HPV16-Positive Skin Metastasis at the Nose 5 Years Following Treatment of an HPV16-Positive Squamous Cell Carcinoma of the Tonsil – A Case Report and Review of Literature
Christiane Stuhlmann-Leisz1*, Asita Fazel2*, Lukas Lükewille2, Christoph Röcken1, Martin Laudien2,3, Elgar Susanne Quabius2,3, Markus Hoffmann2,3**
1Institute of Pathology, Christian-Albrechts-University Kiel, Germany
2Department of Otorhinolaryngology, Head and Neck Surgery, Christian-Albrechts-University Kiel, Germany
3Quincke-Forschungszentrum, Christian-Albrechts-University Kiel, Germany
*contributed equally
*Corresponding Author: Markus Hoffmann, Department of Otorhinolaryngology, Head and Neck Surgery, Christian-Albrechts-University Kiel, Arnold-Heller-Straße 3, Building 27, D-24105 Kiel, Germany.
Received: 04 August 2022; Accepted: 19 September 2022; Published: 06 October 2022
Article Information
Citation: Christiane Stuhlmann-Leisz, Asita Fazel, Lukas Lükewille, Christoph Röcken, Martin Laudien, Elgar Susanne Quabius, Markus Hoffmann. HPV16-Positive Skin Metastasis at the Nose 5 Years Following Treatment of an HPV16-Positive Squamous Cell Carcinoma of the Tonsil – A Case Report and Review of Literature. Journal of Cancer Science and Clinical Therapeutics 6 (2022): 344-348.
View / Download Pdf Share at FacebookAbstract
This case report presents the disease course of a patient who developed HPV16-positive skin metastasis to the nose of HPV16-positive tonsillar carcinoma treated 5 years previously. The report critically reviews the available literature, particularly the comparable case reports available to date. In light of conflicting findings, it now appears unclear whether the occurrence of skin metastases reflecting an unusual pattern of metastasis is more typical of the course of HPV-positive HN/OSCC. There are reports suggesting this association, including our case report. On the other hand, there are retrospective study results that rather do not point to an association of skin metastases explicitly with (HPV-positive) OSCC. Rather, it must be critically questioned whether there is indeed a significant association between a specific metastatic pattern including unusual metastatic sites such as the skin, or whether merely increased attention to HPV-positive cancers leads to a higher publication rate of these cases. Extensive epidemiologic evidence should be obtained on a retrospective and prospective basis before recommendations for surveillance of corresponding patients are derived from the assumption of an atypical or different metastatic pattern in HPV-positive HNSCC.
Keywords
<p>HPV; Nose; Skin Metastases; TSCC; OSCC; Pattern of Metastases; Tonsil</p>
Article Details
1. Introduction
The majority of head and neck squamous cell carcinomas (HNSCC) is causally linked to enhanced consumption of tobacco and alcohol [1]. However, there also is a subgroup of HNSCCs that is causally linked to the infection with human papillomaviruses (HPV), specifically HPV16 [2]. The latter predominantly holds true for oropharyngeal (OSCC), precisely tonsillar SCC (TSCC), which show causative HPV infections in 60-90% of the cases, the divergence of which most likely is due to the geographical region the patients live in [3]. Since the onset of the knowledge on HPV-driven carcinogenesis not only in the anogenital but also in the upper aerodigestive tract starting from the late 1980s, it could be established that HPV-positive compared to HPV-negative head and neck cancers express a different biological behavior with i) an earlier onset of disease consecutively with patients being younger in many, yet not all regions of the world, ii) different mechanisms of underlying malignant transformation reflected by a smaller proportion of smokers, iii) patients suffering from a higher burden of disease especially of the lateral neck, i.e. HNSCCs being diagnosed at a higher tumor stage as a sign of a more aggressive biological behavior of the tumor, iv) a better response to therapy in terms of both, surgery and radio(chemo)therapy with v) a significant better prognosis [lit. in 4]. It might be assumed that due to the established more aggressive biological behavior of HPV-positive tumors and due to the longer overall and or progression free survival times, HPV-positive patients will more often experience the development of distant metastases (DM). However, when compared to HPV-negative cases patients with HPV-positive OSCCs carry a similar risk of DM (approx. 10%) at three or five years after diagnosis [5,6]. While thus the DM rate of OSCC is unaffected by HPV status dissemination of HPV-positive OSCC is more often observed at multiple organs and unusual sites and likely occurs after longer intervals. Whereas the most common sites of DM in OSCC, regardless of HPV status, includes lunge, lymph nodes, bone, and liver, HPV-positive OSCC has also resulted in atypical metastatic patterns, including dural and brain metastases [7,8] as well as metastases of the skin. Distant skin metastases is a rare event in cancer progression in general as depicted by a retrospective study of 4020 patients with metastatic cancer revealing that only 10% of the patients developed skin metastases; the most common malignancies manifesting with skin metastases were breast cancer and indeed melanoma [9]. Another review retrospectively analyzing 798 patients with HNSCC found that only 19 (2.4%) patients developed skin metastases, none of the primary tumors were of tonsillar origin [10]. Moreover, only 15% of skin metastases in the latter study group occurred outside the head and neck region and all occurred in the chest. Therefore, it seems of note that there appears to be a rise of reports describing skin metastases of O/TSCC in recent years, with one case report in 2002 [11] and further six cases described between 2014 and August 2021 [12-17]. Despite the increasing knowledge regarding the role of HPV in head and neck carcinogenesis, only 2 of the aforementioned publications reported on the HPV status of the primary tumor (PT) and the skin metastases [15,17] with one solely applying p16 immunohistochemistry (IHC) [15], known as a surrogate marker for the HPV status. The authors of the one other article [17] performed direct HPV detection methods using tissue specimens derived from the PT and skin metastases, however detecting HPV only in the PT whereas p16 IHC revealed strong staining in both, PT and skin metastases. Hence, the present report on the 8th case of a skin metastases 5 years after treatment of an TSCC is the first i) applying both, p16 IHC and PCR based HPV DNA detection methods and ii) detecting HPV16 DNA in both, PT and skin metastases, paralleled by p16 overexpression determined by IHC.
2. Case Report
We report on a male non-smoking patient, 56 years of age at diagnosis of the primary TSCC, without relevant previous diseases, who presented for the first time in February 2016 in the Department of Otorhinolaryngology, Head and Neck Surgery, University-Clinic of Schleswig-Holstein, Campus Kiel, Germany, with a painful cervical swelling of the lateral neck. The patient reported to suffer from the well-palpable node for 8 months at first presentation. The endoscopic examination remained inconspicuous except for a prominent tonsil on the right side. Ultrasound examination of the lateral neck described several enlarged cervical nodes on the right side suspicious of malignancy. The cytopathologic diagnostic of a fine needle aspiration derived from the largest lymph node detected atypical cells, again suspicious of the presence of a malignancy. A panendoscopy performed in general anesthesia did not detect further lesions of the mucosa and the histopathological work-up of the collected tissue biopsy confirmed the presence of low differentiated, partly keratinizing squamous cell carcinoma. Staging diagnostics performed did not indicate the presence of any distant metastases. The by the tumor board recommended transoral endoscopic laser-assisted resection of the right tonsil with 0.5 cm margins was performed with synchronous neck dissection of the regions II to IV. To protect from peri- and post-operative bleeding, the A. lingualis, A. facialis, and pharyngea ascendens were ligated. The latter is indicated when tumor resection of the tonsils must be performed laterally well beyond the tonsillar capsule. The ligation of the arterial vessels supplying the tumor is not associated with any treatment depending morbidity of the patient, but protects from potentially lethal peri- and postoperative bleeding. The histopathologic work-up confirmed the above described findings of the PT, and additionally described in 3 of 20 lymph nodes the presence of a here none-keratinizing SCC with moderate differentiation and without perinodal spread. HPV diagnostic of the PT found the tissue specimens to be p16INK4A- and HPV16-positive. Again, according to the tumor board recommendation the patient received adjuvant radiochemotherapy (54Gy, 300 mg Cisplatin; end of therapy: 05/31/2016). The patient tolerated the therapy well and in the following years tumor follow-up was inconspicuous until he referred himself to his outpatient doctor because of swelling of the columella and recurrent epistaxis in March 2021. Initially the patient was treated for inflammation and due to progression with the clinical impression of an abscess in the region of the columella an incision of the lesion was performed demasking suspicious tissue. The histopathological work-up of biopsies taken from the tissue confirmed the presence of an invasive, keratinizing, moderate differentiated squamous cell carcinoma of the nose (Figure 1). The patient was referred to our Department and after again inconspicuous staging had surgery on the SCC of the nose in terms of ablation of the nose. The TNM stage regarding this assumed primary tumor was pT3 N0 M0 L0 V0 G2 pR0. Further molecular-pathologic analysis on the tissue specimens, however, detected a HPV16-infection going along with intensive p16-staining determined by IHC. Hence, tissue specimens derived from the tonsillar SCC diagnosed in 2016 were compared to the tissue specimens derived from the nose and finally the latter were interpreted as skin metastasis of the tonsillar SCC, specifically since SCC of the skin in general are not associated with a HPV16 infection (Figure 1). Therefore, TNM-classification of the TSCC was changed to pT2 pN2b (3/20) M0c2 L0 V0 Pn0 G3 pR0) M1 (HPV-positive, p16-positive). According to the recommendation of the tumor board the patient was referred to surveillance without further adjuvant treatment. The patient received a temporary bone-anchored epithesis and by now decided not to have a reconstruction of the nose with a frontal skinflap but prefers to stay with an epithetical reconstruction (Figure 2). Thus, the temporary epithesis will be replaced by a permanent one. In August 2022, the patient still is in tumor surveillance without any signs of recurrence of the disease.
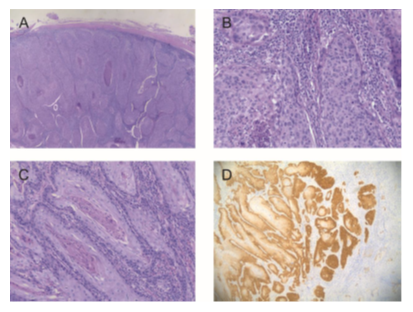
Figure 1: Histology of the primary tonsillar primary tumor and the skin metastasis of the tonsillar SCC. Figure 1A shows a H&E stained overview of the primary tonsillar tumor at 4x magnification and Figure 1B a more detailed view at 20x magnification. Figure 1C shows a H&E staining of the skin metastasis at 10x magnification. Figure 1D shows strong (≥70% of tumor cells show nuclear and cytoplasmic expression) p16INK4A overexpression in the metastasis determined by IHC (10x magnification).
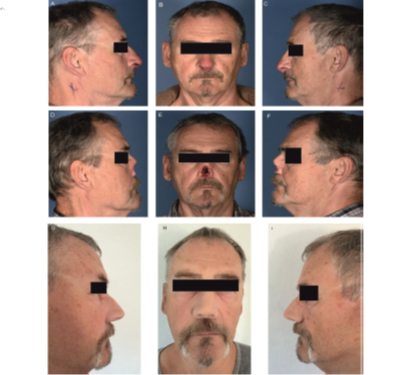
Figure 2: Photographs of the patient. Figures 1A – 1C show lateral (left, right) and frontal views of the patient with the nasal skin metastasis in situ. Figures 1D – 1F show pictures of the patient after the surgery on the nasal SCC (ablation of the nose) and Figures 1G – 1I show the patient with his temporary epithesis which will in due course be replaced by a permanent one.
3. Discussion
The here presented case of a HPV16-positive skin metastasis at the nose derived from a HPV16-positive tonsillar carcinoma treated 5 years previous is, to the best of our knowledge, the first with direct HPV16 detection in both, the primary tumor and the metastasis. Seven other cases with skin metastases attributed to oropharyngeal SCC have been described to date [11-17], but have used no (n=5) [11-14, 16] or only indirect HPV detection methods (p16 IHC, n=2)[15,17]. As already highlighted in the introduction the occurrence of skin metastasis in general is a rare event and, thus, it might be discussed whether or not skin metastases is one of the characteristics that contribute to the different biological behavior of HPV-positive versus HPV-negative SCC, namely the atypical pattern of metastases in terms of unusual anatomical sites of metastasis and a longer interval between PT treatment and the occurrence of any distant metastases (DM). Ferlito and co-workers [18] pointed out that i) the incidence of DM is directly related to the stage of the PT and locoregional control and that ii) SCC with advanced T stage in the oropharynx (along with SCC of the oral cavity and hypopharynx) are the HNSCC entities, most likely to develop DM. However, it should be considered that these parameters were defined for synchronous and metachronous occurrence of DM which generally appear in the order lung, bone or liver. There are, yet, no clear features characterizing a specific subgroup of (HPV-positive or HPV-negative) patients developing skin metastases. Table 1, accordingly, shows rather diverse patient and tumor characteristics: there are three cases with earlier tumor stages according to the 7th edition of the TNM classification (UICC/AJCC) [which needs to be applied to all described cases since all cases were diagnosed before 2017, the year the 8th edition of the TNM classification was published]. The other four cases were diagnosed with tumor stage IV, including ours which, however, would have been staged as pT2pN1pM0, stage II, if the 8th edition of the TNM classification would have been applied. Additionally, all but two [14,17] cases developed skin metastases despite locoregional control of their disease in regard of the OSCC including our case. Additionally, the cases reported by Li and co-workers [17] and Ramser and co-workers [15] suffered from laryngeal and prostate cancer, respectively, prior to OSCC diagnosis. Another two cases developed additionally to the CM, metastases in the brain [16], and in the axilla [15] representing oligo-metastases. In the light of the latter heterogeneity and the partly missing molecular proof that the skin metastases derived from the PT in the here summarized cases, substantial bias of the perception that skin metastases could underline atypical metastatic patterns of (HPV-positive) OSCC cannot be excluded. A remarkable retrospective study by Huang and co-workers performed on 318 patients with known HPV-status of the tumors [19] shows on the one side that there is no difference in DM rate when HPV-negative cases were compared to HPV-positive OSCC. They did, on the other side, find hints that metastases derived from HPV-positive OSCC were more likely to involve multiple organs and unusual sites, such as the skin. Indeed, they reported on 7 skin metastases out of the 318 patients with OSCC, all of them were HPV-positive. On the other side, the already mentioned retrospective study on altogether 4020 patients with metastatic disease published by Lookingbill and co-workers in 1993 [9] reported on altogether 427 patients with skin metastases with 7, 3, and 7 derived from the larynx, esophagus, and oral cavity, respectively. Thus, in that study population there was no oropharyngeal SCC which was associated to distant metastases in terms of skin metastases. Alike, Yoskovitch and co-workers [10] retrospectively analyzing 798 consecutive HNSCC patient records, found 19 skin metastases of which 10, 5, 2, and 1 each were derived from the oral cavity, larynx, oropharynx, hypopharynx and paranasal cavity, respectively. Additionally, the authors reported that the average survival time among patients after the development of skin metastases was 7.2 months; the 1-year survival rate 0%. Nonetheless, the patient described here might indeed represent HPV-attributable tumor characteristics with several noteworthy biological features: Even though the patient was diagnosed with an advanced tumor stage he by now survived for 6 years without locoregional recurrence; 75 months later, i.e. with a comparably long interval to PT treatment, he developed a skin metastasis to the nose which was surgically removed and he is to date, more than one year later, still alive and without recurrence of disease. Combined with HPV-infection in both, PT and skin metastases, the latter course of disease is remarkable. Whether such disease courses are after all typical for HPV-positive OSCC needs further clarification.
|
Study |
TNM*/stage |
Age/Sex |
smoking |
HPV |
p16 |
Time interval** |
CM treatment |
|
Desmajumdar [11] |
T3N2cM0/IV |
40/M |
S |
- |
- |
26 |
Pall. RT |
|
Chikkannaiah [12] |
T2N0M0/II |
70/M |
S |
- |
- |
16 |
none |
|
Singh [13] |
T3N1M0/III |
55/M |
S |
- |
- |
6 |
Pall. MTX |
|
Bari [14] |
n.a./IV |
59/M |
NS |
- |
- |
54 |
Refused |
|
Ramser [15] |
T4N2bM0/IV |
70/M |
NS |
- |
p16+ in CM |
24 |
Nivolumab |
|
Banerjee [16] |
T2N0M0/II |
34/M |
S |
- |
- |
20 |
Pall. RT |
|
Li [17] |
n.a. |
77/M |
S |
HPV16 in PT |
p16+ in PT/CM |
46 |
Pembrol. |
|
Present case |
T2N2bM0/IV |
56/M |
NS |
HPV16 in PT/CM |
p16+ in PT/CM |
75 |
Surgery |
Table 1: Characteristics of patients with CM after treatment of TSCC.
*TNM of PT at first time diagnosis, all cases were staged according to the 7th edition of TNM-classification;
**time in months between treatment of primary tumor and occurrence of CM; Abbr.: S: smoker; NS: non-smoker; n.a.: not available; RT: radiotherapy; M: male;
4. Conclusions
Nowadays, against the background of inconsistent evidence, it seems unclear whether the occurrence of skin metastases as an expression of an unusual metastatic pattern is more typical of the course of a HPV-positive HN/OSCC. There are reports in favor of this association, including our case report. On the other hand, retrospective study results exist that rather do not suggest a relation of skin metastases explicitly to (HPV-positive) OSCC. In fact, it must be critically questioned whether there is really a significant relationship between a particular metastatic pattern including unusual metastatic sites such as the skin, or if there is just an increased awareness regarding HPV-positive cancers resulting in a higher rate of publishing these cases. Before recommendations for surveillance of respective patients are derived from the assumption of an atypical, respectively different metastatic pattern in HPV-positive HNSCC, extensive epidemiological knowledge should be generated on a retrospective and prospective basis.
Informed Consent Statement
Informed consent was obtained from the patient including the permission to publish pictures of the patient himself.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Marur S, Forastier AA. Head and neck squamous cell carcinoma: Update on epidemiology, diagnosis, and treatment. Mayo Clin Proc 91 (2016): 386-936.
- Gillison, ML, Koch WM, et al. Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J Natl Cancer Inst 92 (2000): 709-720.
- Quabius ES, Haag J, Kühnel A, et al. Geographical and anatomical influences on human papillomavirus prevalence diversity in head and neck squamous cell carcinoma in Germany. Int J Oncol 46 (2015): 414-422.
- Hoffmann M, Quabius ES. Relevance of Human papillomaviruses in Head and Neck Cancer – What remains in 2021 from a Clinician´s Point of View? Viruses 13 (2021): 1173.
- Ang K, Harris J, Wheeler R, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med 36 (2010): 24-35.
- O´Sullivan B, Huang SH, Prez-Ordonez B, et al. Outcomes of HPV-related oropharyngeal cancer patients treated by radiotherapy alone using altered fractionation. Radiother Oncol 103 (2012): 49-56.
- Al-Khudari S, Guo S, Chen Y, et al. Solitary dural metastasis at presentation in a patient with untreated human papillomavirus-associated squamous cell carcinoma of the oropharynx. Head Neck 36 (2014): 103-105.
- Ley J, Wildes T, El-Mofty S, et al. Metastasis occurring eleven years after diagnosis of human papillomavirus-related oropharyngeal squamous cell carcinoma. Ecancdermedicalscience 8 (2014): 480.
- Lookingbill DP, Spangler N, Helm KF. Cutaneous metastases in patients with metastatic carcinoma: a retrospective study of 4020 patients. J Am Acad Dermatol 29 (1993): 228-236.
- Yoskovitch A, Hier MP, Okrainec A, et al. Skin metastases in squamous cell carcinoma of the head and neck. Otolaryngol Head Neck Surg 124 (2001): 248-252.
- Dasmajumdar SK, Gairola M, Sharma DN, et al. Cutaneous metastasis from carcinoma of tonsil. J Pastgrad Med 48 (2002): 32-33.
- Chikkannaiah P, Boovalli MM, Kumar P, et al. Unusual cases of carcinoma of palatine tonsil. J Oral Maxillofac Pathol 19 (2015): 242-246.
- Singh GK, Yadav V, Singh P, et al. Multiple cutaneous metastases in a patient of carcinoma tonsil – report of a rare case. J Egypt Natl Canc Inst 28 (2016): 191-193.
- Bari O, Cohen PR. Cutaneous metastasis from tonsillar squamous cell carcinoma: report and review of the literature. Cureus 9 (2017): e1122.
- Ramser AE, Craig EM, Delost GR, et al. Cutaneous metastasis of tonsillar carcinoma: report of a rare Case. JAAD Case Reports 4 (2018): 1-4.
- Banerjee S, Kundu D, Mukherjee M, et al. Early stage squamous cell carcinoma or the tonsil presenting with multiple organ-metastases including skin and brain after successful local treatment. JCMT 1 (2015): 31.
- Li P, Barieri A, Walther Z, et al. Epidermotropic metastasis of squamous cell carcinoma of the tonsil: A case report with molecular confirmation. J Cutan Pathol 48 (2021): 1514-1519.
- Ferlito A, Shaha RS, Silver CE, et al. Incidence and sites of distant metastases from head and neck cancer. ORL 63 (2001): 202-207.
- Huang SH, Perez-Ordonez B, Liu F, et al. Atypical clinical behaviour of p16-confirmed HPV-related oropharyngeal squamous cell carcinoma treated with radical radiotherapy. Int Radiation Oncology Biol Phys 82 (2012): 276-283.


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 