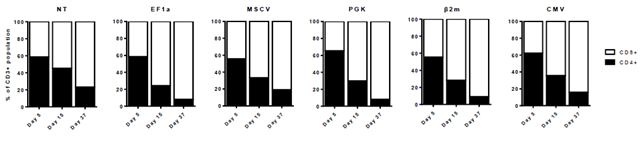
CAR123-T cells were generated and the ΔCD19 protein expression was monitored following transduction step at day 5, 15 and 37 on CD3+ T cells by flow cytometry (Figure 3A). Within the promoters, two different groups were identified: EF1α and β2m showing a significantly higher expression compared to the other group, including the MSCV, PGK and CMV promoters. Expression level slightly decreased at day 15 for EF1α and β2m promoters, reaching 60% of the original expression level, and increased at day 37. For MSCV, PGK and CMV promoters, expression of ΔCD19 remains low all along the experiment. Noteworthy, the nature of the recombinant protein influenced the promoter efficacy. In terms of expression stability, a pattern is shared by all promoters with maximum expression at day 5 and a decrease in expression at day 15, which remained stable until day 37 (Figure 3B). However, EF1α and β2m are the most stable promoters preserving 80% of the CAR-expressing population between day 5 and day 15. At day 37, β2m is the most stable promoter without loss of expression, whereas EF1α decreases to 60%. Strikingly, CMV promoter did not allow stable CAR expression, maintaining only 40% of the original CD8+ ΔCD19+ CAR-T cells. Such expression levels were observed for either CD8+ T cells (Figure 3C) or CD4+ T cells (Supplementary Figure 1), demonstrating that CD8+ and CD4+ T cells express comparable levels of CAR constructs. However, the CD4+/CD8+ T cells ratio strongly decreased from day 5 to day 37, as previously observed during CAR-T cells expansion [35]. Then, the remaining T cells at day 37 are essentially CD8+, independently of the promoter used (Supplementary Figure 2).
These results confirm that EF1α promoter presents better yields for CAR expression. Nevertheless, β2m promoter also ensures high and stable CAR expression in primary human T cells in comparison to CMV, MSCV and PGK promoters.
3.3 CAR expression level is associated to target binding and cytotoxic activity
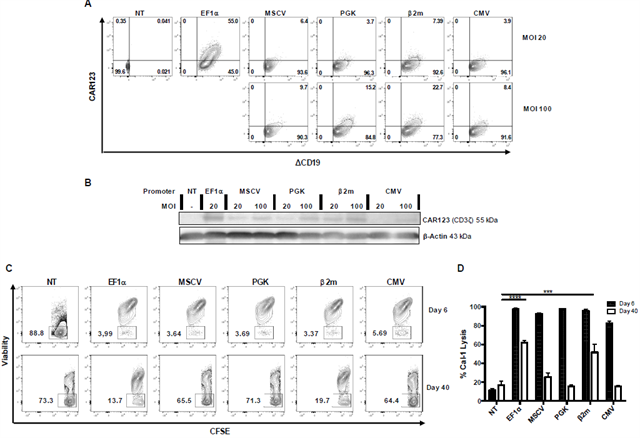
We determined that the expression level of ΔCD19 reporter was notably different between all promoters assessed, with a primacy for EF1α and β2m promoters for CAR expression. This implies that CAR expression level should variate similarly, affecting the functionality of CAR123-T cells. To verify this, CAR123-T cells were co-incubated with biotinylated human CD123 protein. Cell-surface expression of ΔCD19 and CD123-bound cells were simultaneously determined by flow cytometry.
All CAR123-T cells transduced with a multiplicity of infection [36] of 20 displayed the ΔCD19 at their surface for all promoters. Yet only T cells transduced with EF1α CD123 CAR-chain cells showed a “high binding” for CD123 protein with 55% of ΔCD19+ CD123+. On the contrary, MSCV, PGK, β2m and CMV were below 5% of labelling and were considered as “low binding” (Figure 4A). To study if the binding capacity was directly linked to CAR expression level, we sought to improve CAR surface expression of ΔCD19Low CAR-T cells. We transduced T cells with a MOI of 100 instead of 20 for the “low binding” promoter group. By increasing the CAR cell-surface expression, we expected to improve the binding rates of CD123 protein (Figure 4A). PGK and β2m had an important increase from 3.7% and 7.39% to 15.2% and 22.7% respectively, whereas MSCV and CMV had a weaker increase from 6.4% and 3.9% to 9.7% and 8.4% respectively. As a result, increasing the MOI allowed the obtention of CAR-T cells with higher expression levels of CAR-chain, as confirmed by western-blot (Figure 4B). Though, none of the “low-binding” promoters were able to equal EF1α promoter yields with this approach.
These results confirmed that CAR binding capacity is tightly related to the CAR expression level. And only EF1α promoter allows the generation of CAR-T cells with a “high-binding" capacity due to a high expression level. Among “low-binding” group, β2m promoter is the most interesting promoter with a slightly superior binding capacity compared to the others.
To study the incidence of expression stability of CAR-chain on the cell function, we performed a cytotoxicity assay against CAL-1 tumor cells, a BPDCN cell line, at an E:T ratio of 10:1 at day 6 and day 40 (Figure 4C and D). These time points corresponded to the maximum level of CAR expression during activation phase, or to the low and stable expression during resting phase, respectively.
At day 6, complete lysis was observed for all CAR123-T cells used (>90% of tumor lysis), except for CAR123 under the regulation of CMV promoter, which demonstrated a slightly lower efficiency against CAL-1 tumor cells (82% of tumor lysis). However, at day 40 only CAR123-chain regulated by EF1α and β2m promoters were able to eliminate CAL-1 target cells (60% and 52% of tumor lysis respectively). For MSCV, PGK and CMV promoters, tumor cell lysis could not be discriminated from NT T-cells alloreactivity. This implies that specific cytotoxic function was lost at day 40 post-transduction for CAR-T cells generated with these promoters.
This decrease in cytotoxicity for PGK, MSCV and CMV promoters was correlated with the loss of CAR expression observed between day 6 and day 37 (Figure 3A). However, β2m promoter showed a singular profile. Though it shared the “low-binding” profile with PGK, MSCV and CMV promoters, it demonstrated to maintain a stable protein expression, with higher expression level, and was able to sustain a cytotoxic activity comparable to EF1α at day 40.
Thereby, among all promoters tested, this observation points out the advantage of using either EF1α to generate CAR with a “high-binding” profile, or β2m promoters for a “low-binding” profile. Both promoters ensure an efficient level of expression of CAR123-chain in human T-cells to preserve a prolonged efficiency.
3.4 Differentiation of CAR123-T cells is not promoter-dependent after CAL-1 co-culture
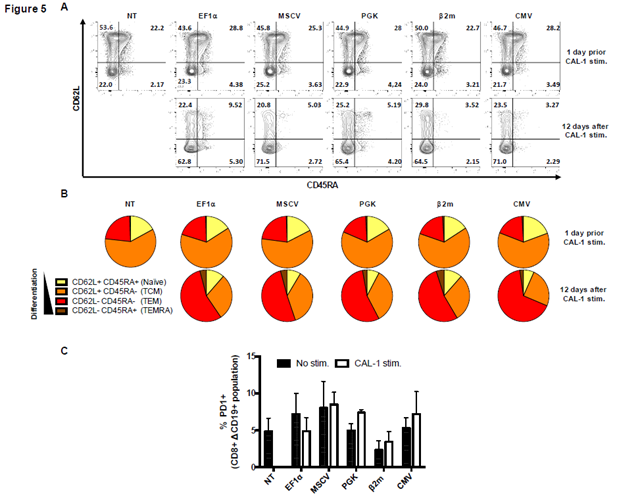
Strong or weak expression levels of CAR-chains might be related to the magnitude of activation signals. So, we investigated if these CAR-T cells with different binding profiles induced diverse differentiation profiles when they were confronted to their target cells. For that, we studied the expression of CD62L and CD45RA markers on CAR-T cells by flow cytometry prior and 12 days after co-incubation with CAL-1 cells.
Before co-culture we observed that (i) around 50% of CAR-T cells were CD62L+/CD45RA- (central memory T cells, TCM), (ii) around 20% of CAR-T cells were CD62L+/CD45RA+ (naïve T cells) and (iii) around 20% were CD62L-/CD45RA- (effector memory T cells, TEM) (Figure 5A). Terminally differentiated memory T (TEMRA) cells CD62L-/CD45RA+ represented less than 5% of total CAR-T cells. 6 days post-transduction, no differences were observed between CAR123-T cells and NT T-cells, suggesting that CAR123-chain expression did not influence T-cells differentiation. However, following a 12-day co-incubation with CAL-1 tumor cells, we observed a major modification in phenotype profile for all CAR-T cells: around 60% of CAR-T cells turned out to be TEM and only 20% of CAR-T cells were still TCM. Naïve and TEMRA populations still represented less than 5% of total T cells population. Nonetheless, promoters were not responsible for variation on CAR-T cells differentiation either before or after co-incubation with CAL-1 tumor cells (Figure 5B).
Probably antigen binding and cytotoxic function could be dampened by CAR-T cells exhaustion. PD-1 up-regulation is known as a CAR-T cell exhaustion marker. Therefore, we analyzed its expression level on CAR123-T cells prior and after co-incubation with CAL-1 tumor cells (Figure 5C). We observed that there was no significant difference in PD-1 cell-surface expression among all CAR123-T cells, independently of the chosen promoters, and less than 5% of CAR123-T cells express PD-1.
Taken together, phenotype and differentiation of CAR-
T cells were not modified by the different promoters assessed. After a single stimulation, all CAR123-T cells were demonstrated to differentiate from CD62L+/CD45RA- T cells to CD62L+/CD45RA+ without of CAR-T cells exhaustion.
Representation of a 2nd generation lentiviral vector production. pCMV-VSV-G plasmid encoded an amphotropic envelope Glycoprotein from Vesicular Stomatitis Virus (VSV-G) Indiana serotype. p8,74 plasmid coded for Gag-pol essential protein and tat-rev accessory proteins. Tat protein acted as trans-activator of U3 native HIV promoter ensuring expression of Vector RNA. The vector was flanked in 5’ by LTR (U3RU5) and in 3’ by truncated LTR (RU5) ensuring self-inactivation. φ: encapsidation sequence, RRE (Rev Response Element), cPPT/CTS (central polypurin tracts/central termination sequence), WPRE (WHV Posttranscriptional Regulation Element), BGH polyA (bovine Growth Hormone polyadenylation signal). hCD123 corresponded to the full length human CD123. CD123 CAR-chain composition: signal peptide (SP) of mouse Igk; single-chain variable fragment (ScFv) from B4D5 anti-CD123 monoclonal antibody [15]; hinge of human IgG4 and 28BBz backbone [55]. Reporter ΔCD19 was a truncated version of CD19 protein (1-313aa) co-expressed through a P2A cleavage site.
(A) The expression level of promoters in HEK-293T production cell line was measured as Relative Luminescent Unit per Viral Copy Number (RLU/VCN). Columns represent mean ± SEM of 4 different transductions. Significance was determined using a Kruskal-Wallis test (*: P<0.05). (B) The titer of lentiviral vectors encoding CD123-CAR protein under control of different promoters was determined by qPCR after HEK-293T cell transduction and values were expressed as Transduction Unit per ml (TU/ml). qPCR primers targeted U5 region of lentiviral vector LTR and CD3 genes. Standard curve was carried out with a quantified vector plasmid carrying CD3 gene. Columns represent mean ± SEM of 4 different productions Kruskal-Wallis test (*: P<0.05). (C) The titer of lentiviral vector encoding different transgene under the control of CMV or β2m promoter was determined by qPCR. “CAR” refers to CD123 CAR protein, “hCD123” refers to Human CD123 protein, “Luc” to Luciferase Firefly protein, “e GFP” to enhanced Green Fluorescent Protein. Columns represent mean ± SEM of 3 different productions. Significance was determined using Mann-Whitney test (*: P<0.05).
Expression of ΔCD19 in CD8+ T cell population was determined at day 5, 15 and 37 after transduction. (A) The expression level of ΔCD19 under the control of different promoters was studied by flow cytometry and represented by dot plots at different time points (same donor). Populations were previously gated on CD8 expression. (B) The stability of CD8+ ΔCD19+ population over time was calculated as the percentage of ΔCD19+ among CD8+ cells. (C) Expression level was expressed as Median Fluorescent Intensity (MFI). For (B) and (C), columns represent mean ± SEM of 3 different donors, significance was determined using a Kruskal-Wallis test (*: P<0,05; **: P<0,01; ***: P<0,001).
(A) CAR123-T cells were labelled using CD123-biotynilated protein and were analyzed by flow cytometry 12 days post transduction with either MOI 20 or 100. Activated but non-transduced T cells (NT) served as control. (B) A western blot analysis of the expression level of B4D5 CAR in Human T cell was carried out with an anti-CD3z antibody, and β-actin was labelled as a loading control. (C) Different promoter/CAR-T cells were co-incubated 24h with CAL-1 cells at an E: T of 10:1. Cell lysis was determined on the basis of remaining living cells (Viability) using a live/dead marker on CAL-1 cells (CFSE+). Cell death from the same donor was studied by flow cytometry and represented by dot plots. (D) Columns represent mean ± SEM of 3 different donors of % of cell lysis. At day 40, significance was determined using a Kruskal-Wallis test (***: P<0,001; ****: P<0,0001).
(A) CAR123-T cells expression under the control of different promoters (EF1α, MSCV, PGK, β2m and CMV) or non-transduced (NT), before and 12 days post activation was studied by flow cytometry and represented by dot plots. (B) Differentiation phenotype was determined through CD62L and CD45RA cell surface expression, 6 days after transduction and 12 days after being exposed to CAL-1 at a ratio E:T of 10:1. (C) PD-1 expression in CD8+ ΔCD19+ population before and after 12 days of stimulation. Columns represent mean ± SEM of 3 different donors. Significance was determined using a Mann-Whitney test (ns: non-significant).
4. Discussion
In this work, we investigated whether promoters with different activity could modulate CAR-chain production, expression, antigen recognition and subsequent cytotoxic function against the targeted antigen. Several constitutive promoters were tested to
determine their effect on CAR-T cells reactivity against CD123 antigen using our CD123 CAR construct.
Firstly, we evidenced that lentiviral vector titer is affected by transgene nature and promoter activity: type I membrane-bound proteins, such as the CAR construct,
were more deleterious to functional titer of lentiviral vector than intracellular proteins. Moreover, this effect is emphasized by promoter activity: strong promoters like EF1α or CMV generate 20 times lower CAR vector than weak promoters like PGK and β2m. This correlation between promoter strength and transgene expression level was previously shown for different transgenes and in the CAR-T therapy context [22, 23, 37].
We confirm the importance of the promoter’s choice for immunotherapy strategies based on lentiviral vector gene transfer, particularly during large-scale manufacturing process. For CAR expression, a weak promoter is optimal for lentiviral vector in a production cell line. However, concerning primary human T-cells, a strong promoter is required. This was the case with β2m promoter, which has shown low expression levels as PGK promoter in HEK-293T, but almost as strong as EF1α in T-cells. Such result was expected since β2m is a housekeeping protein mainly expressed in immune cells but poorly expressed in HEK-293T, becoming particularly relevant for driving CAR expression, especially in the context of CD123 targeting [30].
Regarding lentivirus production, we highlighted multiples critical points affecting the functional titer. At a transcriptional level, we first demonstrated a correlation between the strength of the promoter in a production cell line and the functional titer. Such correlation was previously determined on a g-retroviral vector. Indeed, the introduction of a tetracycline-inducible promoter that was not functional during production, instead of a constitutive promoter, leads to an increase of the vector functional titer [37]. Secondly, in the plasmid used to encode the RNA vector, both the U3 promoter (from HIV 5’LTR) and the constitutive promoter (driving CAR expression) are functional. Thus, these two transcriptional complexes could compete during the transcription phase for DNA matrix or shared transcription factors. For example, the ubiquitous transcription factor SP1 is critical for both CMV and HIV U3 promoters [38, 39]. This was demonstrated in SIN lentiviral vectors, where the deletion of the U3 promoter in HIV 3’LTR led to an increase of the transgene expression after transduction [40]. Taken together, we can hypothesize that this competition could be present during the production of second-generation lentiviral vector, leading to a potential decrease of RNA vector production. Moreover, at the protein level, we observed that the nature of the transgene could affect the functional titer, probably due to an interference between membrane-located proteins and VSV-G envelope during production. In the case of CAR production, we observed that both type-1 membrane-associated proteins follow the same secretion pathway and are accumulated at the membrane. In the context of a strong CAR expression, VSV-G membrane concentration could be impoverished resulting in a decrease of transduction efficiency and, consequently to a lower functional vector titer.
These two events have critical consequences on lentiviral vector-based CAR therapies, especially for large-scale production. Such optimizations of CAR expression at a transcriptional level can both increase therapy efficiency and reduce the global cost of an already expensive therapy.
CD123 presents a differential level of expression between most of BPDCN blasts and normal hematopoietic cells [11, 41]. In this context, “on-target, off-tumor” toxicity can be deleterious for endothelial cells subsets reported to express low level of CD123 [42]. The lack of tumor specificity can have dramatic consequences: a CAR-T therapy clinical trial with CAR-T cells targeting HER2 (ERBB2), led to patient death 5 days after injection because of the basal target expression on the epithelial cells of the lung [43]. Another example was a CAR targeting GD2 that induced fatal encephalitis on in vivo models of neuroblastoma [44]. Even if the first trial with CAR-T cells specific for CD123 in humans (4SCAR123, NCT03125577) showed a rapid remission with minimal cytokine release syndrome (CRS) and without off-target effect, this safety issue must be addressed.
By decreasing the expression level of the CAR-chain at the cell-surface, the binding capacity of CAR-T cells can be modulated and the safety of CAR therapies could be increased. Our studies show that CD123 CAR-T cells differential binding was directly related to the promoter used to regulate the CAR expression, both by its intrinsic level of expression and by its number of copies, evidenced through the increase of the MOI. Only EF1α promoter was able to generate cells with a high binding potential to CD123 proteins. Furthermore, we observed a decrease in CAR expression overtime that we relate to a transition between activation and resting/memory states. Thus, variation overtime in binding capacity is expected and could dampen CAR-T cells cytotoxicity against targeted tumors. On cytotoxic assays at different time points, only CAR construct under the control of the two strongest promoters tested in this study, β2m and EF1α were able to eradicate CAL-1 cells, both during their activation (day 6) and resting/memory (day 40) states.
Evaluation of CAR-T cells functionality during resting/memory phases is crucial to ensure long-term functionality. Classical CAR-T cells culture protocols require high doses of IL2 (>100U/ml) to sustain strong proliferation and expansion of CAR-T cells to obtain enough cells for clinical application. However, this concentration is not compatible with physiological conditions, altering both their phenotype and their cytotoxicity [45]. Actually, many cytotoxic assays or in vivo experiments are performed rapidly after transduction step when T cells activity is optimal. In our long-term culture experiments, only low doses of IL-2 (50U/ml) were used, and activation/memory state was clearly defined by CAR expression overtime. Here we demonstrate that, following long-term culture, cytotoxicity considerably vary according to the promoter’s nature. Thereby, promoter selection could be crucial to maintain CAR-T cytotoxicity in vivo.
Variations in binding capacity related to the promoter’s nature could also be affected by the phenotype of CAR-T cells after specific activation. Indeed, classical T cell activation through TCR engagement induce a fast T cell differentiation associated with exhaustion profile during strong and persistent TCR engagement [46, 47]. However, after 12 days of CAL-1 cells stimulation, differentiation profile was the same for all promoters used with a majority of CAR-T cells being TEM cells (CD45RA-/CD62L-) [48] without up-regulation of PD-1 at their cell-surface.
New generation of “armored” CARs consist of introducing co-expression strategies with an endogenous production of cytokine to mimic their activation pathway [49, 50]. Activation signaling with high levels of cytokines secretion and enhanced CAR-T cells proliferation can be exacerbated by high cell-surface expression of CAR [51]. These approaches could have a great impact on the surrounding immune cells. Thus, a finer regulation of transgenes expression in the CAR vector could be necessary.
Also, it has been demonstrated that introduction of a CAR in a TRAC locus, that mimics TCR regulation, prevents tonic CAR signaling with a basal low level of expression and allows its modulation after stimulation [48]. Thus, β2m promoter, regulated by inflammation signals like Interferons α/β and γ through ISRE motifs, could ensure a CAR expression profile closer to a natural TCR regulation [28, 29].
Our work demonstrates that not all promoters are suited for CAR-T cells development. EF1α remains a robust choice for further development in the context of CD123 B4D5 CAR-T cells. Nevertheless, regarding production, expression level and binding capacity, β2m promoter should be considered as a fine-tuned approach to address the “on-target off-tumor” issue, using low affinity paratopes [52]. Although this study focuses on the CD123 targeting, our results could also be profitable to other CAR sharing the same targeting problem, such as CD33 on hepatic Kupffer cells [53] or CD44 on keratinocytes [54].
Acknowledgements
We would like to thank Maryline Bourgine for comments that greatly improved the manuscript. This study was supported by Programme de Recherche Translationnelle INCa (PRTK N°PRT-K-15-175).
Authorship Contributions Statement
EBR, FA, JMC, PS and MF performed the experiments; FA and EBR analyzed data and performed statistical analysis; JMC, PS FGO, PC, ML, OA, PLD, JC provided guidance and expertise in their respective areas of study. FA, EBR, ML and JM wrote the manuscript; ML, JC supervised research. All authors provided input, edited, and approved the final version of the manuscript.
Conflict of Interest Disclosures
The authors declare no competing financial interests.
References
- Fuzzi B, Rizzo R, Criscuoli L, et al. HLA-G expression in early embryos is a fundamental prerequisite for the obtainment of pregnancy. Eur J Immunol 32 (2002): 311-315.
- Hartmann J, Shubler-Lenz M, Bondanza A, et al. Clinical development of CAR T cells-challenges and opportunities in translating innovative treatment concepts. EMBO Mol Med 9 (2017): 1183-1197.
- Park JH, MB Geyer, RJ Brentjens. CD19-targeted CAR T-cell therapeutics for hematologic malignancies: interpreting clinical outcomes to date. Blood 127 (2016): 3312-3320.
- Boyiadzis MM, Dhodapkar MV, RJ Brentjens, et al. Chimeric antigen receptor (CAR) T therapies for the treatment of hematologic malignancies: clinical perspective and significance. J Immunother Cancer 6 (2018): 137.
- Larson RC, MV Maus. Recent advances and discoveries in the mechanisms and functions of CAR T cells. Nat Rev Cancer 21 (2021): 145-161.
- Taussig DC, Pearce DJ,Simpson C, et al. Hematopoietic stem cells express multiple myeloid markers: implications for the origin and targeted therapy of acute myeloid leukemia. Blood 106 (2005): 4086-4092.
- Bole-Richard E, Fredon M, Biichle S, et al. CD28/4-1BB CD123 CAR T cells in blastic plasmacytoid dendritic cell neoplasm. Leukemia 34 (2020): 3228-3241.
- Spurny C, Kailayangiri S, Altvater B, et al. T cell infiltration into Ewing sarcomas is associated with local expression of immune-inhibitory HLA-G. Oncotarget 9 (2018): 6536-6549.
- Du X, M Ho, I Pastan. New immunotoxins targeting CD123, a stem cell antigen on acute myeloid leukemia cells. J Immunother 30 (2007): 607-613.
- Garnache-Ottou F, Feuillard J, Ferrand C, et al. Extended diagnostic criteria for plasmacytoid dendritic cell leukaemia. Br J Haematol 145 (2009): 624-636.
- Testa U, E Pelosi, A Frankel. CD 123 is a membrane biomarker and a therapeutic target in hematologic malignancies. Biomark Res 2 (2014): 4.
- Gill S, Tasian SK, Ruella M, et al. Preclinical targeting of human acute myeloid leukemia and myeloablation using chimeric antigen receptor-modified T cells. Blood 123 (2014): 2343-2354.
- Mardiros A, Santos CD, Mc Donald T, et al. T cells expressing CD123-specific chimeric antigen receptors exhibit specific cytolytic effector functions and antitumor effects against human acute myeloid leukemia. Blood 122 (2013): 3138-3148.
- Pizzitola I, F Anjos-Afonso, K Rouault, et al. Chimeric antigen receptors against CD33/CD123 antigens efficiently target primary acute myeloid leukemia cells in vivo. Leukemia 28 (2014): 1596-1605.
- Bole-Richard E, Fredon M, Biichle S, et al. CD28/4-1BB CD123 CAR T cells in blastic plasmacytoid dendritic cell neoplasm. Leukemia (2020).
- Cavazzana-Calvo M, Hacein-Bey C, de Saint Basile G, et al. Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science 288 (2000): 669-672.
- Cartier N, Hacein-Bey-Abina S, Bartholomae CC, et al. Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy. Science 326 (2009): 818-823.
- Hacein-Bey-Abina S, Sung-Yun P, Bobby Gaspar H, et al. A modified gamma-retrovirus vector for X-linked severe combined immunodeficiency. N Engl J Med 371 (2014): 1407-1417.
- Hacein-Bey-Abina S, Garrigue A,Wang GP, et al. Insertional oncogenesis in 4 patients after retrovirus-mediated gene therapy of SCID-X1. J Clin Invest 118 (2008): 3132-3142.
- Mitchell RS, Beitzel BF, Schroder RW, et al. Retroviral DNA integration: ASLV, HIV, and MLV show distinct target site preferences. PLoS Biol 2 (2004): E234.
- Hematti P, Bum-Kee H, Ferguson C, et al. Distinct genomic integration of MLV and SIV vectors in primate hematopoietic stem and progenitor cells. PLoS Biol 2 (2004): e423.
- Rad SMA, Poudel A, Tan GMY, et al. Promoter choice: Who should drive the CAR in T cells? PLoS One 15 (2020): e0232915.
- Ramezani A, TS Hawley, RG Hawley. Lentiviral vectors for enhanced gene expression in human hematopoietic cells. Mol Ther 2 (2000): 458-469.
- Jones S, Peng PD, Yang S, et al. Lentiviral vector design for optimal T cell receptor gene expression in the transduction of peripheral blood lymphocytes and tumor-infiltrating lymphocytes. Hum Gene Ther 20 (2009): 630-640.
- Qin JY, zhang L, Clift KL, et al. Systematic comparison of constitutive promoters and the doxycycline-inducible promoter. PLoS One 5 (2010): e10611.
- Swindle CS, HG Kim, CA Klug. Mutation of CpGs in the murine stem cell virus retroviral vector long terminal repeat represses silencing in embryonic stem cells. J Biol Chem 279 (2004): 34-41.
- Weber EL, PM Cannon. Promoter choice for retroviral vectors: transcriptional strength versus trans-activation potential. Hum Gene Ther 18 (2007): 849-860.
- Gobin SJ, Peijnenburg A,Keijesers V, et al. Site alpha is crucial for two routes of IFN gamma-induced MHC class I transactivation: the ISRE-mediated route and a novel pathway involving CIITA. Immunity 6 (1997): 601-611.
- Gobin SJ, P Biesta, PJ Van den Elsen. Regulation of human beta 2-microglobulin transactivation in hematopoietic cells. Blood 101 (2003): 3058-3064.
- Fagerberg L, Hallstrom BM, Kampf C, et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol Cell Proteomics 13 (2014): 397-406.
- Zennou V, Serguera C, sarkis C, et al. The HIV-1 DNA flap stimulates HIV vector-mediated cell transduction in the brain. Nat Biotechnol 19 (2001): 446-450.
- Gussow D, Rein R, Ginjaar I, et al. The human beta 2-microglobulin gene. Primary structure and definition of the transcriptional unit. J Immunol 139 (1987): 3132-3138.
- Coutant F, et al. Protective antiviral immunity conferred by a nonintegrative lentiviral vector-based vaccine. PLoS One 3 (2008): p. e3973.
- Beltrame MH, Catarino SJ, Goeldner I, et al. The lectin pathway of complement and rheumatic heart disease. Front Pediatr 2 (2014): 148.
- Aleksandrova K, Leise J, Priesner C, et al. Functionality and Cell Senescence of CD4/ CD8-Selected CD20 CAR T Cells Manufactured Using the Automated CliniMACS Prodigy(R) Platform. Transfus Med Hemother 46 (2019): 47-54.
- Peranzoni E, Lemonie J, Vimeux L, et al. Macrophages impede CD8 T cells from reaching tumor cells and limit the efficacy of anti-PD-1 treatment. Proc Natl Acad Sci U S A 115 (2018): E4041-E4050.
- Schambach A, Mueller D, Galla M, et al. Overcoming promoter competition in packaging cells improves production of self-inactivating retroviral vectors. Gene Ther 13 (2006): 1524-1533.
- Yurochko AD, Hwang ES, Rasmussen L, et al. The human cytomegalovirus UL55 (gB) and UL75 (gH) glycoprotein ligands initiate the rapid activation of Sp1 and NF-kappaB during infection. J Virol 71 (1997): 5051-5059.
- Yurochko AD, Mayo MW, Poma EE, et al. Induction of the transcription factor Sp1 during human cytomegalovirus infection mediates upregulation of the p65 and p105/p50 NF-kappaB promoters. J Virol 71 (1997): 4638-48.
- Zufferey R, Dull T, Mandel RJ, et al. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery. J Virol 1998. 72 (1998): 9873-9880.
- Munoz L, Nomdedeu JF, Lopez O, et al. Interleukin-3 receptor alpha chain (CD123) is widely expressed in hematologic malignancies. Haematologica 86 (2001): 1261-1269.
- Gilliet M, Conrad C, Geiges M, et al. Psoriasis triggered by toll-like receptor 7 agonist imiquimod in the presence of dermal plasmacytoid dendritic cell precursors. Arch Dermatol 140 (2004): 1490-1495.
- Morgan RA, Yang JC, Kitano M, et al. Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol Ther 18 (2010): 843-851.
- Richman SA, Nunez-Cruz S, Moghimi B, et al. High-Affinity GD2-Specific CAR T Cells Induce Fatal Encephalitis in a Preclinical Neuroblastoma Model. Cancer Immunol Res 6 (2018): 36-46.
- Kaartinen T, Luostarinen A,Maliniemi P, et al, Low interleukin-2 concentration favors generation of early memory T cells over effector phenotypes during chimeric antigen receptor T-cell expansion. Cytotherapy 19 (2017): 1130.
- Schietinger A, PD Greenberg. Tolerance and exhaustion: defining mechanisms of T cell dysfunction. Trends Immunol 35 (2014): 51-60.
- Wherry EJ, M Kurachi. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol 15 (2015): 486-499.
- Eyquem J, Mansilla-Soto J, Giavridis T, et al. Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection. Nature 543 (2017): 113-117.
- Yeku OO, Purdon TJ,Koneru M, et al. Armored CAR T cells enhance antitumor efficacy and overcome the tumor microenvironment. Sci Rep 7 (2017): 10541.
- Shum T, Omer B,Tashiro H, et al. Constitutive Signaling from an Engineered IL7 Receptor Promotes Durable Tumor Elimination by Tumor-Redirected T Cells. Cancer Discov 7 (2017): 1238-1247.
- Frigault MJ, Lee J, Basil MC, et al. Identification of chimeric antigen receptors that mediate constitutive or inducible proliferation of T cells. Cancer Immunol Res 3 (2015): 356-367.
- Drent E, Themeli M, Poels R, et al. A Rational Strategy for Reducing On-Target Off-Tumor Effects of CD38-Chimeric Antigen Receptors by Affinity Optimization. Mol Ther 25 (2017): 1946-1958.
- Sievers EL, Larson RA, Stadtmauer EA, et al. Efficacy and safety of gemtuzumab ozogamicin in patients with CD33-positive acute myeloid leukemia in first relapse. J Clin Oncol 19 (2001): 3244-3254.
- Casucci M, Nicolis di Robilant B, Falcone L, et al. CD44v6-targeted T cells mediate potent antitumor effects against acute myeloid leukemia and multiple myeloma. Blood 122 (2013): 3461-3472.
- Zhong XS, Matsushita M, Plotkin J, et al. Chimeric antigen receptors combining 4-1BB and CD28 signaling domains augment PI3kinase/AKT/Bcl-XL activation and CD8+ T cell-mediated tumor eradication. Mol Ther 18 (2010): 413-420.
Supplementary Material
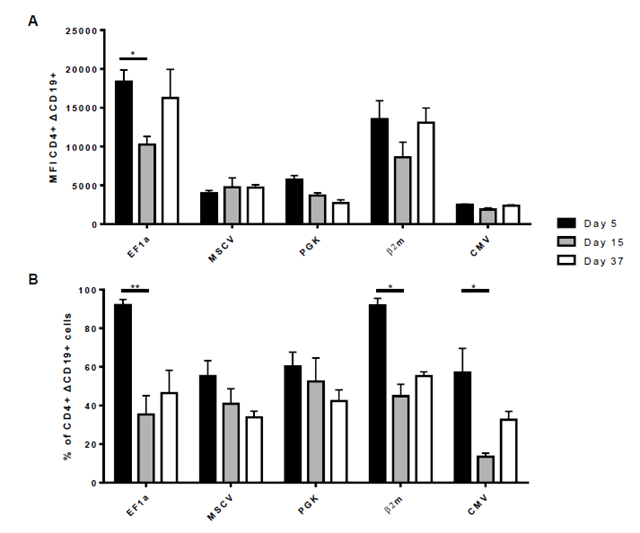
Expression of ΔCD19 in CD4+ T cell population was determined at day 5, 15 and 37 after transduction. (A) Expression
level was expressed as Median Fluorescent Intensity (MFI). (B) The stability of CD8+ ΔCD19+ population over time was calculated as the percentage of ΔCD19+ among CD8+ cells. For (A) and (B), columns represent mean ± SEM of 3 different donors, significance was determined using a Kruskal-Wallis test (*: P<0,05; **: P<0,01).
Percentages of CD3+ CD4+ and CD3+ CD8+ T cells were determined at day 5, 15 and 37 after transduction.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 