Prospects of novel foods as complete Human Nutrition
Sharadha Arun, Tomal Dattaroy, Badrish Soni*, Santanu Dasgupta
Synthetic Biology Group, Reliance Corporate Park, Reliance Industries Limited, Ghansoli, Navi Mumbai - 400701, India
*Corresponding Author: Badrish Soni, Synthetic Biology Group, Reliance Corporate Park, Reliance Industries Limited, Ghansoli, Navi Mumbai - 400701, India
Received: 17 June 2021; Accepted: 25 June 2021; Published: 30 June 2021
Article Information
Citation: Sharadha Arun, Tomal Dattaroy, Badrish Soni, Santanu Dasgupta. Prospects of Novel Foods as Complete Human Nutrition. Journal of Food Science and Nutrition Research 4 (2021): 175-180.
DOI: 10.26502/jfsnr.2642-11000070
View / Download Pdf Share at FacebookAbstract
Food is one of the most basic and unavoidable nutritional requirements of all living organisms. Humans have acquired the art of cooking since decades and have mastered themselves in culinary skills. Food is consumed to suffice macro and micro-nutrients of body. With increasing population and lifestyle changes, the pattern and composition of food is also rapidly altered. The novel foods being rapidly developed are more focused not only on novel varieties but also on sustainability, nutritional balance, consumer acceptability, scalability, and cost. Global regulatory policies have intervened positively and have provided conducive environment for production of such novel foods to meet increasing global demands.
Keywords
<p>Novel foods, Sustainability, Regulatory policies, Nutrition</p>
Article Details
Introduction
Malnutrition and hunger are globally prevalent in alarming numbers. According to UNICEF, about a quarter of all children globally are stunted. Undernutrition contributes to 45% of death of children under 5 years worldwide [1]. Across the world, around 462 million adults are underweight [2]. In micronutrient malnutrition, it is estimated that 42% of children less than 5 years of age and 40% of pregnant women are anemic [3]. Therefore, there is an immediate requirement to address the global nutrition gap. On April 1, 2016, the United Nations proclaimed 2015-2025 as the ‘United Nations Decade of Action on Nutrition’ [4,5]. This calls for eradicating all forms of malnutrition with a focus on sustainable food production. Research towards novel foods and novel technologies would significantly contribute to bridging the gap between food availability and nutrition. Currently, various global policies like food fortification, supplementation, and food distribution address the malnutrition load. However, sustainable food production still remains a challenge. This is where scientists around the world are actively trying to develop sustainable foods with novel ingredients and technology.
Novel food research for sustainable nutrition and food production
Efforts are underway to develop sustainable novel foods globally. In the recent decade, emergence of plant-based as well as cell-cultured dairy and meat substitutes are examples of novel foods penetrating the market. Examples include plant-based burgers from leading successful brands such as Impossible Foods and Beyond Meat [6]. According to the April 16, 2021 Bloomberg reports [7], plant burgers are increasingly accepted by consumers and this growing demand has led to competition between companies. The impact of animal meat production on environment and food security has increased the need to research for sustainable alternatives. The plant based dairy and meat alternatives are emerging out as a sustainable solution. However, there is still very less awareness on plant based dairy and meat substitutes [8-11]. Research is still on-going in terms of making the alternate dairy and meat substitutes nutritionally complete. An independent organization, Safefood, investigated the nutritional composition of alternate meats in the market and observed that one out of every four products do not have enough protein. This opens up further opportunities to study and enhance the nutritional as well as the sensory quality of meat alternatives [12]. Research is on to develop novel foods as functional ingredients and nutraceuticals. These products aim at providing benefits for overall health, when consumed as a supplement to the regular diet. Several micro-organisms are being explored to produce novel functional foods such as probiotics. Microbial ingredients, when tapped to potential can be a sustainable, nutritionally complete part of the human nutrition. Production of microbial ingredients require lesser space and energy [13].
Nutrient delivery through novel foods
To bridge the nutritional gap, targeted novel food research can be conducted. This involves studying the nutritional profile of the population, identifying the gaps, studying the food systems and food supply chain. Crop varieties such as golden rice offer a promising solution to micronutrient delivery [14]. Genetically modified varieties have shown promise to improve yield and quality of crops [15-18]. Technologies such as genome editing can utilize microbes to provide natural and bio-available micronutrients that are both sustainable and can be used as a food fortificant [19]. This principle can also be applied to develop micronutrient supplements which can be administered through government prophylaxis programs [20].
Safety of novel food
Novel foods require in-depth safety assessment and most regulatory frameworks require in-depth safety assessment. In India, a technical dossier needs to be submitted in compliance to the safety and product information detailed out in the Food Safety and Standards [21]. The technical dossier would then be reviewed by the government’s scientific panel and an approval would be issued to commence the commercial manufacture of the product. The United States Food and Drug Administration (US FDA) states that any food additive that is intended to be added in food is subject to Pre-Market Approval (PMA) through the food and color additive petition program to US FDA. The PMA process can be exempted if the substance has a Generally Recognized as Safe (GRAS) status by qualified experts [22]. A novel food derived from a genetically modified organism will have to go through the same process in the US FDA system and it is appropriate to get a GRAS status by qualified experts who will verify the biological safety tests as applicable for products derived from recombinant DNA technology. US regulations qualify foods derived via genome-editing technology as such derived using new breeding technology and are therefore considered safe. Foods derived via CRISPR mediated technology will therefore be subjected to the same scrutiny in a GRAS submission as any other novel food product [23]. The regulation EU 2015/2283 of the European Food Safety Authority encompasses the approval and listing process for novel foods within Europe. This procedure requires a hazard assessment and safety studies to be completed and a product dossier to be submitted to EFSA. It must be noted that foods derived using CRISPR would be considered as genetically modified foods in the EU and therefore, the regulations governing any novel food using this technology will be the same as GMOs. Table 1 provides consolidated requirements for novel food approval whereas Figure 1 gives a flow diagram that outlines the regulatory overview from product approval to the scaling up of the production of a novel food.
Cost, Accessibility, and consumer acceptance of novel foods
Cost of many novel foods remain a premium and less accessible owing to production scales, regulatory framework, and on-going market research. It is essential for novel foods to have a cost benefit too for better market reach. Information Education and Communication (IEC) campaigns should be conducted for consumer acceptance of any novel food. A study indicated that consumers are generally willing to try novel foods and understand the nutrition information [24]. Recently published US FDA trend on consumer behaviour indicate that 87% of people look at nutritional information of food [25]. Hence novel foods must also be developed taking cognizance of nutritional adequacy and sensory acceptance. In summary, it is imperative that more novel foods and technologies are researched and made accessible to increasingly vulnerable population. Sustainable nutrition plays a tremendous role in overall development. Global policies must emphasize on research towards sustainable food systems and embrace novel technologies which offer comprehensive solution to global nutrition gap.
|
Requirement |
India (FSSAI Novel Foods Regulation, 2017) |
United States Food and Drug Administration (GRAS) |
European Food Safety Authority (EFSA) |
|
Intended use and levels |
Yes |
Yes |
Yes |
|
Identity of the protein (Complete taxonomic identity of the source of protein along with identification methods) |
Yes |
Yes |
Yes |
|
Detail production process |
Yes |
Yes |
Yes |
|
Detailed compositional data - Ingredients |
Yes |
Yes |
Yes |
|
Stability |
Yes |
No |
Yes |
|
Technical Specification - Proximate, microbiological specification, contaminants and residues |
Yes |
Yes |
Yes |
|
History of the source of the novel protein |
Yes |
Yes |
Yes |
|
Global regulatory status - whether the protein in question is approved elsewhere |
Yes |
No |
Yes |
|
Target population |
Yes |
Yes |
Yes |
|
Dietary Exposure - How much of the product is likely to be exposed to the population at a given time, based on statistical modeling |
Yes |
Yes |
Yes |
|
Maximum tolerable limits of consumption |
Yes |
Yes |
Yes |
|
Absorption, Distribution, Metabolism and Excretion (ADME) pathway |
No |
Yes |
Yes |
|
Nutritional information/benefits |
Yes |
Yes |
Yes |
|
Toxicological Studies - Conducted in terms of end use and exposure |
|||
|
Genotoxicity - Bacterial reverse mutation assay, in vitro mammalian chromosomal aberration, mutagenicity |
Yes |
Yes |
Yes |
|
Sub chronic toxicity |
No |
Yes |
Yes |
|
Reproductive toxicity |
Yes |
Yes |
Yes |
|
Allergenicity |
Yes |
Yes |
Yes |
|
Neurotoxicity |
No |
Required in some cases |
No |
Table 1: Regulatory requirements for novel food approval
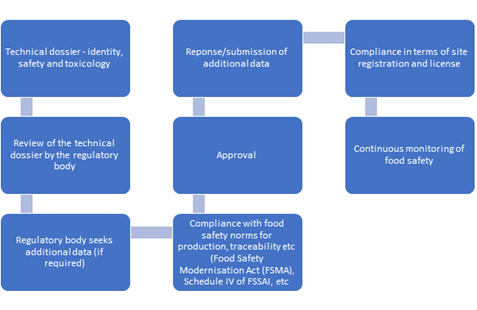
Figure 1: Regulatory overview - novel food approval to production scale up
References
- Global Nutrition Report 2020: Action on equity to end malnutrition (2020).
- Malnutrition, WHO (2021).
- Anaemia, WHO (2021).
- The un decade of action on nutrition 2016-2025 (2021).
- FAO and WHO. Sustainable healthy diets - Guiding principles. Rome. 2019.
- Massimo De Marchi, Angela Costa, Marta Pozza, et al. Manuelian. Detailed characterization of plant-based burgers. Scientific reports 11 (2021): 20-49.
- Impossible and beyond slash prices as fake-meat market heats up, Bloomberg reports (2020).
- The science of plant-based meat (2020).
- Stephan van Vliet, Scott L. Kronberg, Frederick D. Provenza. Plant-Based Meats, Human Health, and Climate Change. Frontiers in sustainable food systems 4 (2020): 128.
- Konstantina Kyriakopoulou, Birgit Dekkers, Atze Janvan der Goot. Chapter 6 - Plant-based meat analogues. Sustainable meat production and processing. Academia Press (2019): 103-126.
- Jiang He, Natasha Marie Evans, Huaizhi Liu, et al. A review of research on plant-based meat alternatives: Driving forces, history, manufacturing, and consumer attitudes. Comprehensive Reviews in Food Science and Food Safety 5 (2019): 2639-2656.
- Quarter of vegetarian meat substitutes contain insufficient protein - Safefood (2021).
- Brodmann, Theodor, Akihito Endo, et al. Safety of novel microbes for human consumption: practical examples of assessment in the European Union. Frontiers in microbiology 8 (2017): 17-25.
- Golden Rice Project (2020).
- Hyung-Keun Ku and Sun-Hwa Ha. Improving Nutritional and Functional Quality by Genome Editing of Crops: Status and Perspectives. Frontiers in plant science 11 (2020).
- Surekha Agarwal, Satendra Mangrauthia, Neelamraju Sarla. Chapter 14 - Genomic approaches for micronutrients biofortification of rice. In plant micronutrient use efficiency molecular and genomic perspectives in crop plants. Academia Press (2018): 245-260.
- Jiemeng Xu, Kai Hua, Zhaobo Lang. Genome editing for horticultural crop improvement. Horticulture Research 6 (2019): 113.
- De Steur H, Mehta S, Gellynck X, et al. GM biofortified crops: potential effects on targeting the micronutrient intake gap in human populations. Current Opinion in Biotechnology 44 (2017): 181-188.
- Rohaya Ali, Khurshid Dijoo Zulaykha, Nasreena Sajjad. Genetically Modified Microbes as Biofertilizers. Bioremediation and Biotechnology 4 (2020): 275-293.
- Abhishek Kulkarni, Monika Khade, Sharadha Arun, et al. An overview on mechanism, cause, prevention and multi-nation policy level interventions of dietary iron deficiency. Critical Reviews in Food Science and Nutrition 5 (2021): 1-15.
- Food Safety and Standards (Non-specified foods and food ingredients) Regulation, 2017, the Food Safety and Standards Authority of India (2017).
- Determining the Regulatory Status of a Food Ingredient, FDA (2018).
- Alice Yuen-TingWong and Albert Wai-KitChan. Genetically modified foods in China and the United States: A primer of regulation and intellectual property protection. Food Science and Human Wellness 5 (2016): 124-140.
- McFarlane Traci and Patricia Pliner. Increasing willingness to taste novel foods: effects of nutrition and taste information. Appetite 28 (1997): 227-238.
- Food Safety and Nutrition Survey Results, FDA (2021).


 Impact Factor: * 3.8
Impact Factor: * 3.8 Acceptance Rate: 77.96%
Acceptance Rate: 77.96%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 