Ketobiotics by Poly-3-Hydroxybutyrate: A Novel Prebiotic Activation of Butyrate-Producing Bacteria through 3-Hydroxybutyrate Donation to the Microbiota
Takumi Satoh*
Department of Antiaging Food Research, School of Bioscience and Biotechnology, Tokyo University of Technology, 1404-1 Katakura, Hachioji 192-0982, Japan
*Corresponding author: Takumi Satoh, Department of Antiaging Food Research, School of Bioscience and Biotechnology, Tokyo University of Technology, 1404-1 Katakura, Hachioji 192-0982, Japan.
Received: 19 August 2022; Accepted: 25 August 2022; Published: 06 September 2022
Article Information
Citation: Takumi Satoh. Ketobiotics by Poly-3- Hydroxybutyrate: A Novel Prebiotic Activation of Butyrate-Producing Bacteria through 3-Hydroxybutyrate Donation to the Microbiota. Journal of Biotechnology and Biomedicine 5 (2022): 158-162.
DOI: 10.26502/jbb.2642-91280056
View / Download Pdf Share at FacebookAbstract
Since the first target of poly-3-hydroxybutyrate (PHB), a simple bioplastic, is the microbiota, it has gained attention from the public and academia because of its promising function as a prebiotic food stuff for butyrate-producing bacteria. Since mammalian digestive enzymes cannot hydrolyze PHB, it passes through the small intestine and reaches the large intestine, allowing the gut microbiota to produce 3-Hydroxybutyrate (3HB) by the depolymerization of PHB around them. As a result, they become an energy substrate that promotes the growth of butyrate-producing bacteria and induces slight acidification of the gut environment. Thus, PHBs may be used as novel prebiotics to improve the gut environment. Based on this background, studies have proposed donating this ketone body to microbiota-mediated prebiotics, termed “ketobiotics”, to act as a microbial activator of the animal gut. Ketobiotics may help maintain the gut health of pets and industrial animals, including enhancing human antiaging in the future. Therefore, this review discusses the potential use of the prebiotic action of ketobiotics for activating butyrate-producing bacteria from other prebiotic strategies, initiated by the direct donation of 3HB to microbiota in the large intestine.
Keywords
<p>Butyrate; Ketone Boy; Microbiota; Propionate; Regulatory T Cells; Short-Chained Fatty Acids</p>
Article Details
Abbreviations:
CRC: Colorectal Cancer; 3HB: 3-Hydroxybutyrate; HMGCS2: Mitochondrial 3-Hydroxy-3-Methylglutaryl-CoA Synthase; KE: Ketone Ester; PHB: Poly-3-Hydroxybutyrate; SCFAs: Short-Chain Fatty Acids; UC: Ulcerative Colitis.
Graphical Abstract:
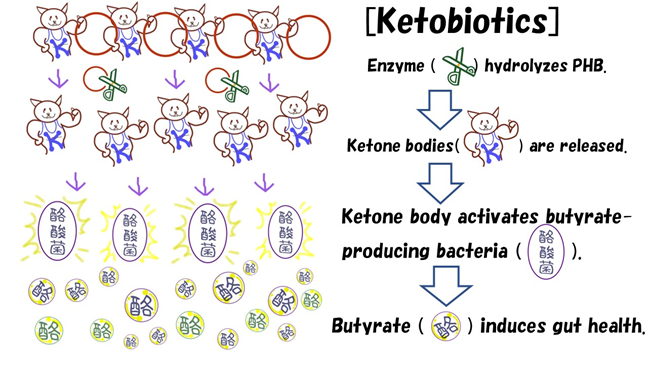
1. Poly-3-Hydroxybutyrate as a Microbial Controller in the Animal Gut
Although poly-3-hydroxybutyrate (PHB, Figure 1) are produced in the cytoplasm of some bacteria as water-insoluble inclusion bodies, they are the most well-known and understudied bioplastic. Therefore, since PHB is an intracellular energy and carbon storage compound produced by bacteria, in addition to its material features suitable for medical applications, it has been focused on as a resource for bio-implants and other medical applications [1,2]. Furthermore, PHB has gained global attention in the biological field because of its potentially new prebiotic actions, initiated by 3-hydroxybutyrate (3HB) donation to microbiota [3,4]. PHB was first reported to protect the small crustacean, Artemia franciscana, from the pathogenic actions induced by Vibrio campbellite [5,6]. Based on these reports, it is considered promising as an alternative anti-infection strategy in aquatic animals [5,6]. These protective effects may be partly through the induction of heat shock protein-70 [6]. Additionally, in the large crustacean, pacific white shrimp (Litopenaeus vannamei), PHB increases growth performance [7]. This growth enhancement may be induced by the upregulation of digestive enzymes, immune functions, and short-chain fatty acid (SCFA) contents, such as acetate, propionate, and butyrate [8]. Hence, these effects can be applied to the protection of fish. Similarly, in the European sea bus (Dicentrarchus labrax) [9], Siberian sturgeon (Acipenser baerii) [10], and soiny mullet (Liza haematocheila) [11], PHB changed the bacterial community composition and slightly reduced gut pH, beneficially affecting fish growth performance. Also, Wang et al. reported that PHB increased the growth rate of the yellow croaker, a fish. However, this effect was absent in piglets and did not affect them adversely [12]. Nevertheless, they estimated that PHB had some beneficial effects on piglets [12]. Accordingly, PHB was recently found to induce an increase in SCFAs, such as acetate, propionate, and butyrate in micro-mini pigs and slightly reduce gut PH [3], suggesting that it acts as an intestinal regulator in mammals (Figure 2). Moreover, while PHB had anti-tumor activity against rat colorectal cancer (CRC) through gut microbiota modulation [4], it also significantly reduced tumor growth associated with an increase in SCFAs (acetate, propionate, and butyrate) in the gut environment, plasma, and brain, including butyrate-producing microbiota populations [4]. The induced microbiota by PHB were including many butyrate-producing bacteria (genus) such as Clostridium, Eubacterium, Roseburia, Ruminococcus and Faecalibacterium [4]. These bacteria may produce butyrate and other SCFAs and modulate gut eveironment to excert protective effects against CRC [4]. This prebiotic effects (ketobiotics) may be protective against other inflammative and infectious deseases such as ulcerative colitis (UC). Therefore, these results indicate that PHB is a gut microbiota controller in mammals, which regulates gut health [3,4].
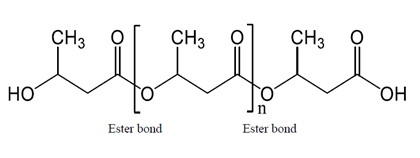
Figure 1: Chemical structure of PHB. Since PHB can be hydrolyzed by the PHB depolymerase of microbiota in the large intestine, it can increase the concentration of 3HB in an insulin spike-independent manner.
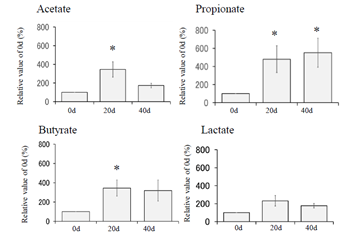
Figure 2: Increase in the concentrations of SCFAs [3]. Micro-mini pigs at 90d after birth were used for this experiment. PHB (2%) had been administered to them for 40days. Fecal samples at 0d, 20d and 40d were subjected to measuring of SCFAs (acetate, propionate, butyrate, and lactate) by use of high-performance liquid chromatography. Although acetate, propionate, and butyrate were increased in response to PHB addition, lactate was not increased. Nevertheless, pH significantly reduced from 8.2 to 7.4, suggesting that their gut environment had significantly improved.
2. Ketbiotics as a Novel Prebiotics
Some bacteria produce PHB molecules as inclusion bodies in the cytoplasm, which can be easily purified to yield a tasteless and odorless powder (purity > 85%) [13]. This powder can subsequently be used as a microbiota controller in mammals and humans (Figure 3) since, although the digestive enzymes of mammals cannot hydrolyze PHB, the microbiota in the large intestine can hydrolyze this molecule with their PHB depolymerases [1,2]. As a result, 3HB is produced around the microbiota, which can be used as an energy growth substrate [3,4]. Due to these events, microbiota growth, especially butyrate-producing bacteria, is upregulated, and microbiota diversity increases [3,4], indicating that ketobiotics by PHB can help human and animal gut health by inducing a butyrate-producing bacteria-dominant gut environment. Therefore, this research aims to improve human and animal gut environments using PHB-based ketobiotics (Figure 3). Although the pH of an ideal gut environment is slightly acidic [14,15], SCFAs released from the microbiota help regulate the mammalian gut environment to ideal conditions [3,4]. Notably, four SCFAs: acetate (C=2), propionate (C=3), lactate (C=3), and butyrate (C=4), are the main SCFA components produced by microbiota communities [14,15]. However, while many microbiota produce SCFAs, the intestinal bacteria determine which SCFA is preferentially produced and released outside the cells [14,15]. For example, in butyrate-producing bacteria-dominant gut environment, regulatory T cells are continuously activated to regulate excessive inflammation [16,17]. In these communities, butyrate activates differentiation into regulatory T cells and is directly involved in improving gut health [16,17]. A study has also reported that PHB allows microbiota to use 3HB as an energy substrate, activating butyrate-producing bacteria [3]. As a result, PHB activates the differentiation of regulatory T cells and controls immune functions [3]. Subsequently, the 3HB spreads into the gut space and is absorbed into mammals by active transportation on epithelial cells of the large intestine to increase the concentrations of 3HB in the blood [18-20]. Consequently, 3HB can function as an energy substrate or a physiologically active compound through specific receptors [20,21].
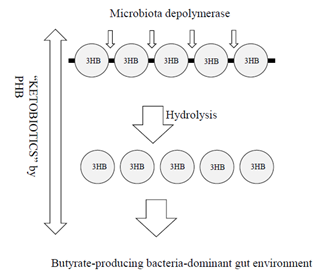
Figure 3: Basic concept of ketobiotics by PHB. PHB, a polyester compound of 3HB, can be hydrolyzed by PHB depolymerase of microbiota to release 3HB in the large intestine. In turn, 3HB activates the microbiota, allowing them to release SCFAs, such as acetate, propionate, and butyrate. In turn, 3HB is used as an energy substrate to allow the release of SCFAs, after which it is absorbed into mammals to induce various health effects.
3. Sustained 3HB Production to Maintain Gut Health
The expression of mitochondrial 3-hydroxy-3-methylglutaryl-CoA synthase (HMGCS2), involved in the synthesis of 3HB from long chain fatty acids [22,23] in the epithelial cells of digestive tracts, especially in the large intestine, has been reported as a highly critical regulator of gut health against UC [24,25], CRC [26], and intestinal stem cell homeostasis [27]. Based on this background, studies have reported that while the exogenous administration of 3HB effectively protected rodent models of UC [24], the expression of HMGCS2 significantly decreased in patients with UC [25] and CRC [26], suggesting that protein downregulation was well correlated with disease progression. Furthermore, another study also reported that the expression of HMGCS2 was required to maintain epithelial stem cells in the large intestine [27], proposing that sustained low levels of 3HB by intestinal epithelial cells expressing HMGCS2 may influence the gut health of humans [24]. Overall, since the exogenous administration of 3HB to the large intestine is considered highly effective for gut health [24], compounds that sustainedly release 3HB at low levels in the large intestine are highly required for sustaining gut health [24]. For example, a ketone donor as a molecular tool for providing 3HB to the large intestine can replace the physiological roles of HMGCS2 in the large intestine [13], of which just PHB can satisfy these physiological human gut health requirements [3].
4. PHB as a Unique Ketone Donor to Micro biota
A ketone donor is a compound that releases 3HB into the digestive tract [3,13]. Studies have reported that although insulin spikes should be suppressed by reducing carbohydrate intake in order to increase the 3HB concentrations in the body since these spikes can inhibit the synthesis of 3HB mainly through the shutdown of HMGCS2 [28,29]. In contrast, ketone donors are not affected by insulin spikes. Therefore, they do not need carbohydrate restriction [3,13]. So far, three ketone donors have been identified: 3HB, ketone ester (KE) [30,31], and PHB [3,13]. Just as PHB can induce ketobiotics since it passes through the small intestine and donates 3HB to microbiota to change its structure [3,13], 3HB and KE can also donate 3HB to mammals since they are absorbed in the small intestine and cannot reach the large intestine [20,30]. I summerized the differential actions of ketone donators in Table 1.
|
Ketone donors |
Number of ketone bodies |
Site of ketone donation |
The first target |
Mode of action |
|
3HB |
1 |
Small intestine |
Mammals |
Transient |
|
KE |
2 |
Small intestine |
Mammals |
Transient |
|
PHB |
>1000 |
Large intestine |
Microbiota |
Sustained |
Table 1: PHB as a ketone donor to microbiota. Although 3HB and KE donate ketone dody to mammals in the small intestine and never reach the large intestinge, PHB donates it to microbiota. Because of large capacity of digestion of mammalian small intestine, 3HB and KE produces transient increase in ketone bodies in the systemic circulation. In contrast, PHB can induce sustained increase in ketone bodies because of limited capacity of microbiota depplymerase [3,13].
4.1. 3HB
3HB is the most essential energy substrate in addition to glucose [20,21]. It can function directly as an energy substrate for mitochondrial oxidative metabolism. It also activates specific receptors to induce various physiological actions, improving cognitive functions, and enhancing lipid metabolism/antiaging activities [20,21]. 3HB can be orally administered, increasing several minutes after oral intake and reducing to baseline within several hours to induce a transient increase in 3HB concentrations. Subsequently, it becomes a free-acid form under slightly alkaline condition of the small intestine, after which it is absorbed into the blood through specific transporters on the epithelial cells of the small intestine [20,21].
4.2 KE
KE is an ester compound between 3HB and 1,3-butandiol [30,31]. However, this ester bond can be easily hydrolysed by a digestive enzyme (esterase) in the small intestine to produce 3HB and 1,3-butandiol in the small intestine. Subsequently, it is absorbed into mammals through transporters on the epithelial cells of the small intestine [30,31]. 1,3-butandiol is oxidized to 3HB in the liver.
4.3 PHB
Since PHB, a 3HB polyester, cannot be digested by mammalian enzymes but can be hydrolysed by enterobacterial enzymes in the large intestine [1,2], the patterns of 3HB increase in the blood differ from those of 3HB and KE (Figure 4) [3,13]. Thus, while KE can produce a sharp increase in blood 3HB levels to mM levels and returns to the baseline within several hours [3,13], 3HB is absorbed through the specific transportation of epithelial cells of the large intestine after microbial enzymes hydrolyse PHB. Therefore, PHB [3,13] needs several hours to raise the concentrations of 3HB and induce a sustained increase in 3HB. Subsequently, after PHB is absorbed into the microbiota, it serves as an energy substrate to induce various physiological effects initiated by microbiota activation [3,13], starting with the liberation of organic acids (acetate, propionate, and butyrate) [3,4]. As a result, PHB can change the microbiome’s structure, transforming it into a butyrate-producing bacteria-dominant environment, thereby protecting mammals against CRC and UC [3,4].
Figure 4: Increase in 3HB concentrations by ketone donors [3]. While KE could increase 3HB concentrations immediately after administration, then return to the basal line, PHB induces a sustained increase in 3HB concentrations.
5. Conclusions
Many reports have shown that the butyrate-producing bacteria-dominant gut environment is a key to maintaining gut health and sustaining antiaging in humans and other mammals [14-18]. Therefore, future research should focus on finding practical methodologies for inducing a butyrate-producing bacteria-dominant gut environment. Dietary fiber is one of the possible methodologies. Additionally, food customs could be another focus since dietary fiber is in daily food. In a previous study, Naito et al. (2019) also demonstrated that several compositional changes in the gut microbiota were associated with urbanization, as an increase in the butyrate-producing bacteria was observed in the rural Kyotago city, a long-lived province, suggesting that the gut environment is the key to human antiaging [32]. Furthermore, using PHB to directly donate 3HB to microbiota for the prebiotic activation of butyrate-producing bacteria has also been proposed as another method. Thus, the sustained application of low-dose PHB can change the microbiota to butyrate-producing bacteria-dominant structures in humans, pets, and industrial animals [3].
References
- Tan D, Wang Y, Tong Y, et al. Grand Challenges for Industrializing Polyhydroxyalkanoates (PHAs). Trends Biotechnol 39 (2021): 953-963.
- Tokiwa Y, Calabia BP. Degradation of microbial polyesters. Biotechnol Lett 26 (2004): 1181-1189.
- Satoh T. Propionate acid releaser, acetic acid releaser, growth promoter and intestinal functional regulator (2022).
- Fernández J, Saettone P, Franchini MC, et al. Antitumor bioactivity and gut microbiota modulation of polyhydroxybutyrate (PHB) in a rat animal model for colorectal cancer. Int J Biol Macromol 203 (2022): 638-649.
- Defoirdt T, Halet D, Vervaeren H, et al. The bacterial storage compound poly-beta-hydroxybutyrate protects Artemia franciscana from pathogenic Vibrio campbellii. Environ Microbiol 9 (2007): 445-452.
- Baruah K, Huy TT, Norouzitallab P, et al. Probing the protective mechanism of poly-ß-hydroxybutyrate against vibriosis by using gnotobiotic Artemia franciscana and Vibrio campbellii as host-pathogen model. Sci Rep 5 (2015): 9427.
- Duan Y, Zhang Y, Dong H, et al. Effects of dietary poly-β-hydroxybutyrate (PHB) on microbiota composition and the mTOR signaling pathway in the intestines of litopenaeus vannamei. J Microbiol 55 (2017): 946-954.
- Duan Y, Zhang Y, Dong H, et al. Effect of dietary poly-β-hydroxybutyrate (PHB) on growth performance, intestinal health status and body composition of Pacific white shrimp Litopenaeus vannamei (Boone, 1931). Fish Shellfish Immunol 60 (2017): 520-528.8.
- De Schryver P, Sinha AK, Kunwar PS, et al. Poly-beta-hydroxybutyrate (PHB) increases growth performance and intestinal bacterial range-weighted richness in juvenile European sea bass, Dicentrarchus labrax. Appl Microbiol Biotechnol 86 (2010): 1535-1541.
- Najdegerami EH, Tran TN, Defoirdt T, et al. Effects of poly-β-hydroxybutyrate (PHB) on Siberian sturgeon (Acipenser baerii) fingerlings performance and its gastrointestinal tract microbial community. FEMS Microbiol Ecol 79 (2012): 25-33.
- Qiao G, Xu C, Sun Q, et al. Effects of dietary poly-β-hydroxybutyrate supplementation on the growth, immune response and intestinal microbiota of soiny mullet (Liza haematocheila). Fish Shellfish Immunol 91 (2019): 251-263.
- Wang X, Jiang XR, Wu F, et al. Microbial Poly-3-Hydroxybutyrate (PHB) as a Feed Additive for Fishes and Piglets. Biotechnol J 14 (2019): e1900132.
- Satoh T. Blood glucose spike suppressor, food product, and method for producing blood glucose spike suppressor (2019).
- Louis P, Flint HJ. Formation of propionate and butyrate by the human colonic microbiota. Environ Microbiol 19 (2017): 29-41.
- Furusawa Y, Obata Y, Fukuda S, et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 504 (2013): 446-450.
- Farkas AM, Panea C, Goto Y, et al. Induction of Th17 cells by segmented filamentous bacteria in the murine intestine. J Immunol Methods 421 (2015): 104-111.
- Sato Y, Atarashi K, Plichta DR, et al. Novel bile acid biosynthetic pathways are enriched in the microbiome of centenarians. Nature 599 (2012): 458-464.
- Stilling RM, van de Wouw M, Clarke G, et al. The neuropharmacology of butyrate: The bread and butter of the microbiota-gut-brain axis? Neurochem Int 99 (2016): 110-132.
- Sikder K, Shukla SK, Patel N, et al. High Fat Diet Upregulates Fatty Acid Oxidation and Ketogenesis via Intervention of PPAR-γ. Cell Physiol Biochem 48 (2018): 1317-1331.
- Veech RL. The therapeutic implications of ketone bodies: the effects of ketone bodies in pathological conditions: ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins Leukot Essent Fatty Acids 70 (2004): 309-319.
- Newman JC, Verdin E. Ketone bodies as signaling metabolites. Trends Endocrinol Metab 25 (2014): 42-52.
- Hegardt FG. Mitochondrial 3-hydroxy-3-methylglutaryl-CoA synthase: a control enzyme in ketogenesis. Biochem J 338 (1999): 569-582.
- Fukao T, Mitchell G, Sass JO, et al. Ketone body metabolism and its defects. J Inherit Metab Dis 37 (2014): 541-551.
- Huang C, Wang J, Liu H, et al. Ketone body β-hydroxybutyrate ameliorates colitis by promoting M2 macrophage polarization through the STAT6-dependent signaling pathway. BMC Med 20 (2022): 148.
- Zou K, Hu Y, Li M, et al. Potential Role of HMGCS2 in Tumor Angiogenesis in Colorectal Cancer and Its Potential Use as a Diagnostic Marker. Can J Gastroenterol Hepatol 2019 (2019): 8348967.
- Helenius TO, Misiorek JO, Nyström JH, et al. Keratin 8 absence down-regulates colonocyte HMGCS2 and modulates colonic ketogenesis and energy metabolism. Mol Biol Cell 26 (2015): 2298-2310.
- Cheng CW, Biton M, Haber AL, et al. Ketone Body Signaling Mediates Intestinal Stem Cell Homeostasis and Adaptation to Diet. Cell 178 (2019): 1115-1131.e15.
- Nakamura MT, Yudell BE, Loor JJ. Regulation of energy metabolism by long-chain fatty acids. Prog Lipid Res 53 (2014): 124-144.
- Rescigno T, Capasso A, Tecce MF. Involvement of nutrients and nutritional mediators in mitochondrial 3-hydroxy-3-methylglutaryl-CoA synthase gene expression. J Cell Physiol 233 (2018): 3306-3314.
- Cox PJ, Kirk T, Ashmore T, et al. Nutritional Ketosis Alters Fuel Preference and Thereby Endurance Performance in Athletes. Cell Metab 24 (2016): 256-268.
- Monzo L, Sedlacek K, Hromanikova K, et al. Myocardial ketone body utilization in patients with heart failure: The impact of oral ketone ester. Metabolism 115 (2021): 154452.
- Naito Y, Takagi T, Inoue R, et al. Gut microbiota differences in elderly subjects between rural city Kyotango and urban city Kyoto: an age-gender-matched study. J Clin Biochem Nutr 65 (2019): 125-131.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 