Mammary SLAMF3 Regulates Store-Operated Ca2+ Entry and Migration Through STIM1 in Breast Cancer Cells and Cell Lines
Grégory Fouquet1, Constance Marié1#, Mehdi Badaoui2#, Baptiste Demey1#, Sylviya Radoslavova2, Marie-Sophie Telliez2, Isabelle Dhennin-Duthille2, Jagadeesh Bayry3, Henri Sevestre2,4, Halima Ouadid-Ahidouch2*, Ingrid Marcq1*, Hicham Bouhlal1*
#Authors contributed equally
1INSERM 1247-GRAP, Centre Universitaire de Recherche en Santé CURS, Université de Picardie Jules Verne, CHU Sud, Amiens, France
2Laboratoire de Physiologie Cellulaire et Moléculaire, LPCM UR-4667-UPJV, Université de Picardie Jules Verne, UFR Sciences, 80000, Amiens, France
3INSERM, Centre de Recherche des Cordeliers-Equipe Immunopathologie et immune-intervention thérapeutique, Sorbonne Université, Université de Paris, Paris, France
4Service Anatomie pathologie, Centre Hospitalier Universitaire Nord, Amiens, France
*Corresponding Authors: Dr. Hicham Bouhlal, INSERM 1247-GRAP, Centre Universitaire de Recherche en Santé CURS, Université de Picardie Jules Verne, CHU Sud, Amiens, France
Dr. Ingrid Marcq, INSERM 1247-GRAP, Centre Universitaire de Recherche en Santé CURS, Université de Picardie Jules Verne, CHU Sud, Amiens, France
Received: 02 July 2020; Accepted: 03 August 2020; Published: 09 October 2020
Article Information
Citation: Grégory Fouquet, Constance Marié, Mehdi Badaoui, Baptiste Demey, Sylviya Radoslavova, Marie-Sophie Telliez, Isabelle Dhennin-Duthille, Jagadeesh Bayry, Henri Sevestre, Halima Ouadid-Ahidouch, Ingrid Marcq, Hicham Bouhlal. Mammary SLAMF3 Regulates Store-Operated Ca2+ Entry and Migration Through STIM1 in Breast Cancer Cells and Cell Lines. Journal of Cancer Science and Clinical Therapeutics 4 (2020): 462-479.
View / Download Pdf Share at FacebookArticle Details
Abstract
Store Operated Calcium Entry (SOCE) is the main route for calcium entry in breast cells. After it’s activation by STromal Interaction Molecule (STIM) during endoplasmic reticulum store depletion, membrane channels ORAI are the main actors of this cell calcium entry. STIM, ORAI and SOCE alterations might contribute to Breast Cancer (BC) carcinogenesis. Recently, we reported the tumor suppressor role of Signaling Lymphocytic Activation Molecule Family member 3 (SLAMF3) on HepatoCellular Carcinoma (HCC) progression. SLAMF3 has been shown to regulate the activity of immune cells by modulating the calcium influx. In this report, we aimed at exploring the role of SLAMF3 in regulating SOCE and migration of BC cells. We quantified and compared the expression of SLAMF3 and STIM1 by quantitative RT-PCR in tumor and healthy resections of 14 patients followed at the University Hospital of Amiens. The expressions of SLAMF3 and STIM1 were also quantified and compared in non-invasive T47D and invasive MDA-MB-231 cell lines by quantitative RT-PCR, Western blot and flow cytometry. We determined the Ca2+ basal entry as well as SOCE by Mn2+ quenching and calcium imaging, respectively, in T47D and MDA-MB-231 cells overexpressing SLAMF3 ectopically. The cell proliferation and migration/invasion were investigated by MTT, wound healing assay and Boyden chambers tests, respectively. First, we report the expression of SLAMF3 in mammary epithelial cells. We highlight the complete loss of SLAMF3 expression in invasive BC cell lines compared to non-invasive cells. In addition, we show that the forced expression of SLAMF3 in invasive cells down-regulate specifically the STIM1 expression in invasive compared to non-invasive mammary cell lines. Interestingly, an inverse correlation is observed between the low expression of SLAMF3 and the high expression of STIM1 in primary human BC tissues. Our results indicate that SLAMF3 reduces SOCE and therefore restricts BC cell migration by decreasing STIM1 expression. Therefore, SLAMF3 might be used as a predictive marker of BC evolution and aggressiveness.
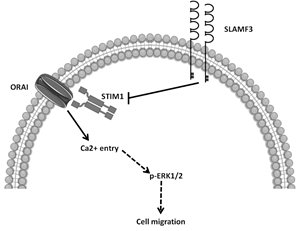
Proposed model of SLAMF3 implication in the regulation of Ca2+ entry through its action on STIM1/ORAI pathway. The STIM1-dependent action of SLAMF3 decreases SOCE, controls ERK1/2 expression and activation, and BC cell migration.
Keywords:
SLAMF3; Breast cancer; Calcium influx; SOCE; STIM1; Migration; Aggressiveness marker
Abbreviation:
SLAMF3: Signaling Lymphocytic Activation Molecules Family 3; STIM1: STromal Interaction Molecule; SOCE: Store Operated Calcium Entry; BC: Breast Cancer; ERK1/2: Extracellular signal-Regulated Kinases ½; HCC: HepatoCellular Carcinoma; RTqPCR: Real Time quantitative Polymerase Chain Reaction; 5 mRNA: messenger RiboNucleic Acid; WB: Western Blot; MTT: Methylthiazolyldiphenyl-tetrazolium bromide; mTOR: mammalian Target Of Rapamycin; RB: RetinoBlastoma; PLK-1: Polo-Like Kinase-1; IFN-γ: InterFeroN-γ; NK: Natural Killer; EAT-2: Ewing's sarcoma-associated transcript 2; MAPK: Mitogen-Activated Protein Kinase
1. Introduction
Breast cancer (BC) is one of the most common malignancies, which caused almost 627.000 deaths worldwide in 2018 [1]. Despite advancement in the treatment options, this disease remains lethal, especially when it turns invasive. Eight our of ten BC cases are diagnosed as invasive and presence of metastasis reduces the overall survival [2]. Understanding the regulatory mechanisms and main actors implicated in the pathogenesis of BC is essential to develop new targeted therapies, predictive or diagnostic markers, and optimized patient care. Over the past years, ion channels have emerged as important players in cell proliferation, survival and migration of BC cells [3]. Calcium (Ca2+) is one of the major second messengers and its cytoplasmic concentration ([Ca2+]i) regulates many cell properties [4] including metastatic mechanisms [5]. In unexcitable cells including mammary cells, store-operated calcium entry (SOCE) is the main way of calcium entry. During calcium store depletion of endoplasmic reticulum in BC cells, SOCE is mediated by the Ca2+ sensor STIM1 (STromal Interaction Molecule 1), which is localized on reticular membrane [6-8], and the pore-forming proteins ORAI1/3 (Ca2+ channels) situated on the plasma membrane [9-12]. STIM1 is known to promote Ca2+-dependent proliferation [13-17] and migration [17-20] in many cancers including BC.
We have recently identified Signaling Lymphocytic Activation Molecule (SLAM) family member, the SLAMF3 receptor, as a key factor, which controls the proliferation and apoptosis of cancer cells, and the progression of tumor masses in liver tissue [21]. We have also reported the loss of SLAMF3 expression in HepatoCellular Carcinoma (HCC) cells, and that induction of SLAMF3 expression inhibited the proliferation and blocked the cell cycle at the G2/M stage by suppressing the MAPK ERK1/2/JNK, mTOR and RB/PLK-1 pathways [21, 22]. SLAMF3 is also expressed in immune cells [23] where it triggers both activatory and inhibitory signals [24]. Indeed, in CD3-activated T cells, SLAMF3 activation decreases the phosphorylation of ERK1/2 and production of IFN-γ [25]. In 3 of 17 parallel, defects in T cell activation and IL-2 production in Ly9 (SLAMF-3)-deficient mice (Ly9-/-) suggests a crucial role of Ly9 in T cell functions [26]. Besides, SLAM family members are also involved in the regulation of immune response through the regulation of Ca2+ influx. In Natural Killer cells (NK), SLAM, via Ewing's sarcoma-associated transcript 2 (EAT-2), a SLAM intracellular adaptor protein, induces activation of MAPK ERK1/2 by recruitment of PLC-γ that evokes Ca2+ influxes [27].
In the present work, we investigated the link between SLAMF3 expression and SOCE modulation in BC. We show a low expression of SLAMF3 in breast tumor tissues compared to the healthy ones. Moreover, in BC cell lines, we highlighted the specific regulatory effect of SLAMF3 on the expression of STIM1 and on SOCE. In addition, high expression of SLAMF3 reduced the migratory potential of invasive cell lines, which is Ca2+-dependent. Taken together, our results provide evidence on the implication of SLAMF3 receptor in the regulation of SOCE through STIM1 pathway in mammary tissue. We also propose high expression of SLAMF3 in cancerous tissue as a therapeutic strategy to control the progression and invasiveness of cancerous mass.
2. Results
2.1. Inverse correlation between SLAMF3 and STIM1 expression in human breast cancer tissues
We have previously described that in HCC tissues [21], the expression of SLAMF3 is lost in tumor tissues compared to peri-tumor tissues. Here, we observed a significantly reduced expression of SLAMF3 mRNA in eight of fourteen (60%) breast tumor tissues compared to peri-tumor tissues (n=14; p<0.01; Figure 1A). Moreover, STIM1 expression was not significantly modified in tumor tissues (n=14; Figure 1B). We highlight a weak but significant, inverse correlation between SLAMF3 and STIM1 expression (n=14; R2=0.356; p<0.05; Figure 1C). Given the limited number of patients, SLAMF3 and STIM1 expression did not reach significance when patients were grouped according to either tumor grade or metastasis.
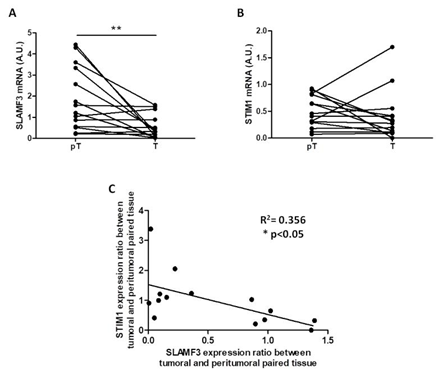
Figure 1: Inverse correlation between SLAMF3 and STIM1 expression in primary human breast cancer tissue samples. SLAMF3 (A) and STIM1 (B) mRNA expressions were quantified by quantitative RT-PCR in same extracts of resected breast cancer tissues. The mRNA expression was compared between peritumor (pT) and tumor (T) tissues (n=14; **p<0.01, ns: non-significant). (C) The correlation curve between SLAMF3 and STIM1 expression (R2=0.356; *p<0.05).
2.2. SLAMF3 is expressed in human breast epithelial cells
The results obtained with breast tumor tissues prompted us to confirm the expression of SLAMF3 in BC cell lines. Until now, SLAMF3 is known to be expressed in lymphocytes [23] and in hepatocytes [21] but not in epithelial breast cells. We investigated SLAMF3 expression in human breast epithelial cancer cell lines by flow cytometry and western blot. In cancerous cells, the cell surface phenotype analysis showed that SLAMF3 is weakly expressed on the cell surface (7.64 ± 1.80% and 0.92 ± 0.07% on non-invasive T47D and invasive MDA-MB-231 triple negative cells, respectively; Figure 2A). Despite this low expression, SLAMF3 expression appears to be significantly lower in invasive human BC cell lines (MDA-MB-231) compared to non-invasive cell lines (T47D) (Figure 2B, N=3, p<0.01). Western blot analysis confirmed the expression of SLAMF3 in human breast epithelial cells. We found a lower SLAMF3 expression in invasive MDA-MB-231 cells (Figure 2C). Indeed, semi-quantitative analysis of proteins showed a reduction of SLAMF3 protein level by 37 ± 7.8% in MDA-MB-231 compared to non-invasive T47D cells (Figure 2D, N=3; p<0.01).
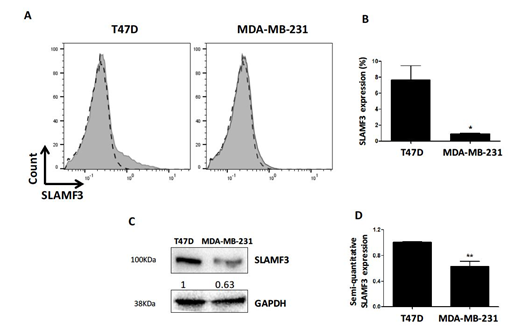
Figure 2: Expression of SLAMF3 in human breast epithelial cells. (A) Analysis of SLAMF3 expression by flow cytometry in T47D and MDA-MB-231 human BC cell lines. SLAMF3 staining (in grey) is overlaid by the negative control (in dashed lines). Representative of three independent experiments. SLAMF3 expression (mean ± SEM) as percent positive cells (%). (N=3; *p<0.05). (C) Western blot analysis of proteins extracted from T47D and MDA-MB-231 cells stained with specific antibody directed against SLAMF3 (mAb K12) or an anti-GAPDH antibody. Representative blots of three independent experiments. The density as measured by semi-quantitative analysis of bands is also presented. (D) SLAMF3 expression (mean ± SEM) from three independent experiments (N=3; **p<0.01).
2.3. SLAMF3 decreases basal Ca2+ influx only in invasive breast cancer cells
Previous studies have demonstrated that SLAMF3 is a regulator of Ca2+ entry in immune cells [27]. Moreover, Ca2+ homeostasis has been shown to regulate various cellular processes [4] and in particular, metastatic mechanisms [5]. Identification of SLAMF3 expression by human BC cells encouraged us to investigate whether SLAMF3 regulates Ca2+ influx in human BC cells. We determined the Ca2+ basal entry by Mn2+ quenching with Fura Red in both T47D and MDA-MB-231cell lines overexpressing SLAMF3 ectopically. First, we confirmed that forced expression of SLAMF3 resulted in higher level of SLAMF3 mRNA and protein than mock plasmid in T47D (increase by 29 ± 1375.508 fold) and MDA-MB-231 cells (increase by 43.733 ± 15.102 fold) (Figure 3A-B-C). We found that the ectopic SLAMF3 expression reduced Mn2+ quenching slope significantly from -0.3345 ± 0.0299 to -0.1665 ± 0.0213 (n=74 and 81 cells respectively; p<0.001) only in invasive MDA-MB-231 cells (Figure 3E). In contrast, SLAMF3 overexpression failed to modify Mn2+ slope quenching in non-invasive T47D cells (from -0.3914 ± 0.0355 to -0.4059 ± 0.0387; n=97 and 118 cells respectively; p>0.05; Figure 3D. Our data suggest that SLAMF3 controls basal Ca2+ influx only in invasive breast cancer cells.
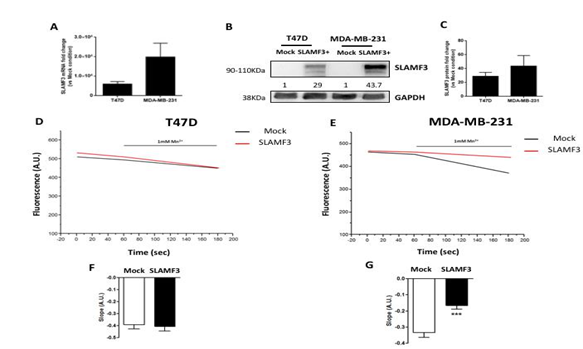
Figure 3: Effect of SLAMF3 overexpression on basal Ca2+ entry in human breast epithelial cells. (A-C) Confirmation of induction of SLAMF3 overexpression by quantitative RT-PCR (A) and Western blot (B-C), in T47D and MDA-MB-231 cell lines. For quantitative RT-PCR experiments, results (mean ± SEM) from three independent experiments are presented. For Western blot, representative of two independent experiments (B), and fold changes (mean ± SEM, N=2 independent experiments) in SLAMF3 expression (C) are presented (Mock: free-plasmid transfected cells; SLAMF3+: SLAMF3- plasmid transfected cells, GAPDH: protein loading control). (D-G) Mean traces and quantification of Mn2+ quenching in T47D (D, F) and MDA-MB-231 (E, G) cells transfected with SLAMF3 plasmid (SLAMF3: n=97; n=74, respectively) or free plasmid (Mock; n=118; n=81, respectively). Representative data (D-E) and mean ± SEM (F-G) values from three independent experiments are presented. *** p<0.001.
2.4. SLAMF3 decreases the cell motility but not cell proliferation only in MDA-MB-231 cells
Ion channels and in particular Ca2+ are known to be altered in numerous cancers and play an essential role in cell proliferation and migration [29]. Moreover, SLAMF3 has been demonstrated as an inhibitor of cell proliferation and migration in HCC [21, 22]. Interestingly, SLAMF3 overexpression significantly reduced the cell motility (Figure 4A). Indeed, wound-healing area is larger in SLAMF3-over expressed condition at 24 hours (from 69.54 ± 3.84% to 82.85 ± 2.93% in Mock and SLAMF3 conditions, respectively) and at 48 hours (from 33.17 ± 7.25% to 56.36 ± 3.69% respectively; N=3; p<0.05; Figure 4B). Migration assays were performed by using transwell system 48 hours post transfection of SLAMF3 and confirmed the lower cell motility of MDA-MB-231 cells overexpressing SLAMF3 (Figure 4C). In fact, migrating cell number was significantly decreased by 44.91 ± 5.77% in SLAMF3 overexpressing conditions (N=3; p<0.001) compared to mock (Figure 4D). As increased cell migration could be also attributed to proliferative capacity of cells, transfected cells were subjected to colorimetric MTT test assay. MTT assay confirmed that cell proliferation was similar in both the conditions 48 hours post transfection of SLAMF3 (Figure 4E), as well as at 24, 72 and 96 hours (supplementary figure). Our results thus suggest that SLAMF3 represses cell migration in invasive BC cells without affecting cell proliferation.
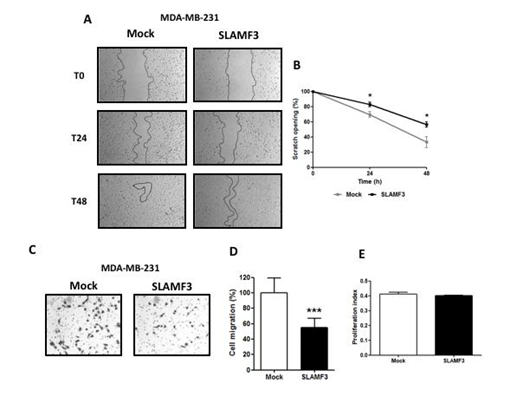
Figure 4: SLAMF3 decreases MDA-MB-231 cell motility but not proliferation. Migratory capacity of SLAMF3 overexpressing MDA-MB-231 cells compared to Mock transfected cells in wound healing (A and B) and transwell migration assays (C and D): (A) photographs of same areas performed at 0 (T0), 24 and 48 hours; (B) The migratory index corresponding to the normalization of the area scratched. Values are presented as percentage compared to T0 and presented as the mean ± SEM (N=3; *p<0.05); (C) Transwell migration assays were performed in MDA-MB-231 cells 48 hours post transfection of SLAMF3. Photographs of one of the three experiments (C) and (D) quantification (mean ± SEM) of cell migration (n=180; N=3; ***p<0.001). (E) No effect of seeding was observed on cell proliferation as analyzed by MTT test in MDA-MB-231 cells transfected with SLAMF3 plasmid (SLAMF3) or free plasmid (Mock), N=3. Associated Supplementary figure: analysis of cell proliferation by MTT assays in MDA-MB-231 cells at 24, 48, 72 and 96 hours post transfection with SLAMF3 plasmid (SLAMF3) or free plasmid (Mock) Results presented as the mean ± SEM (N=3; ns: p>0.05).
2.5. SLAMF3 decreases store-operated Ca2+ entry through STIM1 expression in invasive MDA-MB-231breast cancer cells
We demonstrated that the ectopic expression of SLAMF3 reduces basal Ca2+ entry and cell migration in invasive BC cells. ORAI1 and STIM1 regulate store-operated Ca2+ entry and are essential for BC cell migration and metastasis [20], while ORAI3 and STIM1 are essential for non-invasive BC cell proliferation [30]. First, we determined the effect of SLAMF3 overexpression on STIM1 expression by quantitative RT-PCR in non-invasive T47D and in invasive MDA-MB-231 cell lines, respectively (Figure 5A-B). We observed that overexpression of SLAMF3 led to significant decrease in STIM1 expression (by 48 ± 14.5%) only in MDA-MB-231 (N=5; p<0.05) but not in T47D cells (STIM1 expression changed from 1 ± 0,252 % to 1.204 ± 0.248 %; N=5; NS). Moreover, neither ORAI1 nor ORAI3 expression was modified in our conditions in T47D cells (N=5; p>0.05; Figure 5C) or MDA- MB-231 cells (N=5; p>0.05; Figure 5D). These observations prompted us to quantify the protein level of STIM1 by western blot. The overexpression of SLAMF3 decreased STIM1 proteins level by 37.25 ± 9.55% (N=4; p<0.05; Figure 5E). We then recorded SOCE in invasive MDA-MB-231 cells 48 hours post transfection with SLAMF3 plasmid by using Ca2+ imaging assays. We exposed cells to thapsigargin (1 µM) in a Ca2+-free solution to deplete intracellular Ca2+ stores and activated store-operated Ca2+ channels. SOCE-dependent influx was then measured in the presence of 10 mM extracellular Ca2+. We observed a reduction in SOCE (31.03 ± 6.86% reduction) in SLAMF3 overexpressed cells compared to Mock ones (SOCE changed from 15.18 ± 0.66 % to 10.47 ± 1.04 %; n=40 and n=39 respectively; N=3; p<0.001; Figure 5F-G).
SLAMF3 activation negatively controls MAPK ERK 1/2 and JNK activation as reported in HCC [21]. Constitutive activity of MAPK has been reported in aggressive triple-negative BC. To explore the possible link between the SLAMF3 over-expression, SOCE and MAPK pathway, we analyzed ERK1/2 expression and activity in SLAMF3 over-expressing invasive MDA-MB-231 cell lines. We found that SLAMF3 decreased the expression (reduction up to 18±3%, p<0.05) and phosphorylation of ERK1/2 (reduction up to 24 ± 1,76%; N=3; p<0.05; Figure 5 H-I).
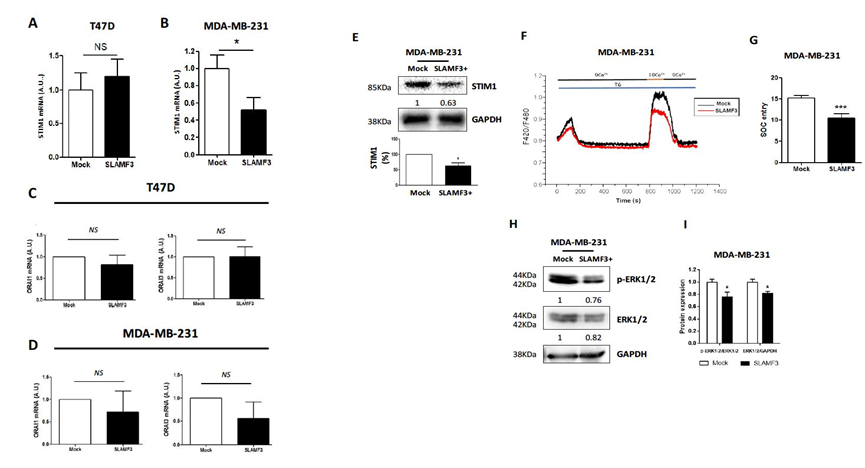
Figure 5: SLAMF3 decreases store-operated Ca2+ entry (SOCE) through STIM1 expression in invasive MDA-MB-231 breast cancer cells. (A-B) Effect of SLAMF3 overexpression on STIM1 expression as analyzed by quantitative RT-PCR in T47D (A) and MDA-MB-231 (B) cells transfected with SLAMF3 plasmid (SLAMF3) or free plasmid (Mock). The results are presented as the mean ± SEM from five independent experiments (N=5; ns: p>0.05 for T47D and *p<0.05 for MDA-MB-231). (C-D) Expression of ORAI1 and ORAI3 mRNA in T47D (C) and MDA-MB-231 (D) cells transfected with SLAMF3 plasmid (SLAMF3) or free plasmid (Mock) (mean ± SEM, N=5, ns: p>0.05). (E) Western blot analysis of STIM1 in protein extracts from MDA-MB-231 cells transfected with SLAMF3 plasmid (SLAMF3+) or free plasmid (Mock). GAPDH was used as a protein loading control. Representative blot and % reduction in STIM1 expression (mean ± SEM, N=4 independent experiments, *p<0.05). (F) Mean traces of SOCE in SLAMF3-transfected cells (SLAMF3) or free plasmid (Mock) transfected cells and (G) quantification of the Ca2+-bound Fura Red fluorescence showing thapsigargin-activated (TG) SOCE in MDA-MB-231 cells transfected with SLAMF3 plasmid (SLAMF3; n=39 analyzed cells) or free plasmid (Mock; n=40 analyzed cells) in three independent experiment (N=3; *** p<0.001). (H) Western blot and semi-quantitative, analysis of protein extracts from MDA-MB-231 cells transfected with SLAMF3 plasmid (SLAMF3+) or free plasmid (Mock). Membranes were probed with mAbs against ERK1/2 and phospho-ERK1/2. GAPDH was used as a protein loading control. Data from one of three independent experiments is presented. (I) The calculated ratio of phospho-ERK1/2/ERK1/2 and ERK1/2/GAPDH (mean ± SEM, N=3 independent experiments, *p<0.05).
3. Discussion
Recently, we identified the expression of SLAMF3 in hepatocytes. We highlighted its implication 236 in controlling the proliferation, apoptosis and migration of HCC [21]. The re-introduced expression of SLAMF3 in HCC cells reduced the proliferative index and motility, induced apoptosis by a caspases-dependent manner and blocked the cell cycle [21, 22]. These effects were also confirmed in vivo in mice where neither tumor appearance nor tumor progression were observed when HCC cells overexpressing SLAMF3 were xenografted. The potential tumor suppressive role of SLAMF3 obtained in HCC prompted us to check the implication of this receptor in other solid tumors. In the present pilot study realized on 14 BC patients, we evidenced, for the first time, the expression of SLAMF3 in epithelial BC cells.
BC cells originate as epithelial cells to form the primary tumor, which might acquire cellular motility and form a more invasive secondary tumor [31]. Several mechanisms and factors have been reported to explain the invasiveness behavior of cancerous cells. In the present work, we show that SLAMF3 is expressed at very low level at cancerous cell surface compared to the total SLAMF3 protein at intracellular level. This result suggests possible regulatory factors in cancerous cells that prevent the membrane expression of SLAMF3 and its tumor suppressive effect. This mechanism might explain, in part, the immune escape behavior of cancerous cells against the processes that control cell proliferation and apoptosis. In addition, the baseline expression of SLAMF3 is different in invasive and non-invasive BC cells. SLAMF3 expression appears to be significantly lower in invasive MDA-MB-231 cells compared to non-invasive T47D cells. It has been reported that SLAMF3 activation through homophilic interactions controls negatively MAPK ERK 1/2 and JNK activation [21]. The MAPK Ras-ERK pathway is dysregulated in approximately one third of human cancers, particularly those of epithelial origin. In aggressive triple-negative BC, most tumors display increased constitutive phosphorylation of MEK1/2 and ERK1/2 [32]. Taken together, our results suggest that reduced membrane expression of SLAMF3 in cancerous breast cells might prevent SLAMF3 homophilic interactions and its ability to control MAPK pathway. In this sense, we checked the ERK1/2 activation state in SLAMF3 overexpressed BC cells. Ectopic introduction of high SLAMF3 expression decreased ERK protein expression and its activation. Considering the fact that STIM1 regulates Ca2+ entry in mammary cells, our results highlight a new pathway by which SLAMF3 controls ERK1/2 activation by controlling SOCE (see graphical abstract).
During carcinogenesis, Ca2+ signaling in some malignant cells is significantly remodeled in a way that compromises normal physiological functions. Various studies strongly suggest the important role of Ca2+ remodeling in carcinogenesis process [33]. Remodeling of Ca2+ signaling is the prime reason for such a transformation in cell behavior and is caused by genetic as well as epigenetic factors [4]. In fact, in cancerous cells, Ca2+ remodeling promotes activation of Ca2+-dependent transcription factors such as the Nuclear Factor of Activated T cells (NFAT), c-myc, c-jun and c-fos. All these factors promote hypertrophic growth via induction of G1 and G1/S transition by cyclins (D and E) associated to cyclin-dependent kinases (CDK2 and CDK4) [34]. In addition, this remodeling is implicated in conferring apoptosis resistance to cancerous cells and in promoting cell migration and metastasis. In breast cells, SOCE is mediated by the Ca2+ sensor STIM1 [6–8] and the pore-forming proteins ORAI1/3 [9–12]. SOCE and its STIM/ORAI constituents have emerged as important regulators of malignant cell migration. Enhanced STIM1-ORAI1-mediated SOCE promotes higher rate of focal adhesion turnover and fast migration of metastatic BC cells via activation of GTPases Ras and Rac [20]. Here, we provide evidence on the negative regulation of SLAMF3 towards STIM1 expression. On one hand, it is further demonstrated that metastasis potency of MDA-MB231 is drastically reduced upon STIM1 or ORAI1 knockdown [35]. On the other hand, we show that overexpression of SLAMF3 decreases the STIM1 expression selectively in invasive MDA-MB-231 cell line. Taken together, our results strongly support the potential role of SLAMF3 expression in determining the metastatic behavior of BC.
Our results suggest that SLAMF3 controls more efficiently the motility of invasive cells by reducing the STIM1 expression, which is highly expressed in cancerous cells. However, the effect of SLAMF3 on SOCE is weak in non-invasive cells compared to its effect in invasive cells. This result strongly suggests that SLAMF3 might regulate the Ca2+ remodeling by controlling the STIM1 expression.
Metastatic dissemination from the primary tumor site to multiple tissues is the main cause of mortality in cancer. Tumor cells become invasive by acquiring high migratory potential along with the increased ability to degrade ExtraCellular Matrix (ECM). Ca2+ signaling is critical for regulating the cell migration and invasion. In malignant cells Ca2+ signaling is remodeled in such a way that it promotes the turnover of focal adhesions, enhances contractile forces and facilitates proteolysis of ECM components [36]. In our work, we report that high expression of SLAMF3 decreases the motility of the invasive cells and slow down the repopulation of empty spaces in the performed wound test. Similarly, this expression modulates the cell migration through the trans-well pores and confers invasive cells a decreased ability to migrate. However, the links between the expression and function of certain Ca2+ permeable channels, and cancer cell migration, invasion and metastasis remain unclear [37, 38].
The SOC constituents, STIM1 and ORAI1, are reported to be implicated in the enhanced migration of BC cells [19, 34]. In addition to ORAI channels, it has been reported that high pro- migratory and pro-invasion potentials have been also ascribed to TRPV1, TRPV2, TRPV6 and TRPM8 channels. Higher expression of TRPV2 in prostate cancer cells or TRPM8 in squamous cell carcinoma correlated with the induction of MMP-2, MMP-9 and cathepsin B [39, 40]. Additional analysis is needed to explore the effect of SLAMF3 expression on the expression of metalloproteases and their enzymatic activity, which might explain in part modulatory effect of SLAMF3 on the migratory and motility behavior of BC cells.
Altogether, our results describe, for the first time, the expression of the SLAMF3 in mammary tissues and its potential effect on STIM1/SOCE-dependent Ca2+ influx and suggests its implication in the regulation of motility and migration in BC cells. More importantly, a pilot study based on resection samples from 14 women with BC was realized by quantifying SLAMF3 and STIM1 mRNA expression. Our results evidenced potential inverse correlation between the expression of SLAMF3 and STIM1 in the same tissue sample. This result suggests reciprocal regulation of SLAMF3 and STIM1 expression, which promotes cancer cell escape, apoptosis resistance, aggressive behavior and metastasis propagation. Additional experiments are required to identify the molecular actors implicated in this negative regulation. Moreover, the present study must be complemented with a bigger cohort of patients with different stages of BC and thorough analyses of role of SLAMF3-STIM1 according to the cancer stage, treatment and global survival. The present work also suggests that the SLAMF3 loss, combined with the acquired high expression of STIM1 in epithelial mammary cells could be used as a potential diagnosis marker for tumor transformation. Moreover, restoration of high membrane expression of SLAMF3 in cancerous cells, which controls the Ca2+ influx and prevents, in part, the metastatic cell migration could be exploited for the anti-cancer therapy.
4. Material and Methods
4.1. Cell lines
Human breast epithelial cancer cell lines T47D and MDA-MB-231 were obtained from ATCC numbers HTB-133 and HTB-26 respectively. T47D and MDA-MB-231 BC cells were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% Fetal Calf Serum (FCS), 2 mM of L-Glutamine and 100 µg/mL of penicillin/ streptomycin (EUROBIO, Les Ulis, France). Cells were grown at 37°C in a humidified incubator with 5% CO2.
4.2. Patient samples
Fourteen (n=14) pairs of tumor (T) tissues and matched peritumor (pT) tissues were obtained from BC patients undergoing surgical resection at Amiens University Hospital (Amiens, France). Our protocol was approved by the local independent ethics committee (Comité de Protection des Personnes (CPP) Nord-Ouest, Amiens, France) (Table 1). Patients were provided with information on the study procedures and objectives, and all gave their written consent for the participation. Total mRNA was extracted using specific kits and used to test SLAMF3 and STIM1 expression.
|
Patients |
Age |
Metastasis |
Infiltration |
Grade |
|
1 |
41 |
Y |
Y |
II |
|
2 |
41 |
N |
Y |
III |
|
3 |
70 |
Y |
Y |
III |
|
4 |
49 |
Y |
Y |
I |
|
5 |
50 |
Y |
Y |
II |
|
6 |
50 |
N |
Y |
II |
|
7 |
49 |
Y |
Y |
III |
|
8 |
47 |
N |
Y |
II |
|
9 |
39 |
N |
Y |
I |
|
10 |
31 |
N |
Y |
II |
|
11 |
45 |
Y |
Y |
I |
|
12 |
NC |
Y |
Y |
III |
|
13 |
NC |
Y |
Y |
III |
|
14 |
NC |
Y |
Y |
III |
Age: Years ; NC: non-communicated ; Y: yes ; N : no.
Table 1: The age and characteristics of the the patients whose samples were used in our study.
4.3. Flow cytometry analysis
Cells were collected in cold PBS/0.5% BSA, washed and then incubated with fluorescent- 346 conjugated primary or isotype-matched antibodies for 20 min at 4°C. Following extensive washing (with PBS), cells were fixed (in 1% paraformaldehyde) and 5,000 viable events were analyzed (MACSQuant cytometer running MACSQuantify software; Miltenyi Biotec, Paris, France). Results were expressed as the percentage of positive cells and the MFI ratio (the MFI obtained in the presence of specific antibody divided by the MFI obtained with a non-specific, matched isotype). For SLAMF3 staining, monoclonal FITC-coupled SLAMF3 antibody (clone HLy9.1.25) and the corresponding IgG1-FITC isotype were used (purchased from AbD Serotec) at 1/10 dilution.
4.4. Western blot analysis
Cells (3 x 106 per assay) were lysed in Nonidet P40 (NP40) buffer (1% NP40, 50 mM Tris pH 7.5, 10% glycerol, 150 mM NaCl, 1 mM EDTA, 100 mM Na3VO6, 0.5 mM phenylmethanesulphonyl fluoride (PMSF), 5 mg/mL aprotinin, 5 mg/ml leupeptin and 2 mg/mL pepstatin) containing protease and phosphatase inhibitors (Roche, Meylan, France). Equal amounts of each (total extracted) protein sample were separated by SDS-PAGE. Proteins were transferred on to PVDF membranes (Bio-Rad, Munich, Germany). Following blockade step, blots were incubated with primary antibodies against SLAMF3 (monoclonal clone K12 or 3H1998; Santa Cruz) used at 1/200; STIM1 (monoclonal anti- STIM1 (clone D88E10) used at 1/1000; ERK1/2 (monoclonal, clone 137F5), phospho-ERK1/2 (monoclonal, Thr202/Tyr204; clone D13.14.4E) used at 1/1000 and GAPDH (monoclonal, clone D16H11) used at 1/1000 all purchased from Cell Signaling Tech. The secondary antibodies were HRP- coupled anti-mouse and anti-rabbit used at 1/10000 (Santa Cruz Biotechnology). Blots were developed with the Enhanced ChemiLuminescence (ECL) system (Bio-Rad, Munich, Germany).
4.5. RNA extraction, quantitative real time PCR and plasmid construction
Total RNA was extracted using RNeasy kit (Qiagen, Courtaboeuf, France) and 1 µg of total RNA was reverse transcribed into cDNA with multiscribe reverse transcriptase (Applied Biosystems, Villebon-sur-Yvette, France). Quantitative PCR was performed according to the Taqman Gene Expression protocol (Applied Biosystems) using SLAMF3 primers previously described [21]. The transcription of GAPDH (HUMAN GAPDH (DQ) Mix (20x), Applied Biosystems®) was measured as an endogenous housekeeping control. For STIM1 quantification, SYBRGreen gene expression protocol was used with the following primers: STIM1 and GAPDH [28]. Transfection of SLAMF3 was performed with pIRES-eGFP-SLAMF3 plasmid in BC cells and with pIRES-eGFP as negative control. The pIRES-eGFP-SLAMF3 plasmid permits both the gene of interest and EGFP gene to be translated from a single bicistronic mRNA.
For SLAMF3 over-expression, 0.3 x 106 of T47D or MDA-MB-231 cells were first seeded into six- well plates for 24 hours prior to transfection. Cells were transfected with 0.5 µg of plasmid DNA using the FuGENE HD Transfection Reagent Kit (Promega, Charbonnières-les-bains, France) according to the manufacturer’s instructions and were incubated for 48 hours at 37°C before all experiments.
4.6. Calcium imaging
After transfection, 0.1 x 106 MDA-MB-231 cells were directly seeded on glass coverslips, and grown for an additional 48 hours. Cells were then incubated in culture medium containing 2 µM Fura Red/AM (Sigma Aldrich) for 45 minutes at 37°C before Ca2+ measurement. After Fura Red/AM loading, cells were washed and kept in extracellular saline solution containing the Ca2 -free solution (in mM): NaCl 145, KCl 5, MgCl2 1, HEPES-NaOH 10, glucose 5 and EGTA 1 at pH 7.4 The glass coverslip was mounted in a chamber on a Zeiss (Charly-le-Roi, France) microscope equipped for fluorescence. Fura Red/AM fluorescence was collected upon alternate excitation at 420 nm for the Calcium-bound Fura Red and 480 nm for the Calcium-free Fura Red, using a monochromator (Polychrome IV; TILL Photonics, Planegg, Germany), and captured by a Cool SNAPHQ camera (Princeton Instruments, Evry, France) after filtration through a long-pass filter (660 nm). Metafluor software (version 7.0, Molecular Devices, St. Grégoire, France) was used for acquisition and analysis. The [Ca2+]i variation is represented as the mean of the fluorescence ratio (F405/F480). We used classical protocol for measurement of SOCE. Cells were perfused by Thapsigargin at 1 µM in free calcium extracellular medium to induce reticular calcium release, the cells were then perfused by extracellular medium containing 10 mM Ca2+. SOCE was quantified by the formula ΔFluo = Fmax-Fmin of the measured fluorescence after the excitation at 420 nm for calcium-bound Fura Red, during the perfusion of the 10 mM Ca2+ (FMax) followed by 0 mM Ca2+ (Fmin). All recordings were performed at room temperature. Cells were continuously perfused with the saline solution and chemicals were added as indicated. Thapsigargin was purchased from CalbioChem. The flow rate of the whole cell chamber perfusion system was set to 1 mL/min and the chamber volume was 500 µL.
4.7. Manganese quenching
To estimate divalent influx through the plasma membrane, we used the Mn2+-induced quenching of Fura Red approach. T47D and MDA-MB-231 cells (0.1 x 106) were seeded on glass coverslips after transfection. Briefly, cells were loaded with 2 µM Fura Red/AM (Sigma-Aldrich Chemie, St Quentin Fallavier, France) in the growth medium at 37°C for 45 minutes. After a period of one minute, the Ca2+ (2 mM) present in the perfusion medium was replaced with 1 mM Mn2+. To measure Mn2+ flux, cells were excited at 420 nm and the emission signal was recorded at 660 nm at one second intervals. The Mn2+ influx was estimated from the quenching of 420 nm fluorescence.
4.8. Proliferation assay
The methylthiazole tetrazolium (MTT) salt assay was used to check the anti-proliferative effect of SLAMF3 expression in BC cells. At 24, 48, 72 and 96 hours, transfected cells were rinsed and exposed for 1 hour to a 0.5 mg/mL solution of MTT suspended in culture medium. Reduced purple Formazan crystals were extracted with DMSO: Isopropanol (50:50) and analyzed at a wavelength of 570 nm.
4.9. Migration assays
Cell migration was assessed using a wound healing assay and Boyden chambers test. 24 hours after transfection, MDA-MB-231 cells were scratched with a sterile micropipette tip and then washed to remove floating cells and debris. The cells were cultured in complete medium and wound closure was photographically monitored at 0 hour (T0), 24 hours (T24) and 48 hours (T48) after the scratch. Wound closure analyses were performed by measuring wound area. Wound area at 24 hours and 48 hours was normalized by the wound opening at T0 for each condition. For the Boyden chambers tests, 4 x 104 MDA-MB-231 transfected cells were seeded in the upper side of Boyden chambers (BD Biosciences, 24-wells plates, 8 µm pore size) in 200 µL of DMEM 10% FCS and the lower were filled with 800 µL of DMEM 10% FCS. Cells were incubated for 16 hours at 37°C. Then, cells were washed twice with PBS, fixed with methanol 10min, stained with crystal violet 5 minutes and washed thrice in water. Cells remaining in the upper side were removed using a swab. Migrating cells were observed with an inverted microscope. The number of migrating cells was counted in 20 separate fields for three Boyden chambers per condition. For migration analysis, the number of migrating cells of SLAMF3 condition was normalized by the number of Mock migrating cells.
4.10. Statistical analysis
Independent Student’s t-test was used to compare SLAMF3 and STIM1 mRNA expression in T and pT samples. Comparison between STIM1 and SLAMF3 mRNA expressions was performed by the Spearman correlation test. The Mann-Whitney U test was also used to compare STIM1 or SLAMF3 mRNA expression in Mock and overexpressing SLAMF3 cells. Unless otherwise stated, results are expressed as the mean ± SEM. Statistical analysis were performed with Sigma Plot software (version 11.0, Systat Software Inc., San Jose, CA, USA). The threshold for statistically significant was set to p <0.05 for all analysis.
5. Conclusions
Our results indicate that SLAMF3 reduces SOCE and therefore migration of BC cells by decreasing the STIM1 expression. SLAMF3 could be used as a predictive marker of BC evolution and aggressiveness.
Funding
All experiments were financed by Inserm 1247 and LPCM UR-4667-UPJV funds.
Acknowledgements
We thank Pr Loïc Garcon and Dr Hakim Ouled-Haddou (Hemateam, CURS-UPJV) for analysis and logistic help for flow cytometry as well as ICAP platform (Cytométrie, Centre universitaire de recherché en santé CHU Amiens) for the help.
Conflicts of Interest
The authors have no financial conflicts of interest.
Ethical Approval and consent to participate
Our study was a retrospective essay. Our protocol was approved by the local independent ethics committee (Comité de Protection des Personnes (CPP) Nord-Ouest, Amiens, France) and all patients were provided with information on the study procedures and objectives, and all gave their written consent to participating.
Consent for publication
All authors of this work have agreed to the publication.
References
- Bray F, GLOBOCAN 2012 v1.0 Cancer Incidence and Mortality Worldwide: IARC CancerBase 11 (2018).
- de Boer M, van Dijck J a. a. M, Bult P, et al. Breast cancer prognosis and occult lymph node metastases isolated tumor cells and micrometastases. J Natl Cancer Inst. 102 (2010): 410-425.
- Lastraioli E, Iorio J, Arcangeli A, Ion channel expression as promising cancer biomarker. Biochim Biophys Acta. 1848 (2015): 2685-2702.
- Monteith GR, McAndrew D, Faddy HM, et al. Calcium and cancer: targeting Ca2+ Nat Rev Cancer 7 (2007): 519-30.
- Chen YF, Chen YT, Chiu WT, et al. Remodeling of calcium signaling in tumor progression. J Biomed Sci 20 (2013): 23.
- Liou J, Kim ML, Heo WD, et al. STIM is a Ca2+ sensor essential for Ca2+-store-depletion-triggered Ca2+ influx. Curr Biol CB 15 (2005): 1235-1241.
- Roos J, DiGregorio PJ, Yeromin AV, et al. STIM1 an essential and conserved component of store-operated Ca2+ channel function. J Cell Biol 169 (2005): 435-445.
- Zhang SL, Yu Y, Roos J, Kozak JA et al. STIM1 is a Ca2+ sensor that activates CRAC channels and migrates from the Ca2+ store to the plasma Nature 437 (2005): 902-905.
- Faouzi M, Hague F, Potier M, et al. Down-regulation of Orai3 arrests cell-cycle progression and induces apoptosis in breast cancer cells but not in normal breast epithelial cells. J Cell Physiol 226 (2011): 542-51.
- Feske S, Gwack Y, Prakriya M, et al. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature441 (2006): 179-185.
- Vig M, Peinelt C, Beck A, et al. CRACM1 is a plasma membrane protein essential for store-operated Ca2+ entry. Science 312 (2006): 1220-1223.
- Zhang SL, Yeromin AV, Zhang XHF, et al. Genome-wide RNAi screen of Ca(2+) influx identifies genes that regulate Ca(2+) release-activated Ca(2+) channel activity. Proc Natl Acad Sci USA 103 (2006): 9357-9362.
- Cheng H, Wang S, Feng R. STIM1 plays an important role in TGF-β-induced suppression of breast cancer cell proliferation. Oncotarget 7 (2016): 16866-16878.
- Kim JH, Lkhagvadorj S, Lee MR, et al. Orai1 and STIM1 are critical for cell migration and proliferation of clear cell renal cell carcinoma. Biochem Biophys Res Commun 448 (2014): 76-82.
- Umemura M, Baljinnyam E, Feske S, et al. Store-operated Ca2+ entry (SOCE) regulates melanoma proliferation and cell migration. PloS One 9 (2014): e89292.
- Wu Z, Qing J, Xia Y, et al. Suppression of stromal interaction molecule 1 inhibits SMMC7721 hepatocellular carcinoma cell proliferation by inducing cell cycle arrest. Biotechnol Appl Biochem 62 (2015): 107-111.
- Xia J, Wang H, Huang H, et al. Elevated Orai1 and STIM1 expressions upregulate MACC1 expression to promote tumor cell proliferation metabolism migration and invasion in human gastric cancer. Cancer Lett 381 (2016): 31-40.
- Zhang Z, Liu X, Feng B, et al. STIM1 a direct target of microRNA-185 promotes tumor metastasis and is associated with poor prognosis in colorectal cancer. Oncogene 34 (2015): 4808-4820.
- Wang Y, Wang H, Pan T, et al. STIM1 silencing inhibits the migration and invasion of A549 cells. Mol Med Rep 16 (2017): 3283-3289.
- Yang S, Zhang JJ, Huang XY. Orai1 and STIM1 are critical for breast tumor cell migration and metastasis. Cancer Cell 15 (2009): 124-134.
- Marcq I, Nyga R, Cartier F, et al. Identification of SLAMF3 (CD229) as an Inhibitor of Hepatocellular Carcinoma Cell Proliferation and Tumor Progression. PLoS ONE (2013): 8.
- Bouhlal H, Ouled-Haddou H, Debuysscher V, et al. RB/PLK1-dependent induced pathway by SLAMF3 expression inhibits mitosis and control hepatocarcinoma cell proliferation. Oncotarget 7 (2016): 9832-9843.
- Calpe S, Wang N, Romero X, et al. The SLAM and SAP Gene Families Control Innate and Adaptive Immune Responses. Advances in Immunology (2008): 177-250.
- Romero X, Zapater N, Calvo M, et al. CD229 (Ly9) lymphocyte cell surface receptor interacts homophilically through its N-terminal domain and relocalizes to the immunological synapse. J Immunol Baltim Md 174 (2005): 7033-7042.
- Martin M, Romero X, de la Fuente MA, et al. CD84 functions as a homophilic adhesion molecule and enhances IFN-gamma secretion: adhesion is mediated by Ig-like domain 1. J Immunol Baltim Md 167 (2001): 3668-3676.
- Graham DB, Bell MP, McCausland MM, et al. Ly9 (CD229)-deficient mice exhibit T cell defects yet do not share several phenotypic characteristics associated with SLAM- and SAP-deficient mice. J Immunol Baltim Md 176 (2006): 291-300.
- Pérez-Quintero LA, Roncagalli R, Guo H, et al. EAT-2 a SAP-like adaptor controls NK cell activation through phospholipase Cγ Ca++ and Erk leading to granule polarization. J Exp Med 211 (2014): 727-742.
- Gueder N, Allan G, Telliez MS, et al. sp2 -Iminosugar α-glucosidase inhibitor 1-C-octyl-2-oxa-3-oxocastanospermine specifically affected breast cancer cell migration through Stim1 β1-integrin and FAK signaling pathways. J Cell Physiol 232 (2017): 3631-3640.
- Déliot N, Constantin B. Plasma membrane calcium channels in cancer: Alterations and consequences for cell proliferation and migration. Biochim Biophys Acta 1848 (2015): 2512-2522.
- Motiani RK, Abdullaev IF, Trebak M. A novel native store-operated calcium channel encoded by Orai3: selective requirement of Orai3 versus Orai1 in estrogen receptor-positive versus estrogen receptor-negative breast cancer cells. J Biol Chem 285 (2010): 19173-1983.
- Roodman GD. Mechanisms of bone metastasis. N Engl J Med 350 (2004): 1655-1664.
- Donovan JC, Milic A, Slingerland JM. Constitutive MEK/MAPK activation leads to p27(Kip1) deregulation and antiestrogen resistance in human breast cancer cells. J Biol Chem 276 (2001): 40888-40895.
- Prevarskaya N, Ouadid-Ahidouch H, Skryma R, et al. Remodeling of Ca2+ transport in cancer: how it contributes to cancer hallmarks? Philos Trans R Soc Lond B Biol Sci 369 (2014): 20130097.
- Roderick HL, Cook SJ. Ca2+ signalling checkpoints in cancer: remodeling Ca2+ for cancer cell proliferation and survival. Nat Rev Cancer 8 (2008): 361-375.
- Vashisht A, Trebak M, Motiani RK. STIM and Orai proteins as novel targets for cancer therapy. A Review in the Theme: Cell and Molecular Processes in Cancer Metastasis. Am J Physiol Cell Physiol 309 (2015): C457-469.
- Prevarskaya N, Skryma R, Shuba Y. Calcium in tumor metastasis: new roles for known actors. Nat Rev Cancer 11 (2011): 609-618.
- Jardin I, Lopez JJ, Salido GM, et al. Store-Operated Ca2+ Entry in Breast Cancer Cells: Remodeling and Functional Role. Int. J. Mol. Sci 19 (2018): 4053.
- Latour S, Mahouche I, Cherrier F, et al. Calcium Independent Effect of Orai1 and STIM1 in Non-Hodgkin B Cell Lymphoma Dissemination. Cancers 10 (2018): 402.
- Okamoto Y, Ohkubo T, Ikebe T, et al. Blockade of TRPM8 activity reduces the invasion potential of oral squamous carcinoma cell lines. Int J Oncol 40 (2012): 1431-1440.
- Monet M, Lehen’kyi V, Gackiere F, et al. Role of cationic channel TRPV2 in promoting prostate cancer migration and progression to androgen resistance. Cancer Res 70 (2010): 1225-1235.


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 