A Concise Overview of Chemotherapeutic Drugs in Gastrointestinal Cancers: Mechanisms of Action and Resistance
Shreya Sharma1, Goutam Chowdhury2, Anindita Chakrabarty1*
1Department of Life Sciences, School of Natural Sciences, Shiv Nadar University, Greater Noida, India
2Independent Scientist, Greater Noida, India
*Corresponding Author: Dr. Anindita Chakrabarty, Department of Life Sciences, School of Natural Sciences, Shiv Nadar University, Greater Noida, UP 201314, India
Received: 25 November 2020; Accepted: 03 December 2020; Published: 04 January 2021
Article Information
Citation: Shreya Sharma, Goutam Chowdhury, Anindita Chakrabarty. A Concise Overview of Chemotherapeutic Drugs in Gastrointestinal Cancers: Mechanisms of Action and Resistance. Journal of Cancer Science and Clinical Therapeutics 5 (2021): 011-035.
View / Download Pdf Share at FacebookAbstract
Gastrointestinal (GI) cancers are malignancies of the gastrointestinal tract and accessory organs of the digestive system including the esophagus, pancreas, stomach, colon, rectum, anus, liver, gallbladder, biliary system, and small intestine. These account for 28% of global cancer incidence and 35% of cancer-related mortalities. The most common type of GI cancers: colorectal and gastric cancers, rank among the top five cancers. Surgery, radiotherapy, chemotherapy, targeted therapy, and immunotherapy are available options for GI cancer treatment. With surgery being the first-line of choice, pre or post-operative chemotherapy is the second most-often used. Nevertheless, the long-term survival rate in GI cancer patients is modest due to the development of drug resistance, which can be overcome by administering various combinations of chemotherapeutic and targeted therapeutic drugs. The classic example is fluoropyrimidine, a class of cytotoxic drug used in combination with irinotecan, gemcitabine, and docetaxel to increase overall and progression-free survival in colorectal, pancreatic, and gastric cancers, respectively. Although in-depth expert reviews on select chemotherapies for the most common GI cancer types can be found, a concise overview of different drugs approved for all GI cancers is not available. In this review, we have compiled a list of the most common chemotherapeutic drugs used for GI cancers and summarized their major modes of action, intrinsic/adaptive resistance, and a few pre-clinical/clinical approaches to overcoming such resistance. We hope this manuscript will be useful for non-expert readers interested in a general overview of GI cancer chemotherapies.
Keywords
Chemotherapy drugs; Resistance mechanisms; Gastrointestinal cancers
Chemotherapy drugs articles; Resistance mechanisms articles; Gastrointestinal cancers articles
Chemotherapy drugs articles Chemotherapy drugs Research articles Chemotherapy drugs review articles Chemotherapy drugs PubMed articles Chemotherapy drugs PubMed Central articles Chemotherapy drugs 2023 articles Chemotherapy drugs 2024 articles Chemotherapy drugs Scopus articles Chemotherapy drugs impact factor journals Chemotherapy drugs Scopus journals Chemotherapy drugs PubMed journals Chemotherapy drugs medical journals Chemotherapy drugs free journals Chemotherapy drugs best journals Chemotherapy drugs top journals Chemotherapy drugs free medical journals Chemotherapy drugs famous journals Chemotherapy drugs Google Scholar indexed journals Resistance mechanisms articles Resistance mechanisms Research articles Resistance mechanisms review articles Resistance mechanisms PubMed articles Resistance mechanisms PubMed Central articles Resistance mechanisms 2023 articles Resistance mechanisms 2024 articles Resistance mechanisms Scopus articles Resistance mechanisms impact factor journals Resistance mechanisms Scopus journals Resistance mechanisms PubMed journals Resistance mechanisms medical journals Resistance mechanisms free journals Resistance mechanisms best journals Resistance mechanisms top journals Resistance mechanisms free medical journals Resistance mechanisms famous journals Resistance mechanisms Google Scholar indexed journals Gastrointestinal cancers articles Gastrointestinal cancers Research articles Gastrointestinal cancers review articles Gastrointestinal cancers PubMed articles Gastrointestinal cancers PubMed Central articles Gastrointestinal cancers 2023 articles Gastrointestinal cancers 2024 articles Gastrointestinal cancers Scopus articles Gastrointestinal cancers impact factor journals Gastrointestinal cancers Scopus journals Gastrointestinal cancers PubMed journals Gastrointestinal cancers medical journals Gastrointestinal cancers free journals Gastrointestinal cancers best journals Gastrointestinal cancers top journals Gastrointestinal cancers free medical journals Gastrointestinal cancers famous journals Gastrointestinal cancers Google Scholar indexed journals colorectal articles colorectal Research articles colorectal review articles colorectal PubMed articles colorectal PubMed Central articles colorectal 2023 articles colorectal 2024 articles colorectal Scopus articles colorectal impact factor journals colorectal Scopus journals colorectal PubMed journals colorectal medical journals colorectal free journals colorectal best journals colorectal top journals colorectal free medical journals colorectal famous journals colorectal Google Scholar indexed journals immunotherapy articles immunotherapy Research articles immunotherapy review articles immunotherapy PubMed articles immunotherapy PubMed Central articles immunotherapy 2023 articles immunotherapy 2024 articles immunotherapy Scopus articles immunotherapy impact factor journals immunotherapy Scopus journals immunotherapy PubMed journals immunotherapy medical journals immunotherapy free journals immunotherapy best journals immunotherapy top journals immunotherapy free medical journals immunotherapy famous journals immunotherapy Google Scholar indexed journals stomach cancers articles stomach cancers Research articles stomach cancers review articles stomach cancers PubMed articles stomach cancers PubMed Central articles stomach cancers 2023 articles stomach cancers 2024 articles stomach cancers Scopus articles stomach cancers impact factor journals stomach cancers Scopus journals stomach cancers PubMed journals stomach cancers medical journals stomach cancers free journals stomach cancers best journals stomach cancers top journals stomach cancers free medical journals stomach cancers famous journals stomach cancers Google Scholar indexed journals Oesophageal articles Oesophageal Research articles Oesophageal review articles Oesophageal PubMed articles Oesophageal PubMed Central articles Oesophageal 2023 articles Oesophageal 2024 articles Oesophageal Scopus articles Oesophageal impact factor journals Oesophageal Scopus journals Oesophageal PubMed journals Oesophageal medical journals Oesophageal free journals Oesophageal best journals Oesophageal top journals Oesophageal free medical journals Oesophageal famous journals Oesophageal Google Scholar indexed journals metastatic disease articles metastatic disease Research articles metastatic disease review articles metastatic disease PubMed articles metastatic disease PubMed Central articles metastatic disease 2023 articles metastatic disease 2024 articles metastatic disease Scopus articles metastatic disease impact factor journals metastatic disease Scopus journals metastatic disease PubMed journals metastatic disease medical journals metastatic disease free journals metastatic disease best journals metastatic disease top journals metastatic disease free medical journals metastatic disease famous journals metastatic disease Google Scholar indexed journals genitourinary articles genitourinary Research articles genitourinary review articles genitourinary PubMed articles genitourinary PubMed Central articles genitourinary 2023 articles genitourinary 2024 articles genitourinary Scopus articles genitourinary impact factor journals genitourinary Scopus journals genitourinary PubMed journals genitourinary medical journals genitourinary free journals genitourinary best journals genitourinary top journals genitourinary free medical journals genitourinary famous journals genitourinary Google Scholar indexed journals gastroesophageal junction articles gastroesophageal junction Research articles gastroesophageal junction review articles gastroesophageal junction PubMed articles gastroesophageal junction PubMed Central articles gastroesophageal junction 2023 articles gastroesophageal junction 2024 articles gastroesophageal junction Scopus articles gastroesophageal junction impact factor journals gastroesophageal junction Scopus journals gastroesophageal junction PubMed journals gastroesophageal junction medical journals gastroesophageal junction free journals gastroesophageal junction best journals gastroesophageal junction top journals gastroesophageal junction free medical journals gastroesophageal junction famous journals gastroesophageal junction Google Scholar indexed journals
Article Details
1. Introduction
Cancers of the organs that constitute our digestive system are collectively known as the gastrointestinal (GI) cancers. Oesophageal, liver, stomach, gallbladder and biliary tract, pancreatic, small bowel, anal cancers as well as gastrointestinal stromal tumors (GISTs) and neuroendocrine tumors (NETs), all fall into the broad category of GI cancers (https://gicancer.org.au/gi-cancer-explained/). According to Globocon 2018 report (www.uicc.org/news/new-global-cancer-data-globocan-2018), GI cancers represent one of the greatest public health issues worldwide, with the colorectal and stomach cancers alone being among the top five cancer types in terms of newly diagnosed cases (1.8 and 1.0 million, respectively) and overall mortality rates (881,000 and 783,000, respectively) [1]. In the USA, the prevalence and fatality rate of GI cancers have now surpassed that of lung cancers (333,680 and 167,790 versus 228,820 and 135,720) [2]. Treatment modality of GI cancers depends on the type of cancer, stage, and other general health factors. Although, early-stage GI cancers are amenable to surgery, their five-year relapse rate is quite high, which only marginally improves after the addition of chemo or radiation therapies. Unfortunately, about 25% of GI cancer patients are diagnosed at advanced stages and the other 25-50% of patients develop metastatic disease during treatment. In the last decade or so, the introduction of various molecularly targeted drugs including cetuximab, panitumumab, bevacizumab, regorafenib, sorafenib, irinotecan has improved the prognosis of metastatic GI cancers, however, the death rate continues to be quite high. Since the success of immunotherapy in melanoma, genitourinary, non-small cell lung, and hematological cancers, numerous clinical trials have been launched for GI cancers, especially with gastric, gastroesophageal junction, oesophageal, hepatic, and colorectal cancers. However, many of these are still in clinical development [3].
Until the 1990s, before the advent of molecular targeted therapies, cytotoxic drugs, especially fluoropyrimidine-based agents were the standard of care for GI cancers. Many of these drugs are still being used for GI cancer treatment but in combination with targeted therapies. For instance, in metastatic colorectal cancer, anti-angiogenic drugs (example, bevacizumab or aflibercept) are used in combination with fluoropyrimidines (5-FU or capecitabine) or irinotecan or oxaliplatin. A similar combination is also used in oesophageal and gastric cancers. However, the poorly vascularized pancreatic ductal adenocarcinomas are relatively resistant to a combination of chemotherapy (gemcitabine) and antiangiogenic therapies. More recently, anti-angiogenic therapies have been replaced by drugs targeting EGFR (like cetuximab or erlotinib) or multi-kinase inhibitors (like sorafenib or sunitinib or regorafenib) [4].
Drug resistance (against chemo or targeted therapy) is an inevitable problem in cancer treatment and emergence of therapeutic resistance in GI cancers is no exception to this phenomenon. Tumor drug resistance can be intrinsic (primary) or adaptive (secondary) and may be contributed by one or more of the following mechanisms including altered drug transport, mutations to drug target, enhanced DNA damage repair, rewiring of signalling pathways, changes in drug metabolism, resistance to apoptosis, enrichment of cancer stem cells and transformed tumor microenvironment. Several comprehensive reviews discussing these general mechanisms of anti-cancer drug resistance are available [5, 6]. In this review, we will summarize the most well-known mechanisms of action and resistance of common chemotherapeutic drugs against different GI cancer types. We will also briefly mention alternate therapeutic strategies that have been proposed or adapted in the clinic to avoid and/or overcome GI cancer chemoresistance. Our goal is to make a concise overview of GI cancer chemotherapies available to non-expert readers.
|
Drug Name |
Used in Cancer types |
Broad Mode of action |
|
5-Fluorouracil |
Stomach, colon and anal |
Anti-metabolite |
|
Capecitabine |
Colorectal, Esophageal, hepatobiliary, neuroendocrine, pancreatic |
Anti-metabolite |
|
SI |
Gastric and pancreatic |
Anti-metabolite |
|
Tegafur-uracil |
Colorectal |
Anti-metabolite |
|
Irinotecan |
Colorectal, gastric, pancreatic |
Topoisomerase I inhibitor |
|
Oxaliplatin |
Colorectal |
DNA damage |
|
Cisplatin |
Esophageal and stomach |
DNA damage |
|
Gemcitabine |
Pancreatic |
Anti-metabolite |
|
Trifluridine/tipiracil |
Esophageal and stomach |
Anti-metabolite |
|
Docetaxel |
Stomach |
Microtubule inhibitor |
|
Leucovorin |
Esophageal and colorectal |
Anti-metabolite |
|
Etoposide (VP-16) |
Stomach |
Topoisomerase II inhibitor |
Table 1: List of chemotherapies, their most common uses, and broad modes of action in GI cancers.
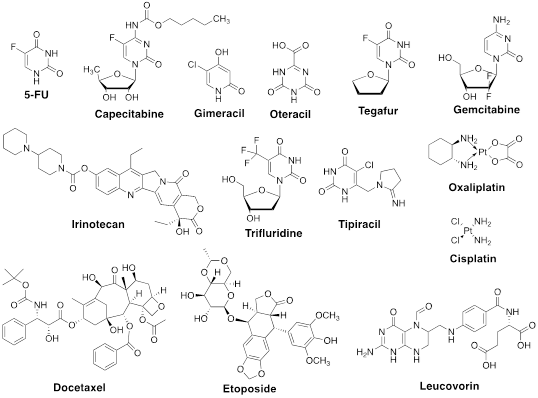
Figure 1: Structures of chemotherapeutic drugs discussed herein.
2. Fluoropyrimidine
5-Flurouracil (5-FU) and its prodrug capecitabine, known as fluoropyrimidines (FPs) kill tumor cells by creating imbalances in the nucleotide pool, resulting in inhibition of DNA replication, transcription, and repair [7]. Although used in several cancer types, FPs exerts the most impact on colorectal cancer treatment [8]. Below, we will briefly discuss their mechanisms of action and resistance.
2.1 5-FU
5-FU, a prototypical FP, one of the first chemotherapeutic drugs reported to have anti-cancer activities is a synthetic fluorinated analog of pyrimidine base. Administered intravenously, 5-FU’s cellular import relies on the same facilitated transport mechanism used by uracil. When inside the cell, it is readily converted into several active metabolites: fluorodeoxyuridine monophosphate (FdUMP), fluorodeoxyuridine triphosphate (FdUTP), and fluorouridine triphosphate (FUTP). The primary metabolite FdUMP inhibits thymidylate synthase (TS), an important enzyme involved in the generation of DNA nucleotide dTMP (deoxythymidine monophosphate). This impairs DNA replication and repair. Other 5-FU metabolites FUTP and FdUTP are incorporated into DNA as false nucleotides, interfering with transcription. Collectively, these result in apoptotic cell death. The majority (80%) of the 5-FU is catabolized into dihydrofluouracil (DHFU) in the liver that abundantly expresses the enzyme dihydropyrimidine dehydrogenase (DPD). This decreases the bioavailability of the drug [9].
One of the most well-known modes of 5-FU resistance involves overexpression of its target enzyme TS. In fact, TS expression level has long been recognized as a primary predictor of 5-FU therapy success. Either copy number variation or alteration in the promotor region of the gene encoding TS, TYMS results in intrinsic resistance to 5-FU. Free TS controls its expression level by binding to its mRNA and constituting a negative feedback control on its translation. In 5-FU-treated tumor cells, TS binding to FdUMP disrupts this feedback loop, increasing the TS level, and decreasing the sensitivity to 5-FU. This plays an important role in the emergence of adaptive resistance to 5-FU [10]. Formation of an inactive ternary complex between FdUMP and 5,10-methylenetetrahydrofolate (MTTHF) is required for the inhibitory effect of FdUMP on TS. An elevated level of MTHF is important for appropriate inhibition of TS, which is regulated by the enzyme methylenetetrahydrofolate reductase (MTHFR). Therefore, along with TS, MTHFR activity is also a determinant of 5-FU sensitivity in patients. Genetic polymorphisms affecting expression levels and activities of both TS and MTHFR have been reported. Another mechanism of 5-FU resistance is associated with the activities of three enzymes thymidine phosphorylase (TP), uridine phosphorylase (UP), and orate phosphoribosyl transferase (OPRT), necessary for the conversion of 5-FU to 5-FdUTP and 5-FUTP metabolites. However, multiple studies have produced mixed results and require further confirmation [11-16]. Conversion of 5-FU to DHFU by the enzyme DPD is required for increasing its solubility and urinary excretion of the drug. Higher DPD expression in tumor cells has been associated with intrinsic 5-FU resistance. However, definite proof for the same mechanism leading to adaptive 5-FU resistance is lacking.
2.2 Capecitabine
Capecitabine, an orally bioavailable FP was developed to mimic the continuous infusion of 5-FU. It is bioactivated when inside the tumor cells. Capecitabine is a prodrug that is metabolized to 5-FU in different steps. Thymidine phosphorylase (TP), one of the enzymes involved in this process is abundantly expressed in the tumor compared to healthy tissues. This led to the expectation that this drug would have less systemic toxicity. However, in clinical settings this has not been proven correct [17]. Since capecitabine is converted into 5-FU within the tumor, these two drugs share several common mechanisms of resistance. Specifically, DPD expression has been associated with capecitabine resistance [18]. TP, the enzyme that converts capecitabine into 5-FU, is expressed in the tumor microenvironment, rather than the tumor cells themselves. High TP expression correlates with better response to capecitabine, while its loss of function is associated with capecitabine resistance. This could happen by abnormal splicing of TP mRNA due to increased expression of the splicing factors [19, 20].
2.3 S1 and tegafur-uracil
S1, consisting of tegafur (UFT), gimeracil (5-chloro-2, 4-dihydroxypryridine), and oteracil (potassium oxanate) is a fourth-generation oral FP. Tegafur is a prodrug that is converted into FU within tumor cells, while gimeracil interferes with its metabolism by inhibiting DPD and oteracil reduces the toxic side effect of 5-FU by reducing its phosphorylation in the GI tract through inhibition of the enzyme orate phosphoribosyl transferase [21]. Tegafur-uracil consists of tegafur attached to uracil that blocks DPD-mediated catabolism. It is metabolized by the cytochrome P-450 CYP2A6 gene product. Tegafur’s high systemic toxicity in white patients than in East Asian patients is likely associated with its altered metabolism due to polymorphism in the cytochrome P-450 CYP2A6 gene. This is caused by the different extent of conversion of tegafur into 5-FU, responsible for its cytotoxic effects [22]. This drug is not approved in the USA because of its high toxicity [23].
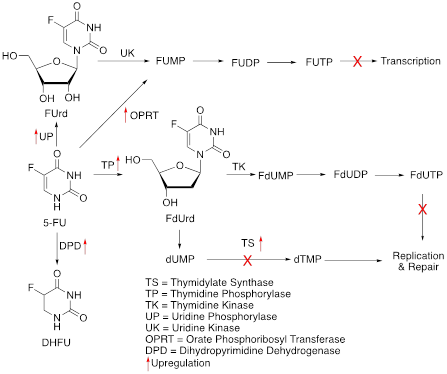
Figure 2: Schematics of 5FU mechanisms of action and resistance. Red arrows indicate altered expression leading to resistance.
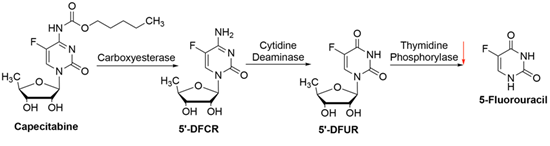
Figure 3: Schematics of capecitabine conversion to FP and its resistance mechanisms. The red arrow indicates altered expression leading to resistance.
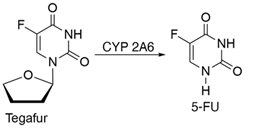
Figure 4: Schematics of tegafur conversion into FP.
3. Irinotecan
Irinotecan is a semi-synthetic derivative of natural DNA-topoisomerase inhibitor camptothecin. DNA topoisomerase-I cleaves one strand of a double-stranded DNA relaxing its supercoiled structure and further re-ligates it. Camptothecin, by forming a complex with topoisomerase I, prevents the re-ligation step causing DNA strand breakage and ultimately, cell death [24]. Irinotecan must be converted to its active metabolite SN-38 by the enzyme carboxylesterase, which reversibly binds to DNA topoisomerase-I, stabilizing it. Irinotecan causes obstruction in DNA synthesis and transcription and works more effectively in combinatorial therapy with FPs, 5-FU and capecitabine, and/or oxaliplatin than as a single agent [25].
Overexpression of the ABC-family of transporter proteins such as multidrug resistance protein (MRP) and P-glycoprotein (Pgp) plays a crucial role in the emergence of resistance by decreasing the intracellular concentration of irinotecan and its metabolite SN-38 [26]. The active involvement of Pgp and MRP in the efflux of SN-38 and irinotecan has been shown in several human epidermoid carcinoma KB-3-1-derived cell lines overexpressing the said transporter proteins, implying their role in the development of intrinsic resistance [27]. SN-38 is converted to SN-38 glucuronosyl (SN-38G) by the action of uridine diphosphate glucuronosyl transferase (UGT) in the liver. This is a step in the drug detoxification process. Enhanced clearance through UGT confers irinotecan resistance by reducing SN-38 concentration in the tumor cells [28]. Another mode of adaptive resistance to irinotecan is the alterations identified in the gene encoding DNA topoisomerase I enzyme. Point mutations within this gene decrease the binding affinity of SN-38 to the enzyme [29, 30]. More specifically, these mutations include changes in the catalytic tyrosine which renders the enzyme catalytically inactive [31, 32]. Insufficient levels of the topoisomerase gene expression, a result of transcriptional silencing due to hypermethylation could attribute to intrinsic resistance. This diminished yield of the enzyme stems from a rearrangement in the Topo-1 genome [20, 25]. Lastly, a lower level of acetylated histone H4K16 is associated with the resistance to irinotecan. H4K16 acetylation is inversely linked to the levels of p53-binding protein (53BP1) repair factor which is responsible for regulating the repair mechanisms of DNA double-stranded breaks. A diminished amount of acetylation implicates dysregulation of H4K16Ac steady levels and a greater accumulation of 53BP1 factor, resulting in mitigating irinotecan’s anti-cancer effects [33].
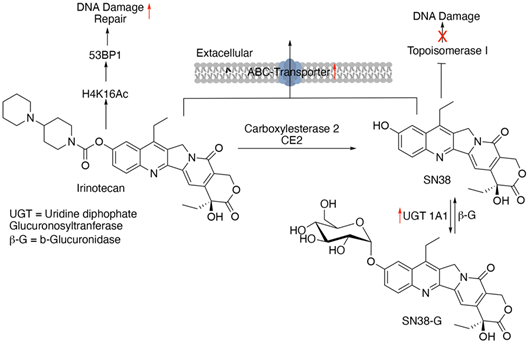
Figure 5: Schematics of conversion of irinotecan to SN-38, their mechanism of action, and resistance. Red arrows indicate altered expression leading to resistance.
4. Oxaliplatin
Oxaliplatin is a third-generation platinum-based chemotherapeutic drug which contains a bidentate (chelating) ligand 1,2-diamniocyclohexane. It has shown about 50% higher progression-free survival (PFS) and overall survival (OS) when given in combination with 5-FU and leucovorin (folinic acid) [34]. Oxaliplatin induces apoptosis by cross-linking (both inter and intra) DNA strands, preventing DNA replication and transcription [35].
Glutathione (GSH) is one of the most potent endogenous antioxidants that prevents the binding of reducing agents such as free radicals and electrophiles to DNA and proteins, thereby preventing them from causing cellular damage. Cancer cells often express increased levels of GSH which is essential for their survival and cell cycle progression [36]. Platinum based compounds like oxaliplatin form conjugates with GSH which facilitates their efflux from the cells via the ABC transporter proteins [37]. Hence, certain tumor cells could become resistant due to the excess GSH levels, in oxaliplatin-based therapy [38]. Nucleotide excision repair (NER) mechanism is responsible for eliminating DNA-Pt adducts particularly the intra-strand cross-links, which are responsible for the antitumor effects of oxaliplatin. Upregulation of genes encoding excision repair cross-complementation group 1 and 2 (ERCC1 and 2) proteins, X-ray cross-complementing group 1 (XRCC1), and xeroderma pigmentosum group D (XDP), primarily involved in the NER pathway are also contributing factors for oxaliplatin acquired resistance [39, 40]. Elevated mRNA expression of ERCC1 in oxaliplatin-resistant tumours and SNPs identified in ERCC1 (354C>A) along with XRCC1 (1196A>G) have been recorded in retrospective analyses of tumor samples [41, 42]. However, forthcoming studies of analyses are still awaited. The breast cancer gene 1, BRCA1 and its interacting partner SRBC (Serum-deprivation response factor-related gene product that binds to c-kinase) are involved in the homology-directed repair (HDR) of double-stranded breaks. Oxaliplatin-induced double-stranded breaks are repaired by these two proteins. Inactivation or depletion of BRCA1 interactor SRBC as a result of methylation is associated with oxaliplatin resistance in colorectal cancer cells [41, 43]. In fact, hypermethylation of SRBC1 has been linked to poor outcome of oxaliplatin treatment [43]. Concrete evidence for the involvement of epigenetic changes specifically in CRC resistance is however, lacking. Finally, cytokine transforming growth factor β1 (TGF-β1), secreted abundantly by tumor cells and known to promote epithelial to mesenchymal transition (EMT) has also been associated with the emergence of oxaliplatin resistance [44].
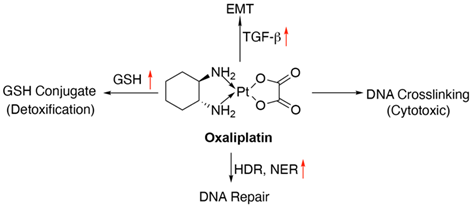
Figure 6: Schematics of oxaliplatin mode of action and resistance mechanisms. Red arrows indicate altered expression leading to resistance.
5. Cisplatin
Cisplatin is a well-known platinum-based chemothe-rapeutic drug used for the treatment of several GI cancers including oral and oropharyngeal, gall bladder and biliary duct cancer (https://www.cancer.net/cancer-types). Its mode of action is associated with altering DNA repair mechanisms once it is absorbed into the cell leading to DNA damage by the formation of DNA strand cross-linking, which ultimately results in apoptotic cell death [45]. It binds to the N7 atoms of purines (mainly guanine) and form a complex called as the 1,2-intrastand adduct.
DNA repair pathway alterations modulate cisplatin efficacy. Expression of ERCC1, a key player in the NER pathway is enhanced by cisplatin, via the MAPK signalling [46, 47]. This leads to an increased recognition of DNA lesions, counteracting the drug’s effect. Dual Specificity Phosphatase-1 (DUSP1) is one of many chemotherapy resistance-associated genes, and a potential therapeutic target to enhance the cisplatin treatment efficacy in gall bladder cancer. Overexpression of DUSP1 is associated with attenuation of p38 MAPK signalling, preventing tumor cell apoptosis by cisplatin [48]. Cisplatin resistance has also been associated with EMT induction in tongue squamous cell carcinoma (TSCC) cells along with a decrease in the expression of microRNAs miR-200b and miR-15b. These microRNAs functions in metastasis inhibition by obstructing EMT [41, 49].
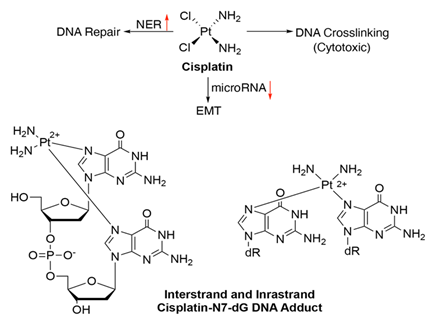
Figure 7: Schematics showing the mechanisms of action and resistance for cisplatin. Structure of cisplatin intra- and inter-strand N7-guanine DNA adducts. Red arrows indicate altered expression leading to resistance.
6. Gemcitabine
Gemcitabine, also referred to as dFdC or 2’,2’-difluorodeoxycitidine is the standard choice of treatment for locally advanced and metastatic pancreatic cancers [50]. It is also used in advanced gallbladder cancers [51]. Being a deoxycytidine nucleoside analogue it hinders DNA synthesis and cell cycle at the G1/S-Phase junction [52]. Once imported into the cells by the nucleoside transporters (NTs), it is converted into three metabolites by sequential phosphorylation-first to gemcitabine monophosphate (dFdCMP), then to gemcitabine diphosphate (dFdCDP) and finally to gemcitabine triphosphate (dFdCTP) by the action of deoxycytidine kinase (dCK), pyrimidine nucleoside monophosphate kinase (NMPK) and nucleoside diphosphate kinase (NDPK), respectively [53, 54]. During replication, dFdCTP competes with deoxycytidine triphosphate (dCTP) to integrate into DNA, blocking DNA synthesis and ultimately resulting in cell death [55].
Bioavailability of gemcitabine or dFdC is highly reduced by cytidine deaminase (CDA) that deaminates dFdC to produce less active metabolite-2',2'-diflurodeoxyuridine (dFdU) [56]. Even the phosphorylated metabolites of gemcitabine are converted into their inactivated forms, for example: dFdCMP is converted to 2'-deoxy-2',2'-difluorouridine monophosphate (dFdUMP) by deoxycytidylate deaminase (DCTD) [54]. Inadequate expression of NTs such as human equilibrative nucleoside transporters (hENT1) in pancreatic cancer cells have been associated with gemcitabine resistance due to insufficient transport into the cells [57, 58]. Deoxycytidine kinase (dCK) is the major rate limiting enzyme in the intracellular activation pathway of gemcitabine’s metabolites [59]. Inactivation of dCK gene has also been exhibited in human pancreatic cancer cell lines with acquired resistance [60]. Increase in the concentration of dFdU, the inactive metabolite of Gemcitabine is another main cause of diminished effect of dFdC activity [59]. It was successfully demonstrated in in vitro studies that overexpression of CDA which generates such metabolites is responsible for emergence of gemcitabine resistance [61-63].
Similar to cisplatin resistance, DUSP1 overexpression is also associated with gemcitabine insensitivity through modulation of p38 MAPK in pancreatic ductal carcinoma cells and DUSP1 inhibition can enhance gemcitabine sensitivity [64]. The enzyme ribonucleotide reductase (RR) is crucial for DNA synthesis [65]. It regulates the concentration of NTP pool and is responsible for converting CDP to dCDP, followed by its phosphorylation to dCTP, which finally gets incorporated into the DNA. The gemcitabine metabolite, dFdCDP inhibits RR leading to a disruption in the nucleotide pool and facilitating the addition of dFdCTP in place of dCTP into the DNA. Moreover, reduced concentration of dCTP fails to activate the deaminase DCTD, rendering it unable to degrade dFdCMP to dFdUMP [53, 66, 67]. Therefore, RR inhibition or activation plays a vital role in determining gemcitabine sensitivity. It has been reported that enhanced expression of the catalytic subunit of RR, RRM1 is linked to poor overall survival (OS) in pancreatic cancer patients, implying its role in intrinsic resistance to gemcitabine [68-70].
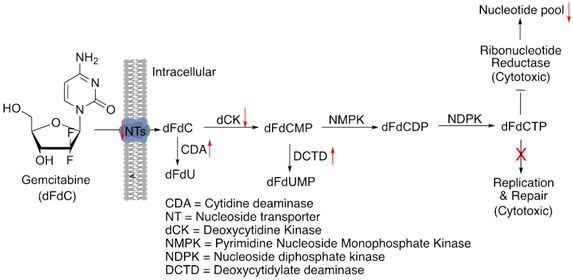
Figure 8: Schematics of gemcitabine’s mechanisms of action and resistance. Red arrows indicate altered expression leading to resistance.
7. Trifluridine/tipiracil
Trifluridine (FTD) is a novel drug used to treat patients with metastatic colorectal cancer who have developed resistance to standard chemotherapies such as fluoropyrimidine, oxaliplatin and irinotecan-based chemotherapy, and also to EGFR and VEGFR targeted therapies [71]. It is also approved for advanced gastric cancers refractory to several lines of treatments [72]. More recently, it is being tested with immune checkpoint inhibitor in microsatellite-stable (MSS) refractory metastatic colorectal cancers [73]. Trifluridine is a thymidine analogue which acts in combination with tipiracil, a thymidine phosphorylase inhibitor [74]. Trifluridine is incorporated into the DNA as its phosphorylated metabolite, triflourothymidine-5'-triphosphate (F3dTTP) in place of thymidine, obstructing DNA synthesis and tumor cell growth. However, it is also rapidly degraded by the enzyme thymidine phosphorylase (TPase), which is prevented by tipiracil, resulting in the increased bioavailability of trifluridine [72]. This unique feature of trifluridine/tipiracil combination compared to other FPs has rendered it a preferred choice against most 5-FU/FP-resistant GI cancer cell lines and clinical samples [73].
Absence of a functional thymidine kinase 1 (TK1) protein in human colorectal cancer cell lines was shown as an adaptive mechanism of FTD resistance. TK1 is responsible for the conversion of FTD to F3dTMP [75]. Downregulation of Let-7, a famous tumor suppressor microRNA has been correlated with FTD-induced acquired resistance in the colorectal adenocarcinoma cell line DLD-1. There is an inverse relationship between expression of Let-7d-5p miRNA from chromosome 9 and FTD-induced anti-proliferative effects [74].
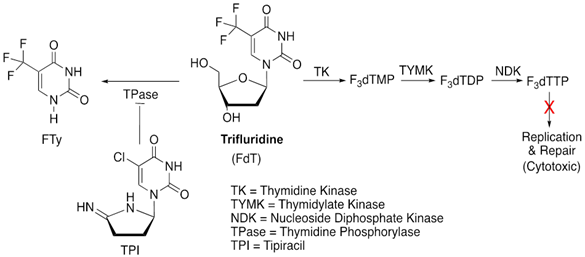
Figure 9: Schematics for the mode of action of trifluridine/tipiracil (TPI).
8. Docetaxel
Chemotherapies for gastric cancer patients include different combinations of 5-FU, platinum based agents like cisplatin and oxaliplatin, irinotecan and taxanes such as docetaxel and paclitaxel (https://www.cancer.net/cancer-types/stomach-cancer/types-treatment). Docetaxel is a second generation taxane that arrests cell cycle by inhibiting mitosis. It stimulates the polymerisation of tubulin by stabilizing the binding of the microtubules to β-actin. As a result, microtubule depolymerisation is inhibited and cell division is arrested at the G2-M Phase, ultimately inducing apoptosis [76].
With an increase in the efficacy of docetaxel as a third-line treatment for refractory gastric cancer, there is also an increasing emergence of resistance against this drug. Forkhead box protein M1 (FOXM1) is a transcription factor that plays an important role in cell cycle progression from the G1 to S phase and promotes the cell’s transition into mitosis [77, 78]. Overexpression of FOXM1 has been associated with acquired resistance to docetaxel in gastric cancers via the direct upregulation of the microtubule-destabilising protein Stathmin [79]. Stathmin overexpression promotes microtubule depolymerisation, counteracting docetaxel action and preventing tumor cell apoptosis [80-83]. Low ratio of soluble to polymerized tubulin was observed in cells overexpressing FOXM1 which is a prominent indication of decreased docetaxel sensitivity [80]. Overexpression of class III β-tubulin (TUBB3) has been studied in depth in non-small cell lung cancer (NSCLC) as a contributor to taxane resistance [84, 85]. However, its significance in gastric cancer is unclear. In a small cohort of advanced gastric cancer patients, enhanced TUBB3 expression was associated with a lack of the desired clinical effect of (preoperative) docetaxel-based treatment [86]. Using cell line models of docetaxel-resistant gastric cancer, expression levels of CXCR4, a chemokine receptor for stromal cell-derived factor-1/SDF-1 was found as a predictor of docetaxel sensitivity. This was verified in gastric cancer specimens as well. In this study, a CXCR4 antagonist, AMD3100 was used to restore docetaxel sensitivity [87]. In another study with gastric cancer cell lines and patient samples, BAK (a proapoptotic member of the BCL2 family) expression was found as a strong predictor of docetaxel sensitivity. Patients with BAK index (as measured by immunohistochemical analysis) equal to or more than 3 had better progression-free and overall survival [88]. In a number of gastric cancer cell lines, docetaxel sensitivity was correlated with the miRNA Let-7a expression level, with lower expression found in the drug-resistant line [89].
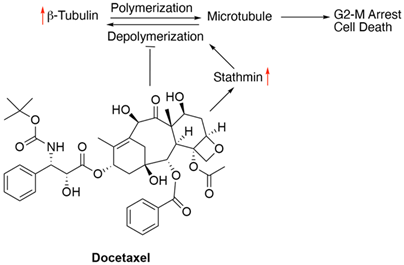
Figure 10: Schematics of docetaxel’s mechanism of action and resistance. Red arrows indicate alteration of expression leading to resistance.
9. Leucovorin
Leucovorin (LV) is a 5-formyl derivative of tetrahydrofolic acid (THF) and is best known to facilitate the action of 5-FUs and its derivatives, which inhibit the enzyme TS responsible for DNA synthesis and repair. Administration of LV leads to an increase in the intracellular concentration of 5,10-methylene tetrahydrofolate (CH2THF) which is responsible for an improved stabilisation of the ternary complex of CH2THF with TS and FdUMP, via its polyglutamation [90]. LV is used as an adjuvant for many first-line treatments for metastatic colorectal cancer, including FOLFOX (5-FU, LV and oxaliplatin) [23].
In their proteomic studies, Carloni, V. et al demonstrated that in metastatic colon cancer cells, ADAM metallopeptidase domain 10 (ADAM10), GTP-binding protein α13 (Gα13) and Ras homolog family member A (RHOA) promote tumor cell fusion, which increases in cells resistant to both 5-FU and oxaliplatin [91]. Therefore, this phenomenon was implicated in the development of multidrug resistance to FOLFOX and poor prognosis. In another study with a large number of advanced gastric cancer clinical samples roles of oncogenes encoding the RAS and b-catenin in the resistance and recurrence following FOLFOX treatment was indicated. In patient-derived xenografts (PDX) from the same clinical samples, resistance to FOLFOX was further correlated with increase in cancer stem cell (CSC)-specific marker expression in addition to enhancement of RAS and b-catenin levels. Treatment with KYA1797K, a small molecule capable of degrading both RAS and b-catenin suppressed the PDX tumors with acquired resistance to FOLFOX and reduced CSC marker expression [92]. In advanced colorectal cancer, another mechanism of poor sensitivity to 5-FU plus LV is associated with p53 overexpression [93]. This could be due to the effect of TS on p53 expression. However, the exact nature of the relationship between these two is still ambiguous. Multiple studies have demonstrated contrasting roles of ABC family drug transporters in FOLFOX resistance in colorectal cancers. While some correlated high expression levels of ABC family members with reduced drug sensitivity, others did not find a significant association between the two. It is possible that the biological role of ABC transporters in FOLFOX response may vary with cancer stages [94].
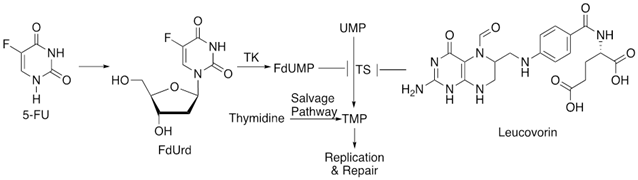
Figure 11: Schematics of leucovorin’s mechanism of action.
10. Etoposide (VP-16)
Etoposide is an eukaryotic topoisomerase-II inhibitor which stabilizes the TopoII cleavage complexes and inhibits DNA re-ligation during DNA replication, repair, transcription and chromatin remodelling [95]. The primary mechanism of etoposide resistance is the decrease in the expression level of its target enzyme, TopoII [96]. Additionally, the altered expression of the multidrug resistance protein 1 (MRP1), a transporter protein encoded by ABCC1 gene which serves multiple functions such as carrying out efflux of drugs, organic anions and several lipid-derived mediators from cells, is implicated in developing etoposide resistance [97]. An increased expression of MRP1 protein is directly correlated with etoposide resistance in stomach cancer cell lines as well oral squamous cell carcinoma (OSCC) [46, 98]. Furthermore, as demonstrated in vitro, down regulation of pro-apoptotic proteins, Bid and/or Bax contributed to lower sensitivity to etoposide in a hypoxic (low oxygen) environment, which decreased with further reduction in oxygen level [99].
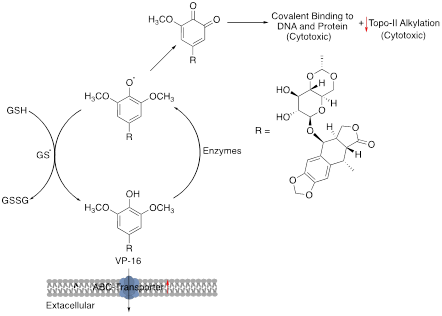
Figure 12: Schematics of etoposide mechanism of action and resistance. Red arrows indicate alteration of expression leading to resistance.
11. Strategies of Overcoming Chemotherapy Resistance
Development of chemoresistance is a major clinical challenge for managing any cancer type. In the previous section, we have discussed several modes of resistance mechanisms of various drugs used for GI cancer treatment and the molecular players involved in these processes. Several of these have been the focus of clinical studies and trials since many years. Some of these have even been adapted in clinic for overcoming chemoresistance in GI cancers. Below, we will briefly mention a handful of strategies that according to us are quite interesting.
DPD-inhibitory fluoropyrimidines (DIF) have been in use as neoadjuvant and/or adjuvant chemotherapy in cases of advanced gastric carcinoma [100] to combat high DPD level-induced 5-FU resistance. Melatonin, a hormone involved in regulating the 24-hour internal clock (circadian rhythm), has been proposed to enhance anticancer effects of 5-FU by downregulating the expression of TYMS via upregulation of the microRNA miR-215-5p, making it a promising therapeutic agent to overcome chemoresistant CRC [101]. Quantifying the expression of OPRT in patients using sandwich ELISA has been suggested to help monitor the efficacy and sensitivity of S-1 based chemotherapy and predict the need of reversal of resistance strategies in advanced gastric carcinomas [102]. Combination treatment with histone deacetylase (HDAC) inhibitor is a an FDA approved clinical strategy to mechanistically overcome irinotecan chemoresistance by enriching H4K16Ac levels and thus maintaining optimum concentrations of 53BPI [36]. A recent study has shown to enhance the effect of gemcitabine in argininosuccinate synthetase-1 negative (ASS1-) tumors of pancreas and few other cancer cell lines, such as sarcoma and melanoma. Starving the tumor cells of arginine deiminase (ADI-PEG20) and priming it with docetaxel treatment leads to the transport of c-MYC into the nucleus resulting in the increased expression of hENT1, thereby counteracting one of the factors which confers resistance, by increasing the uptake of gemcitabine by tumor cells [103]. As gemcitabine resistance is associated with overactivation of the NF-kB signaling pathway, use of melatonin to block this pathway and overcome drug resistance has been proposed in pre-clinical setting (https://doi.org/10.1111/jpi.12285). Pre-clinical and clinical studies also tested the efficacy of blocking the tyrosine kinase receptor c-Met signaling in gemcitabine-resistant pancreatic cancer, based on the effect of this pathway on CSC population [104]. Over expression of TOP2A to increase the sensitivity to TopoII inhibitory agents like etoposide has been extensively studied [105-107]. However, it requires further investigation to achieve its full therapeutic potential. There is also evidence that MK571, an MRP inhibitor restores etoposide sensitivity of MRP1-expressing stomach cancer cell lines by enhancing the cell-to-medium ratio of etoposide [97]. Since the discovery of the first pgp inhibitor in 1980’s, many other inhibitors have been developed and some of those even been tested in clinic for reversal of multidrug resistance. Lack of selectivity and potency, high toxicity are some of the reasons why none of these have yet been approved for patient use [108]. Majority of chemotherapeutics drugs exert cytotoxicity by inducing apoptosis and apoptosis suppression is a common feature of most drug-resistant cancer cells. Reversal of apoptosis resistance by reactivating apoptotic signaling pathways and/or activating alternate cell death pathways are popular approaches for overcoming chemoresistance in GI cancers. For example, several of the therapeutic agents among BH3 mimetics, EGFR inhibitors, autophagy inducers have already been introduced in clinic for treatment of advanced colorectal cancers [108].
12 Conclusion
In this review, we have discussed the major chemotherapeutic drugs used in the treatment of GI cancers, namely 5-FU, capecitabine, S1 (tegafur-uracil), irinotecan, oxaliplatin, cisplatin, gemcitabine, trifluridine, docetaxel, leucovorin and etoposide as well as some of their most noteworthy modes of resistance. We have also touched upon a few approaches of overcoming resistance to some of these drugs. Since the discovery of several targeted therapies and their proven efficacies in clinic for GI cancers such as metastatic CRC, pancreatic cancer and hepatocellular carcinoma (HCC), a majority of chemotherapeutic drugs have been replaced by or used in combination with the anti-EGFR therapies cetuximab [109, 110], panitumumab [111, 112] and erlotinib [113, 114] or anti-angiogenic therapies bevacizumab [115, 116], regorafenib [117, 118], aflibercept [119, 120] and ramucirumab [110] or multi-kinase inhibitors sorafenib [121] and sunitinib [122]. Recently, the success of immunotherapy in lung cancer [123] and melanoma [124, 125] has prompted scientists and oncologists to deploy the use of the same in GI cancers. We believe that the next generation therapy for GI cancer will be a combination of chemotherapy and targeted therapy or immunotherapy. In conclusion, exciting discoveries will continue to pave the way for more effective treatment options to improve the survival rate in GI cancer patients.
Acknowledgements
None.
Authors’ contribution
SS, AC and GC wrote the manuscripts.
Financial Support
None.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68 (2018): 394-424.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin 70 (2020): 7-30.
- Tannapfel A, Reinacher-Schick A. Immunotherapy in gastrointestinal cancer: where do we stand?. Visc Med 35 (2019): 1-2.
- Neuzillet C, Rousseau B, Kocher H, et al. Unravelling the pharmacologic opportunities and future directions for targeted therapies in gastro-intestinal cancers Part 1: GI carcinomas. Pharmacol Ther 174 (2017): 145-172.
- Cree IA, Charlton P. Molecular chess? Hallmarks of anti-cancer drug resistance. BMC Cancer 17 (2017): 10.
- Housman G, Byler S, Heerboth S, et al. Drug resistance in cancer: an overview. Cancers (Basel) 6 (2014): 1769-1792.
- Wilson PM, Danenberg PV, Johnston PG, et al. Standing the test of time: targeting thymidylate biosynthesis in cancer therapy. Nat Rev Clin Oncol 11 (2014): 282-298.
- Longley DB, Harkin DP, Johnston PG. 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer 3 (2003): 330-338.
- Diasio RB, Harris BE. Clinical pharmacology of 5-fluorouracil. Clin Pharmacokinet 16 (1989): 215-237.
- Chu E, Voeller DM, Jones KL, et al. Identification of a thymidylate synthase ribonucleoprotein complex in human colon cancer cells. Mol Cell Biol 14 (1994): 207-213.
- Freund M, Cazenave JP, Courtney M, et al. Inhibition by recombinant hirudins of experimental venous thrombosis and disseminated intravascular coagulation induced by tissue factor in rats. Thromb Haemos 63 (1990): 187-192.
- Metzger R, Danenberg K, Leichman CG, et al. High basal level gene expression of thymidine phosphorylase (platelet-derived endothelial cell growth factor) in colorectal tumors is associated with nonresponse to 5-fluorouracil. Clin Cancer Res 4 (1998): 2371-2376.
- Koopman M, Venderbosch S, van Tinteren H, et al. Predictive and prognostic markers for the outcome of chemotherapy in advanced colorectal cancer, a retrospective analysis of the phase III randomised CAIRO study. Eur J Cancer 45 (2009): 1999-2006.
- Tokunaga Y, Sasaki H, Saito T. Clinical role of orotate phosphoribosyl transferase and dihydropyrimidine dehydrogenase in colorectal cancer treated with postoperative fluoropyrimidine. Surgery 141 (2007): 346-353.
- Isshi K, Sakuyama T, Gen T, et al. Predicting 5-FU sensitivity using human colorectal cancer specimens: comparison of tumor dihydropyrimidine dehydrogenase and orotate phosphoribosyl transferase activities with in vitro chemosensitivity to 5-FU. Int J Clin Oncol 7 (2002): 335-342.
- Marrangoni AG, Marcelli G, Culig M, et al. Vascular grafts in microvascular surgery. An experimental study. Am J Surg 155 (1988): 258-262.
- Cassidy J, Saltz L, Twelves C, et al. Efficacy of capecitabine versus 5-fluorouracil in colorectal and gastric cancers: a meta-analysis of individual data from 6171 patients. Ann Oncol 22 (2011): 2604-2609.
- Podoba J. Etiologic factors of endemic goiter and problems of its prevention in Slovakia. Bratisl Lek Listy 59 (1973): 385-396.
- Petrioli R, Bargagli G, Lazzi S, et al. Thymidine phosphorylase expression in metastatic sites is predictive for response in patients with colorectal cancer treated with continuous oral capecitabine and biweekly oxaliplatin. Anticancer Drugs 21 (2010): 313-319.
- Stark M, Bram EE, Akerman M, et al. Heterogen-eous nuclear ribonucleoprotein H1/H2-dependent unsplicing of thymidine phosphorylase results in anticancer drug resistance. J Biol Chem 286 (2011): 3741-3754.
- Sakuramoto S, Sasako M, Yamaguchi T, et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med 357 (2007): 1810-1820.
- Wang H, Bian T, Liu D, et al. Association analysis of CYP2A6 genotypes and haplotypes with 5-fluorouracil formation from tegafur in human liver microsomes. Pharmacogenomics 12 (2011): 481-492.
- Hammond WA, Swaika A, Mody K. Pharmacologic resistance in colorectal cancer: a review. Ther Adv Med Oncol 8 (2016): 57-84.
- Xu Y, Villalona-Calero MA. Irinotecan: mechanisms of tumor resistance and novel strategies for modulating its activity. Ann Oncol 13 (2002): 1841-1851.
- Shimada Y, Rougier P, Pitot H. Efficacy of CPT-11 (irinotecan) as a single agent in metastatic colorectal cancer. Eur J Cancer. 32A Suppl 3 (1996): S13-S17.
- Loe DW, Deeley RG, Cole SP. Biology of the multidrug resistance-associated protein, MRP. Eur J Cancer 32a (1996): 945-957.
- Chu XY, Suzuki H, Ueda K, et al. Active efflux of CPT-11 and its metabolites in human KB-derived cell lines. J Pharmacol Exp Ther 288 (1999): 735-741.
- Cummings J, Boyd G, Ethell BT, et al. Enhanced clearance of topoisomerase I inhibitors from human colon cancer cells by glucuronidation. Biochem Pharmacol 63 (2002): 607-613.
- Tsurutani J, Nitta T, Hirashima T, et al. Point mutations in the topoisomerase I gene in patients with non-small cell lung cancer treated with irinotecan. Lung Cancer 35 (2002): 299-304.
- Gongora C, Vezzio-Vie N, Tuduri S, et al. New Topoisomerase I mutations are associated with resistance to camptothecin. Mol Cancer 10 (2011): 64.
- Li XG, Haluska P Jr, Hsiang YH, et al. Identification of topoisomerase I mutations affecting both DNA cleavage and interaction with camptothecin. Ann N Y Acad Sci 803 (1996): 111-127.
- Fujimori A, Harker WG, Kohlhagen G, et al. Mutation at the catalytic site of topoisomerase I in CEM/C2, a human leukemia cell line resistant to camptothecin. Cancer Res 55 (1995): 1339-1346.
- Meisenberg C, Ashour ME, El-Shafie L, et al. Epigenetic changes in histone acetylation underpin resistance to the topoisomerase I inhibitor irinotecan. Nucleic Acids Res 45 (2017): 1159-1176.
- de Gramont A, Figer A, Seymour M, et al. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol 18 (2000): 2938-2947.
- Hector S, Bolanowska-Higdon W, Zdanowicz J, et al. In vitro studies on the mechanisms of oxaliplatin resistance. Cancer Chemother Pharmacol 48 (2001): 398-406.
- Traverso N, Ricciarelli R, Nitti M, et al. Role of Glutathione in Cancer Progression and Chemoresistance. Oxidative Medicine and Cellular Longivity 2013 (2013).
- Ishikawa T, Ali-Osman F. Glutathione-associated cis-diamminedichloroplatinum(II) metabolism and ATP-dependent efflux from leukemia cells. Molecular characterization of glutathione-platinum complex and its biological significance. J Biol Chem 268 (1993): 20116-20125.
- Kelland LR. New platinum antitumor complexes. Crit Rev Oncol Hematol 15 (1993): 191-219.
- Baba H, Watanabe M, Okabe H, et al. Upregulation of ERCC1 and DPD expressions after oxaliplatin-based first-line chemotherapy for metastatic colorectal cancer. Br J Cancer 107 (2012): 1950-1955.
- Gnoni A, Russo A, Silvestris N, et al. Pharmacokinetic and metabolism determinants of fluoropyrimidines and oxaliplatin activity in treatment of colorectal patients. Curr Drug Metab 12 (2011): 918-931.
- Chen CC, Feng W, Lim PX, et al. Homology-directed repair and the role of BRCA1, BRCA2, and related proteins in genome integrity and cancer. Annu Rev Cancer Biol 2 (2018): 313-336.
- Liang J, Jiang T, Yao RY, et al. The combination of ERCC1 and XRCC1 gene polymorphisms better predicts clinical outcome to oxaliplatin-based chemotherapy in metastatic colorectal cancer. Cancer Chemother Pharmacol 66 (2010): 493-500.
- Moutinho C, Martinez-Cardús A, Santos C, et al. Epigenetic inactivation of the BRCA1 interactor SRBC and resistance to oxaliplatin in colorectal cancer. J Natl Cancer Inst 106 (2014): djt322.
- Mao L, Li Y, Zhao J, et al. Transforming growth factor-β1 contributes to oxaliplatin resistance in colorectal cancer via epithelial to mesenchymal transition. Oncol Lett 14 (2017): 647-654.
- Wang L, Mosel AJ, Oakley GG, et al. Deficient DNA damage signaling leads to chemoresistance to cisplatin in oral cancer. Mol Cancer Ther 11 (2012): 2401-2409.
- Wang C, Liu XQ, Hou JS, et al. Molecular mechanisms of chemoresistance in oral cancer. Chin J Dent Res 19 (2016): 25-33.
- Li W, Melton DW. Cisplatin regulates the MAPK kinase pathway to induce increased expression of DNA repair gene ERCC1 and increase melanoma chemoresistance. Oncogene 31 (2012): 2412-2422.
- Fang J, Ye Z, Gu F, et al. DUSP1 enhances the chemoresistance of gallbladder cancer via the modulation of the p38 pathway and DNA damage/repair system. Oncol Lett 16 (2018): 1869-1875.
- Sun L, Yao Y, Liu B, et al. MiR-200b and miR-15b regulate chemotherapy-induced epithelial-mesenchymal transition in human tongue cancer cells by targeting BMI1. Oncogene 31 (2012): 432-445.
- 3rd HAB, Moore MJ, Andersen J, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. Journal of Clinical Oncology 15 (1997): 2403-2413.
- Nakamura M, Nakashima H, Abe T, et al. Gemcitabine-based adjuvant chemotherapy for patients with advanced gallbladder cancer. Anticancer Res 34 (2014): 3125-3129.
- Huang P, Chubb S, Hertel LW, et al. Action of 2',2'-difluorodeoxycytidine on DNA synthesis. Cancer Res 51 (1991): 6110-6117.
- Mini E, Nobili S, Caciagli B, et al. Cellular pharmacology of gemcitabine. Ann Oncol 5 (2006): v7-v12.
- Moysan E, Bastiat G, Benoit JP. Gemcitabine versus Modified Gemcitabine: a review of several promising chemical modifications. Mol Pharm 10 (2013): 430-444.
- Gandhi V, Plunkett W. Modulatory activity of 2',2'-difluorodeoxycytidine on the phosphorylation and cytotoxicity of arabinosyl nucleosides. Cancer Res 50 (1990): 3675-3680.
- Shipley LA, Brown TJ, Cornpropst JD, et al. Metabolism and disposition of gemcitabine, and oncolytic deoxycytidine analog, in mice, rats, and dogs. Drug Metab Dispos 20 (1992): 849-855.
- Mackey JR, Mani RS, Selner M, et al. Functional nucleoside transporters are required for gemcitabine influx and manifestation of toxicity in cancer cell lines. Cancer Res 58 (1998): 4349-4357.
- García-Manteiga J, Molina-Arcas M, Casado FJ, et al. Nucleoside transporter profiles in human pancreatic cancer cells: role of hCNT1 in 2',2'-difluorodeoxycytidine- induced cytotoxicity. Clin Cancer Res 9 (2003): 5000-5008.
- Fujita H, Ohuchida K, Mizumoto K, et al. Gene expression levels as predictive markers of outcome in pancreatic cancer after gemcitabine-based adjuvant chemotherapy. Neoplasia 12 (2010): 807-817.
- Saiki Y, Yoshino Y, Fujimura H, et al. DCK is frequently inactivated in acquired gemcitabine-resistant human cancer cells. Biochem Biophys Res Commun 421 (2012): 98-104.
- Weizman N, Krelin Y, Shabtay-Orbach A, et al. Macrophages mediate gemcitabine resistance of pancreatic adenocarcinoma by upregulating cytidine deaminase. Oncogene 33 (2014): 3812-3819.
- Funamizu N, Okamoto A, Kamata Y, et al. Is the resistance of gemcitabine for pancreatic cancer settled only by overexpression of deoxycytidine kinase?. Oncol Rep 23 (2010): 471-475.
- Eda H, Ura M, K FO, et al. The antiproliferative activity of DMDC is modulated by inhibition of cytidine deaminase. Cancer Res 58 (1998): 1165-1169.
- Liu F, Gore AJ, Wilson JL, et al. DUSP1 is a novel target for enhancing pancreatic cancer cell sensitivity to gemcitabine. PLoS One 9 (2014): e84982.
- Amrutkar M, Gladhaug IP. Pancreatic cancer chemoresistance to gemcitabine. Cancers (Basel) 9 (2017).
- Heinemann V, Xu YZ, Chubb S, et al. Cellular elimination of 2',2'-difluorodeoxycytidine 5'-triphosphate: a mechanism of self-potentiation. Cancer Res 52 (1992): 533-539.
- Heinemann V, Xu YZ, Chubb S, et al. Inhibition of ribonucleotide reduction in CCRF-CEM cells by 2',2'-difluorodeoxycytidine. Mol Pharmacol 38 (1990): 567-572.
- Nakano Y, Tanno S, Koizumi K, et al. Gemcitabine chemoresistance and molecular markers associated with gemcitabine transport and metabolism in human pancreatic cancer cells. Br J Cancer 96 (2007): 457-463.
- Giovannetti E, Del Tacca M, Mey V, et al. Transcription analysis of human equilibrative nucleoside transporter-1 predicts survival in pancreas cancer patients treated with gemcitabine. Cancer Res 66 (2006): 3928-3935.
- Nakahira S, Nakamori S, Tsujie M, et al. Involvement of ribonucleotide reductase M1 subunit overexpression in gemcitabine resistance of human pancreatic cancer. Int J Cancer 120 (2007): 1355-1363.
- Tsunekuni K, Konno M, Asai A, et al. MicroRNA profiles involved in trifluridine resistance. Oncotarget 8(2017): 53017-53027.
- Roda D, Roselló S, Huerta M, et al. Trifluridine/tipiracil in earlier lines of chemotherapy for advanced colorectal cancer. Ann Oncol 31 (2020): 1097-1098.
- Peeters M, Cervantes A, Moreno Vera S, et al. Trifluridine/tipiracil: an emerging strategy for the management of gastrointestinal cancers. Future Oncol 14 (2018): 1629-1645.
- Kish T, Uppal P. Trifluridine/Tipiracil (Lonsurf) for the treatment of metastatic colorectal cancer. P t 41 (2016): 314-325.
- Edahiro K, Iimori M, Kobunai T, et al. Thymidine kinase 1 loss confers trifluridine resistance without affecting 5-fluorouracil metabolism and cytotoxicity. Mol Cancer Res 16 (2018): 1483-1490.
- Kraus LA, Samuel SK, Schmid SM, et al. The mechanism of action of docetaxel (Taxotere®) in xenograft models is not limited to bcl-2 phosphorylation. Investigational New Drugs 21 (2003): 259-268.
- Wang IC, Chen YJ, Hughes D, et al. Forkhead box M1 regulates the transcriptional network of genes essential for mitotic progression and genes encoding the SCF (Skp2-Cks1) ubiquitin ligase. Mol Cell Biol 25 (2005): 10875-10894.
- Costa RH. FoxM1 dances with mitosis. Nature Cell Biology 7 (2005): 108-110.
- Li X, Yao R, Yue L, et al. FOXM1 mediates resistance to docetaxel in gastric cancer via up-regulating Stathmin. J Cell Mol Med 18 (2014): 811-823.
- Golouh R, Cufer T, Sadikov A, et al. The prognostic value of Stathmin-1, S100A2, and SYK proteins in ER-positive primary breast cancer patients treated with adjuvant tamoxifen monotherapy: an immunohistochemical study. Breast Cancer Res Treat 110 (2008): 317-326.
- Zheng P, Liu YX, Chen L, et al. Stathmin, a new target of PRL-3 identified by proteomic methods, plays a key role in progression and metastasis of colorectal cancer. J Proteome Res 9 (2010): 4897-4905.
- Jeon TY, Han ME, Lee YW, et al. Overexpression of stathmin1 in the diffuse type of gastric cancer and its roles in proliferation and migration of gastric cancer cells. Br J Cancer 102 (2010): 710-718.
- Mitra M, Kandalam M, Sundaram CS, et al. Reversal of stathmin-mediated microtubule destabilization sensitizes retinoblastoma cells to a low dose of antimicrotubule agents: a novel synergistic therapeutic intervention. Invest Ophthalmol Vis Sci 52 (2011): 5441-5448.
- Sève P, Isaac S, Trédan O, et al. Expression of class III {beta}-tubulin is predictive of patient outcome in patients with non-small cell lung cancer receiving vinorelbine-based chemotherapy. Clin Cancer Res 11 (2005): 5481-5486.
- Sève P, Mackey J, Isaac S, et al. Class III beta-tubulin expression in tumor cells predicts response and outcome in patients with non-small cell lung cancer receiving paclitaxel. Mol Cancer Ther 4 (2005): 2001-2007.
- Urano N, Fujiwara Y, Doki Y, et al. Clinical significance of class III beta-tubulin expression and its predictive value for resistance to docetaxel-based chemotherapy in gastric cancer. Int J Oncol 28 (2006): 375-381.
- Xie L, Wei J, Qian X, et al. CXCR4, a potential predictive marker for docetaxel sensitivity in gastric cancer. Anticancer Res 30 (2010): 2209-2216.
- Kubo T, Kawano Y, Himuro N, et al. BAK is a predictive and prognostic biomarker for the therapeutic effect of docetaxel treatment in patients with advanced gastric cancer. Gastric Cancer 19 (2016): 827-838.
- Shekari N, Asghari F, Haghnavaz N, et al. Let-7a could serve as a biomarker for chemo-responsiveness to docetaxel in gastric cancer. Anticancer Agents Med Chem 19 (2019): 304-309.
- Danenberg PV, Gustavsson B, Johnston P, et al. Folates as adjuvants to anticancer agents: Chemical rationale and mechanism of action. Crit Rev Oncol Hematol 106 (2016): 118-131.
- Carloni V, Mazzocca A, Mello T, et al. Cell fusion promotes chemoresistance in metastatic colon carcinoma. Oncogene 32 (2013): 2649-2660.
- Ryu WJ, Lee JE, Cho YH, et al. A therapeutic strategy for chemotherapy-resistant gastric cancer via destabilization of both β-catenin and RAS. Cancers (Basel) 11 (2019).
- Liang JT, Huang KC, Cheng YM, et al. P53 overexpression predicts poor chemosensitivity to high-dose 5-fluorouracil plus leucovorin chemotherapy for stage IV colorectal cancers after palliative bowel resection. Int J Cancer 97 (2002): 451-457.
- Han SH, Kim JW, Kim M, et al. Prognostic implication of ABC transporters and cancer stem cell markers in patients with stage III colon cancer receiving adjuvant FOLFOX-4 chemotherapy. Oncol Lett 17 (2019): 5572-5580.
- Montecucco A, Zanetta F, Biamonti G. Molecular mechanisms of etoposide. Excli j 14 (2015): 95-108.
- Jaffrézou JP, Chen KG, Durán GE, et al. Mutation rates and mechanisms of resistance to etoposide determined from fluctuation analysis. J Natl Cancer Inst 86 (1994): 1152-1158.
- Cole SP. Multidrug resistance protein 1 (MRP1, ABCC1), a "multitasking" ATP-binding cassette (ABC) transporter. J Biol Chem 289 (2014): 30880-30888.
- Obuchi W, Ohtsuki S, Uchida Y, et al. Identification of transporters associated with etoposide sensitivity of stomach cancer cell lines and methotrexate sensitivity of breast cancer cell lines by quantitative targeted absolute proteomics. Molecular Pharmacology 83 (2013): 490-500.
- Erler JT, Cawthorne CJ, Williams KJ, et al. Hypoxia-mediated down-regulation of Bid and Bax in tumors occurs via hypoxia-inducible factor 1-dependent and -independent mechanisms and contributes to drug resistance. Mol Cell Biol 24 (2004): 2875-2889.
- Schöffski P. The modulated oral fluoropyrimidine prodrug S-1, and its use in gastrointestinal cancer and other solid tumors. Anticancer Drugs 15 (2004): 85-106.
- Sakatani A, Sonohara F, Goel A. Melatonin-mediated downregulation of thymidylate synthase as a novel mechanism for overcoming 5-fluorouracil associated chemoresistance in colorectal cancer cells. Carcinogenesis 40 (2018): 422-431.
- Sakurai Y, Sakamoto K, Sugimoto Y, et al. Orotate phosphoribosyltransferase levels measured by a newly established enzyme-linked immunosorbent assay in gastric carcinoma. Cancer science 97 (2006): 492-498.
- Prudner BC, Rathore R, Robinson AM, et al. Arginine starvation and docetaxel induce c-Myc-Driven hENT1 surface expression to overcome gemcitabine resistance in ASS1-negative tumors. Clin Cancer Res 25 (2019): 5122-5134.
- Hage C, Rausch,V, Giese,N, et al. The novel c-Met inhibitor cabozantinib overcomes gemcitabine resistance and stem cell signaling in pancreatic cancer. Cell Death Dis 4 (2013): e627.
- Heestand GM, Schwaederle M, Gatalica Z, et al. Topoisomerase expression and amplification in solid tumours: Analysis of 24,262 patients. Eur J Cancer 83 (2017): 80-87.
- Varis A, Wolf M, Monni O, et al. Targets of gene amplification and overexpression at 17q in gastric cancer. Cancer Res 62 (2002): 2625-2629.
- Nitiss JL. Targeting DNA topoisomerase II in cancer chemotherapy. Nat Rev Cancer 9 (2009): 338-350.
- Hu T, Li Z, Gao CY, et al. Mechanisms of drug resistance in colon cancer and its therapeutic strategies. World J Gastroenterol 22 (2016): 6876-6889.
- Cunningham D, Humblet Y, Siena S, et al. Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med 351 (2004): 337-345.
- Tabernero J. The role of VEGF and EGFR inhibition: implications for combining anti-VEGF and anti-EGFR agents. Mol Cancer Res 5 (2007): 203-220.
- Yang BB, Lum P, Chen A, et al. Pharmacokinetic and pharmacodynamic perspectives on the clinical drug development of panitumumab. Clin Pharmacokinet 49 (2010): 729-740.
- Giannopoulou E, Antonacopoulou A, Matsouka P, et al. Autophagy: novel action of panitumumab in colon cancer. Anticancer Res 29 (2009): 5077-5082.
- Kelley RK, Ko AH. Erlotinib in the treatment of advanced pancreatic cancer. Biologics 2 (2008): 83-95.
- Miyabayashi K, Ijichi H, Mohri D, et al. Erlotinib prolongs survival in pancreatic cancer by blocking gemcitabine-induced MAPK signals. Cancer Res 73 (2013): 2221-2234.
- Hurwitz H, Fehrenbacher L, Novotny W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350 (2004): 2335-2342.
- Saltz LB, Clarke S, Díaz-Rubio E, et al. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol 26 (2008): 2013-2019.
- Grothey A, Van Cutsem E, Sobrero A, et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381 (2013): 303-312.
- Arai H, Battaglin F, Wang J, et al. Molecular insight of regorafenib treatment for colorectal cancer. Cancer Treat Rev 81 (2019): 101912.
- Mitchell EP. Targeted therapy for metastatic colorectal cancer: role of aflibercept. Clin Colorectal Cancer 12 (2013): 73-85.
- Van Cutsem E, Tabernero J, Lakomy R, et al. Addition of aflibercept to fluorouracil, leucovorin, and irinotecan improves survival in a phase III randomized trial in patients with metastatic colorectal cancer previously treated with an oxaliplatin-based regimen. J Clin Oncol 30 (2012): 3499-3506.
- Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 359 (2008): 378-390.
- Zhu AX, Sahani DV, Duda DG, et al. Efficacy, safety, and potential biomarkers of sunitinib monotherapy in advanced hepatocellular carcinoma: a phase II study. J Clin Oncol 27 (2009): 3027-3035.
- Brahmer JR, Pardoll DM. Immune checkpoint inhibitors: making immunotherapy a reality for the treatment of lung cancer. Cancer Immunology Research 1 (2013): 85-91.
- Lugowska I, Teterycz P, Rutkowski P. Immunotherapy of melanoma. Contemp Oncol (Pozn) 22 (2018): 61-67.
- Yu C, Liu X, Yang J, et al. Combination of immunotherapy with targeted therapy: theory and practice in metastatic melanoma. Frontiers in Immunology 10 (2019).


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 