Investigating the Quality of Life of Retinoblastoma Survivors: An Initial Exploration
Krishnasri Padamandala1, Santosh G Honavar2, Rolika Bansal2, Surya Durga Prasad M1, Padmaja Gadiraju3, Geeta K Vemuganti1*
1School of Medical Sciences, University of Hyderabad, Hyderabad, Telangana, India
2Ocular Oncology and Oculoplasty, Centre for Sight Eye Hospital, Hyderabad, Telangana, India
3Associate Professor, Centre for Health Psychology, School of Medical Sciences, University of Hyderabad, Hyderabad, Telangana, India
*Corresponding Author: Geeta K Vemuganti, School of Medical Sciences, University of Hyderabad, Prof CR Rao Road, Hyderabad, Telangana 500046, India.
Received: 14 February 2023; Accepted: 12 May 2023; Published: 19 May 2023
Article Information
Citation: Krishnasri Padamandala, Santosh G Honavar, Rolika Bansal, Surya Durga Prasad M, Padmaja Gadiraju, Geeta K Vemuganti. Investigating the Quality of Life of Retinoblastoma Survivors: An Initial Exploration. Journal of Cancer Science and Clinical Therapeutics. 7 (2023): 118-126.
View / Download Pdf Share at FacebookAbstract
Background: The purpose of this study was to assess the viability of using the quality-of-life questionnaire and structured interview in South Indian tertiary eye care hospital survivors, age-matched controls, and their parents.
Procedure: 15 RB survivors, 15 age-matched controls, and 30 primary carers were included. Peds QL 4.0 generic core scale and structured interview were administered. Physical, social, emotional, and school health were also examined.
Results: Cases and controls had a mean age of 5.9 years, SD + 2.4, based on 60 samples. The questionnaire was completed by children in 15.1 + 0.4 minutes, whereas it took controls in 12.2 + 0.6 minutes. RB survivors (15/15) and controls (3/15) of all ages and genders need parental help to complete the questionnaire. The physical health of RB survivors was 0.3 SD +0.96, the social health was 0.32 SD +1.0, the emotional health was 0.56 SD +1.06, and the school health was 1.6 SD +2.1. Physical health was 0.3 SD +0.7, social health 0.06 SD +0.1, emotional health 0.2 SD +0.5, and school health 0.2 SD +0.4. The P-value for the two-tailed t-test is 95% CI, P0.05.
Conclusion: The pilot research showed that participants, especially parents, understood and accepted the quality of life measuring instrument (Peds QL 4.0), making it suitable for a larger study. This age group needed parents and researchers. The tendency suggests a reduction in survivors' quality of life, emphasizing the necessity for psychoeducation and counseling before and after treatment.
Keywords
<p>Feasibility; Quality of Life Questionnaire; Retinoblastoma; Tertiary Eye Care Center; Pediatric Eye Cancer</p>
Article Details
1. Introduction
RB is the most common intraocular malignancy in childhood globally, with a prevalence of 1 in every 15000-20000 live births across the population correlates with 9000 new cases every year globally [1, 2]. Rapid progress has been made in diagnosing and treating the disease by considering saving life and vision in order of priority [3, 4]. Together 43% of the global burden of RB lives in these countries 1486 children in India, 1103 children in China and 277 children in Indonesia, 260 children in Pakistan, 184 children in Bangladesh, and 142 children in the Philippines [5]. Recently retinoblastoma has exceeded uveal melanoma to become the most ocular malignancy globally [2]. It is important to study the quality of life in retinoblastoma as it is potentially fatal but has a good prognosis and survival rates with early diagnosis and treatment. It is one of the most easily detectable cancers constituting 3% of pediatric cancers [6]. However, Quality of life is the most neglected and important aspect of survivorship. It is necessary to create awareness of the affected quality of life domains and allow timely advocacy of initiatives for addressing the issues at an individual level [2].
Few studies have evaluated the QoL in children affected with RB. In one study, the pediatric quality of life inventory 4.0 generic core scales (PedsQLTM 4.0) among enucleated subjects revealed that the social and academic dimensions were statistically insignificant compared to healthier children and influenced by the laterality of the illness, age, and satisfaction with the ocular prosthesis [7]. Because there is a dearth of scientific research and disparities in a wide range of technological and socioeconomic elements that could potentially affect clinical diagnosis and life expectancy, the researchers contend that knowing the impact of retinoblastoma in the Asia Pacific area is crucial [8]. Unlike in other pediatric tumors, visual disability, physical appearance, multiple extensive treatments, and challenges in counseling on prognosis and consequences could affect the quality of life and well-being. The observations in the clinical practice of the researcher also indicate that there appears to be a difference between children and the perceptions of parents of children with RB as compared to other children with non-RB eye pathology. With this as rationale before embarking on a larger study, the authors aimed to evaluate the feasibility of implementing the Paediatric quality of life questionnaire, PEDS QL 4.0 generic score.
The researchers administered and observed the difficulties faced during the pilot process. It would also generate preliminary data at the tertiary eye care hospital in south India. We hypothesized that there will be a difference in the quality of life in children who are RB survivors and parents, in comparison with control group children (non-RB) and the perception of their parents.
To evalute our hypothesis we aimed to assess the quality of life of RB and non-RB survivors, as well as their parent’s perceptions with regard to the QoL of their children in both the groups. We also attempted to compare the differences in QoL between RB survivors and non-RB survivors, as well as the perceptions of their parents.
2. Methodology
2.1 Study sampling technique
The authors of this study utilized a “convenience sampling” technique to access the data already there in the study setting (a tertiary care hospital) to make data collecting easier, as retinoblastoma is a rare condition.
2.2 Study population
Two groups of participants were considered for the study. The group of children with RB as per the information and details provided by the hospital comprises the treatment group children. Their parents were also considered for the administration of the questionnaire and the interview.
On the other hand, the children who were non-RB were considered as a control group, their parents were also considered to administer the questionnaire and interview as shown below in supplementary table 1.
|
Total sample size N=60 |
Sample Size |
Cases |
Controls |
Study tools implemented |
|
Children Sample size (A1+A2) N=30 |
A1 (N=15) |
A2 (N=15) |
Quality of life questionnaire only |
|
|
Parent groups sample size (B1+B2) N=30 |
B1 (N=15) |
B2 (N=15) |
Perceptions of QoL of their children questionnaire + structured interview |
Supplementary Table 1: This table shows the distribution of questionnaires between cases and controls as well as the care givers (parents) of these subjects.
The sample was collected during the pandemic phase from the Center for Sight (CFS) eye hospital. The cooperating institutes conducted an ethical evaluation of subjects who underwent retinoblastoma treatment, including unilateral or bilateral enucleation, chemotherapy, plaque irradiation, freezing, laser eye coagulation, and usage of supporting and intensive services and ocular prostheses. The subject or the subject’s legal representative provided their informed written consent and approval. As per the ethical procedures, informed consent was obtained from the parents of the treatment group & control group as well as assent from the children of both groups.
2.3 Parameters collected for Population Clinical features/parents data
Demographics such as Age, Gender, Geographical region, Type of treatment, Laterality of the disease, Disease-free survival, BCVA, Parents’ income, Parent education level, and Occupation of the parent, time is taken to complete the questionnaire, comprehensibility of the questionnaire were included.
2.4 QoL Questionnaire details/ content
The Standardised PEDS QL 4.0 Generic core scare(Telugu, Hindi, English) versions were used to assess the Quality of Life, which contains the Physical domain (8 items), Social health domain (5 items), School domain (5 items), and Emotional (5 Items) were analysed to check the feasibility of implementing questionnaire time and, documenting the problems faced while collecting the same. Additionally, the researcher administered a questionnaire also implemented includes detailed treatment history, electronic gadgets usage, nearest blind school presence, financial expenditure of the survivors, and whether the child required special attention are not.
2.5 Study process
This pilot study was conducted from September 2021 to October 2021 and could include 60 patients, of which 15 were RB survivors, 15 were controls and 15 were RB case primary caregivers, and 15 were (non-RB) control primary caregivers who are parents. Following Institutional Ethics approval, the authors started recruiting the subjects. As shown in (Figure 1) the study process began with the study’s eligibility criteria of 2-18 years, having completed two years of treatment at the Centre for Sight Super Specialty Eye Hospital.
The authors have considered a minimum of 1-2 years of disease-free survival to avoid the treatment-related co-morbidities that might influence the questionnaire response. Age-matched subjects acted as controls, and whoever could not consent and comprehend the questionnaires, parents were offered the participation (for the 2-4 years age group) and alone to study parents’ perspectives also included. The current study followed the WHO definition of quality of life which states the personal understanding and perspective towards their life physically, mentally, emotionally, and socially. Participants who were not willing to participate and were outside the defined age group were excluded from the study.
Figure-1: The Pilot process demonstrates the entire feasibility study procedure where the study invited 60 participants from both the cases and controls group following informed consent. Those who did not consent continued to avail of the regular services at the hospital. The study involved implementing the validated Peds QoL 4.0 Generic Core scale (Figure 2) without modification and structured interviews (Figure 3) with the recruited subjects. The time it took to complete the questionnaire during their hospital visit and the difficulties children or parents encountered during data collection were recorded. It was made certain that their participation did not interfere with any of the procedures or protocols for which their visit was scheduled.
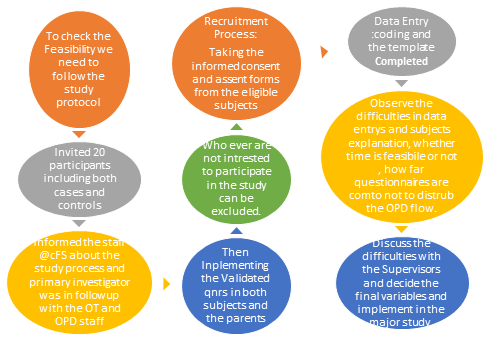
Figure 1: The Pilot process of the study.

Figure 2: PedsQL-4.0-Core-PYC - India/English - Version of 08 Apr 2019 - Mapi.I Copyright © 1998 JW Varni, Ph.D.(2-4 age format).

Figure 3: Investigator-focused Structured Interview format.
2.6 Statistical Analysis
This pilot study checked the feasibility of the tools and compared the QoL among cases and control groups. The questionnaire responses were measured on a 5 (i.e. 0-4) point Likert scale. And the analyses were conducted using Microsoft- Excel 2010 version. Descriptive statistics such as mean and standard deviation and the time taken to complete the implemented questionnaires and interviews; socio-demographic profiles were analysed using excel. Additionally, the authors analysed how far the questionnaires and the interview variables were understandable and at which part of the questionnaire subjects felt uncomfortable and sought help also noted. The quality of life variables among self-reported and parent proxy responses was observed, and compared the p- values were by performing a two-tailed t-test in addition to the descriptive statistics.
3. Results
3.1 Study population demographics
Of the 60 RB survivors and controls (N= 15 cases, N=15 controls, N=15 case parents, and N=15 control parents), the female-to-male ratio was 6:9 and controls (N=7) 8:7, respectively. The mean age of the RB survivors was 5.9 + 2.4 years and the controls were 5.9 + 2.4 years. The disease was unilateral in 9 and bilateral RB cases in 6 cases (Table 1-2). The treatments received in the 15 RB survivors were chemotherapy =7, External beam radiation (EXBRT)=2, Brachytherapy=1, and Enucleation=5. The mean time taken by cases to complete the questionnaire was 15.1 + 0.4 min, and controls were 12.5+ 0.5 min. All 15 RB survivors and three controls opted for parents’ help while filling up the questions of emotional and social health groups irrespective of age and gender. All the parents were able to understand the questionnaire and structured interview. The time taken by the parents to fill out the questionnaire was 11.4 + 1.3 min, and for the controlling parent, 10.5 + 0.5 min. Vision in bilateral RB cases was having one seeing eye where the Best corrected visual acuity in the better eye (BCVA) was (FFL) fixing and following the light 6, and 6/12 and better 4, 6/18, and worse five as per the clinical documentation (Table 4).
|
Variable |
Cases (RB Survivors) ( n=15) |
Control group (n=15) |
|||||
|
Age in yrs (Mean+SD) |
5.9+2.4 |
5.9 + 2.4 |
|||||
|
Disease-free survival in yrs Mean+SD |
2.6years+1.3 |
Not applicable |
|||||
|
Gender |
Males |
9 (60 %) |
8 (53.4%) |
||||
|
Females |
6 (40%) |
7 ( 46.6%) |
|||||
|
Disease laterality |
Unilateral |
9 (60 %) |
Not applicable |
||||
|
Bilateral |
6 (40%) |
Not applicable |
|||||
|
Type of treatment availed |
Enucleation |
5 (33.3%) |
Emmetropia |
5 (33.4%) |
|||
|
Chemotherapy |
7 (46.6%) |
Myopia |
9 (60 %) |
||||
|
Brachytherapy |
1 (6.6%) |
Hyperopia |
0 |
||||
|
EXBRT |
2 (13.3%) |
Amblyopia |
1 (6 %) |
||||
|
BCVA in the better eye |
Fixed and Following light |
6 (40%) |
6 / 6 in Both eyes |
14 (93.4%) |
|||
|
6/12 to 6/6 |
4 (26.6%) |
< 6 / 9 in one eye |
1 (6.6 %) |
||||
|
6/60 to 6/18 |
5 (33.3%) |
0 |
|||||
|
Parents Education Level |
Father |
High-school 5th to 9th |
1 (6.6%) |
0 |
|||
|
SSC |
4 (26.6%) |
0 |
|||||
|
Intermediate |
4 (26.6%) |
0 |
|||||
|
UG |
2 (13.3%) |
5 (33.4%) |
|||||
|
PG |
2 (13.3%) |
10 (66.6%) |
|||||
|
Mother |
High-class 5th to 9th |
1 (6.6%) |
0 |
||||
|
SSC |
3 (20%) |
0 |
|||||
|
Intermediate |
3 (20%) |
0 |
|||||
|
UG |
4 (26.6%) |
5 (33.4%) |
|||||
|
PG |
2 (13.3%) |
10 (66.6%) |
|||||
|
Socioeconomic status |
Upper lower class |
6 ( 40 %) |
0 |
||||
|
Lower middle class |
6 (40%) 1 (6.6%) 1 (6.6%) 1 (6.6%) |
0 5 (33.3%) 10 (66.4%) 0 |
|||||
|
Upper class |
|||||||
|
Upper middle class |
|||||||
|
Lower class |
|||||||
|
Diagnosis age |
In months |
18.5 months + 16.9 |
Not applicable |
||||
|
Average time to complete Questionnaire |
Mean+ SD |
15.1 + 0.4 min |
12.2+ 0.6 min |
||||
|
Average time is taken by the cases parent group |
Mean+ SD |
11.4+1.3 min |
10.5 + 0.5 min |
||||
|
Electronic gadgets usage (yes, no) |
Yes |
13 (86.6%) 2 (13.3%) |
15 (100 %) 0 |
||||
|
No |
|||||||
|
Internet Access availability ( yes=1, no=2) |
Yes |
13 (86.6%) 2 (13.3%) |
15 (100%) 0 |
||||
|
No |
|||||||
|
Nearest Blind school presence (yes or no) |
Yes |
4 (26.6%) 7 (46.6%) 4 (26.6%) |
0 2 ( 13.3%) 13 ( 86.6%) |
||||
|
No |
|||||||
|
Not aware |
|||||||
|
Special attention required or not (yes or no) |
Yes |
7 (46.6%) |
0 |
||||
|
No |
8 (53.3%) |
15 ( 100%) |
|||||
|
Using ocular prosthesis (yes or no ) |
Yes |
5 (100%) |
0 |
||||
|
No |
0 |
0 |
|||||
Table 1: Comparison of structured interview study variables among cases and controls.
|
Quality of life Variable |
Cases Mean+SD |
Controls Mean+SD |
P-Value |
|
Physical health |
0.3+0.96 |
0.3+0.7 |
<0.05 |
|
Social Health |
0.32+1.0 |
0.06+0.1 |
<0.05 |
|
Emotional Health |
0.56+1.06 |
0.2+0.5 |
<0.05 |
|
School Health |
1.6+2.1 |
0.2+0.4 |
<0.05 |
Table 2: Quality of life scores among cases and controls children.
3.2 QoL questionnaire analysis
was done using the SPSS version 25; all the domains, including subgroups such as walking, running, sports activity, helping in household chores, lifting heavy objects, taking a shower, feeling pain, having low energy, feeling afraid, sad, or angry and trouble in sleeping, worrying about future, and getting along with other children, other children not wanting to be their friends, teasing by others, keeping up with other children while playing, not able to do the thing that other children can do, and also paying attention in the class, trouble keeping up with the school work, unable to visit the School due to doctor/ hospital visit, unable to go to the School due to unwell were analyzed using descriptive statics- mean and standard deviation and significance (P value) also calculated.
|
Quality of life Variable |
Cases Mean+SD |
Controls Mean+SD |
P-Value |
|
Physical health |
1.9+0.6 |
0.2 + 0.5 |
<0.05 |
|
Social Health |
2.1 + 0.4 |
0.2 + 0.6 |
<0.05 |
|
Emotional Health |
1.2 + 1.1 |
0.3 + 0.6 |
<0.05 |
|
School Health |
1.5 + 1.2 |
0.1 + 0.4 |
<0.05 |
Table 3: Quality of life scores among parents of cases and controls children.
|
Factors |
N |
(N %) |
||
|
Disease laterality |
Unilateral |
9 6 |
60% 40% |
|
|
Bilateral |
||||
|
Socioeconomic status |
Upper lower class |
6 6 1 1 1 |
40% 40% 6.6% 6.6% 6.6% |
|
|
Lower middle class |
||||
|
Upper class |
||||
|
Upper middle class |
||||
|
Lower class |
||||
|
Gender |
Male |
9 6 |
60% 40% |
|
|
Female |
||||
|
Type of treatment availed |
Enucleation |
5 7 2 1 |
33.3% 46.6% 13.3 % 6.6% |
|
|
Chemotherapy |
||||
|
ExBRT |
||||
|
Brachytherapy |
||||
|
Diagnosis age of Rb survivors in Months (Mean+ SD) |
18.5 + 16.9 |
|||
Table 4: Influencing factors of Quality of Life in Rb Survivors.
In Addition, QoL components were analyzed (Table-2) where RB survivor’s mean of Physical health was 0.3 SD +0.96, Social health 0.32 SD +1.0, and Emotional health 0.56 SD +1.06, School health 1.6 SD+2.1, where controls mean health score recorded as Physical health 0.3 SD +0.7, Social health 0.06 SD +0.1 and Emotional health 0.2 SD +0.5, School health 0.2 SD + 0.4. The P-value for the two-tailed t-test shows P=0.03 with 95% confidence interval, which is less than alpha =0.05 in all the domains of quality of life.
In (Table-3) where RB and non RB- parents (control group) show the mean of Physical Health was 1.9 SD +0.6, Social Health 2.1 SD +0.6, and Emotional Health 1.2 SD +1.1, School Health 1.5 SD +1.2, where control parents mean health score recorded as Physical Health 0.2 SD +0.5, Social Health 0.2 SD +0.6 and Emotional Health 1.2 SD +1.1, School Health 0.1 SD + 0.4. The P-value for the two-tailed t-test shows P=0.03 with a 95% confidence interval, which is less than alpha =0.05 in all the domains of quality of life.
Five RB survivors and their parents reported compromised quality of life due to enucleation. And mean age of the disease diagnosis was 18.5 months SD+ 16.9 (Table 3). Notably, the disease laterality, socioeconomic status, gender, type of treatment, and diagnosis age of survivors warranted the variation in the quality of life in the current feasibility study. Structural Interviews consisted of age, disease-free survival, type of treatment, use of ocular prosthesis need for special attention, ability to use electronic gadgets, and access to the internet (Table 1). Additional features noted were level of education, rural or uRBan background, parents’ education, access and availing of blind schools in their area, and time is taken to complete the survey.
4. Discussion
Health-related quality of life in pediatric practice has a multidimensional role where the subject and the parent’s perception of disease and the treatment side effects can be addressed at the individual level. It is important to study the quality of life in retinoblastoma as it is potentially fatal but has a good prognosis and survival rates with early diagnosis and treatment. It is one of the most easily detectable cancers constituting 3% of pediatric cancers [6]. The results demonstrated that the QoL of RB survivors (M 0.6 SD +1.2) was slightly lower than the control group (M 0.2 SD + 0.4), and the P-value =0.03 was significant. Similarly, the perceptions of parents of QoL of RB survivors (M 1.6 SD + 0.8) were lower than the perception of the parent of the control group(M 0.2 SD + 0.5) and the difference was significant P-value =0.03.
However, Quality of life is the most neglected and important aspect of survivorship. Unlike in other pediatric tumors, visual disability, physical appearance, multiple extensive treatments, and challenges in counseling on prognosis and consequences could affect the quality of life and well-being. The observations in the clinical practice of the researcher also indicate that there appears to be a difference between children and the perceptions of parents of children with RB as compared to other children with non-RB eye pathology. With this as rationale before embarking on a larger study, the authors aimed to evaluate the feasibility of implementing the Pediatric quality of life questionnaire, PEDS QL 4.0 generic score. Previous cross-sectional studies have not identified the feasibility of the Peds Ql-4.0 Generic core scale (Paediatric Quality of Life Inventory Questionnaire) and the supplement interviews like structured interviews to assess the quality of life among RB survivors. Literature has stipulated that childhood cancer survivors may experience poor quality of life in adulthood [5]. In a recent study, Belson et al. 2020 reported that less than six studies had reported a compromised quality of life out of fifteen eligible studies that were reviewed.
Our research shows that the QoL, as mentioned above and structured interview questionnaires may be used with no adverse outcomes. Time spent filling out the questionnaires was much less than required for the Retinoquest, with the average time for cases being 15.1 + 0.4 minutes and the average time for controls being 12.5 + 0.5 minutes [9]. However, Himani Dhingra et al. found that it took 13-15 minutes for participants to complete the Peds QL independently, which is quite close to our findings. In addition, we have found fewer variations in RB survivors’ abilities to walk, run, take showers, assist their parents with everyday chores, and carry anything heavy (Table 4).
The researchers had certain interesting observations while checking the feasibility of the instrument for QoL. Comprehension was seen among the research participants as they walked through the study, with reference to the questionnaire and interview using the PEDS QL 4.0 generic core scale within the target age range [9, 10]. While parents were concerned about their children’s well-being, few were asked about their treatment options or expected length of stay in the hospital, and the information needs were evident. Therefore, the present study points to the need to educate patients, caregivers, and parents on retinoblastoma and its post-treatment care. There was a significant difference in the quality of life of those who survived enucleation versus controls, as well as reports from parents acting as proxies for their children.
The present study’s findings are superior, lending credence to the idea that, for RB survivors, the kind of therapy is crucial in preserving a Normal Quality of life after treatment, mainly because a delay in diagnosis may affect QoL. However, illness laterality and disease diagnostic time play a critical role in sustaining quality of life. Early disease identification might save the visual prognosis post-treatment owing to recency bias. These factors have not been seen to alter significantly among [2, 11] RB survivors.The present research found that utilizing age-matched controls as subjects to compare the RB survivor’s quality of life as supported by previous research is preferable [2]. Because of scheduling constraints, locating the age-matched control is challenging. Himani et al. recommend using age-matched controls to adjust for any socioeconomic differences that could affect research results. However, the same study revealed that factors including SES, gender, and illness diagnosis had no bearing on the QoL of RB survivors [2].
We adjusted the approach by contacting the Age-matched controls by phone or e-mail to compensate for the research’s shortcomings and increase the number of participants in the primary trial. Patient-reported outcomes from parents and cases in a tertiary eye care setting were analyzed. The author believes this study may be the first to manually demonstrate the feasibility of the Peds QoL 4.0 generic core scale. This could shed light on the QoL questionnaire admitting problems and how to modify and proceed further.
Problems were faced during the QoL questionnaire implementation, which will be modified and addressed with adequate counseling
- Finding age-matched controls- The time-taking process must wait until the age-matched subject gets into the OPD
- The subjects and the parents of RB survivors are distressed and not able to answer the questions - cases>controls. Still, it can be improved with supportive prior counseling sessions
- Misguided information in the demographic sheets, like the wrong phone no and addresses and e-mails
- Addressing the patients when the questionnaire was implemented through phone calls
- Require two to three times mock rounds to make patients/ subjects understand the questionnaire answering process
5. Limitation and Future Scope
Recruiting fewer controls due to time constraints and the prevailing pandemic, finding the Participants as well as age-matched young children as controls were difficult in an active comprehensive Opd. The subjects do not represent varied socioeconomic status as the sample is not large. This may influence the perspective differences towards the questionnaire. And this study supports a larger sample to gain some deeper insights into the quality of life of RB survivors, which includes Focused group discussions and in-depth interviews, which leads to giving an understanding of the quality of life of RB survivors in developing countries on a large magnitude.
Implications for Policy and Practise
- The results of the pilot study indicated a lower quality of life among the survivors compared to the control group children
- The perception of the parents of the survivors with regard to the quality of life of the survivors was lower compared to the parents of a control group
- We believe that primary research should be modified so that when the questionnaire is given to children in addition to the researcher's support, assistance from their parents is sought, accurate information is obtained, and participants who are unable to answer the questions easily required more time given more time to complete the survey
- Counseling and helping parents and caregivers understand the changes in survivors’ quality of life before and after treatment and answering their questions concerning financial incentives are recommended
6. Conclusion
The pilot study findings indicate that the instrument of quality of life measurement (Peds QL 4.0) was accepted and understood by the participants, specially the parents, thus making it a feasibility tool for a larger study.As anticipated in this age group, the children required assistance of parents and researchers. Though the larger studies are warranted for interpretation, the trend suggests lower quality of life among the survivors, as well as the perception of parents, thus emphasizing the need for pertinent psychoeducation and counseling of the cases as well as parents before and after the treatment.
Statements
Acknowledgment
The author feels great pleasure in extending thanks to the entire group of CFS staff and the School of medical sciences department staff at the University of Hyderabad for their valuable support throughout the study. The author acknowledges my gratitude to my family for encouraging me all the time and for supporting me in completing my study. The authors are also so grateful for their contribution and guidance in the research. The author feels delighted to acknowledge the authors/publishers/editors’ contributions to the articles referred to in the manuscript. Last but not least, I would like to acknowledge all my study participants for participating.
Ethics Approval
IEC Number: UH/IEC/2021/146 Approved on 26-08-2021.
Conflict of Interest Statement
“The authors have no conflicts of interest to declare.”
Funding Sources
This research is self-funded and received no specific grant from any public, commercial, or not-for-profit funding agency.
Author Contributions
Krishnasri Padamandala (KSP): Study Designing, Analyzing, and interpreting data, and wrote the first draft of the manuscript.
Dr Santosh G Honavar (SH): Provided all clinical resources and supported and guided throughout the study.
Dr. Padmaja Gadiraju (PG): Revised and Reviewed the Manuscript.
Dr Rolika Bansal (RB): Supervision and Guidance throughout the study period.
Dr Surya Durga Prasad (SDP): Revised the proposed work and manuscript content critically, data analysis.
Prof Geeta K Vemuganti (GKV): Supervision and final approval of the study and revised the clinically drafted content critically for important intellectual content.
References
- Retinoblastoma - Definiti | OpenMD.com [Internet] (2021).
- Dhingra H, Arya D, Taluja A, et al. A study analysing the health-related quality of life of retinoblastoma survivors in India. Indian J Ophthalmol 69 (2021): 1482-1486.
- Kivelä T. The epidemiological challenge of the most frequent eye cancer: retinoblastoma, an issue of birth and death. Br J Ophthalmol 93 (2009): 1129-1131.
- Dimaras H, Kimani K, Dimba EAO, et al. retinoblastoma. Lancet Lond Engl 379 (2012): 1436-1446.
- Belson PJ, Eastwood JA, Brecht ML, et al. A Review of Literature on Health-Related Quality of Life of Retinoblastoma Survivors. J Pediatr Oncol Nurs 37 (2020): 116-127.
- Retinoblastoma - PubMed [Internet] (2021).
- Abramson DH, Beaverson K, Sangani P, et al. Screening for retinoblastoma: presenting signs as prognosticators of patient and ocular survival. Pediatrics 112 (2003): 1248-1255.
- Zhang L, Gao T, Shen Y. Quality of life in children with retinoblastoma after enucleation in China. Pediatr Blood Cancer 65 (2018): e27024.
- McNeill NA, Kors WA, Bosscha MI, et al. Feasibility of RetinoQuest: e-health application to facilitate and improve additional care for retinoblastoma survivors. J Cancer Surviv Res Pract 11 (2017): 683-690.
- Krieger JL, Neil JM, Duke KA, et al. A Pilot Study Examining the Efficacy of Delivering Colorectal Cancer Screening Messages via Virtual Health Assistants. Am J Prev Med 61 (2021): 251-255.
- Batra A, Kumari M, Paul R, et al. Quality of Life Assessment in Retinoblastoma: A Cross-Sectional Study of 122 Survivors from India. Pediatr Blood Cancer 63 (2016): 313-317.


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 