Interactions Between Adenosine Receptors and Cordycepin (3'- Deoxyadenosine) from Cordyceps Militaris: Possible Pharmacological Mechanisms for Protection of the Brain and the Amelioration of Covid-19 Pneumonia
Jing Du1,2*, Weijing Kan1, Hongkun Bao2, Yue Jia2, Jian Yang1, Hongxiao Jia1
1The National Clinical Research Center for Mental Disorders and Department of Chinese Traditional Medicine, Beijing Anding Hospital, and Advanced Innovation Center for Human Brain Protection, Capital Medical University, Beijing, 100088, China
2Yunnan University, School of Medicine, 2 Cuihu North Road, Kunming, Yunnan, 650091, China
*Corresponding Author: Jing Du, The National Clinical Research Center for Mental Disorders, Beijing Anding Hospital, Capital Medical University, Beijing, 100088, China.
Received: 26 May 2021; Accepted: 04 June 2021; Published: 21 June 2021
Article Information
Citation:
Jing Du, Weijing Kan, Hongkun Bao, Yue Jia, Jian Yang, Hongxiao Jia, Interactions Between Adenosine Receptors and Cordycepin (3-Deoxyadenosine) from Cordyceps Militaris: Possible Pharmacological Mechanisms for Protection of the Brain and the Amelioration of Covid-19 Pneumonia. Journal of Biotechnology and Biomedicine 4 (2021): 26-62.
DOI: 10.26502/jbb.2642-91280035
View / Download Pdf Share at FacebookAbstract
At present, the novel Covid-19 pneumonia is prevalent, affecting millions of people. Here, we summarized the pharmacological basis of adenosine, adenosine receptors, adenosine agonist cordycepin (3'-deoxyadenosine), and Cordyceps product in the brain protection and amelioration of pneumonia to provide useful information to cope with the global pandemic of novel coronavirus (COVID-19). Adenosine, a mediator of innate immunity, is abundantly secreted by the injured lung tissues during inflammation. Through the activation of adenosine receptors A1, A2A, A2B and A3, adenosine plays an important role in protecting against acute lung injury and brain injury. Cordycepin (3-deoxyadenosine) is an activator of adenosine receptors. It can enhance human immunity, promote anti-inflammatory processes, inhibit RNA virus reproduction, protect against brain, lung, liver, heart, and kidney damage, and ameliorate lung-fibrosis in clinical and animal models. Cordyceps and cordycepin products could be used as a potential medicinal adenosine receptor agonist that can play a beneficial role in the amelioration of Covid-19 pneumonia and protection of brain.
Keywords
<p>Adenosine receptor; Brain protection; Covid-19; Cordycepin; pneumonia</p>
Article Details
Abbreviations:
ARDS- Acute respiratory distress syndrome; ATP- Adenosine triphosphate; AMP- Adenosine monophosphate; Aβ- Beta amyloid protein; ALI- Acute lung injury; A1R- Receptor; A2AR- A2A receptor; AD- Alzheimer's disease; ADA- Adenosine deaminase; AMPA- Aminomethyl phosphonic acid; Akt/PKB- Protein Kinase B; BLM- bleomycin; Covid-19- Coronavirus disease 2019; CAMP- Cyclic Adenosine monophosphate; COX-2- Cyclooxygenase-2; CCL2- Chemokine (C-C motif) ligand 2; CNS- Central nervous system; DNA- Deoxyribonucleic acid; DA- Dopamine; GSH-Pox- glutathione peroxidase; IL-1β- Interleukine-1 beta; IL-6- Interleukin-6; IFN-γ- γ-Interferon; IL-2- Interleukin-2;IL-4- Interleukin-4; IL-6- Interleukin-6; IL-8- Interleukin-8; IL-10- Interleukin-10; IL-12- Interleukin-12; IL-13- Interleukin-13; IgA- Immunoglobulin A; IgM- Immunoglobulin M; IgG- Immunoglobulin G; IBO- Ibotenic acid; INOS- Inducible nitric oxide synthase; LPS- Lipopolysaccharides; mRNA- Messenger RNA; MIP-1α-Macrophage inflammatory protein-1α; MIP-2- Macrophage inflammatory protein-2; MDD- Major depression disease; MCP-1- Monocyte chemotactic protein 1; MPTP- Methyl 4-phenyl 1- 2- 5-6 tetrahydropyridine; NF-κB- Nuclear factor kappa-B; NK- Natural killer; 6-OHDA- 6-Hydroxydopamine Hydrobromide; PI3K- Phosphatidylinositol 3-kinase; PD- Parkinson's disease; p-Tau- protein Tau; PI- Proliferation index; PD- Parkinson's Disease; RNA- Ribonucleic acid; SOD- Superoxide dismutase; SARS-CoV-2- Severe acute respiratory syndrome coronavirus 2;TNF-β- Tumor Necrosis Factor-β; TLR- Toll-like receptors; TGF-β- Transforming growth factor-β; Th1- T helper cell 1; TNF-α- Tumor necrosis factor α; TAA- Thioacetamide.
1. Introduction
At present, there exists a global urgency in identifying supportive medication for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the disease caused by Covid-19. This review discusses the adenosine receptor-mediated pharmacological effects of Cordyceps and cordycepin on acute and chronic pneumonia and the subsequent organ damage.
Adenosine is produced in injured lung tissues and plays multiple roles in the regulation of inflammation and tissue remodeling. Adenosine acts as an anti-inflammatory molecule through suppressing the production of cytokine storm, protecting against organ damage, and repairing damaged tissue from acute lung diseases. The activation of adenosine receptors A1, A2A, A2B, and A3 benefits the recovery of lung diseases and is of great significance for the amelioration of pneumonia [1]. Cordycepin (3’-deoxyadenosine), the most important active ingredient in Cordyceps militaris or Cordyceps sinensis, is proposed as an agonist of adenosine receptors. The "Pharmacopoeia of the People's Republic of China" notes the ability of Cordyceps as an herb “to nourish the lungs and kidney, stop bleeding, and reduce phlegm” [2]. Cordyceps as a family of eatable mushroom have been found mainly in North America, Europe, and Asia [3], and have a history of medicinal use spanning millennia in Asia [4]. However, exploitation of Cordyceps has significantly reduced its wild occurrence [5], the manufacturers make efforts to artificially cultivate this mushroom by surface and submerged fermentation techniques.
In this article, we give a detailed and objective review of research on Cordyceps and cordycepin and on their interactions with adenosine receptors for the prevention and amelioration of acute and chronic pneumonia, such as that observed in Covid-19. We discuss cordycepin’s ability to 1) enhance human immunity in the lung; 2) inhibit virus replication; 3) exert anti-inflammatory, reparative, and regenerative effects; 4) inhibit cytokine storm; 5) protect the brain, lung, liver, heart, and kidney; and 6) protect against pulmonary fibrosis.
2. Efficacy and chemical structure of cordycepin
Cordycepin’s chemical name is 3’-deoxyadenosine (Figure 1). It has a molecular weight of 251.24kD and is soluble in water and ethanol. The rotation of natural cordycepin is unique and determines its efficacy. Most of the bioactive cordycepin products on the market are produced biosynthetically from Cordyceps militaris. Cordycepin is higher in Cordyceps militaris, up to approximately 0.12% [6]. Cordycepin in Cordyceps militaris that is artificially cultivated by some manufacturers can reach up to 1-3%.
2.1. Cordycepin is a specific activator of adenosine receptors
Adenosine acts in anti-bacterial, anti-viral, anti-neoplastic, and immune repair and recovery mechanisms [7]. The family of adenosine receptors includes four members: adenosine receptors A1, A2A, A2B, and A3 [7]. Adenosine is a metabolite of the energy metabolism pathway of ATP and is an important marker of the body's energy exhaustion. Adenosine activates sleep, immune system, tissue repair, and energy regeneration. Currently, there are reports that cordycepin and adenosine receptor subtypes such as A1, A2A, A2B, and A3 can interact to promote anti-inflammatory effects and cell repair, as well as to protect the lung, liver, kidney, heart, and brain [8-10]. Adenosine has high affinity for adenosine receptor subtypes A1 and A2A and a low affinity for A2B and A3 [1]. Cordycepin, however, has good affinity for all four subtypes [8-10].
A growing body of studies showed that the interaction of cordycepin with four types of adenosine receptors was shown in various organs and cell lines. Cordycepin could induce apoptotic cell death in a couple of tumor cell lines, including a mouse Leydig tumor cell line MA-10 and a follicular thyroid carcinoma cell line CGTH W-2. In both tumor cell lines the specific antagonists to four AR subtypes A1AR, A2AAR, A2BAR, and A3AR, all blocked cordycepin-induced apoptosis to different degrees [11, 12]. Cordycepin also stimulated mouse Leydig cell testosterone production, regulated the mRNA expression of the A1, A2A, A2B, and A3 adenosine receptors, and that antagonists of A1, A2A, and A3 suppressed 20-50% testosterone production in the mouse Leydig cells [13]. In the CNS, cordycepin reduced sleep-wake cycles and increased non-rapid eye movement (NREM) sleep, and the protein levels of AR subtypes (A1, A2A, and A2B) were increased after the administration of cordycepin in the rat hypothalamus [8]. In addition, cordycepin remarkably alleviated LTP impairment and protected pyramidal cell of hippocampal CA1 region against cerebral ischemia and excitotoxicity, and the effect was blocked by A1 specific antagonist DPCPX (8-cyclopentyl-1, 3-dipropylxanthine) [14]. Several additional studies also showed that cordycepin exerts neuroprotective effect through activation of A1, and the neuroprotective effects of cordycepin were blocked by DPCPX [15, 16]. Furthermore, it has been shown that the anti-tumor, anti-inflammation, and anti-fibrosis effects of cordycepin were mediated through A3 receptors [9, 17-19].
Adenosine receptor A1 is widely expressed throughout the body, but its highest level of expression is in the brain, especially at the excitatory nerve endings. This receptor regulates the activity of neurons and reduces the firing rate by blocking the release of neurotransmitters, protecting the brain, regulating sleep, and protecting the heart muscle when the blood oxygen concentration decreases and during myocardial ischemia [7].
The A2A subtype is expressed in many organs and cells, such as the striatum, spleen, thymus, heart, lung, blood, white blood cells, and platelets. A2A receptors play a regulatory role in peripheral tissues, in brain during exercise, mental behavior, sleep, and others, and in controlling inflammation, myocardial oxygen consumption, coronary blood flow, and angiogenesis in cancer and other diseases [7].
The adenosine receptor A2B subtype is widely expressed in vivo and is found in almost all organs, but its expression level is low. Under the condition of elevated adenosine levels, such as in hypoxia and ischemia, it has a certain protective effect on organs and tissues. Therefore, the tissue can survive without oxygen [7].
In normal tissues, the adenosine receptor A3 subtypes are mainly distributed in the brain, lung, liver, aorta, testis, and heart. After activation of the adenosine receptor A3 subtype, it mainly plays a protective role in the tissues in which it resides [7, 20-23].
The expression level of the adenosine receptor A3 subtypes in cancer tissues and inflammatory tissues is extraordinarily high. A large number of adenosine receptor A3 subtypes are activated by cordycepin in inflammatory tissues and have a good effect on eliminating inflammation, including cytokine storm. It is worth noting that cordycepin, with its high affinity for the adenosine receptor A3 subtypes, plays an important role in the anti-inflammatory protection of organs [24, 25]. A large number of adenosine receptors A3 can cause cancer cell apoptosis after cancer cell activation, which is one of the main drug mechanisms of cordycepin's anti-inflammatory and anti-tumor properties [9, 10, 26].
2.2. Cordycepin is a strong antioxidant
Studies have shown that a low concentration of cordycepin can effectively inhibit the oxidation reaction of free radicals. During the process of viral pneumonia, super-oxidative free radicals can damage mitochondrial functions and cause mitochondrial dysfunction. This results in damage to lung cells during acute respiratory failure, which are important causes of lung dysfunction [27]. Therefore, cordycepin has a scavenging effect on free radicals and can delay, inhibit, and block the oxidative damage of active oxygen/oxygen-free radicals to protect mitochondrial function and cells and tissues from oxidative damage [16, 28, 29].
2.3. Cordycepin can selectively inhibit the formation of messenger RNA polynucleotide A chains
Cordycepin, which is also named 3’-deoxyadenosine, can be phosphorylated to generate 3’-deoxytriphosphateadenosine. This molecule can interact with RNA polymerase to stop the synthesis of polyadenylated RNA strands, which is important for inhibiting RNA viruses (Figure. 2).
Cumulative studies have confirmed that cordycepin effectively inhibited the replication and reproduction of a variety of RNA and DNA viruses. Such RNA viruses include influenza virus [30], poliovirus [31], rhinovirus [32], Epstein-Barr virus [33], hepatitis C virus (HCV) [34], Hantaan virus [35], picornavirus [36], type-c RNA tumor viruses [37], Semliki Forest virus [38], western equine encephalitis virus [39] and others. Cordycepin can also inhibit the proliferation of DNA viruses, such as herpes simplex virus [40], vaccinia virus [41] and others. The effect of cordycepin on the virus that causes Covid-19 has not yet been reported. However, since cordycepin has an inhibitory effect on the reproduction of many RNA viruses, it is likely to also to effectively inhibit this new virus, as the replication and reproduction mechanisms of RNA viruses in humans are nearly the same for all viruses.
Cordycepin blocks the reproduction of RNA viruses because the RNA replication of many viruses requires a polyadenylated (poly A) tail. Cordycepin, or 3’-deoxyadenosine, lacks a hydroxyl group in the 3’ position, which allows it to interfere with the elongation of the poly A tail of the RNA [31, 36, 38] to block RNA virus replication. The Covid-19 coronavirus is a positive, single-stranded RNA virus with a polyadenylic acid tail, similar to poliovirus, which experiments have shown that its reproduction can be inhibited by cordycepin [31]. Cordycepin inhibits virus replication and synthesis, which is of great significance for the amelioration of viral pneumonia (Figure. 2).
3. Adenosine receptors and Cordyceps enhance the functioning of the human immune system
3.1 Adenosine increase is an important natural mechanism to relieve lung inflammation
In patients with lung disease and in animal models of lung injury, adenosine levels in the lungs increase significantly [42-46]. During the progression of acute lung injury, adenosine elevation in injured lung tissue plays an important anti-inflammatory role. Both lung tissue and epithelial cells express adenosine receptors that activate G-proteins that cause changes in the intracellular cAMP and Ca2+ levels [47-49]. The key immune cells associated with lung disease include lymphocytes, neutrophils, dendritic cells, and macrophages. These cells all express adenosine receptors and are involved in regulating various aspects of the innate and adaptive immunity [50, 51].
Elevated adenosine levels during acute tissue injury often have anti-inflammatory and tissue-protective effects. Under normal circumstances, the concentration of adenosine in extracellular fluid varies from 40-600 nM [52]. It is noteworthy that in acute pathological conditions, such as sepsis or ischemia, patients can have adenosine concentrations as high as 10 μM [53]. In chronic diseases such as arthritis [54], asthma, and chronic obstructive pulmonary disease [43], concentrations of adenosine reach 100 μM. In the case of tissue damage, the main source of extracellular adenosine comes from the breakdown of released adenine nucleotides [46, 55] or from infiltrating inflammatory cells such as mast cells [56], eosinophils [57], neutrophils [58] and others. These nucleotides are subsequently dephosphorylated to AMP by exonucleoside triphosphate diphosphate hydrolase CD39 [59], and AMP is subsequently dephosphorylated to adenosine by exonucleoside 5'-nucleotidase CD73 [60]. Increases in CD39 and CD73are often observed in inflammatory states [61].
3.2 Cordyceps or cordycepin enhances the immune cell function of lymphocytes and monocytes
An increasing number of studies have shown that cordycepin can enhance immunity [62-67]. Studies have shown that cordycepin can enhance the proliferation and secretion of T and B lymphocytes. Cordycepin can enhance the function of T lymphocytes, regulate effect of T cells, and release immune-active lymphokines: interleukins, interferon, and others [68, 69]. Lymphokines mostly exert their immune effect by strengthening the action of various related cells. Cordycepin directly induced a proliferation response in B lymphocytes or amplify and regulate the response of B lymphocytes. Cordycepin also promoted the secretion of γ-interferon (IFN-γ), which is a highly effective anti-viral substance with extensive immune-regulatory effects [70], the enhancement and enlargement of B lymphocytes, and the strengthening of the body's resistance to bacteria, viruses, and other harmful substances.
Cordycepin enhanced the activity of natural killer (NK) cells and the phagocytic index of monocytes [68, 69, 71-73]. NK cells synthesize and secrete a variety of cytokines, exerting a role in regulating immune functions, and directly killing target cells [68, 71]. Cordycepin also increased the NK activity and IL-2 secretion in mouse spleen cells and can enhance the secretion of Tumor Necrosis Factor-β (TNF-β) by human tonsil-activated T cells [74]. In macrophages, cordycepin transformed their pro-inflammatory M1 status into an anti-inflammatory M2 status, which plays a role in cell protection and repair [75].
Kang et al. evaluated the effect and safety of Cordyceps militaris on the cellular immune function of 79 healthy adult men [64]. These subjects took equal amounts of Cordyceps militaris or placebo capsules for 4 consecutive weeks. After treatment, NK cell activity, the lymphocyte proliferation index (PI), and T helper cell 1 (Th1) cytokines (including IFN-γ, interleukin-12, interleukin-2, and tumor necrosis factor α) were measured at weeks 0, 2, and 4. Compared with the placebo, NK cell activity (P=0.0010), lymphocyte PI (P ≤ 0.0001), IL-2 (P=0.0096), and IFN-γ (P=0.0126) increased significantly in the Cordyceps militaris-treated group. Thus, Cordyceps militaris enhanced NK cell activity and lymphocyte proliferation, partially increased Th1 cytokine secretion, and was safe and effective for improving cellular immunity in healthy adult males [64].
4. Role of adenosine receptor and Cordyceps in acute lung injury
4.1 Protective role of activated adenosine receptors A2A and A2B in acute lung injury
Acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) may be caused by pneumonia (including viral pneumonia), acid inhalation, severe trauma, or prolonged mechanical ventilation [76]. Experimental and clinical studies have shown that the pathogenesis of ALI and ARDS is characterized by the massive production of inflammatory cytokines and the transport of inflammatory neutrophils into the lung [77].
Extracellular adenosine has an important anti-inflammatory effect in acute lung injury. Pharmacological and genetic studies have shown that the adenosine receptor A2A is the major signaling pathway that mediates the anti-inflammatory properties of adenosine in LPS-induced lung injury [78]. Up-regulation of A2A may improve the healing process after acute LPS-induced lung injury [79]. A2A also down-regulated the expression of IL-12 [80], which in turn promoted the development of an anti-inflammatory cytokine environment that stimulates repair. A recent study showed that inhalation of the selective adenosine receptor A2A agonist ATL202 reduced LPS-induced neutrophil migration, microvascular permeability, and chemokine release, making it a possible clinical amelioration for acute lung injury amelioration [81].
Studies have also shown that the adenosine receptor A2B mediates adenosine protection in acute lung injury models and that A2B agonists can reduce lung injury. Measurements of alveolar fluid clearance indicated that the activation of adenosine A2B receptors enhanced alveolar fluid transport clearance after hypoxia, suggesting that A2B agonist amelioration was accomplished by promoting fluid clearance in the lung and protecting the lung barrier [61, 82, 83]. It has also been reported that A2B receptor activation had a tissue protective effect on ischemic lung injury [84]. Taken together, these studies indicate that adenosine signaling via A2B plays important roles in decreasing inflammation, clearing alveolar fluid, and protecting lung tissue in ALI. Clinically, doctors recommend inhaled A2B agonists for the amelioration of acute lung injury [85].
It is noteworthy that althoughA2BAR signaling serves important anti-inflammatory functions in acute lung injury [83], however, in chronic lung diseases multiple studies demonstrated the ability of A2BAR engagement to promote the expression of pro-inflammatory mediators from various cell types [86]. Moreover, increases in A2BAR have been described as a feature of individuals with accelerated pulmonary fibrosis, suggesting A2BAR antagonists may have utility in the treatment of chronic lung diseases where fibrosis was a major component [87]. Therefore, the timing of A2BAR agonist or antagonist treatment is very important.
In short, the anti-inflammatory effect of the adenosine receptor A2A and the effect of the A2B receptor on the clearance of alveolar fluid and protection of the lung barrier are very beneficial for acute lung injury. Cordycepin, as an activator of both these receptors, may also have a beneficial effect on acute lung injury. Currently, clinical trials targeting these receptors have entered the second phase.
4.2 Activation of the adenosine receptor A3 resists the formation of cytokine storm
Acute inflammation is triggered when virus-infected cells are apoptotic or necrotic, which is characterized by directing plasma and leukocytes to the site of injury outside the blood vessel and activating pro-inflammatory cytokines or chemokines [88, 89]. These cytokine and chemokine signals led to the accumulation of inflammatory cells, increased expression of inflammatory, antiviral, and apoptotic genes, and immune cell infiltration and tissue damage [90, 91]. At the same time, the regeneration process and the recovery of the injury began. In most cases, this repair process completely restored lung function [80, 88, 89]. However, when cytokine storms occur, severe pathological changes can be observed, such as diffuse alveolar injury, transparent membrane formation, fibrin exudation, and fibrotic healing.
Cytokine storm has the potential to result in multi-organ dysfunction. The release of inflammatory cytokines enhances the immune response, activates immune cell proliferation, and further secretes inflammatory cytokines. This series of events leads to a cycle between inflammatory cytokines and immune cells, which can potentiate a cytokine storm [88]. A severe cytokine storm has significantly higher levels of pro-inflammatory cytokines, especially tumor necrosis factor-alpha (TNF-α), interleukin 1β (IL-1β), and interleukin 6 (IL-6). Studies have shown that cytokine storms are at least partially IL-6-mediated [92-94].
A3 activation effectively inhibited the production of IL-6 and IL-8 [95, 96]. It also led to the inhibition of PI3K/Aktto cause powerful anti-inflammatory effects [97]. The use of A3 adenosine receptor agonists for lung injury significantly reduced the levels of TNF-α, IL-1b, IL-6, and IL-12, as well as immune cell infiltration [98]. This may play an important role in the regulation of cytokine secretion. Phase I and II clinical data showed that the highly selective A3AR agonists namodenoson and piclidenoson have good safety and pharmacokinetics profiles [99-101]. This suggests that they may possibly replace hormones and become candidates for the amelioration of inflammatory factor storms. Moreover, the A3R plays complex roles in inflammation, with both pro- and anti-inflammatory functions being described in multiple cellular and animal models with varying roles being dictated largely by species differences [86]. It is likely that the usefulness of A3AR agonists and antagonists in the treatment of acute lung diseases will only be revealed following appropriate clinical trials with such compounds [1].
4.3 Cordycepin as an adenosine receptor agonist improves respiratory tract inflammation
The protective effect of Cordyceps sinensis extract on experimental LPS-induced acute lung injury mice was studied by Fu and colleagues. This study demonstrated that giving Cordyceps sinensis extract (10, 30, 60 mg/kg) to mice 4-6 hours after LPS injection significantly reduced the number of total cells, neutrophils, and macrophages in bronchoalveolar lavage fluid (P <0.05). Additionally, Cordyceps could significantly reduce the increase of TNF-α, IL-1β, IL-6 and NO levels after LPS in bronchoalveolar lavage fluid (P <0.05). Cordyceps sinensis extract also significantly reduced the protein and mRNA levels of iNOS and COX-2 and the NF-κBp65 DNA-binding ability in the LPS group (P <0.05) [102].
Cordycepin, as an A3 adenosine receptor agonist, achieves its anti-inflammatory effects by inhibiting the expression of pro-inflammatory cytokines. Studies have shown that cordycepin inhibited the production of NO and pro-inflammatory cytokines (IL-1β, IL-6, TNF-α) in macrophages from LPS-induced animals [75]. Cordycepin also increased the expression of the anti-inflammatory interleukin-10 (IL-10) in human peripheral blood mononuclear cells to play an anti-inflammatory repair role [69]. Cordycepin inhibited NF-κB function, thereby attenuating tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), IL-12, and macrophage inflammatory protein-1α (MIP-1α). Cordycepin enhanced the expression of MIP-2, which has the effect of inhibiting autoimmune inflammation [78, 81].
Zheng and colleagues studied the clinical effect of Cordyceps sinensis in preventing and treating respiratory infections in elderly patients. Eighty patients were randomly divided into three groups: 30 in the Cordyceps sinensis decoction group (stewed twice a week, 10g/dose), 26 in the Cordyceps powder group (1.5g/dose, 2 times/day), and 24 in the levamisole control group. The groups received their respective ameliorations for 2 months. The statistical analysis of the immunoglobulin levels of the Cordyceps sinensis group showed that levels of immunoglobulin IgG, IgA, and IgM were better than the control group [103]. These studies have laid the foundation for the possible application of Cordyceps or cordycepin in acute lung injury.
5. Adenosine receptors and cordycepin from Cordyceps showed a brain-protection efficacy
5.1 The role of adenosine receptors in brain protection
Studies have shown that adenosine is an essential neuro-modulatory molecule in the brain and plays an important role in multiple physiological and pathophysiological processes [104, 105]. Adenosine exerts its effects throughout the brain through a family of four G protein-coupled adenosine receptors, A1, A2A, A2B, and A3 [106]. These receptors can affect crucial processes such as normal neuronal signaling [107, 108], astrocytic function [109-111], learning and memory [107, 112-114], motor function [115], feeding [116], control of sleep [117], and normal aging processes [114, 118, 119]. Of the four adenosine receptors, the A1 receptor and A2A receptor are both highly expressed throughout the brain, and their effects in the brain have been extensively studied [112, 120].
A1 receptors, which have high affinity for adenosine, are distributed both pre- and postsynaptically in the brain. When presynaptically localized, they specifically inhibited the release of excitatory and/or inhibitory neurotransmitters, e.g., glutamate, dopamine, serotonin, and acetylcholine [105]. When situated postsynaptically, A1 receptors inhibited neuronal signaling by hyperpolarization and reduced excitability via the regulation of potassium channels. The potential role of A1receptor in protecting against brain damage from ischemia was investigated in terms of its ability to control calcium, glutamate release, membrane potential, and metabolism after ischemic damage [121-124]. Cumulative studies have shown that A1receptor was enriched in excitatory synapses, where it inhibited glutamate release and decreased glutamatergic responsiveness and the hyperpolarization of neurons to reduce the hyperexcitability associated with epilepsy [125, 126].
A2A receptors are highly expressed on striato-pallidal neurons, with a lower presence in other parts of the brain such as the cortex and the hippocampus. These receptors can form heteromers with A1 receptors [127-129] and with dopamine D2 receptors [130], which enabled adaptive responses in the regulation of synaptic plasticity [131]. Adenosine A2B and A3receptors may play a protective role in brain ischemia [132] and excitotoxicity [133]. Extracellular adenosine concentrations in the brain are determined by the hydrolysis of ATP released from neurons or astrocytes and by transport through equilibrative nucleoside transporters [134]. Under neuropathological conditions (e.g., ischemia, trauma, excitotoxicity, neurodegeneration, neuro-inflammation, and epilepsy), the extracellular concentration of adenosine in the brain can rise rapidly from nanomolar to micromolar levels, which can have both beneficial and detrimental effects on the course of the illness [104, 135, 136].
5.2 The neuroprotective role of cordycepin from Cordyceps in diseases of the central nervous system
Cordycepin showed neuroprotective effects on cerebral ischemia-reperfusion injury during inflammation, which included improving the behaviors in mice, reducing the area of cerebral infarction, inhibiting the expression of the pro-inflammatory factors IL-1β and TNF-α, and increasing the expression of the inflammatory factors IL-10 and TGF-β1[137, 138]. Our previous studies also found that cordycepin significantly ameliorated cuprizone-induced motor dysfunction, demyelination, glial cell activation and pro-inflammatory cytokine (IL-1β and IL-6) expression in the corpus callosum and hippocampus in a mouse model of demyelination [139], which demonstrated that cordycepin may protect against demyelination via suppression of neuroinflammation.
Covid-19 pneumonia may provoke systemic inflammation [140], leading to psychiatric problems, including anxiety, depression, guilt, stigma, and anger. Cordycepin also plays an important role in the amelioration of the psychiatric disorders, including major depression disease (MDD) and anxiety disorder. Studies on depression showed that an injection of cordycepin led to a rapid and robust antidepressant effect, which may be modulated at multiple beneficial mechanisms, particularly in regulating the prefrontal AMPA receptor signaling pathway [141]. Our previous studies also found that cordycepin exhibited a stronger and faster anxiolytic effect in behavioral tests and that IL-4 expression showed a strong positive correlation with reduced anxiety behaviors. RIL-4Rα (an IL-4 specific inhibitor) can completely block the anxiolytic effects induced by cordycepin, providing a novel and common anxiolytic IL-4 signaling pathway and an innovative drug with a novel neuroimmune mechanism for the amelioration of anxiety disorder [142].
Cordycepin also showed neuroprotective properties in other neurotoxicity disease models, such as Alzheimer's disease (AD) and Parkinson's disease (PD). Studies on Aβ-induced toxicity in primary hippocampal cultured neurons showed that cordycepin significantly inhibited Aβ-induced apoptosis and decreased the upregulated p-Tau expression in hippocampal neurons [15]. Studies in an Aβ plus ibotenic acid (IBO)-induced injury model of cultured hippocampal neurons showed that cordycepin significantly delayed Aβ plus IBO-induced excessive neuronal membrane depolarization. Furthermore, the suppressive effect of cordycepin against Aβ plus IBO-induced excessive neuronal membrane depolarization was blocked by an antagonist of adenosine A1 receptor [143]. Moreover, cordycepin protected PC12 cells against 6-OHDA-induced neurotoxicity through its potent antioxidant activity, including inhibition of 6-OHDA-induced cell apoptosis, mitochondrial dysfunction, and enhancement of antioxidant enzymes, such as superoxide dismutase (SOD) and glutathione peroxidase (GSH-Pox) [144]. Studies in a rotenone-induced PD rat model showed that cordycepin significantly protected dopamine neurons against rotenone-induced apoptosis by improving mitochondrial dysfunction [145]. In addition, cordycepin effectively alleviated motor dysfunction, the loss of DA neurons, and the activation of the TLR/NF-κB signaling pathway in an MPTP-induced PD model [146]. In summary, cordycepin protected neuronal functions and cell apoptosis in many different neurotoxic animal models, which may be relevant to the psychiatric disorders during Covid-19 infection.
6. Adenosine receptor and cordycepin from Cordyceps protect important organs such as the heart, liver, and kidney during hypoxemia
6.1 Elevated adenosine in hypoxemia or bacteremia activates adenosine receptors to protect the functions of multiple organs
Adenosine is a signaling molecule produced after injury, which promotes wound healing and tissue protection. It exerts significant effect on the regulation of angiogenesis, stromal formation, and inflammation [147]. Studies have shown that in the case of ischemia [148] and sepsis [149], the extracellular adenosine concentration rises and then sends a signal through the adenosine receptor on the tissue’s cells to provide systemic protection and avoid causing host tissue damage. Activation of the adenosine receptor A2A has also been shown to provide extensive organ protection against ischemic damage, including for the heart [150], lung [151], liver [152], kidney [153], and spinal cord [154]. The A2A adenosine receptor agonist has a good anti-inflammatory effect, especially for lung ischemia-reperfusion injury. A2A agonists significantly reduced ischemia-reperfusion injury and block neutrophil-mediated inflammatory responses in lung transplantation models [101, 151, 155, 156]. A mouse model of LPS-induced injury also exhibited the A2AR-mediated protection of lung tissue [157].
At the same time, it was discovered that adenosine receptor A2B can maintain tissue functional integrity in the heart [61] and kidney [158]. In addition, gene knockout or antagonism of the adenosine A1 and A3 receptors increased cecal ligation-induced systemic inflammation and mortality [159, 160]. A3 receptors activated anti-inflammatory pathways in lung ischemia-reperfusion injury [161], while A3 receptor agonists protected against reperfusion lung injury and reduce apoptosis [161, 162]. It is noteworthy that activation of the A1 receptors mediated the protective properties of ischemic preconditioning and adenosine preconditioning against pulmonary ischemia-reperfusion injury [163].
6.2 Cordyceps and cordycepin protect the lung, liver, and kidney in hypoxemia or inflammation
When the human body is attacked by germs, viruses, or pathogens, it shows an inflammatory pattern that the activities of pro-inflammatory cytokine (IL-1β, IL-6, TNF-α) and chemokine (CCL2) increased, causing functional cell death, and eventually leading to tissue fibrosis [74, 164]. At this time, cordycepin can transform the human immune inflammatory status (M1) into an immune repair and regeneration status(M2) to stimulate immune cells to release anti-inflammatory cytokines (IL-4, IL-10, IL-13) and to increase the phagocytosis of macrophages. The phagocytosis of necrotic tissue and the secretion of cytotropic factors promote tissue repair [69, 75, 165].
Cordycepin's mechanism of action involves its role in protecting organs and tissues, including against pulmonary fibrosis, liver fibrosis, and renal fibrosis. First, cordyceps and cordycepin can prevent pulmonary fibrosis [166-169]. Studies have shown that the continuous intragastric feeding of Cordyceps sinensis for 14 days improved the intratracheal injection of bleomycin, which results in an increased lung coefficient (lung weight/body weight) in rats, a reduced weight-bearing swimming time, and decreased arterial oxygen pressure, and lung tissue fibrotic lesions [166]. When using cordycepin to treat bleomycin (BLM)-induced pulmonary fibrosis in rats, it was found that cordycepin reduced the infiltration of inflammatory cells, fibroblast deposition, and prevented pulmonary fibrosis [168]. Clinical experiments have shown that Cordyceps sinensis dilated the bronchi, worked as an expectorant, and moderated asthma [170-172]. Zhang and colleagues treated 20 patients with pulmonary interstitial disease with Cordyceps sinensis capsules and found that the beneficial effect was significant [173].
Cordyceps can reduce myocardial oxygen consumption, increase myocardial nutritional blood flow, improve myocardial oxygen supply, and demand balance, and support the improvement of the pathophysiological status of myocardial ischemia and hypoxia. A Cordyceps water extract (2.5 g/kg) was able to enhance the ability of mice to withstand hypoxia at normal pressure. In addition, intraperitoneal injection and intragastric administration of cordycepin to mice significantly reduced myocardial oxygen consumption, counteracted the effect of isoproterenol on increasing myocardial oxygen consumption, improved hypoxia tolerance, and extended the survival time of hypoxic mice [174, 175].
Cordycepin also protects against liver fibrosis. Studies have shown that cordycepin and adenosine have anti-fibrotic effects in mice with hepatic fibrosis induced by intraperitoneal injection of thioacetamide (TAA) [176]. It was also found that 200 μ mol/L cordycepin amelioration significantly inhibited the increase in mRNA and protein expression of MCP-1 in cells under LPS stimulation in order to reduce the inflammatory phenotype and fibrosis response [177].
Cordycepin showed good protection in kidney diseases. Cordycepin inhibited high glucose-induced renal tubular epithelial-mesenchymal transition in rats. The mechanism may be achieved by down-regulating transforming growth factor-β (TGF-β) [178]. Cordycepin can protect the kidney by inhibiting renal tubular epithelial cell apoptosis [17, 179]. Studies have found that the mechanism by which cordycepin worked on membranous nephropathy was to protect the foot processes and cytoskeleton structures of the podocyte and to suppress complement-mediated signaling pathways, and to protect complement-mediated podocyte damage [180]. This also has related clinical findings. Shen and colleagues showed that clinical observation of 31 cases of acute renal failure with the addition of Cordyceps sinensis demonstrated that urine osmotic pressure increased after the addition of Cordyceps sinensis compared with the control group. Glucosidase decreased significantly, confirming that Cordyceps sinensis had a good effect on renal tubular epithelial cell repair in patients with acute renal failure [181]. Two other related studies have also found that Cordyceps sinensis was effective in treating acute renal damage due to epidemic hemorrhagic fever [182, 183].
7. Conclusions and Remarks
For a long time, the western medical community has been working on the development of adenosine receptor-selective agonists and antagonists [100]. Selective adenosine receptor A1 agonists may have some clinical effects in patients with heart failure and patients with renal failure [184, 185]. Medical institutions are developing clinical applications and continuing research on A2A activators for the amelioration of lung disease and Parkinson's disease [186, 187]. A2B and A3 selective agonists have also been studied in clinical trials of inflammatory and autoimmune excitatory diseases, respectively [51, 100]. It is noteworthy that adenosine receptors have also been studied as potential therapeutic targets for acute respiratory stress syndrome and acute lung injury [188, 189]. For example, the protective effect of A2B signaling can prevent ischemic lung injury [84]. A3 activation also has protective effects in reducing reperfusion-induced lung injury [161]. In addition, adenosine kinase inhibition can also reduce acute lung injury [190].
Table 1: The beneficial effects of the Cordyceps products in clinical studies.
Cordyceps militaris can activate the adenosine receptors A1, A2A, A2B, A3, and its affinity for these receptors is very similar [8-10, 26]. Studies have shown that it has the effects of enhancing human immunity, inhibiting virus reproduction, reducing inflammation, inhibiting the generation of cytokine storms, protecting the lungs, liver, heart, and kidney, and resisting pulmonary fibrosis [206]. Cordyceps militaris and cordycepin are also very safe. We summarized the clinical trials which used Cordycepin or Cordyceps for the treatment of various diseases (Table 1). Cordyceps is a new food resource approved by the Chinese government. There are almost no significant side effects. Only approximately 0.5% of people may have a fungal allergic reaction. Although there is no clear research on its use in pregnant women and children, it is recommended for safety reasons to not be used in these populations. With all its beneficial properties, why hasn’t cordycepin been developed as a medicine?
In fact, cordycepin has been approved as a new drug for leukemia in the United States [207]. With the application of an artificially purified monomer cordycepin, it is extremely easy to lose activity in the human body due to adenosine deaminase (ADA) activity. The pharmacokinetic reports suggest that the presence of ADA in the human body causes cordycepin into be rapidly deaminated to form a non-bioactive metabolite, 3'-deoxyhypoxanthine, and only a small portion of which is phosphorylated to the effective cordycepin triphosphate (the bioactive ingredient). To delay the metabolism time of cordycepin in the body, it must be used in combination with anti-deamination ADA inhibitors (such as pentostatin); however, this is more costly and has limited the widespread use of cordycepin [207].
Since ancient times, people have been treating and curing diseases with unrefined Cordyceps powder. Wang's team recently found the molecular mechanism of the concomitant biosynthesis of cordycepin and pentostatin (ADA inhibitor) in Cordyceps militaris, in which cordycepin and the ADA inhibitor pentostatin
could be simultaneously synthesized from the same gene cluster in Cordyceps militaris [207]. This finding reveals the mystery of why the Cordyceps can be utilized by the human body to produce a medicinal effect, since when taking the cordycepin in the original Cordyceps, the adenosine deaminase (ADA) inhibitor pentostatin, which prevents the deamination of cordycepin, is also present [207]. Cordycepin in Cordyceps militaris is very stable, easily absorbed, and utilized by the human body. Therefore, Cordyceps militaris with high cordycepin (> 1%) can be used as an activator of the adenosine receptors A1, A2A, A2B, and A3. It is safe and reliable and comes with a protective agent, pentostatin. Now, some pharmaceutical companies can produce Cordyceps powder with a high cordycepin content, up to 0.5-3%. The cultivated Cordyceps militaris can be made into tablets or granules and can play its important role for the prevention, adjuvant amelioration and rehabilitation of viral pneumonia.
The role of cordycepin in preventing cytokine storms and protecting alveolar tissue in COVID-19 pneumonia is worth exploring to overcome the devastating disease and to save lives. Because Cordyceps militaris is ideal to use as a food supplement to build up the immune system, and its anti-viral properties may prevent the infections from becoming severe or critical cases. It can be used as an adjunctive therapy to ameliorate the lung inflammation and cytokine storm, to clear alveolar fluid, and to protect the lung tissue. In the long run, it may be able to protect against lung fibrosis and repair lung tissue. A clinical trial for the cordycepin from Cordyceps militaris in the amelioration of COVID-19 pneumonia is warranted.
Acknowledgments
We are thankful for grant support from the National Natural Science Foundation of China (Grant No.31650005) We are also thankful for the scientific editing and assistance from Zhenghua Chen, Liping Shan, Grace YZhang, and Jessica PZhang.
Conflict of Interest
The authors have no conflicts of interest to disclose, financial or otherwise.
References:
- Zhou Y, Schneider DJ, Blackburn MR. Adenosine signaling and the regulation of chronic lung disease. Pharmacol Ther 123 (2009): 105-116.
- Song X. Analysis on the clinical application of Cordyceps in the treatment of respiratory diseases. Nei Mongol Journal of Traditional Chinese Medicine 33 (2014): 91.
- Panda AK, Swain KC. Traditional uses and medicinal potential of Cordyceps sinensis of Sikkim. J Ayurveda Integr Med 2 (2011): 9-13.
- Tuli HS, Sandhu SS, Sharma AK. Pharmacological and therapeutic potential of Cordyceps with special reference to Cordycepin. Biotech 4 (2014): 1-12.
- Negi CS, Koranga PR, Ghinga HS. Yar tsa Gumba (Cordyceps sinensis): A call for its sustainable exploitation. International Journal of Sustainable Development & World Ecology 13 (2006): 165-172.
- Lu W, Tang Y, Tang L. Comparative study on the functional components of nucleosides in Cordyceps sinensis, Cordyceps sinensis and edible fungi. Science & Technology Industry of China (2012): 54-57.
- Sheth S, Brito R, Mukherjea D, et al. Adenosine receptors: expression, function and regulation. Int J Mol Sci 15 (2014): 2024-2052.
- Hu Z, Lee CI, Shah VK, et al. Cordycepin Increases Nonrapid Eye Movement Sleep via Adenosine Receptors in Rats. Evid Based Complement Alternat Med (2013): 840134.
- Cao HL, Liu ZJ, Chang Z. Cordycepin induces apoptosis in human bladder cancer cells via activation of A3 adenosine receptors. Tumour Biol 39 (2017): 1010428317706915.
- Chen Y, Yang SH, Hueng DY, et al. Cordycepin induces apoptosis of C6 glioma cells through the adenosine 2A receptor-p53-caspase-7-PARP pathway. Chem Biol Interact 216 (2014): 17-25.
- Chen Y, Chen YC, Lin YT, et al. Cordycepin induces apoptosis of CGTH W-2 thyroid carcinoma cells through the calcium-calpain-caspase 7-PARP pathwayJ Agric Food Chem 58 (2010): 11645-11652.
- Pan BS, Lin CY, Huang BMThe Effect of Cordycepin on Steroidogenesis and Apoptosis in MA-10 Mouse Leydig Tumor Cells. Evid Based Complement Alternat Med (2011): 750468.
- Leu SF, Poon SL, Pao HY, et al. The in vivo and in vitro stimulatory effects of cordycepin on mouse leydig cell steroidogenesis. Biosci Biotechnol Biochem 75 (2011): 723-731.
- Dong ZS, Cao ZP, Shang YJ, et al. Neuroprotection of cordycepin in NMDA-induced excitotoxicity by modulating adenosine A(1) receptors. Eur J Pharmacol 853 (2019): 325-335.
- Yao LH, Wang J, Liu C, et al. Cordycepin protects against β-amyloid and ibotenic acid-induced hippocampal CA1 pyramidal neuronal hyperactivity. Korean J Physiol Pharmacol 23 (2019): 483-491.
- Jin ML, Park SY, Kim YH, et al. The neuroprotective effects of cordycepin inhibit glutamate-induced oxidative and ER stress-associated apoptosis in hippocampal HT22 cells. Neurotoxicology 41 (2014): 102-111.
- Gu L, Johno H, Nakajima S, et al. Blockade of Smad signaling by 3'-deoxyadenosine: a mechanism for its anti-fibrotic potential. Lab Invest 93 (2013): 450-461.
- Hwang S, Cho GS, Ryu S, et al. Post-ischemic treatment of WIB801C, standardized Cordyceps extract, reduces cerebral ischemic injury via inhibition of inflammatory cell migration. J Ethnopharmacol 186 (2016): 169-180.
- Yoshikawa N, Yamada S, Takeuchi C, et al. Cordycepin (3'-deoxyadenosine) inhibits the growth of B16-BL6 mouse melanoma cells through the stimulation of adenosine A3 receptor followed by glycogen synthase kinase-3beta activation and cyclin D1 suppression. Naunyn Schmiedebergs Arch Pharmacol 377 (2008): 591-595.
- Fishman P, Bar-Yehuda S, Ardon E, et al. Targeting the A3 adenosine receptor for cancer therapy: inhibition of prostate carcinoma cell growth by A3AR agonist. Anticancer Res 23 (2003): 2077-2083.
- Fishman P, Bar-Yehuda S, Liang BT, et al. Pharmacological and therapeutic effects of A3 adenosine receptor agonists. Drug Discov Today 17 (2012): 359-366.
- Madi L, Ochaion A, Rath-Wolfson L, et al. The A3 adenosine receptor is highly expressed in tumor versus normal cells: potential target for tumor growth inhibition. Clin Cancer Res 10 (2004): 4472-4479.
- Ochaion A, Bar-Yehuda S, Cohen S, et al. The anti-inflammatory target A(3) adenosine receptor is over-expressed in rheumatoid arthritis, psoriasis and Crohn's disease. Cell Immunol 258 (2009): 115-122.
- Yan LI, Hongyue XU, Shulin LI, et al. Study on Cordycepin Prevents Obesity and Mediates Prolactin Secretion Through ADORA_1 in GH3 Cells. China Animal Husbandry & Veterinary Medicine (2018).
- Zhang M, Zhou X, Tang L, et al. Role of cordycepin on transforming growth factor-β_1 in a rat model of chronic bronchial asthma. chinese journal of asthma (electronic edition) (2011).
- Nakamura K, Yoshikawa N, Yamaguchi Y, et al. Antitumor effect of cordycepin (3'-deoxyadenosine) on mouse melanoma and lung carcinoma cells involves adenosine A3 receptor stimulation. Anticancer Res 26 (2006): 43-47.
- Grazioli S, Dunn-Siegrist I, Pauchard LA, et al. Mitochondrial alarmins are tissue mediators of ventilator-induced lung injury and ARDS. PLoS One 14 (2019): e0225468.
- Ramesh T, Yoo SK, Kim SW, et al. Cordycepin (3'-deoxyadenosine) attenuates age-related oxidative stress and ameliorates antioxidant capacity in rats. Exp Gerontol 47 (2012): 979-987.
- Zhang XQ, Yue-Pu PU, Yin LH. Study on the scavenging effect on superoxide anion free radical and hydroxyl free radical of cordycep sinensis and mycelium of cultured cordyceps sinensis. Chinese Journal of Gerontology (2003).
- Mahy BW, Cox NJ, Armstrong SJ, et al. Multiplication of influenza virus in the presence of cordycepin, an inhibitor of cellular RNA synthesis. Nat New Biol 243 (1973): 172-174.
- Nair CN, Panicali DL. Polyadenylate sequences of human rhinovirus and poliovirus RNA and cordycepin sensitivity of virus replication. J Virol 20 (1976): 170-176.
- Shirman GA, Maslova SV. The effect of inhibitors of cellular RNA synthesis on stimulation of mouse encephalomyocarditis virus reproduction by poliovirus in HeLa and MIO cells. Acta Virol 22 (1978): 177-182.
- Du Y, Yu J, Du L, et al. Cordycepin enhances Epstein-Barr virus lytic infection and Epstein-Barr virus-positive tumor treatment efficacy by doxorubicin. Cancer Lett 376 (2016): 240-248.
- Ueda Y, Mori K, Satoh S, et al. Anti-HCV activity of the Chinese medicinal fungus Cordyceps militaris. Biochem Biophys Res Commun 447 (2014): 341-345.
- Xu FL, Lee YL, T sai, et al. Effect of cordycepin on Hantaan virus 76-118 infection of primary human embryonic pulmonary fibroblasts--characterization of apoptotic effects. Acta Virol 49 (2005): 183-193.
- Panicali DL, Nair CN. Effect of cordycepin triphosphate on in vitro RNA synthesis by picornavirus polymerase complexes. J Virol 25 (1978): 124-128.
- Richardson LS, Ting RC, Gallo RC, et al. Effect of cordycepin on the replication of type-c RNA tumor viruses. Int J Cancer 15 (1975): 451-456.
- Wittek R, Koblet H, Menna A, et al. The effect of cordycepin on the multiplication of Semliki Forest virus and on polyadenylation of viral RNA. Arch Virol 54 (1977): 95-106.
- Hashimoto, K, Simizu, B. Effect of cordycepin on the replication of western equine encephalitis virus. Arch Virol 52 (1976): 341-345.
- Becker Y, Olshevsky U. Inhibition of Herpes simplex virus replication by cordycepin. Isr J Med Sci 9 (1973): 1581-1585.
- Weiss SR, Bratt MA. Effect of cordycepin (3'-deoxyadenosine) on virus-specific RNA species synthesized in Newcastle disease virus-infected cells. J Virol 16 (1975): 1575-1583.
- Blackburn MR. Too much of a good thing: adenosine overload in adenosine-deaminase-deficient mice. Trends Pharmacol Sci 24 (2003): 66-70.
- Driver AG, Kukoly CA, Ali S, et al. Adenosine in bronchoalveolar lavage fluid in asthma. Am Rev Respir Dis 148 (1993): 91-97.
- Huszár E, Vass G, Vizi E, et al. Adenosine in exhaled breath condensate in healthy volunteers and in patients with asthma. Eur Respir J 20 (2002): 1393-1398.
- Ma B, Blackburn MR, Lee CG, et al. Adenosine metabolism and murine strain-specific IL-4-induced inflammation, emphysema, and fibrosis. J Clin Invest 116 (2006) : 1274-1283.
- Volmer JB, Thompson LF, Blackburn MR. Ecto-5'-nucleotidase (CD73)-mediated adenosine production is tissue protective in a model of bleomycin-induced lung injury. J Immunol 176 (2006): 4449-4458.
- Gessi S, Merighi S, Varani K, et al. The A3 adenosine receptor: an enigmatic player in cell biology. Pharmacol Ther 117 (2008): 123-140.
- Jin X, Shepherd RK, Duling BR, et al. Inosine binds to A3 adenosine receptors and stimulates mast cell degranulation. J Clin Invest 100 (1997): 2849-2857.
- Ryzhov S, Goldstein AE, Biaggioni I, et al. Cross-talk between G(s)- and G(q)-coupled pathways in regulation of interleukin-4 by A(2B) adenosine receptors in human mast cells. Mol Pharmacol 70 (2006): 727-735.
- Haskó G, Cronstein BN. Adenosine: an endogenous regulator of innate immunity. Trends Immunol 25 (2004): 33-39.
- Haskó G, Linden J, Cronstein B et al. Adenosine receptors: therapeutic aspects for inflammatory and immune diseases. Nat Rev Drug Discov 7 (2008): 759-770.
- Arch JR, Newsholme EA. The control of the metabolism and the hormonal role of adenosine. Essays Biochem 14 (1978): 82-123.
- Martin C, Leone M, Viviand X, et al. High adenosine plasma concentration as a prognostic index for outcome in patients with septic shock. Crit Care Med 28 (2000): 3198-3202.
- Sottofattori E, Anzaldi M, Ottonello L. HPLC determination of adenosine in human synovial fluid. J Pharm Biomed Anal 24 (2001): 1143-1146.
- Eltzschig HK, Ibla JC, Furuta GT, et al. Coordinated adenine nucleotide phosphohydrolysis and nucleoside signaling in posthypoxic endothelium: role of ectonucleotidases and adenosine A2B receptors. J Exp Med 198 (2003): 783-796.
- Marquardt DL, Gruber HE, Wasserman SI. Adenosine release from stimulated mast cells. Proc Natl Acad Sci U S A 81 (1984): 6192-6196.
- Resnick MB, Colgan SP, Patapoff TW, et al. Activated eosinophils evoke chloride secretion in model intestinal epithelia primarily via regulated release of 5'-AMP.J Immunol 151 (1993): 5716-5723.
- Madara JL, Patapoff TW, Gillece-Castro B, et al. 5'-adenosine monophosphate is the neutrophil-derived paracrine factor that elicits chloride secretion from T84 intestinal epithelial cell monolayers. J Clin Invest 91 (1993): 2320-2325.
- Kaczmarek E, Koziak K, Sévigny J, et al. Identification and characterization of CD39/vascular ATP diphosphohydrolase. J Biol Chem 271 (1996): 33116-33122.
- Resta R, Yamashita Y, Thompson LF. Ecto-enzyme and signaling functions of lymphocyte CD73. Immunol Rev 161(1998): 95-109.
- Eckle T, Krahn T, Grenz A, et al. Cardioprotection by ecto-5'-nucleotidase (CD73) and A2B adenosine receptors. Circulation 115 (2007): 1581-1590.
- Fan B, Zhu H. Cordycepin: pharmacological properties and their relevant mechanisms. Tang 2 (2012): 14.11-14.17.
- Jiang X, Ge P, Hu X. Resistant Effect of Cordyceps militaris on Immunodeficiency by Cyclophosphamide. Journal of Ocean University of Qingdao 01(2002): 46-50.
- Kang HJ, Baik HW, Kim SJ, et al. Cordyceps militaris Enhances Cell-Mediated Immunity in Healthy Korean Men. J Med Food 18 (2015): 1164-1172.
- Kuo YC, Tsai WJ, Shiao MS, et al. Cordyceps sinensis as an immunomodulatory agent. Am J Chin Med 24 (1996): 111-125.
- Zhong SS, Xiang YJ, Liu PJ, et al. Effect of Cordyceps sinensis on the Treatment of Experimental Autoimmune Encephalomyelitis: A Pilot Study on Mice Model. Chin Med J (Engl) 13 (2017): 2296-2301.
- Zhu SJ, Pan J, Zhao B, et al. Comparisons on enhancing the immunity of fresh and dry Cordyceps militaris in vivo and in vitro. J Ethnopharmacol 149 (2013): 713-719.
- Zhou X, Luo L, Dressel W, et al. Cordycepin is an immunoregulatory active ingredient of Cordyceps sinensis. Am J Chin Med 36 (2008): 967-980.
- Zhou X, Meyer CU, Schmidtke P, et al. Effect of cordycepin on interleukin-10 production of human peripheral blood mononuclear cells. Eur J Pharmacol 453 (2002): 309-317.
- Zhang Y, Chen H, Wang F et al. Study of Cordycepin on Regulation of Mouse Spleen Lymphocytesproceedings of the 4th Chinese Conference on Allergy in Integrated Traditional Chinese and Western Medicine. Xi'an, Shaanxi, China. (2009).
- Black PL, Henderson EE, Pfleiderer W, et al. 2',5'-Oligoadenylate trimer core and the cordycepin analog augment the tumoricidal activity of human natural killer cells. J Immunol 133 (1984): 2773-2777.
- Peng Y, Tao Y, Liu C. Effect of Cordyceps mycelium on liver NK cell function on carbon tetrachloride-induced acute experimental liver injury in miceproceedings of the 20th National Conference on Integrated Traditional Chinese and Western Medicine Liver Disease, Zhangjiajie, Hunan, China (2011).
- Qiu Z, Lin X, Qian L, et al. Effects of Cordyceps sinensis on the activity of NK and LAK cells in leukemia patients. Shanghai. Journal of Immunology 1 (1994): 30-31.
- Jeong MH, Seo MJ, Park JU, et al. Effect of cordycepin purified from Cordyceps militaris on Th1 and Th2 cytokines in mouse splenocytes. J Microbiol Biotechnol 22 (2012): 1161-1164.
- Shin S, Moon S, Park Y, et al. Role of Cordycepin and Adenosine on the Phenotypic Switch of Macrophages via Induced Anti-inflammatory Cytokines. Immune Netw 9 (2009): 255-264.
- Ware LB, Conner ER, Matthay MA. von Willebrand factor antigen is an independent marker of poor outcome in patients with early acute lung injury. Crit Care Med 29 (2001): 2325-2331.
- Abraham E, Carmody A, Shenkar R, et al. Neutrophils as early immunologic effectors in hemorrhage- or endotoxemia-induced acute lung injury. Am J Physiol Lung Cell Mol Physiol 279 (2000): L1137-1145.
- Reutershan J, Cagnina RE, Chang D, et al. Therapeutic anti-inflammatory effects of myeloid cell adenosine receptor A2a stimulation in lipopolysaccharide-induced lung injury. J Immunol 179 (2007): 1254-1263.
- Friebe D, Yang T, Schmidt T, et al. Purinergic signaling on leukocytes infiltrating the LPS-injured lung. PLoS One (2014): e95382.
- Haskó G, Kuhel DG, Chen JF, et al. Adenosine inhibits IL-12 and TNF-[alpha] production via adenosine A2a receptor-dependent and independent mechanisms. Faseb j 14 (2000): 2065-2074.
- Reutershan J, Vollmer I, Stark S, et al. Adenosine and inflammation: CD39 and CD73 are critical mediators in LPS-induced PMN trafficking into the lungs. Faseb j 23 (2009): 473-482.
- Eckle T, Faigle M, Grenz A, et al. A2B adenosine receptor dampens hypoxia-induced vascular leak. Blood 111 (2008): 2024-2035.
- Eckle T, Grenz A, Laucher S, et al. A2B adenosine receptor signaling attenuates acute lung injury by enhancing alveolar fluid clearance in mice. J Clin Invest 118 (2008): 3301-3315.
- Densmore JC, Schaid TR, Jeziorczak PM, et al. Lung injury pathways: Adenosine receptor 2B signaling limits development of ischemic bronchiolitis obliterans organizing pneumonia. Exp Lung Res 43 (2017): 38-48.
- Hoegl S, Brodsky KS, Blackburn MR, et al. Alveolar Epithelial A2B Adenosine Receptors in Pulmonary Protection during Acute Lung Injury. J Immunol 195 (2015): 1815-1824.
- Polosa R, Blackburn MR. Adenosine receptors as targets for therapeutic intervention in asthma and chronic obstructive pulmonary disease. Trends Pharmacol Sci 30 (2009): 528-535.
- Selman M, Carrillo G, Estrada A, et al. Accelerated variant of idiopathic pulmonary fibrosis: clinical behavior and gene expression pattern. PLoS One 2 (2007): e482.
- La Gruta NL, Kedzierska K, Stambas J, et al. A question of self-preservation: immunopathology in influenza virus infection. Immunol Cell Biol 85 (2007): 85-92.
- Shinya K, Gao Y, Cilloniz C, et al. Integrated clinical, pathologic, virologic, and transcriptomic analysis of H5N1 influenza virus-induced viral pneumonia in the rhesus macaque. J Virol 86 (2012): 6055-6066.
- Hussell T, Goulding, J. Structured regulation of inflammation during respiratory viral infection. Lancet Infect Dis 10 (2010): 360-366.
- Tisoncik JR, Korth MJ, Simmons CP, et al. Into the eye of the cytokine storm. Microbiol Mol Biol Rev 76 (2012): 16-32.
- Crayne CB, Albeituni S, Nichols KE, et al. The Immunology of Macrophage Activation Syndrome. Front Immunol 10 (2019): 119.
- Karakike E, Giamarellos-Bourboulis EJ. Macrophage Activation-Like Syndrome: A Distinct Entity Leading to Early Death in Sepsis. Front Immunol 10 (2019): 55.
- Tanaka T, Narazaki M, Kishimoto T. Immunotherapeutic implications of IL-6 blockade for cytokine storm. Immunotherapy 8 (2016): 959-970.
- Ravani A, Vincenzi F, Bortoluzzi A, et al. Role and Function of A(2A) and A3 Adenosine Receptors in Patients with Ankylosing Spondylitis, Psoriatic Arthritis and Rheumatoid Arthritis. Int J Mol Sci 18 (2017).
- Varani K, Padovan M, Vincenzi F, et al. A2A and A3 adenosine receptor expression in rheumatoid arthritis: upregulation, inverse correlation with disease activity score and suppression of inflammatory cytokine and metalloproteinase release. Arthritis Res Ther 13 (2011): R197.
- Lee HS, Chung HJ, Lee HW, et al. Suppression of inflammation response by a novel A3 adenosine receptor agonist thio-Cl-IB-MECA through inhibition of Akt and NF-κB signaling. Immunobiology 216 (2011): 997-1003.
- Rivo J, Zeira E, Galun E, et al. Activation of A3 adenosine receptors attenuates lung injury after in vivo reperfusion. Anesthesiology 101 (2004): 1153-1159.
- Cohen S, Fishman P. Targeting the A(3) adenosine receptor to treat cytokine release syndrome in cancer immunotherapy. Drug Des Devel Ther 13 (2019): 491-497.
- Jacobson KA, Merighi S, Varani K, et al. A(3) Adenosine Receptors as Modulators of Inflammation: From Medicinal Chemistry to Therapy. Med Res Rev 38 (2018): 1031-1072.
- Ross SD, Tribble CG, Linden J, et al. Selective adenosine-A2A activation reduces lung reperfusion injury following transplantation. J Heart Lung Transplant 18 (1999): 994-1002.
- Fu S, Lu W, Yu W, et al. Protective effect of Cordyceps sinensis extract on lipopolysaccharide-induced acute lung injury in mice. Biosci Rep 39 (2019).
- Zheng X. Clinical observation on prevention and treatment of recurrent respiratory infections in 56 elderly patients. Fujian Medical Journal (1999): 58-59.
- Stockwell J, Jakova E, Cayabyab FS. Adenosine A1 and A2A Receptors in the Brain: Current Research and Their Role in Neurodegeneration. Molecules 22 (2017).
- van Calker D, Biber K, Domschke K, et al. The role of adenosine receptors in mood and anxiety disorders. J Neurochem 151 (2019): 11-27.
- Fredholm BB, Chen JF, Cunha RA, et al. Adenosine and brain function. Int Rev Neurobiol 63 (2005): 191-270.
- de Mendonça A, Ribeiro JA. Adenosine and neuronal plasticity. Life Sci 60 (1997): 245-251.
- Dunwiddie TV, Masino SA. The role and regulation of adenosine in the central nervous system. Annu Rev Neurosci 24 (2001): 31-55.
- Florian C, Vecsey CG, Halassa MM, et al. Astrocyte-derived adenosine and A1 receptor activity contribute to sleep loss-induced deficits in hippocampal synaptic plasticity and memory in mice. J Neurosci 31 (2011): 6956-6962.
- Parkinson FE, Xiong W, Zamzow CR. Astrocytes and neurons: different roles in regulating adenosine levels. Neurol Res 27 (2005): 153-160.
- Schmitt LI, Sims RE, Dale N, et al. Wakefulness affects synaptic and network activity by increasing extracellular astrocyte-derived adenosine. J Neurosci 32 (2012): 4417-4425.
- Costenla AR, Cunha RA, de Mendonça A. Caffeine, adenosine receptors, and synaptic plasticity. J Alzheimers Dis 20 (2010): S25-S34.
- Costenla AR, de Mendonça A, Ribeiro JA. Adenosine modulates synaptic plasticity in hippocampal slices from aged rats. Brain Res 851 (1999): 228-234.
- Costenla AR, Diógenes MJ, Canas PM, et al. Enhanced role of adenosine A(2A) receptors in the modulation of LTP in the rat hippocampus upon ageing. Eur J Neurosci 34 (2011): 12-21.
- El Yacoubi M, Ledent C, Ménard JF, et al. The stimulant effects of caffeine on locomotor behaviour in mice are mediated through its blockade of adenosine A(2A) receptors. Br J Pharmacol 129 (2000): 1465-1473.
- Lee K, Li B, Xi X, et al. Role of neuronal energy status in the regulation of adenosine 5'-monophosphate-activated protein kinase, orexigenic neuropeptides expression, and feeding behavior. Endocrinology 146 (2005): 3-10.
- Reichert CF, Maire M, Schmidt C, et al. Sleep-Wake Regulation and Its Impact on Working Memory Performance: The Role of Adenosine. Biology (Basel) 5 (2016).
- Castillo CA, Albasanz JL, León D, et al. Age-related expression of adenosine receptors in brain from the senescence-accelerated mouse. Exp Gerontol 44 (2009): 453-461.
- Chen Z, Stockwell J, Cayabyab FS. Adenosine A1 Receptor-Mediated Endocytosis of AMPA Receptors Contributes to Impairments in Long-Term Potentiation (LTP) in the Middle-Aged Rat Hippocampus. Neurochem Res 41 (2016): 1085-1097.
- Cunha RA. Neuroprotection by adenosine in the brain: From A(1) receptor activation to A (2A) receptor blockade. Purinergic Signal 1 (2005): 111-134.
- de Mendonça A, Sebastião AM, Ribeiro JA. Adenosine: does it have a neuroprotective role after all? Brain Res Brain Res Rev 33 (2000): 258-274.
- Fredholm BB. Adenosine and neuroprotection. Int Rev Neurobiol 40 (1997): 259-280.
- Rudolphi KA, Schubert P, Parkinson FE, et al. Adenosine and brain ischemia. Cerebrovasc Brain Metab Rev 4 (1992): 346-369.
- Von Lubitz DK, Lin RC, Jacobson KA, et al. Cerebral ischemia in gerbils: effects of acute and chronic treatment with adenosine A2A receptor agonist and antagonist. Eur J Pharmacol 287 (1995): 295-302.
- Boison D. The adenosine kinase hypothesis of epileptogenesis. Prog Neurobiol 84 (2008): 249-262.
- Malva JO, Silva AP, Cunha RA et al. Presynaptic modulation controlling neuronal excitability and epileptogenesis: role of kainate, adenosine and neuropeptide Y receptors. Neurochem Res 28 (2003): 1501-1515.
- Ciruela F, Ferré S, Casadó V, et al. Heterodimeric adenosine receptors: a device to regulate neurotransmitter release. Cell Mol Life Sci 63 (2006): 2427-2431.
- Cristóvão-Ferreira S, Navarro G, Brugarolas M, et al. A1R-A2AR heteromers coupled to Gs and G i/0 proteins modulate GABA transport into astrocytes. Purinergic Signal 9 (2013): 433-449.
- Ferre S, Ciruela F, Borycz J, et al. Adenosine A1-A2A receptor heteromers: new targets for caffeine in the brain. Front Biosci 13 (2008): 2391-2399.
- Fuxe K, Ferré S, Genedani S, et al. Adenosine receptor-dopamine receptor interactions in the basal ganglia and their relevance for brain function. Physiol Behav 92 (2007): 210-217.
- Fuxe K, Agnati LF, Borroto-Escuela DO. The impact of receptor-receptor interactions in heteroreceptor complexes on brain plasticity. Expert Rev Neurother 14 (2014): 719-721.
- Pedata F, Dettori I, Coppi E, et al. Purinergic signalling in brain ischemia. Neuropharmacology 104 (2016): 105-130.
- Moidunny S, Vinet J, Wesseling E, et al. Adenosine A2B receptor-mediated leukemia inhibitory factor release from astrocytes protects cortical neurons against excitotoxicity. J Neuroinflammation 9 (2012): 198.
- King N, Lin H, McGivan JD, et al. Expression and activity of the glutamate transporter EAAT2 in cardiac hypertrophy: implications for ischaemia reperfusion injury. Pflugers Arch 452 (2006): 674-682.
- Beamer E, Gölöncsér F, Horváth G, et al. Purinergic mechanisms in neuroinflammation: An update from molecules to behavior. Neuropharmacology 104 (2016): 94-104.
- Eisenstein A, Patterson S, Ravid K. The Many Faces of the A2b Adenosine Receptor in Cardiovascular and Metabolic Diseases. J Cell Physiol 230 (2015): 2891-2897.
- Cheng Z, He W, Zhou X, et al. Cordycepin protects against cerebral ischemia /reperfusion injury in vivo and in vitroEur. J Pharmacol 664 (2011): 20-28.
- Deng C, Yang-Bo OU, Tao R, et al. Cordycepin's protecting brain and reducing inflammation in a mouse model of focal cerebral ischemia reperfusion injury. Chinese Journal of Gerontology (2014).
- Jia Y, Li H, Bao H, et al. Cordycepin (3'-deoxyadenosine) promotes remyelination via suppression of neuroinflammation in a cuprizone-induced mouse model of demyelination. Int Immunopharmacol 75 (2019): 105777.
- Arabi YM, Harthi A, Hussein J, et al. Severe neurologic syndrome associated with Middle East respiratory syndrome corona virus (MERS-CoV) Infection 43 (2015): 495-501.
- Li B, Hou Y, Zhu M, et al. 3'-De-oxyadenosine (Cordycepin) Produces a Rapid and Robust Antidepressant Effect via Enhancing Prefrontal AMPA Receptor Signaling Pathway. Int J Neuropsychopharmacol 19 (2016).
- Gao T, Li B, Hou Y, et al. Interleukin-4 signalling pathway underlies the anxiolytic effect induced by 3-deoxyadenosine. Psychopharmacology (Berl) 236 (2019): 2959-2973.
- Song H, Huang LP, Li Y, et al. Neuro-protective effects of cordycepin inhibit Aβ-induced apoptosis in hippocampal neurons. Neurotoxicology 68 (2018): 73-80.
- Olatunji OJ, Feng Y, Olatunji OO, et al. Cordycepin protects PC12 cells against 6-hydroxydopamine induced neurotoxicity via its antioxidant properties. Biomed Pharmacother 81 (2016): 7-14.
- Jiang X, Tang PC, Chen Q, et al. Cordycepin Exerts Neuroprotective Effects via an Anti-Apoptotic Mechanism based on the Mitochondrial Pathway in a Rotenone-Induced Parkinsonism Rat Model. CNS Neurol Disord Drug Targets 18 (2019): 609-620.
- Cheng C, Zhu X. Cordycepin mitigates MPTP-induced Parkinson's disease through inhibiting TLR/NF-κB signaling pathway. Life Sci 223 (2019): 120-127.
- Valls MD, Cronstein BN, Montesinos MC. Adenosine receptor agonists for promotion of dermal wound healing. Biochem Pharmacol 77 (2009): 1117-1124.
- Sperlágh B, Dóda M, Baranyi M, et al. Ischemic-like condition releases norepinephrine and purines from different sources in superfused rat spleen strips. J Neuroimmunol 111 (2000): 45-54.
- Jabs CM, Sigurdsson GH, Neglen P. Plasma levels of high-energy compounds compared with severity of illness in critically ill patients in the intensive care unit. Surgery 124 (1998): 65-72.
- Yang Z, Day YJ, Toufektsian MC, et al. Myocardial infarct-sparing effect of adenosine A2A receptor activation is due to its action on CD4+ T lymphocytes. Circulation 114 (2006): 2056-2064.
- Reece TB, Ellman PI, Maxey TS, et al. Adenosine A2A receptor activation reduces inflammation and preserves pulmonary function in an in vivo model of lung transplantation. J Thorac Cardiovasc Surg 129 (2005): 1137-1143.
- Day YJ, Marshall MA, Huang L, et al. Protection from ischemic liver injury by activation of A2A adenosine receptors during reperfusion: inhibition of chemokine induction. Am J Physiol Gastrointest Liver Physiol 286 (2004): G285-293.
- Day YJ, Huang L, McDuffie MJ, et al. Renal protection from ischemia mediated by A2A adenosine receptors on bone marrow-derived cells. J Clin Invest 112 (2003): 883-891.
- Li Y, Oskouian RJ, Day YJ, et al. Mouse spinal cord compression injury is reduced by either activation of the adenosine A2A receptor on bone marrow-derived cells or deletion of the A2A receptor on non-bone marrow-derived cells. Neuroscience 141 (2006): 2029-2039.
- Ellman PI, Reece TB, Law MG, et al. Adenosine A2A activation attenuates nontransplantation lung reperfusion injury. J Surg Res 149 (2008): 3-8.
- Lisle TC, Gazoni LM, Fernandez LG, et al. Inflammatory lung injury after cardiopulmonary bypass is attenuated by adenosine A(2A) receptor activation. J Thorac Cardiovasc Surg 136 (2008): 1280-1287.
- Moore CC, Martin EN, Lee GH, et al. An A2A adenosine receptor agonist, ATL313, reduces inflammation and improves survival in murine sepsis models.BMC Infect Dis 8 (2008): 141.
- Grenz A, Osswald H, Eckle T, et al. The reno-vascular A2B adenosine receptor protects the kidney from ischemia. PLoS Med 5 (2008): e137.
- Gallos G, Ruyle TD, Emala CW, et al. A1 adenosine receptor knockout mice exhibit increased mortality, renal dysfunction, and hepatic injury in murine septic peritonitis. Am J Physiol Renal Physiol 289 (2005): F369-376.
- Lee HT, Kim M, Joo JD, et al. A3 adenosine receptor activation decreases mortality and renal and hepatic injury in murine septic peritonitis. Am J Physiol Regul Integr Comp Physiol 291 (2006): R959-969.
- Rivo J, Zeira E, Galun E, et al. Activation of A3 adenosine receptor provides lung protection against ischemia-reperfusion injury associated with reduction in apoptosis. Am J Transplant 4 (2004): 1941-1948.
- Matot I, Weiniger CF, Zeira E, et al. A3 adenosine receptors and mitogen-activated protein kinases in lung injury following in vivo reperfusion. Crit Care 10 (2006): R65.
- Yildiz G, Demiryürek AT, Gümüsel B, et al. Ischemic preconditioning modulates ischemia-reperfusion injury in the rat lung: role of adenosine receptors. Eur J Pharmacol 556 (2007): 144-150.
- Seo MJ, Kim MJ, Lee HH, et al. Effect of cordycepin on the expression of the inflammatory cytokines TNF-alpha, IL-6, and IL-17A in C57BL/6 mice. J Microbiol Biotechnol 23 (2013): 156-160.
- Choi YH, Kim GY, Lee HH. Anti-inflammatory effects of cordycepin in lipopolysaccharide-stimulated RAW 264.7 macrophages through Toll-like receptor 4-mediated suppression of mitogen-activated protein kinases and NF-κB signaling pathways. Drug Des Devel Ther 8 (2014): 1941-1953.
- Chen M, Cheung FW, Chan MH, et al. Protective roles of Cordyceps on lung fibrosis in cellular and rat models. J Ethnopharmacol 143 (2012): 448-454.
- Liu YF, Tang QJ, Yang Y, et al. Purification of Cordycepin from Fermentation Broth by Macroporous Resin and Its Antifibrosis Activity. Journal of Fungal Research (2013).
- Yang J, Liu Z, Guo J et al. Experimental study on Cordyceps sinensis preventing pulmonary fibrosis. The Journal of Practical Medicine, (2008): 1310-1312.
- Zhan R, Feng YZ, Gu ZG, et al. The effects and mechanisms of complex corolyceps on pulmonary fibrosis in experimental mice. Chinese Pharmacological Bulletin 24 (2008): 839-840.
- Cheng X. Clinical intervention of artificial Cordyceps sinensis on pulmonary fibrosis Practical. Journal of Cardiac Cerebral Pneumal and Vascular Disease (2014): 152-153.
- Qu T, Jing X. Application of Cordyceps sinensis in Respiratory Diseases. Strait Pharmaceutical Journal (2005): 140-141.
- Zhu L. Comparison of clinical effects of tetrandrine combined with cordycepin powder and thymosin on idiopathic pulmonary interstitial fibrosis. Journal of Community Medicine (2017): 35-37.
- Zhang Z, Jiang F. Clinical analysis of 20 cases of pulmonary interstitial disease treated by Cordyceps sinensis. Clincal Focus (2000): 518-519.
- Li F, Liu L. Research progress on cardiovascular pharmacological effects of Cordyceps sinensis. Journal of Traditional Chinese Medicine (2002): 55-56.
- Yan S, Liu SH, Bo W. Study advancement about pharmacological actions and clinical application of Cordyceps sinensis SaccSichuan. Journal of Physiological Sciences (2008).
- Hung YP, Lee CL. Higher Anti-Liver Fibrosis Effect of Cordyceps militaris-Fermented Product Cultured with Deep Ocean Water via Inhibiting Proinflammatory Factors and Fibrosis-Related Factors Expressions. Mar Drugs 15 (2017).
- Ouyang YY, Zhang Z, Cao YR, et al. [Effects of cordyceps acid and cordycepin on the inflammatory and fibrogenic response of hepatic stellate cells] Zhonghua Gan Zang Bing Za Zhi 21 (2013): 275-278.
- Liang Z, Zhu S, Chen J, et al. Hengmei. Cordycepin Inhibit Epithelial-mesenchymal-transition of NRK52 E Cells Induced by High Glucose by Downregulate TGF-βExpression. Chinese Journal of Integrated Traditional and Western Nephrology 15 (2014): 773-776.
- Gu LB, Bian RW, Tu Y, et al. [Mechanisms of cordycepin on improving renal interstitial fibrosis via regulating eIF2α/TGF-β/Smad signaling pathway] Zhongguo Zhong Yao Za Zhi 39 (2014): 4096-4101.
- Hong T, Cui LK, Wen J, et al. [Cordycepin protects podocytes from injury mediated by complements complex C5b-9]Sichuan Da Xue Xue Bao Yi Xue Ban 46 (2015): 173-178, 227.
- Shen Y Liu H. A controlled clinical study of Cordyceps sinensis in 31 cases of acute renal failure. Yunnan Journal of Traditional Chinese Medicine and Materia Medica (1996): 36-33.
- Feng Z, Li L, Shen X. Effects of Cordyceps Sinensis in renal dam-age of epidemichemorrhagic fever. CHINESE JOURNAL OF NEPHROLOGY, DIALYSIS & TRANSPLANTATION (1995).
- Gang M, Liu Y, Wang X. Clinical Observation on the Prevention and Cure of Acute Renal Failure with Hemorrhagic Fever with Renal Syndrome Combined with Cordyceps Sinensis. Shandong Medical Journal (2001): 35-36.
- Cleland JG, Chiswell K, Teerlink JR, et al. Predictors of postdischarge outcomes from information acquired shortly after admission for acute heart failure: a report from the Placebo-Controlled Randomized Study of the Selective A1 Adenosine Receptor Antagonist Rolofylline for Patients Hospitalized With Acute Decompensated Heart Failure and Volume Overload to Assess Treatment Effect on Congestion and Renal Function (PROTECT) Study. Circ Heart Fail 7 (2014): 76-87.
- Hocher B. Adenosine A1 receptor antagonists in clinical research and development. Kidney Int 78 (2010): 438-445.
- Jenner P. An overview of adenosine A2A receptor antagonists in Parkinson's disease. Int Rev Neurobiol 119 (2014): 71-86.
- Trevethick MA, Mantell SJ, Stuart EF, et al. Treating lung inflammation with agonists of the adenosine A2A receptor: promises, problems and potential solutions. Br J Pharmacol 155 (2008): 463-474.
- Eckle T, Koeppen M, Eltzschig HK. Role of extracellular adenosine in acute lung injury. Physiology (Bethesda) 24 (2009): 298-306.
- Schepp CP, Reutershan J. Bench-to-bedside review: adenosine receptors--promising targets in acute lung injury? Crit Care 12 (2008): 226.
- Köhler D, Streienberger A, Morote-García JC, et al. Inhibition of Adenosine Kinase Attenuates Acute Lung Injury. Crit Care Med 44 (2016): e181-189.
- Zhu L. Comparison of the therapeutic effects of tetrandrine combine with cordyceps sinensis powder and thymosin in treatment of idiopathic pulmonary interstitial fibrosis. Journal of Community Medicine (2017): 39-41.
- Wang N, Jiang L, Zhang X, et al. Effect of Dongchong Xiacao capsule on airway inflammation of asthmatic patients. China Journal of Chinese Materia Medica 32 (2007): 1566-1568.
- Sun T, Dong W, Jiang G, et al. Cordyceps militaris Improves Chronic Kidney Disease by Affecting TLR4/NF-κB Redox Signaling Pathway. Oxid Med Cell Longev 2019 (2019): 7850863.
- Zhao K, Li Y, Zhang H. Role of dongchongxiacao (Cordyceps) in prevention of contrast-induced nephropathy in patients with stable angina pectoris. J Tradit Chin Med 33 (2013): 283-286.
- Zhoa K, Li Y, Gao S, et al. Effect of Dongchongxiacao (Cordyceps) therapy on contrast-induced nephropathy in patients with type 2 diabetes and renal insufficiency undergoing coronary angiography. J Tradit Chin Med 35 (2015): 422-427.
- Huang J, Li J, Liu T, et al. Effect of Combined Therapy with Hypha Cordyceps and Ginkgo Leaf Tablet on Micro-inflammation in Patients Undergoing Maintenance Hemodialysis. Chinese Journal of Integrated Traditional and Western Medicine (2008): 502-504.
- Lan L. Study on Effect of Cordceps Sinensis and Artemisinin in Preventing recurrence of Lupus Nephritis. Chinese Journal of Integrated Traditional and Western Medicine 22 (2002): 169-171.
- Ding C, Tian PX, Xue W, et al. Efficacy of Cordyceps sinensis in long term treatment of renal transplant patients. Front Biosci (Elite Ed) 3 (2011): 301-307.
- Ding C, Tian P, Jin J, et al. Clinical Application and Exploration on echanism of Action of Cordyceps Sinesis mycelia Prepration for Renal Transplantation Recipients. Chinese Journal of Integrated Traditional and Western Medicine 29 (2009): 975-978.
- Li Y, Xue WJ, Tian PX, et al. Clinical application of Cordyceps sinensis on immunosuppressive therapy in renal transplantation. Transplant Proc 41 (2009): 1565-1569.
- Sun M, Yoga Y, Lu Y, et al. Clinical Study on Application of Bailing Capsule after Renal Transplantation Chinese Journal of Integrated Traditional and Western Medicine 024 (2004): 808-810.
- Wang X, Jiang Y, Zhao C, et al. Clinical Research of Xinganbao Capsule on the Treatment of Chronic Hepatitis B Liver FibrosisChinese Journal of Integrated Traditional and Western Medicine (2012): 325-328.
- Jung SJ, Jung ES, Choi EK, et al. Immunomodulatory effects of a mycelium extract of Cordyceps (Paecilomyces hepiali; CBG-CS-2): a randomized and double-blind clinical trial. BMC Complement Altern Med 19 (2019): 77.
- Chen S, Li Z, Krochmal R, et al. Effect of Cs-4 (Cordyceps sinensis) on exercise performance in healthy older subjects: a double-blind, placebo-controlled trial. J Altern Complement Med 16 (2010): 585-590.
- Chen CY, Hou CW, Bernard JR, et al. Rhodiola crenulata- and Cordyceps sinensis-based supplement boosts aerobic exercise performance after short-term high altitude training. High Alt Med Biol 15 (2014): 371-379.
- Foss FM. Combination therapy with purine nucleoside analogs. Oncology (Williston Park) 14 (2000): 31-35.
- Xia Y, Luo F, Shang Y, et al. Fungal Cordycepin Biosynthesis Is Coupled with the Production of the Safeguard Molecule Pentostatin. Cell Chem Biol 24 (2017): 1479-1489. e1474.

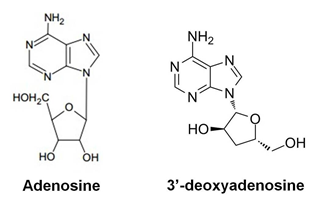
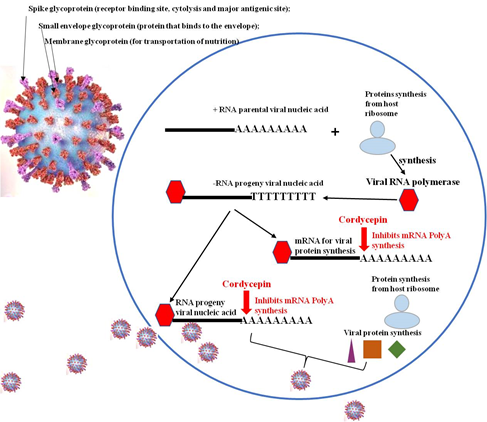

 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 