Cavernoma in Septum Pellucidum: Descriptive Analysis and Review of Existing Literature
Samiul Haque1*, Asraful Islam2, Tyfur Rahman3, Mohammad D Hossain2, Abu Bakar Siddik4,
Md Manjurul Islam Shourav5, Fayad Hasan6, Samar Ikram6, Masum Rahman6
1Sir Salimullah Medical College, Dhaka, Bangladesh
2Jalalabad Ragib Rabeya Medical College and Hospital Sylhet, Sylhet, Bangladesh
3University of Science and Technology Chittagong, Chittagong, Bangladesh
4Department of Pain Medicine, Mayo Clinic, Florida, USA
5Sylhet MAG Osmani Medical College and Hospital, Sylhet, Bangladesh
6Research Fellow, Department of Neurological Surgery, Mayo Clinic, MN, USA
*Corresponding Author: Masum Rahman, Research Fellow, Department of Neurological Surgery, Mayo Clinic, Rochester, Minnesota, USA
Received: 10 July 2021; Accepted: 19 July 2021; Published: 30 July 2021
Article Information
Citation:
Samiul Haque, Asraful Islam, Tyfur Rahman, Mohammad D Hossain, Abu Bakar Siddik, Md Manjurul Islam Shourav, Fayad Hasan, Samar Ikram, Masum Rahman. Cavernoma in Septum Pellucidum: Descriptive Analysis and Review of Existing Literature. Journal of Cancer Science and Clinical Therapeutics 5 (2021): 320-333.
View / Download Pdf Share at FacebookAbstract
Background: Cavernomas are rare central nervous system (CNS) lesions that constitute a distinct type of vascular malformation encountered in the brain parenchyma or ventricular system. A cavernoma can be familial or sporadic forms and exhibit a range of presentation from incidental findings to seizures, headaches, hemorrhage. Septum pellucidum cavernoma is exceedingly rare and should be studied for its unique topographical location and clinical course.
Method: We performed a comprehensive literature search and review using multiple databases. the title/abstract and MeSH keywords used included “cavernoma,” “cavernous hemangioma,” “cavernous malformation,” “cavernous angioma,” “CM,” “septum pellucidum” “SP” and “intraventricular,” along with “AND” and “OR” operators. Demographic and clinical data of each patient were collected for qualitative synthesis.
Result: Reported cases were diagnosed at a median age of 42 years; the most frequent symptom was headaches. The incidence of hemorrhage and hydrocephalus was 30%. Gross total resection was performed in 100% of patients and exhibit clinical improvement.
Conclusion: The unique location of the cavernoma exhibits characteristic clinical presentations. Gross total resection conveys the impression of an optimum management strategy in most cases.
Keywords
<p>Cavernoma; Cavernous hemangioma; Cavernous malformation; Cavernous angioma; Septum pellucidum</p>
Article Details
1. Introduction
Lesions affecting septum pellucidum are broad and include tumors, cysts, hamartomas, and hemorrhages, which usually arise from the nearby structures. Among these broad differentials, cavernomas malformation (CM) in the septum pellucidum is rare. Cavernoma or cavernous malformation is an incidental or hereditary hamartomatous vascular malformation. They consist of an enlarged, lobulated, and thin-walled vascular malformation without cerebral tissue, typically residing within the brain parenchyma. In the general population, the overall prevalence of cavernomas malformation is 0.02-0.13%, whereas 5-13% among CNS vascular malformations. CNS cavernomas are more frequent in the supratentorial region (74–90%) than infratentorial (10–26%), whereas intraventricular cavernomas are a rare variety of CNS cavernomas accounting for 2.5-10% of all intracranial cavernomas [1, 2, 3]. The prevalence of a cavernoma in the septum pellucidum (SP) is infrequent, having characteristic symptomatology and radiological features. The unusual location, relevant clinical manifestations, risk factors, radiological characteristics, and management principles of SP cavernomas are summarized following existing literature review.
We reviewed relevant literature to find the answer to the following questions: 1) What characteristic clinical features manifest with CMs located at septum pellucidum? 2) What are the associations of septum pellucidum cavernoma? 3) How frequently hydrocephalus, seizure and hemorrhage manifest in patients with these lesions? 4) What is the efficacy of surgical resection in the treatment of these lesions? 5) What surgical approaches have been used when treating CMs at this specific location? 6) What are the outcomes of CM of this location in terms of recurrence, and survival?
2. Data Sources and Search Strategies
A comprehensive search of several databases from 1990 to January 15th, 2021, limited to English language and human study, was conducted. The databases included Pubmed, Ovid MEDLINE(R) and Epub Ahead of Print, In-Process & Other Non-Indexed Citations, and Daily, Ovid EMBASE, Ovid Cochrane Central Register of Controlled Trials, Ovid Cochrane Database of Systematic Reviews, and Scopus. The search strategy was designed and conducted by an experienced librarian with input from the study's principal investigator. Controlled vocabulary supplemented with keywords was used to search for patients with septum pellucidum cavernomas. Keywords used, "septum lucidum," "septum pellucidum", "septum pelusidum," "supracommissural septum," "Cerebral ventricle," "neoplas," "tumor," "tumour," "angioma," "hemangioma," "haemangioma," "cavernoma," "cavernous," along with "AND" and "OR" operators.
PICO question narrated for the study, Population: any patient with septum pellucidum cavernoma, Intervention: surgical resection, Comparator: no comparator, Outcomes: symptomatic improvement, survival, recurrence, progression. There were no restrictions imposed on the age, gender, ethnicity of the patient. Two independent authors screened the search results by examining titles and abstracts for relevance. Articles were finally selected if they met inclusion criteria following full-text article review. Disagreements were resolved by a third reviewer. Any conference papers or studies without full texts, review articles, or meta-analyses without new cases were excluded. Following data were collected from included papers in a preformulated electronic database: age, sex, presenting symptoms, site, and characteristics of the lesion, presence of bleeding, management, prior radiation, presence of concurrent or prior CNS cavernoma, presence of hydrocephalus, management details, surgical approaches and results, clinical outcome, and follow-up. To overcome reporter's bias, only patient-reported symptomatic status was compared to preoperative condition.
3. Results
10 studies were selected using the PRISMA flow diagram and were included in the final analysis. The literature included 10 patients with CMs in the septum pellucidum (Table 1) with a median age of 42 years (range: 16 to 74 years) [4, 3, 5, 6, 7-11]. Of these patients, 8 were male (80%) and 2 were female (20%). Among the study population, the most common presenting symptom was headache (70%, n=7). 20% of patients reported memory impairments. Seizure was reported in one patient precipitated by head trauma (patient-1). Hydrocephalus was apparent on imaging in 30% of cases. 30% had intralesional bleeding at the time of presentation. One asymptomatic patient with prior cavernoma in different locations was diagnosed incidentally on a follow-up MRI. None had a positive family history mentioned in the reviewed literature.
In the case of established risk factors of cavernoma formation, one of the patients had a history of prior radiation and another had a history of head trauma. There was insufficient data on other risk factors. The most common finding on MRI of the brain was a heterogeneous contrast-enhancing lesion (n=9), consistent with cavernoma. In 40% of patients (n-4), lesions were limited to septum pellucidum, while 60% (n=5) had extension into either ventricle. All patients managed surgically without radiation, either open procedure (90%) or endoscopic (10%), and the anterior interhemispheric transcallosal approach was used in most of the cases (70%). Gross total resection was achieved in all patients with negligible postoperative complications. Follow-up data were reported in 80% of cases and did not yield any recurrence or progression.
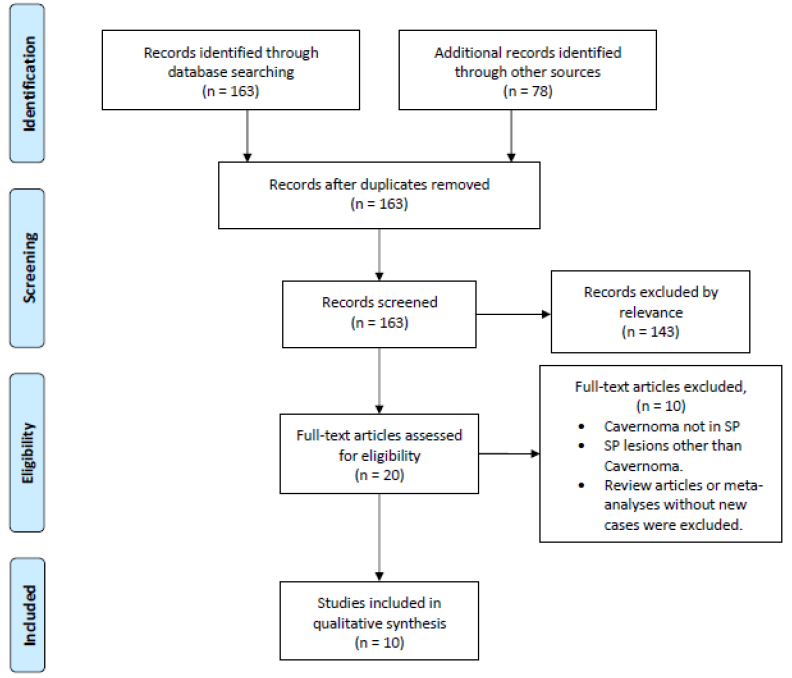
Figure 1: PRISMA flow diagram illustrating the selection process for the studies included in the review for final analysis.
|
No. |
Author |
Patients |
Presenting symptoms |
Family History |
Prior or concurrent CNS cavernoma |
Prior radiation |
|
1 |
Paiva A |
19, M |
Incidental finding following head injury and seizure |
Absent |
Absent |
CNS Radiotherapy |
|
2 |
Baldo S |
32, F |
Asymptomatic, diagnosed by follow-up MRI for other CVs |
Absent |
Multiple, prior |
Absent |
|
3 |
Entezami P |
16, M |
Headache |
Absent |
Absent |
Absent |
|
4 |
Faropoulos K |
56, M |
Headache and lassitude |
Absent |
Absent |
Absent |
|
5 |
31, M |
Headache, loss of consciousness, accompanying vomiting |
Absent |
Absent |
Absent |
|
|
6 |
Muzumdar D |
35, F |
headaches and giddiness |
Absent |
Absent |
Absent |
|
7 |
Katoh M |
44, M |
Hemorrhage, Memory disturbance |
Absent |
Absent |
Absent |
|
8 |
Kasliwal MK |
55, M |
Headache |
Absent |
Absent |
Absent |
|
9 |
Constantinos Picolas |
58, M |
Headache, personality and memory changes, intraventricular hemorrhage |
Absent |
Absent |
Absent |
|
10 |
Motaz Alsereihi |
74, M |
Headache, vomiting, and unsteady |
Absent |
one, in the left putamen. |
Absent |
Table 1: Patient demography, clinical manifestation and relevance.
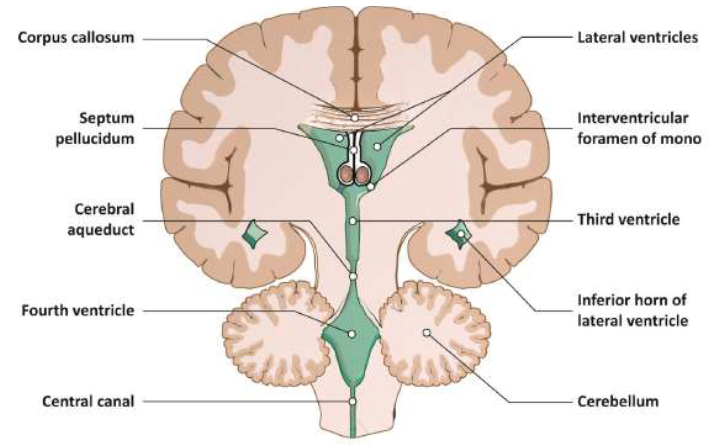
Figure 2: Coronal view ventricles of the brain showing the anatomical location of septum pellucidum.
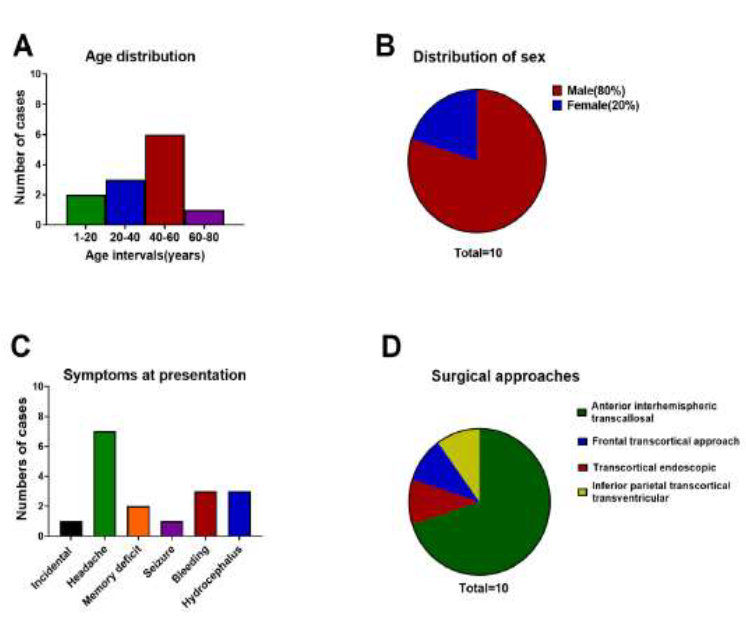
Figure 3: Graphical representation of patient demography, presenting symptoms and surgical approach. A-B: Age and sex distribution. C: Frequency distribution of presenting clinical manifestation. D: Frequency of various approaches used for surgical resection.
4. Discussion
Cavernomas are a cluster of abnormal blood vessels of benign histology. These benign malformations are found in the Central nervous system and throughout the body and have been called Cavernomas, cavernous angiomas, and cavernous hemangiomas. They belong to those cerebrovascular malformations that have no arteriovenous shunt and thus are generally angiographically occult [12]. These malformations are sporadic or occur in relation to an inherited pattern [4]. They are commonly anticipated in the supratentorial region than the infratentorial region exhibiting a varying prevalence of approximately 70-80% and 10-26% respectively [13-15]. Intraventricular cavernomas are rare, commonly found in the lateral ventricle followed by the third ventricle and the fourth ventricle in the CNS. The septum pellucidum (Latin for "translucent wall") is a thin, triangular, vertical double membrane separating the anterior horns of the left and right lateral ventricles of the brain. The first reported case of cavernoma in the septum pellucidum available in the English literature was an intraventricular hemorrhage and the patient presented with headache, nausea [16]. The lesion was resected with no residual signs and symptoms.
4.1 Clinical presentation
Symptoms encircling cavernoma are supposedly induced by a mass effect, raising the intracranial pressure manifesting as headache, nausea, vomiting, papilloedema. CNS cavernomas present with a varying spectrum of clinical signs and symptoms depending on the anatomical site. In the supratentorial region, they present with headaches, seizures, and focal neurological deficits. The severity of such clinical features depends on the location and the extent of the size of the entity. Seizure is a common presentation in supratentorial cavernoma, approximately 39-79% of patients with an annual risk of development of 1.5% [7, 17, 18] In contrast, bleeding is more common in the infratentorial region with a prevalence of 3.8% compared to the supratentorial region, which bears a prevalence of 0.4%. Our analysis demonstrates hemorrhagic complications in 30% and seizure manifestation in 10% of cases, which is quite the opposite of the statistics of supratentorial cavernoma. The manifestation of CNS cavernoma may be delayed, depending on their size and location. Even though relatively small lesions and having enough room on both sides in lateral ventricles, 90% of our patients were symptomatic on presentation. We hypothesize minute intralesional bleeding for symptomatic manifestations. These fragile vascular malformations are susceptible to hemorrhages and are eventually surrounded by hemosiderin deposits and a gliotic membrane when they encounter clinically silent bleeding or may have hemorrhagic stroke-like features. Anatomically septum pellucidum runs as a thin sheet from the corpus callosum down to the fornix. The fornix plays an important role in memory, connecting the hippocampus and other structures involved in memory, including the anterior thalami, septal nuclei, and the mamillary bodies [8, 19]. An impact on the hippocampus can affect the ability to form long-term memories leading to anterograde amnesia.
One study has shown, memory disturbances in a patient with intraventricular cavernoma are more likely (57.1%) when affecting the lateral wall of the third ventricle, mostly secondary to compression on columns of the fornix running through it. [20, 21] A confined hemorrhage within septum pellucidum cavernoma may damage the bilateral fornices. Therefore, even with a small lesion, septum pellucidum cavernoma should undergo immediate surgical removal without further haste. CNS cavernoma may also cause obstructive hydrocephalus when interrupt CSF flow. It has shown that hydrocephalus is more prevalent while affecting the foramen of Monro [20]. In our analysis, hydrocephalus was found in 30% of reported cases of septum pellucidum cavernoma, relatively lower than third ventricular cavernoma (59%). Hydrocephalus likely occurred in these patients owing to downwards extension of the lesion compressing foramen of Monro.
4.2 Imaging
Intraventricular lesions exhibit several differential diagnoses, including arteriovenous malformations, astrocytomas, choroid plexus papillomas, ependymomas, and colloid cysts [4, 6]. Radiologically, cavernoma have been typically described as "popcorn" or "berry" shaped appearance on MRI which represents a histologic pattern of fibrosis, thrombosis, calcification found within these lesions. MRI is the gold standard for imaging cavernomas. Cavernomas are angiographically occult malformations since they are not connected to any major blood vessels [22]. Nevertheless, developmental venous anomalies typically associated with and are visible during angiography and MRI [4]. In the parenchymal form, a hypointense ring is observed on T1 and T2-weighted images due to hemosiderin deposits from recurring microhemorrhages [4]. In our analysis, all cases exhibit characteristic features on MRI includes, well-circumscribed nodular lesion with heterogeneous signal intensity, none to moderate post-gadolinium enhancement with or without hemorrhagic components (Table-2).
4.3 Management
The management of intracerebral cavernomas is related to specific locations and signs and symptoms. Asymptomatic lesions in eloquent areas can be observed. Whereas complete excision is the treatment of choice symptomatic patients with mass effect (headache and hydrocephalus, neuro deficit), seizures, recurrent hemorrhages, intractable seizures, and lesions increasing in size. [3, 7, 20]. Intraventricular cavernomas are usually accessed by transcranial, transcortical approach, which is technically easier requires large ventricles and a craniotomy to enter the brain. Like all procedures, there are potential risks to be discussed as the judgment of benefits versus risk must be considered to proceed with the procedure. In the end, the patient benefit must outweigh the risks. A transcranial transcortical approach can cause epilepsy. The other approach is a transcranial interhemispheric transcallosal route. The anterior transcallosal approach is adequate and is often superior to the ventricular transcallosal approach without hydrocephalus (to prevent blood spillage into the ventricles to prevent meningism and obstructive hydrocephalus. This approach is more anatomical and does not require a large ventricle, but it is technically more demanding [3].
In addition, callosotomy is too posterior and can give away a disconnection syndrome problem. An alternative and much less invasive procedure concerns the endoscopic method, which is increasingly used in the treatment of ventricular tumors. Likewise, it brings about technical dismay and hesitation among surgeons due to the vascular nature of these lesions and possible difficulty in achieving hemostasis [3]. Regardless, minimally invasive surgery permits a better surgical outcome and reduces adverse outcomes and prompts better postoperative recovery. Neuroendoscopy is being increasingly used in the treatment of intraventricular tumors, however its potential for use in the excision of septum pellucidum tumor is still unclear. Since an endoscopic approach leaves out the doubt of achieving adequate hemostasis, microsurgical approach is the viable option to ensure a safe and complete resection, given the anatomy of the region and its intense vascularization [7]. One patient in our cohort (patient-2) underwent endoscopic resection, which was justified by the possibility of heavy scar from previous surgery. Inferior parietal transcortical approach was reported in patient-11 due to presence of ventricular hemorrhage [11].
To summarize the approach and risks. For a lateral ventricle trigonum IVC, transtemporal, posterior transcortical or posterior transcallosal approach is advocated [23-25]. And likewise, each approach bears its own risk. The posterior middle temporal gyrus transcortical approach can induce damage to the optic radiation leading to left lower quadrantanopia or to the dominant hemisphere. Posterior parietal transcortical access can inflict visuospatial apraxia if the mass is located in the dominant hemisphere [6, 26]. Ultimately, in the long run, complete surgical removal of IVC is the treatment of choice with low-rate complications and associated morbidity [6]. Radiotherapy is controversial for managing intraventricular cavernoma. Intraparenchymal cavernomas in high-risk areas have been reported to treat with stereotactic radiotherapy [27, 28]. In few case reports, they received radiation upon misdiagnosis as a tumor and have not shown satisfactory clinical improvement [29, 30]. Moreover, radiation contains the potential risk of bleeding, recurrence, and expansion of the cavernoma. One study has shown that surgical resection of supratentorial cavernomas results in a lower incidence of rebleeding and better control seizures than stereotactic radiotherapy [27]. In our analysis, gross total resection was achieved in all patients with favorable postoperative and long-term outcomes without any radiotherapy.
4.4 Effect of resection on outcome
Our analysis denotes gross total resection is an effective method of treating septum pellucidum cavernoma, as 100% of cases demonstrated significant clinical improvement after surgery (Table-3). Nevertheless, memory disturbances were the most common residual deficit following surgery that was persistent in all patients who initially had. We hypothesize irreversible damage to fornices either due to mass effect or minute bleeding.
|
No. |
Author |
Patients |
Topography |
Radiology |
Evidence of hydrocephalus |
|
1 |
Paiva A |
19, M |
Septum pellucidum |
MRI: a nodular lesion with heterogeneous signal intensity on T1-weighted, T2- weighted, and FLAIR modalities; |
No |
|
2 |
Baldo S |
32, F |
Septum pellucidum |
MRI: Multilobulated mass with heterogeneous signal intensity. |
No |
|
3 |
Entezami P |
16, M |
SP, Rt lateral ventricle |
CT: Hyperdense lesion in the frontal aspect of the septum pellucidum. MRI: Heterogeneously enhanced lesion on T1-sequences with hemorrhagic components |
No |
|
4 |
Faropoulos K |
56, M |
SP, Rt lateral ventricle |
CT: hyperintense mass in the septum pellucidum. T1-weighted MRI: a mixed signal mass; moderate post-gadolinium enhancement. |
No |
|
5 |
31, M |
SP, Rt lateral ventricle |
T1-weighted MRI: 2 -2.3-cm mulberry-like mass which had a low signal, with few high magnetic signal spots. Gadolinium did not cause enhancement. |
No |
|
|
6 |
Muzumdar D |
35, F |
SP, extending to anterior 3rd ventricle, FoM |
T1-weighted MR: a heterogeneous iso- to hypointense well-circumscribed lesion attached to the lower septum pellucidum abutting the FoM leading to left-sided ventricular dilation. |
Yes |
|
7 |
Katoh M |
44, M |
Septum pellucidum |
MRI: hematoma localizing in the septum pellucidum |
No |
|
8 |
Kasliwal MK |
55, M |
Septum pellucidum, Rt lateral ventricle |
MRI: an inhomogeneous lesion in the lateral ventricles, arising from the septum pellucidum |
No |
|
9 |
Constantinos Picolas |
58, M |
Septum pellucidum |
CT: A lesion of the posterior1/3 of the septum pellucidum with intraventricular hemorrhage in right lateral ventricle. MRI: mixed dispersed iso- and hyperintense nodular loci in T1 sequence and hypointense with dispersed nodular hyperintense loci in T2 |
Yes |
|
10 |
Motaz Alsereihi |
74, M |
Septum pellucidum, Lt lateral Ventricle |
CT: a hyperdense lesion within the frontal horn of the left lateral ventricle. MRI: intraventricular hemorrhagic lesion within the frontal horn with a moderate left-sided hydrocephalus from obstruction of FoM. |
Yes |
SP= Septum pellucidum, Rt= Right, Lt= Left, FoM= foramen of Monro, M= Male, F= Female
Table 2: Imaging data.
NA= Not available, *author’s assumption of anterior interhemispheric transcallosal according to the most likely approach with this craniotomy for this lesion.
Table 3: Management and outcome.
4.5 Study limitations
This study bears a couple of inherent limitations. First, it's a review article that summarizes all reported cases without primary patient data. Second, cases of CP cavernoma were retrospectively reviewed with inevitable heterogeneous and limited availability of data regarding imaging or work-up strategies, treatment approaches, as well as follow-up. Third, as unpublished cases and studies are written in non-English languages are excluded; thus publication bias was unavoidable. Lastly, due to the small number of cases and absence of comparator data, this study doesn't draw any broad statistically significant conclusions, including ideal
treatment methods.
5. Conclusion
While common in the parenchyma and ventricle, SP cavernoma is a rare CNS vascular malformation with distinct clinical behavior due to its location. Perilesional lateral ventricular space may allow expansile growth and probably higher hemorrhagic incidences with potential damage to nearby noble structures. Despite smaller lesions, high clinical and radiological suspicion is necessary to expedite early intervention to prevent irreversible neurologic impairment. Complete excision with the long-term excellent and curative outcome is possible and is the treatment of choice. Despite being nonconclusive, this study summarizes all the well-managed CP cavernoma cases and gives a minor but essential clue to diagnostic and therapeutic approaches. This is a highly selected series of patients, and all have been operated. In general, managing CM is very restrictive with surgical treatment, especially in asymptomatic cases. The reported headaches may be coincidental (no-hydrocephalus cases) and oftentimes not caused by a CCM in the septum pellucidum.
Author’s Contribution
Literature search, screening, and data extraction by SH, AI, TR. Data analysis and manuscript writing by MDH, AS, MIS, SI. Conceptualisation, study design, data analysis, and manuscript writing and editing by MR. Illustration by MFH. All authors reviewed and suggested edits to make the final version.
Conflict of Interest
All authors declare no conflict of interest.
Disclosures
The authors have no financial conflicts of interest to declare.
Human/Animal Rights
This article does not contain any studies with human or animal subjects performed by any of the authors.
Acknowledgments
We want to acknowledge and thank Larry J. Prokop, Outreach Librarian, Mayo Clinic Libraries, and CMSR (center for medical study and research) foundation.
References
- Dalyai RT, Ghobrial G, Awad I, et al. Management of incidental cavernous malformations: a review. Neurosurg Focus 31 (2011): E5.
- Tatagiba M, Schönmayr R, Samii M. Intraventricular cavernous angioma. A survey. Acta Neurochir (Wien) 110 (1991): 140-145.
- Baldo S, Magrini S, Tacconi L. Purely Endoscopic Resection of Cavernoma of the Septum Pellucidum. Surg J (New York, NY) 5 (2019): e42-e45.
- Paiva A, Lovato RM, Araujo JO, et al. Septum Pellucidum Cavernoma: A Case Report and Anatomical Consideration of an Extremely Rare Lesion. Turk Neurosurg 30 (2020): 145-148.
- Entezami P, Adamo MA. Rare Pediatric Presentation of a Cavernous Angioma of the Septum Pellucidum. Pediatr Neurosurg 54 (2019): 147-149.
- Faropoulos K, Panagiotopoulos V, Partheni M, et al. Therapeutic management of intraventricular cavernoma: case series and review of the literature. J Neurol Surg A Cent Eur Neurosurg 76 (2015): 233-239.
- Muzumdar D, Avinash KM, Ramdasi R. Cavernoma of the septum pellucidum in the region of foramen of Monro. Neurol India 63 (2015): 68-71.
- Katoh M, Sawamura Y, Moriwaki T, et al. A case of cavernous angioma in the septum pellucidum. Clin Neurol Neurosurg 115 (2013): 1126-1127.
- Kasliwal MK, Sharma BS. Giant intraventricular mass arising from the septum pellucidum. Cavernoma. J Clin Neurosci Off J Neurosurg Soc Australas 18 (2011): 1108-1145.
- Alsereihi M, Turkistani F, Alghamdi F, et al. Apoplexy of a collision tumour composed of subependymoma and cavernous-like malformation in the lateral ventricle: a case report. Br J Neurosurg 33 (2019): 581-583.
- Picolas C, Faropoulos K, Kekempanou K, et al. Case Report of a Septum Pellucidum Cavernoma Surgically Resected via Inferior Parietal Approach and Short Literature Review. Open J Mod Neurosurg (2016).
- Ahlhelm F, Hagen T, Schulte-Altedorneburg G, et al. Cavernous malformations. Radiologe 47 (2007): 863-867.
- Mouchtouris N, Chalouhi N, Chitale A, et al. Management of cerebral cavernous malformations: from diagnosis to treatment. ScientificWorldJournal 2015 (2015): 808314.
- McCormick WF, Hardman JM, Boulter TR. Vascular malformations (“angiomas”) of the brain, with special reference to those occurring in the posterior fossa. J Neurosurg 28 (1968): 241-251.
- Voigt K, Yasargil MG. Cerebral cavernous haemangiomas or cavernomas. Incidence, pathology, localization, diagnosis, clinical features and treatment. Review of the literature and report of an unusual case. Neurochirurgia (Stuttg) 19 (1976): 59-68.
- Narisawa A, Kumabe T, Anzai T, et al. A case of cavernous angioma at the septum pellucidum. No Shinkei Geka 35 (2007): 53-58.
- Zabramski JM, Wascher TM, Spetzler RF, et al. The natural history of familial cavernous malformations: results of an ongoing study. J Neurosurg 80 (1994): 422-432.
- Casazza M, Broggi G, Franzini A, et al. Supratentorial cavernous angiomas and epileptic seizures: preoperative course and postoperative outcome. Neurosurgery 39 (1996): 24-26.
- Moudgil SS, Azzouz M, Al-Azzaz A, et al. Amnesia due to fornix infarction. Stroke 31 (2000): 1418-1419.
- Beechar VB, Srinivasan VM, Reznik OE, et al. Intraventricular Cavernomas of the Third Ventricle: Report of 2 Cases and a Systematic Review of the Literature. World Neurosurg 105 (2017): 935-943.e3.
- Sinson G, Zager EL, Grossman RI, et al. Cavernous malformations of the third ventricle. Neurosurgery 37 (1995): 37-42.
- Pozzati E, Gaist G, Poppi M, et al. Microsurgical removal of paraventricular cavernous angiomas. Report of two cases. J Neurosurg 55 (1981): 308-311.
- Cikla U, Swanson KI, Tumturk A, et al. Microsurgical resection of tumors of the lateral and third ventricles: operative corridors for difficult-to-reach lesions. J Neurooncol 130 (2016): 331-340.
- Asgari S, Engelhorn T, Brondics A, et al. Transcort-ical or transcallosal approach to ventricle-associated lesions: a clinical study on the prognostic role of surgical approach. Neurosurg Rev 26 (2003): 192-197.
- Timurkaynak E, Rhoton ALJ, Barry M. Microsurgical anatomy and operative approaches to the lateral ventricles. Neurosurgery 19 (1986): 685-723.
- Juretschke FR, Güresir E, Marquardt G, et al. Trigonal and peritrigonal lesions of the lateral ventricle-surgical considerations and outcome analysis of 20 patients. Neurosurg Rev 33 (2010): 457-464.
- Shih Y-H, Pan DH-C. Management of supratentorial cavernous malformations: craniotomy versus gammaknife radiosurgery. Clin Neurol Neurosurg 107 (2005): 108-112.
- Nagy G, Razak A, Rowe JG, et al. Stereotactic radiosurgery for deep-seated cavernous malformations: a move toward more active, early intervention. Clinical article. J Neurosurg 113 (2010): 691-699.
- Tu J, Stoodley MA, Morgan MK, et al. Different responses of cavernous malformations and arteriovenous malformations to radiosurgery. J Clin Neurosci Off J Neurosurg Soc Australas 16 (2009): 945-949.
- Ogawa A, Katakura R, Yoshimoto T. Third ventricle cavernous angioma: report of two cases. Surg Neurol 34 (1990): 414-420.


 Impact Factor:
* 4.1
Impact Factor:
* 4.1
 Acceptance Rate:
74.74%
Acceptance Rate:
74.74%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
