Clinical Characteristics and Prognostic Values of PIK3CA Mutation in Colorectal Cancer Patients
Jinming Han1, Jianxiang Shi2, Miao Jiang1, Ruowen Zhang4, Shuiling Jin1,2*, Yongliang Jia3*, Hong Zong1*
1Department of Oncology, the First Affiliated Hospital of Zhengzhou University, NO.1 Eastern Jianshe Road, Zhengzhou 450052, Henan, China
2Precision Medicine Center, Henan Institute of Medical and Pharmaceutical Sciences, Zhengzhou University, 40 Daxue Road, Zhengzhou 450052, China
3BGI College & Henan Institute of Medical and Pharmaceutical Sciences, Zhengzhou University, Zhengzhou 450000, Henan, China
4Department of Medicine, Jiahehongsheng (Shenzhen) Health Industry Group, Stony Brook University, Shenzhen 518081, China
*Corresponding Authors: Shuiling Jin, Department of Oncology, the First Affiliated Hospital of Zhengzhou University, NO.1 Eastern Jianshe Road, Zhengzhou 450052, Henan, China; Yongliang Jia, BGI College & Henan Institute of Medical and Pharmaceutical Sciences, Zhengzhou University, Zhengzhou 450000, Henan, China
Hong Zong, Department of Oncology, the First Affiliated Hospital of Zhengzhou University, NO.1 Eastern Jianshe Road, Zhengzhou 450052, Henan, China.
Received: 28 February 2022; Accepted: 11 March 2022; Published: 18 March 2022
Article Information
Citation:
Jinming Han, Jianxiang Shi, Miao Jiang, Ruowen Zhang, Shuiling Jin, Yongliang Jia, Hong Zong. Clinical Characteristics and Prognostic Values of PIK3CA Mutation in Colorectal Cancer Patients. Journal of Cancer Science and Clinical Therapeutics 6 (2022): 121-132.
View / Download Pdf Share at FacebookAbstract
Objective: This study aimed to investigate the role of PIK3CA mutation in the prognosis of Colorectal Cancer (CRC).
Methods: The clinical data of 386 CRC patients who underwent next-generation sequencing (NGS) test in the First Affiliated Hospital of Zhengzhou University from January 2017 to December 2020 were retrospectively reviewed. The associations between PIK3CA mutation and clinicopathologic characteristics of CRC patients were analyzed by chi-square test and Fisher’s exact test. Kaplan-Meier curves and Cox models were conducted to assess possible prognostic values of PIK3CA mutation.
Results: PIK3CA mutation was found in 88 (22.8%) of the 386 CRC patients. The percentage of MSI-H in PIK3CA mutated patients were higher than that in PIK3CA wild-type patients (P < 0.0001). The Median Overall Survival (mOS) of patients with PIK3CA mutation was significantly higher than that of wild-type patients (P = 0.0112). The progression free survival (PFS) of the mutated patients was also significantly prolonged compared with that of the wild type (P = 0.0132). Results of multivariate analysis showed that PIK3CA wild-type was independent risk factor for the survival of CRC patients.
Conclusion: PIK3CA mutation had an important impact on the clinic pathologic features and the prognosis for CRC patients. The survival of PIK3CA mutated patients was significantly prolonged, which needs further studies of large sample size, and long follow-up period to provide robust evidence.
Keywords
<p>Colorectal cancer; Clinical characteristics; Prognosis; PIK3CA mutation</p>
Article Details
Strengths and Limitations of This Study
- A total of 386 Colorectal Cancer (CRC) patients who underwent next-generation sequencing test in the First Affiliated Hospital of Zhengzhou University in China between January 2017 and December 2020 and 1949 CRC patients from the cBioPortal website (www. http://www.cbioportal.org/) were retrospectively analyzed the role of PIK3CA mutation in the prognosis of CRC.
- The significant differences were also found between the percentage of MSI-H in PIK3CA mutated patients and PIK3CA wild-type patients (P < 0.0001), the median overall survival of patients with PIK3CA mutation and that of wild-type patients (P = 0.0112), and the progression free survival of the mutated patients and that of the wild type (P = 0.0132). Multivariate analysis suggested that PIK3CA wild-type was independent risk factor for the survival of CRC patients. Overall, this study demonstrated that PIK3CA mutation had an important impact on the clinicopathologic features and the prognosis for CRC patients.
- This study was inherently retrospective and based on the CRC data with small sample size, and short follow-up periods.
1. Introduction
The morbidity of Colorectal Cancer (CRC) ranked the third among all types of malignant tumor in China, which is also increasing rapidly worldwide [1]. CRC is asymptomatic and inconspicuous in the early stages. Thirty percent of patients had developed distant metastasis at presentation [2]. In recent years, the advent of targeted therapies for CRC patients has significantly prolonged the overall and progression-free survival. It has been reported that Phosphatidylinositol-3-kinase (PI3K) is involved in the proliferation, differentiation and apoptosis of cells [3]. The PIK3CA gene encodes the P110α catalytic subunit of PI3K [4]. PIK3CA mutation can abnormally Activate the Phosphatidylinositol 3-kinase/protein Kinase B (AKT)/Mammalian Target of Rapamycin (mTOR) signal transduction pathway, and inhibit the expression of the tumor suppressor gene PTEN [5]. The rate of PIK3CA mutation is approximately 10-30% in CRC patients [6]. PIK3CA mutation plays a role in the prognosis and the survival of colorectal cancer patients [7]. Therefore, targeting PIK3CA gene had became a hot spot in the research of anticancer drug development. The efficiency of PI3K inhibitors in CRC patients with PI3KCA mutation was unsatisfied [6, 7]. CRC patients with PIK3CA mutation turned out to resistant to the first-line therapy [8]. The PI3K/AKT/mTOR signaling pathway intersects with other signaling pathways and has many compensatory pathways as well [9], which may be the reason for the weak potency, high toxicity and low efficiency of PI3K inhibitors. A series of inhibitors targeting PI3KCA gene are still researched by clinical trials [10]. However, there are few studies to explore the frequency, clinical characteristics, and prognostic significance of the PIK3CA mutation in CRC patients. Thus, this study retrospectively analyzed the clinical characteristics of PIK3CA mutation in colorectal cancer patients from the First Affiliated Hospital of Zhengzhou University and cBioPortal website, and explored the prognostic value of PIK3CA mutation in CRC patients.
2. Methods
2.1. Study objects and data
The clinical information of 386 CRC patients who underwent NGS testing in the First Affiliated Hospital of Zhengzhou University from January 2017 to December 2020 were retrospectively reviewed. All patients were diagnosed with CRC by biopsy. Data from the cBioPortal website (www. http://www.cbioportal.org/) of 1949 CRC patients were also included. The information including PIK3CA mutation status, age, gender, microsatellite status, pathological type, differentiation, depth of invasion, lymph node metastasis, risk factors, and pathological stage at diagnosis of the patients was retrospectively collected. Data were collected retrospectively and the study was approved by ethics committee (Approved No.2021-KY-1040-002).
2.2. Statistical analysis Progression-Free-Survival
Progression Free Survival (PFS) was measured from the start of first-line therapy to tumor progression or death. PFS was calculated as the follow-up cutoff time for patients who did not progress at the time of follow-up cutoff. Overall Survival (OS) was measured from the date of pathological diagnosis to the date of death with the follow-up cutoff date was March 1, 2021. Chi-square test and Fisher's exact test were used to analyze the differences between gene mutation status and age, gender, microsatellite status, pathological type, differentiation, depth of invasion, lymph node metastasis, risk factors and pathological stage at diagnosis in CRC patients, respectively. Survival analysis between the two groups was compared using the log-rank test. The survival function curves were plotted using the Kaplan-Meier method. Multivariate analysis was used in the Cox proportional hazards model [11, 12]. The OR value was used to estimate the relative risk. Confidence Interval (CI) was to estimate the range of the overall parameters according to a certain probability. The significant level of P-value was set as less than 0.05. The mentioned analyses were produced using SPSS version 25.
3. Results
3.1. The frequency of PIK3CA mutation in CRC patients
According to the data of 386 CRC patients, PIK3CA mutation was detected in 88 (22.8%) patients. Among PIK3CA mutation sites (Figure 1), the most common sites were exon 9 (N=27, 30.7%), exon 10 (N=18, 20.5%), and exon 21 (N=10, 11.4%). After included the data of 1949 CRC patients from cBioPortal, the number increased to 277(14.2%).
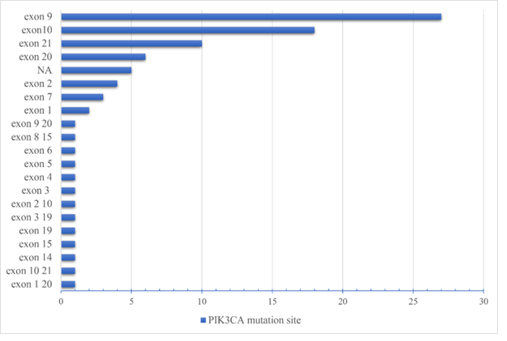
Figure 1: Frequency at different sites of PIK3CA mutation in CRC patients.
3.2. Clinical characteristics of patients with PIK3CA mutation
The information of the clinical features of PIK3CA mutated patients was shown in Table 1.
|
Clinical data |
Database data |
|||||
|
PIK3CA mutation |
Wild type |
P value |
PIK3CA mutation |
Wild type |
P value |
|
|
Age, years median, (range) |
51.5 (25-77) |
54 (20-88) |
67 (35-88) |
70(31-93) |
||
|
Age, years |
0 .736 |
0.041 |
||||
|
< 60 |
59 (67.0%) |
194 (65.1%) |
70 (25.3%) |
250 (15%) |
||
|
≥60 |
29 (33.0%) |
104 (34.9%) |
175 (63.2%) |
863 (51.6%) |
||
|
NA |
0 |
0 |
32 (11.6%) |
559 (33.4%) |
||
|
Sex |
0.651 |
0.451 |
||||
|
Male |
49 (55.7%) |
174 (58.4%) |
116 (41.9%) |
724 (43.3%) |
||
|
Female |
39 (44.3%) |
124 (41.6%) |
112 (40.4%) |
778 (46.5) |
||
|
NA |
0 |
0 |
49 (17.7%) |
170 (10.2%) |
||
|
Pathology |
0.513 |
0.007 |
||||
|
Adenocarcinoma |
73 (83.0%) |
249 (83.6%) |
24 (8.7%) |
522 (31.2%) |
||
|
Mucinous adenocarcinoma |
6 (6.8%) |
12 (4.0%) |
9 (3.2%) |
67 (4.0%) |
||
|
Others |
9 (10.2%) |
36 (12.1%) |
0 |
0 |
||
|
NA |
0 |
1 (0.3%) |
244 (88.1%) |
1065 (63.7%) |
||
|
Differentiation |
0.322a |
NA |
||||
|
Moderately differentiated |
59 (67.0%) |
170 (57.2%) |
NA |
NA |
||
|
Poorly differentiated |
9 (10.2%) |
45 (15.1) |
NA |
NA |
||
|
Moderate-poorly differentiated |
12 (13.6%) |
43 (14.4%) |
NA |
NA |
||
|
Others |
0 |
6 (2.0%) |
NA |
NA |
||
|
NA |
8 (9.1%) |
34 (11.4%) |
277 |
1672 |
||
|
Depth of tumor |
0.102a |
NA |
||||
|
invasion |
||||||
|
T1 |
0 |
5 (1.7%) |
NA |
0 |
||
|
T2 |
11 (12.5%) |
61 (20.5%) |
NA |
16 (1.0%) |
||
|
T3 |
27 (30.7%) |
67 (22.5%) |
NA |
80 (4.8%) |
||
|
T4 |
43 (48.8%) |
117 (39.2%) |
NA |
14 (0.8%) |
||
|
NA |
7 (8.0%) |
48 (16.1%) |
277 |
1562 (93.4%) |
||
|
Lymph node |
0.274 |
0.128 |
||||
|
metastases |
||||||
|
0 |
35 (39.8%) |
100 (33.6%) |
13 (4.7%) |
83 (5.0%) |
||
|
3-Jan |
27 (30.7%) |
60 (20.1%) |
7 (2.5%) |
78 (4.7%) |
||
|
6-Apr |
8 (9.1%) |
29 (9.7%) |
3 (1.1%) |
6 (0.3%) |
||
|
> 6 |
11 (12.5%) |
52 (17.5%) |
1 (0.4%) |
6 (0.3%) |
||
|
NA |
7 (8.0%) |
57 (19.1%) |
253 (91.3%) |
1499 (89.7%) |
||
|
Hazardous factor |
0.273 |
NA |
||||
|
None |
39 (44.3%) |
90 (30.2%) |
NA |
NA |
||
|
Nerve invasion |
12 (13.6%) |
40 (13.4%) |
NA |
NA |
||
|
Vessel carcinoma embolus |
NA |
NA |
||||
|
11 (12.5%) |
52 (17.4%) |
|||||
|
Both |
13 (14.8%) |
41 (13.8%) |
NA |
NA |
||
|
NA |
13 (14.8%) |
75 (25.2%) |
277 |
1672 |
||
|
Stage |
0.052 |
0.503 |
||||
|
I |
6 (6.8%) |
26 (8.7%) |
4 (1.4%) |
8 (0.5%) |
||
|
II |
24 (27.3%) |
44 (14.8%) |
7 (1.4%) |
35 (2.1%) |
||
|
III |
21 (23.9%) |
65 (21.8%) |
12 (4.4%) |
37 (2.2%) |
||
|
IV |
36 (40.9%) |
152 (51.0%) |
6 (2.2%) |
30 (1.8%) |
||
|
NA |
1 (1.1%) |
11 (3.7%) |
248 (89.5%) |
1562 (93.4%) |
||
|
Microsatellite status |
<0.0001 |
<0.0001 |
||||
|
MSS |
57 (64.8%) |
219 (73.5%) |
116 (41.9%) |
491 (29.3%) |
||
|
MSI-H |
23 (26.1%) |
22 (7.4%) |
51 (18.4%) |
79 (4.6%) |
||
|
NA |
8 (9.1%) |
57 (19.1%) |
110 (39.7%) |
1102 (65.9%) |
||
|
a: Fisher's exact test; NA: not available |
||||||
Table 1: Characteristics of CRC patients with PIK3CA gene mutation and wild-type.
According to the data, the median age of the patients with PIK3CA mutation and that of the wild type were 51.5 years and 54 years, respectively. For the patients with the age less than 60 years old, 67.0% were PIK3CA mutant and 65.1% were PIK3CA wild-type. The difference between the two groups was not statistically significant (P = 0.736). The proportion of male patients in PIK3CA subgroup was similar to wild-type subgroup (55.7% vs.58.4% with P = 0.651). Most of the PIK3CA mutant and wild-type patients were diagnosed with adenocarcinoma (83.0% vs. 83.6% with P = 0.513) and moderately differentiated (67.0% vs. 57.2% with P = 0.322). The proportion of the patients at stage IV was high in both PIK3CA mutant and wild-type (40.9% vs. 51.0% with P = 0.052). 39.8% of the patients in the PIK3CA mutation group were no lymph node metastasis, compared with 33.6% of those in the wild-type group (P = 0.274). The proportion of deep invasion into the whole layer patients in PIK3CA subgroup was slightly higher than wild-type subgroup (48.8% vs., 39.2% with P = 0.102). The proportion of MSI-H with PIK3CA mutation (26.1%) was higher than wild group (7.4%) with the difference statistically significant (P < 0.0001). In the database, the median age of the patients with PIK3CA mutant and that with wild-type were 67 and 70 years, respectively. In the PIK3CA mutation and wildtype subgroups, 41.4% and 44.3% of the patients were male (P = 0.451), respectively; 63.2% and 51.6% were older than 60 years (P = 0.041) respectively. The proportion of adenocarcinoma patients in wild-type subgroup was higher than PIK3CA mutation subgroup (8.7% vs. 31.2% with P = 0.013). The proportion of MSI-H was higher in patients with PIK3CA mutation than wild group, 18.4% and 4.6%, respectively, and the difference was statistically significant (P < 0.0001).
3.3. Prognostic value of PIK3CA mutation
In clinical data, 386 CRC patients were followed for a median of 24 months. The OS of PIK3CA mutation group was not reached. The OS of wild group was 54 Month (HR: 0.4827, 95% CI: 0.3038 to 0.7672, P =0.0112). The median PFS in PIK3CA mutant group and wild group were 46 months and 15 months, respectively (HR: 0.656, 95% CI: 0.4819 to 0.893, P= 0.0132) (Figure 2). Validation was also performed in the database (Figure 3). Clinical and pathological characteristics were included in multivariate Cox regression analysis (Table 2), which showed that age older than 60 years old (P=0.006), lymph node metastasis (P=0.001), advanced stage at diagnosis (P=0.006) remained as independent factors for poor prognosis with the difference statistically significant. Both the PIK3CA mutation and the non-PIK3CA mutation were statistically significant (P= 0.043).
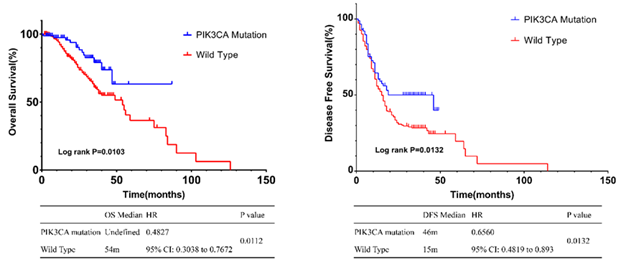
Figure 2: Survival analysis curves for PIK3CA mutant and wild type in clinical data.
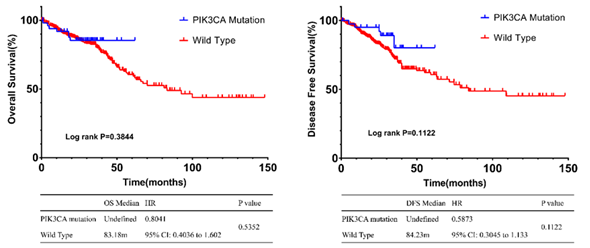
Figure 3: Survival analysis curve of PIK3CA mutant and wild-type in database.
|
B |
SE |
Wald |
Df |
P |
Exp (B) |
95% CI |
|
|
PIK3CA mutation |
-0.858 |
0.424 |
4.092 |
1 |
0.043 |
0.424 |
[0.185, 0.974] |
|
Age > 60 |
0.881 |
0.318 |
7.684 |
1 |
0.006 |
2.413 |
[1.294, 4.499] |
|
Lymph node metastases |
2.211 |
0.642 |
11.841 |
1 |
0.001 |
9.121 |
[2.590, 32.127] |
|
Advanced stage |
1.176 |
0.426 |
7.617 |
1 |
0.006 |
3.24 |
[1.406, 7.468] |
|
B: Partial regression coefficient; SE: Standard Error; Wald: test factor; DF: Degree of Freedom; EXP (B): OR value; CI: Confidence Interval. |
|||||||
Table 2: Multivariate analysis of OS based on clinical and molecular variables.
4. Discussion
The incidence and mortality of CRC in China are still increasing with time going by [13] and most patients were diagnosed at the middle or advanced stages [14]. Gene mutation and signaling pathways abnormity contributed to the occurrence and development of CRC. The PI3K signaling pathway plays an important role in the processes including cell growth, development, differentiation, proliferation and apoptosis with the abnormal activation closely related to the development of tumors [15]. PIK3CA mutation results in enhanced activity of the p110a enzyme, stimulation of the downstream Akt signaling, increased cell invasion and metastasis [16]. Therefore, PIK3CA gene has become one of the hotspots in the research of CRC in recent years. It was reported that the frequency of PIK3CA mutation in CRC was approximately 10%-30% [17-19]. In China, the frequency of PIK3CA mutation in CRC was reported as about 8.2% [19]. This study found that 22.8% (88) of the 386 CRC patients were with PIK3CA mutation. The frequency of PIK3CA mutation in CRC was 14.2% according to the data from cBioPortal. The differences may result from the different tissue types of samples, sample sources, processing methods of samples, storage time of samples, and gene sequencing technologies. PIK3CA mutation most frequently occurred on exon 9 (N=27, 30.7%), exon 10 (N=18, 20.5%) and exon 21 (N=10, 11.4%), which was the same as other reports [20]. It had been shown that simultaneous mutation of exon9 and exon20 in the PIK3CA gene may lead to tumor invasiveness [21].
The results of this study showed that PIK3CA mutation was more likely to occur in patients with microsatellite instability. The proportion of MSI-H in PIK3CA mutation group was 26.1%, which was significantly higher than that in the wild group (7.4%) with the difference statistically significant. The results of this study were similar to the data online. The data from cBioPortal showed that the percentage of MSI-H was higher in the PIK3CA mutation group than in the wild group (18.4 % and 4.6 %, respectively) with the difference statistically significant. Data published by other research group showed that there was no correlation between PIK3CA mutation and clinicopathological features of CRC including gender, age, and stage [22]. One study showed that PIK3CA gene mutation was more likely to occur in well-to-moderately differentiated CRC and mucinous adenocarcinoma [23]. It had also been documented that PIK3CA mutation was not significantly associated with gender or age [19]. Another study showed more females in the patients with PIK3CA mutation [24]. The data included in this study suggested that there was no significant correlation between age, gender, pathological type, differentiation, depth of invasion, lymph node metastasis, risk factors, or pathological stage at diagnosis. A foreign study [17] showed that most CRC patients with PIK3CA mutations were younger than 60 years old. This study found that most patients with CRC were younger than 60 years old while most CRC patients abroad were diagnosed at an age of over 60 years old. The differences between the data in this study and that from the website may result from the sample size and the races. Another possible reason of included population younger than 60 years old is that elderly people inclined to give up the opportunity of surgery and chemotherapy because they cannot tolerate treatment and other reasons. The discrepant source may be the incomplete data from database.
The results showed that the patients with PIK3CA mutation turned out with better prognosis, which may be related to an increased proportion of MSI-H in patients with PIK3CA mutations. It had been reported that patients with microsatellite instability achieved better prognosis from the immunotherapy [25]. The results of the analysis with Cox regression model showed that PIK3CA wild type remained an independent factor for poor prognosis excluding the effects of other clinical and pathological characteristics including age, gender, pathological type, and disease stage. It was also found that older than 60 years old, lymph node metastasis, and advanced stage at diagnosis were independent factors for poor prognosis in CRC patients. The relationship between PIK3CA mutation and CRC prognosis is controversial. Some studies shown that PIK3CA mutations have no significant correlation with the prognosis of CRC [26], while other studies shown that PIK3CA mutations led to worse prognosis of CRC patients[27, 28]. When it comes to the clinical management, PIK3CA mutation was correlated to chemoresistance and recurrence [29-31]. However, one study suggested that PI3KCA mutation was not the main determinant of resistance to cetuximab in patients with metastatic colon cancer [32]. The relationship between PIK3CA mutations and prognosis remains unclear. The relationship between PIK3CA mutation and microsatellite status has not been reported. The better prognosis caused by PIK3CA mutations may be associated with MSI-H. This retrospective study was conducted base on the real-world data. However, the data was a small sample and insufficient that may lead to the deviation of the results. The limitation of this study was the small sample size and low follow-up periods. The correlation between PIK3CA mutation and prognosis of CRC may still be controversial. Although the number of patients included in this study was small, it provided evidence on clinical evaluation of the clinicopathological features in CRC between PIK3CA mutated and unmutated groups. More further studies for PIK3CA mutation with a large sample size from multicenter and long follow-up periods are still warranted to provide the robust evidence on the treatment strategy of targeting PIK3CA for CRC.
5. Conclusion
PIK3CA mutation had an important impact on the clinicopathologic features and the prognosis for CRC patients. The survival of PIK3CA mutated patients was significantly prolonged, which needs further studies of large sample size, and long follow-up period to provide robust evidence.
Author Contributions
JH conceived and designed the paper and drafted the paper. Study implementation and feasibility analysis for SJ and MJ; JS and YJ analyzed and explained the results, and modified the later versions. HZ is responsible for the quality control and proofreading of articles, as well as the overall supervision and management of articles.
Funding
This study was conducted with support from the Education Department of Henan Province (Grant No.15A320090). This study was conducted with support from the National Natural Science Foundation of China (Grant No.82000164). The work of YLJ was financially supported by the Henan Institute of Medical and Pharmacological Sciences (Grant No. 2021BP0113), Zhengzhou University (Grant No. 32212456).
Competing Interests
None declared.
Patient consent for publication
Not required.
Ethical Approval
Data were collected retrospectively and the study was approved by Scientific Research and Clinical Trial Ethics Committee of the First Affiliated Hospital of Zhengzhou University (Approved No.2021-KY-0919-002).
Acknowledgments
I would like to thank all my teachers who have helped me to develop the fundamental and essential academic competence. My sincere appreciation also goes to the teachers and students from Zhengzhou University, who participated this study with great cooperation.
Data Statement
The excel data used to support the findings of this study are included within the supplementary information file. No additional data are available.
Patient and Public Involvement
Patients included in this study were clearly informed with the ethics committee approval (No. 2021-KY-0919-002).
References
- Ferlay J, Colombet M, Soerjomataram I, et al. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer 144 (2019): 1941-1953.
- Andre T, Gramont AD, Vernerey D, et al. Adjuvant Fluorouracil, Leucovorin, and Oxaliplatin in Stage II to III Colon Cancer: Updated 10-Year Survival and Outcomes According to BRAF Mutation and Mismatch Repair Status of the MOSAIC Study. J Clin Oncol 33 (2015): 4176-4487.
- Samuels Y, Wang Z, Bardelli A, et al. High frequency of mutations of the PIK3CA gene in human cancers. Science 304 (2004): 554.
- Katso R, Okkenhaug k, Ahmadi K, et al. Cellular function of phosphoinositide 3-kinases: implications for development, homeostasis, and cancer. Annu Rev Cell Dev Bi 17 (2001): 615-675.
- Papadatos-Pastos D, Rabbie R, Ross P, et al. The role of the PI3K pathway in colorectal cancer. Crit Rev Oncol Hematol 94 (2015): 18-30.
- Whitehall VL, Rickman C, Bond CE, et al. Oncogenic PIK3CA mutations in colorectal cancers and polyps. Int J Cancer 131 (2012): 813-820.
- Kato S, lida S, Higuchi T, et al. PIK3CA mutation is predictive of poor survival in patients with colorectal cancer. Int J Cancer 121 (2007): 1771-8.
- Wang Q, Shi YL, Zhpu K, et al. PIK3CA mutations confer resistance to first-line chemotherapy in colorectal cancer. Cell Death Dis 9 (2018): 739.
- Narayanankutty A. PI3K/ Akt/ mTOR Pathway as a Therapeutic Target for Colorectal Cancer: A Review of Preclinical and Clinical Evidence. Curr Drug Targets 20 (2019: 1217-1226.
- Okkenhaug K, Graupera M, Vanhaesebroeck B. Targeting PI3K in Cancer: Impact on Tumor Cells, Their Protective Stroma, Angiogenesis, and Immunotherapy. Cancer Discov 6 (2016): 1090-1105.
- Lin, Y, Wang Y, Zhng Y, et al. Clinical characteristics and prognostic study of adult acute myeloid leukemia patients with ASXL1 mutations. Hematology 25 (2020): 446-456.
- Mei ZB, Duan CY, Li CB, et al. Prognostic role of tumor PIK3CA mutation in colorectal cancer: a systematic review and meta-analysis. Ann Oncol 27 (2016): 1836-1848.
- Feng RM, Zong YN, Cao Sm, et al. Current cancer situation in China: good or bad news from the 2018 Global Cancer Statistics? Cancer Commun (Lond) 39 (2019): 22.
- Brody H, Colorectal cancer. Nature 521 (2015): 1.
- Kumar A, Carrera AC. New functions for PI3K in the control of cell division. Cell Cycle 6 (2007): 1696-1698.
- Ligresti G, Millitello L, Steelman LS, et al. PIK3CA mutations in human solid tumors: role in sensitivity to various therapeutic approaches. Cell Cycle 8 (2009): 1352-1358.
- Garrido-Laguna I, Hong DS, Janku F, et al. KRASness and PIK3CAness in patients with advanced colorectal cancer: outcome after treatment with early-phase trials with targeted pathway inhibitors. PLoS One 7 (2012): 38033.
- Janku F, Lee JJ, Tsimberidou AM, et al. PIK3CA mutations frequently coexist with RAS and BRAF mutations in patients with advanced cancers. PLoS One 6 (2011): 22769.
- Mao C, Zhou J, Yang Z, et al. KRAS, BRAF and PIK3CA mutations and the loss of PTEN expression in Chinese patients with colorectal cancer. PLoS One 7 (2012): 36653.
- Liao X, Morikawa T, Lochhead P, et al. Prognostic role of PIK3CA mutation in colorectal cancer: cohort study and literature review. Clin Cancer Res 18 (2012): 2257-2268.
- Hsu HC, Thiam TK, Lu YJ, et al. Mutations of KRAS/NRAS/BRAF predict cetuximab resistance in metastatic colorectal cancer patients. Oncotarget 7 (2016): 22257-22270.
- Zhang J, Roberts TM, Shivdasani RA. Targeting PI3K signaling as a therapeutic approach for colorectal cancer. Gastroenterology 141 (2011): 50-61.
- Ye ZL, Qiu MZ, Tang T, et al. Gene mutation profiling in Chinese colorectal cancer patients and its association with clinicopathological characteristics and prognosis. Cancer Med 9 (2020): 745-756.
- Benvenuti S, Frattini M, Arena S, et al. PIK3CA cancer mutations display gender and tissue specificity patterns. Hum Mutat 29 (2008): 284-288.
- Gelsomino F, Barbolini M, Spallanzani A, et al. The evolving role of microsatellite instability in colorectal cancer: A review. Cancer Treat Rev 51 (2016): 19-26.
- Qin A, Qian W. MicroRNA-7 inhibits colorectal cancer cell proliferation, migration and invasion via TYRO3 and phosphoinositide 3-kinase/protein B kinase/mammalian target of rapamycin pathway suppression. Int J Mol Med 42 (2018): 2503-2514.
- Kim HS, Lee SF, Bae YS, et al. PIK3CA amplification is associated with poor prognosis among patients with curatively resected esophageal squamous cell carcinoma. Oncotarget 7 (2016): 30691-30701.
- Naguib A, Cooke JC, Happerfield L, et al. Alterations in PTEN and PIK3CA in colorectal cancers in the EPIC Norfolk study: associations with clinicopathological and dietary factors. BMC Cancer 11 (2011): 123.
- Liao W, Liao Y, Zhou JX, et al. Gene mutations in epidermal growth factor receptor signaling network and their association with survival in Chinese patients with metastatic colorectal cancers. Anat Rec (Hoboken) 293 (2010): 1506-1511.
- Mao C, Yang ZY, Hu XF, et al. PIK3CA exon 20 mutations as a potential biomarker for resistance to anti-EGFR monoclonal antibodies in KRAS wild-type metastatic colorectal cancer: a systematic review and meta-analysis. Ann Oncol 23 (2012): 1518-1525.
- Sclafani F, Wilson SH, Cunningham D, et al. Analysis of KRAS, NRAS, BRAF, PIK3CA and TP53 mutations in a large prospective series of locally advanced rectal cancer patients. Int J Cancer 146 (2020): 94-102.
- Prenen H, Schutter JD, Jacobs B, et al. PIK3CA mutations are not a major determinant of resistance to the epidermal growth factor receptor inhibitor cetuximab in metastatic colorectal cancer. Clin Cancer Res 15 (2009): 3184-3188.


 Impact Factor:
* 4.1
Impact Factor:
* 4.1
 Acceptance Rate:
74.74%
Acceptance Rate:
74.74%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
