The Effect of Different Malting Periods on the Nutritional Composition of Malted Sorghum-Soy Composite Flour
Atamgba Agbor Asuk1,2*, Melvin Nnaemeka Ugwu1, Boaz Idole2
1Department of Medical Biochemistry, Cross River University of Technology, Calabar, Nigeria
2Department of Food Science and Nutrition, Islamic University in Uganda, Mbale, Uganda
*Corresponding Author: Atamgba Agbor Asuk, Department of Medical Biochemistry, Cross River University of Technology, Calabar, Nigeria
Received: 24 June 2020; Accepted: 20 July 2020; Published: 31 August 2020
Article Information
Citation: Atamgba Agbor Asuk, Melvin Nnaemeka Ugwu, Boaz Idole. The Effect of Different Malting Periods on the Nutritional Composition of Malted Sorghum-Soy Composite Flour. Journal of Food Science and Nutrition Research 3 (2020): 217-230.
DOI: 10.26502/jfsnr.2642-11000051
View / Download Pdf Share at FacebookAbstract
Malted sorghum-soy composite flour was developed from sorghum grains and soy beans. The sorghum grains were subjected to different malting periods of 2, 3, 4, and 5 days respectively and then blended with soy in the ratio 2:1. The composite flour was analysed for proximate and specific mineral composition and sensory evaluation was carried out on the porridge made from the composite flour. The results of sensory evaluation showed general decrease in sensory attributes with increased malting period, but day 2 malted sorghum compared well (P≥0.05) with the non-malted period in all attributes. The proximate analysis showed a significant (P<0.05) increase in moisture content at day 2 malting but a significant (P<0.05) decrease in ash and crude fat content with increased malting compared to the non-malted (day 0). While the crude fibre was significantly (P<0.05) increased at day 3, 4 and 5 of malting, total carbohydrate was at day 2 only compared with day 0. The crude protein showed no significant (P≥0.05) difference at day 4 compared with day 0. The specific mineral analysis revealed a significant (P<0.05) increase in Zn, Fe, Mn and Mg but not Ca and Cu on day 3 malting compared with day 0. The study revealed improved mineral and fibre content of the composite with malting and a demonstration of the potential of the malting process in reducing anti-nutrients and increasing the presence of rich phytochemicals. Hence, malted sorghum with soy possibly complements the nutrient demand of the malnourished groups, especially of developing countries.
Keywords
<p>Malnutrition, Complementation, Proximate composition, Malted grains, Specific minerals, Porridge</p> <gdiv></gdiv>
Article Details
1. Introduction
Sorghum is a common and underutilized coarse cereal especially in developing countries and has been considered in the development of sorghum flour fortified with soy flour to produce composite flour with improved nutritional value. Sorghum proves a great deal in feeding programs after its fortification with soybeans [1-3]. Soybeans (Glycine max) are cheap source of high-quality proteins with a good balance of amino acids. It contains a reasonable amount of methionine lacking in sorghum and it is a good supplement to cereals. Soybeans contain all the essential amino acids found in animal proteins; a quality not common with other plants [4]. One way to curb global menace of Protein Energy Malnutrition (PEM) is through food fortification of plant origin [5]. This is aimed to allow people to obtain from their diet all the energy, macro and micronutrients they need to enjoy a healthy and productive life. Soybean legume has been considered as an ideal legume food for meeting protein needs in a population [6].
There is a relationship between diet consumption, socio-economic conditions and health status of individual. Most of the cereal grains namely wheat, barley and rye commonly used as staple foods contain gluten [7]. Gluten is a flexible protein that triggers inflammatory reactions in people with celiac disease or gluten sensitivity causing abdominal pain and digestive issues and eventually leads to intestinal damage [8]. According to epidemiological studies, PEM remains one of the most significant child health problems in developing countries with an estimated 53% of child deaths per year attributed to being underweight [9]. PEM results from deprivation of essential amino acids and total nitrogen and energy substrates. The serious problem associated with PEM is rapid loss of weight and death in severe cases.
Sorghum which is the base material for the composite flour is a principal source of energy, proteins and minerals for the poorest regions [10]. Sorghum starch is gluten free and could be a good substitute for wheat flour for individuals who are unable to digest food made from wheat [11]. The protein matrix and starch structure in sorghum grain are responsible for its slow digestibility than those of other cereals, a trait beneficial for diabetics [12].
Sorghum is a terrific substitute for traditional grains consumed in everyday life in most communities. It is an excellent source of energy containing about 75% carbohydrate. It is also a good to excellent source of iron, zinc and rich in B complex vitamins. Additionally, sorghum provides good to excellent source of phytochemicals such as phenolic acids, anthocyanins, phytosterols and policosanols [13]. Sorghum is deficient in essential amino acids like lysine, methionine and tryptophan [10]. These together with the presence of antinutritional factors like the tannins, phytates and oxalates make it nutritionally inadequate. According to research report by [14], it is believed that malting of cereal grains increases the overall nutrient content. Malting of sorghum may improve its digestibility, sensory and nutritional quality as well as pronounced effect in lowering antinutrients [15]. The increase in total dietary fibre as a result of malting coupled with a decrease in fat content can be of benefit to persons with cardiovascular diseases [14, 16].
The development of composite flour from malted sorghum grains and soy beans might prove a great deal as this initiative puts into consideration protein supplementation and complementation. The high energy composite flour with balanced amino acid profile will help address certain nutritional deficiencies common in both adults and children of the undernourished groups particularly those of the rural communities. Malting of sorghum grains to obtain malted sorghum flour and fortification of the resulting flour with soy flour could be used as one of the strategies to improve the nutrient content in sorghum. The development of malted sorghum-soy composite flour will also enhance proper utilization of sorghum in various forms such as porridge, cookies, cereal extracts and unleavened bread while making available nutritiously cheap composite flour.
2. Materials and Methods
2.1 Equipment, chemicals and reagents
The equipment used included separating funnels, pipettes, muffle furnace, desiccators, pair of tongs, watch glass, and analytical balance, Petri dishes, crucibles, vacuum pump, beakers, volumetric flasks, burettes, digestion flask and Atomic Absorption Spectrophotometer. The chemicals and reagents used in the analysis were of analytical grade.
2.2 Raw materials collection and preparation
The soybeans and sorghum grains used were bought from Odelo market in Kumi town, Uganda.
2.2.1 Preparation of malted sorghum flour: The method of Hallen et al. [17] was used to prepare malted sorghum flour of different days of malting. The sorghum grains (10 kg) were sorted using a winnower to remove dirt, pieces of wood, stones and some extraneous matter. After the grains had been thoroughly washed and steeped for 12 hours in cold water to initiate germination (malting), they were divided into four parts each of 2 kg and subjected to different days of malting. The divided grains were placed in separate sterilized jute bags labelled A, B, C and D and allowed to germinate for different days (2, 3, 4, and 5) respectively. The germinated grains were then dried under the sun separately to a moisture content of around 13%. The withered rootlets were gently brushed off leaving malted grains. The malted grains were dry milled using a hammer mill and resulting flour sieved. The resulting flours were packed in separate polythene bags which were sealed and labelled A, B, C and D respectively.
2.2.2 Preparation of soy flour: The soy flour was prepared according to the method of Ndife et al. [18]. The soy beans (4 kg) were thoroughly cleaned by winnowing to remove stones, sticks, dirt and other extraneous material. The soybeans were then washed with clean water and oven dried at 60°C for 6 hours. The soybeans were roasted at 150°C for 30 minutes and cooled. The soybeans were winnowed after roasting. The soybeans were milled into fine flour using a hammer mill. The soy flour was sieved and packed in a polythene bag which was sealed tightly.
2.2.3 Preparation of malted sorghum-soy composite flour: The malted sorghum-soy composite flour was prepared by blending malted sorghum flour and soy flour in the ratio of 2:1 (Figure 1).
2.2.4 Preparation of malted sorghum-soy composite flour porridge: To aluminium saucepan was added 250 mL of clean cold water. The saucepan with cold water was put in an oven and the water allowed boiling at 100°C. 20 mL of cold water was added into 50 g of composite flour in a plastic cup and mixed thoroughly forming free flowing paste. The paste was then added into the boiling water in the saucepan and stirred at regular intervals until a thick porridge was formed (Figure 2).
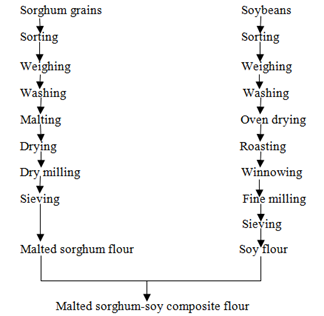
Figure 1: Process flow chart for preparation of malted sorghum-soy composite flour.
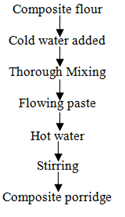
Figure 2: Process flow chart for preparation of malted sorghum-soy composite flour porridge.
2.3 Sensory evaluation
Sensory evaluation of the sample product was done at Islamic University in Uganda from the food science laboratory. The panellists (n=30) were from the Department of Food Science and Nutrition, while others were from the University community. They were recruited on the criteria that they did not have any food allergies, and no information was availed on the amounts and constituents of the product. This was done to minimize biasness. The samples were coded with 1-digit random letter; A, B, C, D for 2, 3, 4 and 5 days of malted sorghum in sorghum-soy composite flour and E for control. Lukewarm water was provided in clean cups and the panellists were asked to rinse their mouths in between tasting of samples. The samples were judged for flavour, texture, taste, colour and overall acceptability. The scaling procedure was based on 9-point hedonic scale [19].
2.4 Proximate analysis
The composite flour mixes were analysed proximately for moisture content, crude protein, crude fat, crude fibre and ash content according to the method described by Association of Official Analytical Chemist [20]. The carbohydrate content of flour mixes was determined by difference method. This was determined by difference method after obtaining the protein, ash, moisture, and crude fat content as below;
%Carbohydrate=100-(%protein +%Ash +%moisture content +%Fat +%crude fibre)
2.5 Energy determination
The energy value of the composite flour was obtained from the formula given below;
Energy value (kJ/100g) = [37 × Fat) + (17 × Carbohydrate) + (17 × Protein)] [21].
2.6 Specific mineral analysis
The minerals were extracted from dry ash samples after first digested using nitric acid and perchloric acid. The filtered aliquots were used for the determination of Zn, Fe, Ca, Mn, Mg and Cu by atomic absorption spectrometric method described by Association of Official Analytical Chemist [20].
2.7 Statistical analysis
Data obtained from the experiment was presented as mean ± SEM after using Microsoft Office Excel 2016. The data was also subjected to a one-way analysis of variance (ANOVA) and post hoc (LSD) for levels of significance using SPSS version 23.0. The levels of significance were accepted at P <0.05.
3. Results
The results of sensory evaluation of sorghum-soy composite porridge, proximate and specific mineral composition of sorghum-soy composite flour after various malting periods of sorghum are presented here.
3.1 Sensory evaluation of malted sorghum-soy composite porridge
Results of sensory evaluation of malted sorghum-soy composite porridge are presented in Table 1. The sensory attributes evaluated include; colour, texture, flavour, taste and overall acceptability for zero day (0 day), two days (2 days), three days (3 days), four days (4 days) and five days (5 days) of sorghum malting. The colour values decreased with increase in sorghum malting periods. There was a significant difference (P<0.05) in colour values between day 0 and day 3 and between day 0 and day 5. There was a decrease in texture values with increase in sorghum malting periods. The decrease was significant (P<0.05) for day 4 and day 5 compared with day 0. There was a slight decrease in flavour values from day 2 to day 4 and further decrease in day 5 when compared with day 0. This decrease was not significant (P≥0.05), however, it showed that flavour preference decreased with increased malting period. The taste preference showed no significant (P≥0.05) difference between day 0, day 2, day 3, and day 4 when compared, however, there was a significant (P <0.05) decrease in taste preference at day 5 of malting compared with day 0 and day 2. The overall acceptability was significantly (P<0.05) decreased at day 3 and day 5 compared with day 0. There was however no significant (P≥0.05) difference in the overall acceptance between day 0, day 2 and day 4. The sensory attributes for day 0 and day 2 compared well with each other as no significant (P≥0.05) difference was observed in all the attributes vis-à-vis colour, texture, flavour, taste and overall acceptability.
3.2 Proximate analysis of malted sorghum-soy composite flour
The results of proximate composition of malted sorghum-soy composite flour are shown in Table 2. The proximate parameters analysed include moisture content, ash content, crude fat, crude fibre, crude protein, total carbohydrate and energy value. The moisture content values of day 2, day3 and day 5 were significantly (P<0.05) higher than day 4 but only day 2 produced a significantly (P<0.05) higher value than day 0. Ash content values showed a decrease with increase in malting days as there was a significant (P<0.05) decrease in the ash content of the malting days compared with day 0. The ash content dropped further at day 3 and then increased slightly at day 4 and5. There was no significant (P≥0.05) difference in ash content values between day 4 and day 5. The crude fat showed a similar pattern as the ash content except that on day 3, there was initial increase before decrease at day 4 and 5.
The values of crude fibre showed an increase with increase in sorghum malting periods. The increase was significant (P<0.05) at day 3, 4 and 5 when compared with day 0. The crude protein values showed a significant (P<0.05) decrease with increase in sorghum malting periods, however, this was not clear-cut as day 4 showed no significant (P≥0.05) difference compared with day 0. There was significant (P<0.05) increase in total carbohydrate in day 2 of sorghum malting compared to day 0, the rest were significantly (P<0.05) decreased compared with day 0. Day 4 and day 5 also compared well with each other as there was no significant (P≥0.05) difference between them. There was a significant (P<0.05) decrease in energy value with increase in malting days compared with day 0. The energy values of day 2 and 3 compared well, similarly day 4 and day 5 also compared well with each other as there was no significant (P≥0.05) difference between them.
3.3 Specific mineral composition of malted sorghum-soy composite flour
The results of specific mineral composition are shown in Table 3. The specific minerals analysed include zinc (Zn), iron (Fe), calcium (Ca), Manganese (Mn), Magnesium (Mg) and Copper (Cu). The result for Zn showed a significant (P<0.05) decrease in day 2 compared with day 0. Day 2 and day 4 compared well with each other. However, the Zn levels in day 3 and 5 were significantly increased compared to day 0, day 2 and day 4. Iron (Fe) was significantly (P<0.05) decreased in day 2, 4, 5 but significantly (P<0.05) increased in day 3 compared with day 0 (non-malted). A similar pattern of Fe was also seen with Mg.
The results for Ca and Cu also showed a significant (P<0.05) decrease in day 2, 4 and 5 compared with day 0. But the Ca levels in day 3, 4, 5 was significantly (P<0.05) higher than day 2, while the Cu of day 2 compared well with day 4, that of day 3 compared well with day 5. The level of Mn was significantly (P<0.05) decreased in day 2 and 4 but significantly (P<0.05) increased in day 3 and 5 compared with day 0. The Mn levels of day 2 and 4 compared well with each other and so were day 3 and 5.
The results of the specific minerals put together revealed that there a significant decrease in all the mineral on day 2 compared with day 0. In day 3, there was significant increase in all the mineral except Ca and Cu compared with day 0. Generally, day 2 and day 4 compared with each other, especially for Zn, Mn and Cu levels, while day 3 and 5 compared well with each other especially for Mn and Cu levels.
Table 1: Sensory evaluation of sorghum-soy composite porridge after various malting periods of sorghum.
|
Malting Days Sensory Attributes |
Day 0 |
Day 2 |
Day 3 |
Day 4 |
Day 5 |
|
Colour |
6.73 ± 0.55a |
6.53 ± 0.38ab |
5.47 ± 0.51bc |
6.20 ± 0.48ab |
4.67 ± 0.62c |
|
Texture |
6.80 ± 0.44a |
6.40 ± 0.48ab |
5.33 ± 0.68bc |
4.93 ± 0.58c |
4.40 ± 0.66c |
|
Flavour |
6.80 ± 0.44a |
6.53 ± 0.41a |
6.13 ± 0.38a |
6.13 ± 0.57a |
5.87 ± 0.65a |
|
Taste |
6.20 ± 0.50a |
6.33 ± 0.36a |
5.53 ± 0.52ab |
5.73 ± 0.56ab |
4.40 ± 0.60b |
|
Overall Acceptability |
7.20 ± 0.44a |
7.07 ± 0.37a |
5.73 ± 0.54bc |
6.53 ± 0.36ac |
5.07 ± 0.69b |
Values are expressed as mean ± SEM (n=30). Values with different superscripts (a, b, c) along the rows are statistically significant at P<0.05.
Table 2: Proximate composition of sorghum-soy composite flour after various malting periods of sorghum [w/w dry mass basis (%)].
|
Malting Days Parameter |
Day 0 |
Day 2 |
Day 3 |
Day 4 |
Day 5 |
|
Moisture content |
6.44 ± 0.19bc |
7.23 ± 0.10a |
7.02 ± 0.17ab |
5.94 ± 0.14c |
7.03 ± 0.10ab |
|
Ash content |
2.90 ± 0.02a |
2.59 ± 0.02b |
2.24 ± 0.01c |
2.46 ± 0.01d |
2.45 ± 0.01d |
|
Crude fat |
7.45 ± 0.01a |
5.31 ± 0.04b |
6.75 ± 0.05c |
4.87 ± 0.00d |
4.86 ± 0.01d |
|
Crude fibre |
6.47 ± 0.21a |
7.53 ± 0.08b |
10.48 ± 0.20c |
13.06 ± 0.50d |
11.35 ± 0.18e |
|
Crude protein |
4.35 ± 0.01a |
4.27 ± 0.01b |
4.30 ± 0.01c |
4.34 ± 0.00a |
4.28 ± 0.01bc |
|
Total carbohydrate |
72.06 ± 0.11a |
72.80 ± 0.09b |
69.19 ± 0.04c |
69.84 ± 0.13d |
70.02 ± 0.11d |
|
Energy (KJ/100g) |
1574.51 ± 2.03a |
1506.54 ± 1.73b |
1499.26 ± 2.01b |
1445.07 ± 2.41c |
1442.91 ± 1.68c |
Values are expressed as mean ± SEM (n=3). Values with different superscripts (a, b, c, d, e) along the rows are statistically significant at P<0.05.
Table 3: Specific mineral composition of sorghum-soy composite flour after various malting periods of sorghum (ppm).
|
Malting Days Specific Minerals |
Day 0 |
Day 2 |
Day 3 |
Day 4 |
Day 5 |
|
Zn |
0.30 ± 0.00a |
0.29 ± 0.00b |
0.32 ± 0.00c |
0.29 ± 0.00b |
0.48 ± 0.00d |
|
Fe |
3.87 ± 0.00a |
1.84 ± 0.00b |
4.01 ± 0.00c |
3.22 ± 0.00d |
3.67 ± 0.00e |
|
Ca |
277.53 ± 0.00a |
187.51 ± 0.00b |
202.92 ± 0.01c |
191.61 ± 0.00d |
196.15 ± 0.00e |
|
Mn |
1.05 ± 0.00a |
0.99 ± 0.00b |
1.07 ± 0.00c |
0.99 ± 0.00b |
1.08 ± 0.00d |
|
Mg |
8.83 ± 0.00a |
8.53 ± 0.01b |
10.05 ± 0.01c |
8.35 ± 0.00d |
8.65 ± 0.00e |
|
Cu |
0.11 ± 0.00a |
0.08 ± 0.00b |
0.09 ± 0.00c |
0.08 ± 0.00b |
0.09 ± 0.00c |
Values are expressed as mean ± SEM (n=3). Values with different superscripts (a, b, c, d, e) along the rows are statistically significant at P<0.05.
4. Discussion and Conclusion
4.1 Discussion
This study considered malting of sorghum for two days (48 hours), three days (72 hours), four days (96 hours) and five days (120 hours) before forming a composite with soy (for each of them) to obtain a sorghum-soy composite porridge. The malted sorghum-soy composite was subjected to organoleptic evaluation as well as proximate and specific mineral analysis. There were changes in sensory attributes in the malting of sorghum in the sorghum-soy composite porridge. There was general decrease in preference towards the malted sorghum-soy composite porridge with increased malting periods, however there was none observed between the malted sorghum-soy composite porridge at day 2 and the non-malted sorghum-soy composite porridge. The decrease in textural preferences with increased malting of sorghum in sorghum-soy composite porridge may be as a result of loss of sponginess [22]. The intensity of the colour was observed to have increased with increased malting from light brown to reddish brown which may have been due to increased presence of certain phytochemicals such as alkaloids and phenols [23]. Some alkaloids have been reported to be produced during plant germination as allelochemicals [23], however alkaloids are also known to exhibit analgesic, antispasmodic and bactericidal properties [24]. The brown sorghum has been reported to contain tannins while the red or reddish-brown sorghum lacks tannins but with moderately high levels of phenols [25]. It can therefore be inferred that the colour change from light brown to reddish-brown with increased malting, is an indication that the tannins present in the sorghum were converted to beneficial phenols. This conversion also causes the release of minerals attached to the tannins hence improving the mineral quality and availability of the sorghum. The proximate and specific mineral compositions assayed showed variations with malting periods. There was increase in moisture content with increased malting period due probably to increased activity of glyoxylate cycle enzymes (a variation of tricarboxylic acid cycle) resulting in the production of metabolic water [26]. This result was comparable to reports by Ogbonna et al. [27], who indicated that in sorghum grains, the moisture content increased with increasing malting periods compared to non-malted grains. The peculiar biochemical or physiological process in the drop of moisture content on the fourth day of malting is unclear. However, there is suggestion by Singh et al. [28] that it may be due to the different treatments the grains were subjected to during drying after germination. High moisture content is undesirable in flours. High moisture in foods could enhance the activities of spoilage microorganisms which in turn reduces quality and shelf life of food products [29].
Enzymes are protein in nature and therefore increased enzyme activity will result in increased protein utilization in the formation of enzymes [30]. This is corroborated by the decrease in protein content as various enzymes demand may have varied during the malting period. The initial decrease in crude protein at day 2 could have been due to the fact that storage nitrogen reserves may have been mobilized during sprouting after hydrolysis by proteolytic enzymes to play a role in synthesis of cellular materials for the rapidly growing roots and shoots during germination [27].
There was progressive decrease in crude fat which could be attributed to increased activity of lipase enzymes during malting that acted on fat to produce fatty acids [31]. In plants the fatty acids are further acted on by polyketide synthase in malted products to form pentadecatrienyl resorcinol intermediates known to act as an allelochemical [23].
General increase in crude fibre was also observed. The increase was probably due to apparent increase in utilization of other constituents such as starch during germination. The inclusion of rootlets and shoots during milling of germinated grains also appears to increase the fibre content [32]. The increase was in agreement with the research work done by Ogbonna et al. [27] which showed increase in crude fibre in sorghum grist after malting. Increased fibre content has positive nutritional implications in that it prevents diverticulosis and aids absorption of trace minerals in the gut as well as helps in the elimination of undigested food materials [24].
The total carbohydrate was increased at the second day of malting thereafter decreased. The increase was probably due to activation of the glyoxylate pathway with preference towards converting fatty acids to four-carbon dicarboxylic acids and incorporated into the gluconeogenesis pathway leading to the production of more carbohydrate [33]. The amylase produced during malting eventually acted on the stored carbohydrate reducing them to maltodextrin molecules and low molecular weight sugars [34], hence the decrease. The low molecular weight sugars produced were then utilized to produce energy required to drive biochemical processes occurring in the grain during germination [28].
The decrease in ash content did not correlate with increase in minerals such as Zn, Fe, Mn and Mg on certain periods of malting, particularly on the third day. This is probable because only two of the major minerals were selected, the rest are trace minerals. Therefore, this difference could be augmented with the presence of other major minerals. Zinc is a component of several enzymes whose demand may have increased during the malting period due to its incorporation to form various enzymes such as polyketide synthase, carbonic anhydrase etc. [35, 36], hence facilitating growth, respiration of embryo and cell constituents’ development during germination [37]. This could be seen on the second day when there was a drop in Zn levels hence corroborating the above statement. Howbeit, it was observed that zinc content increased at the third and fifth day of malting may be as a result of decreased phytate content which may have been partially degraded by endogenous phytase [38]. This trend is also reflected with other minerals such as Fe, Mn and Mg including Ca and Cu that phytate also has a strong binding affinity with [39-41]. The increase in iron content was in accordance with earlier reports on pearl millet by Suma et al. [42]. The high increase especially at the third day of malting may be attributed to decrease in tannin and phytate contents which may have been degraded by endogenous tannase and phytase with eventual leaching into the soaking water [43].
Iron plays critical role in DNA synthesis, respiration and photosynthesis [44]. In plants it is necessary in the synthesis of chlorophyll and maintenance of chloroplast structure and function [44]. Iron is an essential component of haemoglobin and myoglobin required for blood formation. Its deficiency causes anaemia in infants, pregnant women and the elderly with other related diseases [45]. It is important in immune function, cognitive performance and temperature regulation [45]. The calcium content decreased at the second day of malting which may be due to utilization of its reserve for growth and structural development of the plant [46]. At the third day of malting, increased calcium level was probably brought about by the degradation of oxalate and phytate respectively by oxalate decarboxylase and phytase, thereby releasing calcium for plant use [47, 48]. Calcium also plays the role of an intracellular messenger in the cytosol of plants [49]. It performs several roles in human health such as building of strong bones and teeth (proper level over time prevents osteoporosis), muscle contraction, blood clothing, nerve impulse transmission, regulation of heart beat and fluid balance within cells [50].
Manganese plays important role in chlorophyll production and electron transport during photosynthesis and activates several enzymes involved in plant metabolism [51]. It is important in normal human development, metabolism and the antioxidant system of the body, however, excessive exposure or intake may lead to a neurodegenerative disorder [52]. Magnesium is the central atom of chlorophyll and thus is considered critical for plant development and photosynthesis a role similar to manganese [53]. In human health, magnesium is critical as it governs the activity of hundreds of enzymes including about 80% of known metabolic functions [54]. It is also associated with calcium and potassium transport, cell signalling, energy metabolism, genome stability, DNA repair and replication [54].
Though the overall acceptability of the composite tilted towards preference for the non-malted sorghum in the sorghum-soy composite, the chemical analysis showed that the sensory evaluation alone may not be sufficient to determine the beneficial aspects of the sorghum-soy composite as the malted sorghum presents better mineral value, fibre content and rich phytochemicals with reduced antinutritional agents such as phytate, oxalate and tannins. This is particularly observable on the third day of malting. There is also a rich profile of macronutrients in the malted sorghum in sorghum-soy composite flour.
4.2 Conclusion
This work presents possible evidence that malting of sorghum improves on the nutritional content of sorghum-soy composite flour. There was improved mineral and fibre content of the composite with malting and a demonstration of the potential of the malting process in reducing anti-nutrients and increasing the presence of rich phytochemicals. The malting process of cereals including sorghum is largely dependent on the end product desired. Depending on the target needs, the different days of malted sorghum in sorghum-soy composite flour could be blended to obtain a composite that will present a much better nutritional composition. Therefore, the supplementation or complementation of malted sorghum with soy (which is rich in quality proteins) should be advocated for as one of the vital ways of improving nutrient content in cereals aimed at addressing malnutrition in certain groups of local communities, especially of developing countries.
Conflict of Interest
The authors declare that there is no conflict of interest.
References
- Bolarinwa IF, Olaniyan SA, Adebayo LO, et al. Malted Sorghum-Soy Composite Flour: Preparation, Chemical and Physico-Chemical Properties. Journal of Food Processing Technology 6 (2015): 467.
- Mohapatra D, Patel AS, Kar A, et al. Effect of different processing conditions on proximate composition, antioxidants, anti-nutrients and amino acid profile of grain sorghum. Food Chemistry 271 (2019): 129-135.
- Adebo OA. African Sorghum-Based Fermented Foods: Past, Current and Future Prospects. Nutrients 12 (2020): 1111.
- Chatterjee C, Gleddie S, Xiao CW. Soybean bioactive peptides and their functional properties. Nutrients 10 (2018): 1211.
- Abubakar N, Atiku MK, Alhassan AJ, et al. An assessment of micronutrient deficiency: A comparative study of children with protein-energy malnutrition and apparently healthy controls in Kano, Northern Nigeria. Tropical Journal of Medical Research 20 (2017): 61-65.
- De Jager I, Borgonjen-van den Berg KJ, Giller KE, et al. Current and potential role of grain legumes on protein and micronutrient adequacy of the diet of rural Ghanaian infants and young children: using linear programming. Nutrition Journal 18 (2019): 12.
- Sharma N, Bhatia S, Chunduri V, et al. Pathogenesis of celiac disease and other gluten related disorders in wheat and strategies for mitigating them. Frontiers of Nutrition 7 (2020): 6.
- Brouns F, van Rooy G, Shewry P, et al. Adverse Reactions to Wheat or Wheat Components. Comprehensive Reviews in Food Science and Food Safety 18 (2019): 1437-1448.
- Ashaba S, Rukundo GZ, Beinempaka F, et al. Maternal depression and malnutrition in children in southwest Uganda: a case control study. BMC Public Health 15 (2015): 1303.
- Awadalkareem AM, Mustafa AI, Tinay AH. Protein, mineral content and amino acid profile of sorghum flour as influenced by soybean protein concentrate supplementation. Pakistan Journal of Nutrition 7 (2008): 475-479.
- Taylor JRN, Emmambux MN. Gluten-free cereal products and beverages. In Eds.: Arendt EK, Bello FD. Gluten-Free Foods and Beverages from Millets. Elsevier: Amsterdam, The Netherlands (2018): 119-148.
- Poquette NM, Gu X, Lee S. Grain sorghum muffin reduces glucose and insulin responses in men. Food and Function 5 (2014): 894-899.
- Adebo OA, Gabriela Medina-Meza I. Impact of fermentation on the phenolic compounds and antioxidant activity of whole cereal grains: A mini review. Molecules (Basel, Switzerland) 25 (2020): 927.
- Banusha S, Vasantharuba S. Effect of Malting on Nutritional Contents of Finger millet and Mung bean. American Journal of Agriculture and Enviromental Science 13 (2013): 1642-1646.
- Okpalanma EF. Production of Malt-based Sugar Syrup from Enzymatic Hydrolysis of Malted Sorghum and Millet Grains. Asian Food Science Journal 14 (2020): 1-17.
- Adebo OA, Njobeh PB, Adebiyi JA, et al. Fermented pulse-based foods in developing nations as sources of functional foods. In Ed.: Hueda MC. Functional Food-Improve Health through Adequate Food. INTECH: Rijeka, Croatia (2017): 77-109.
- Hallen E, Ibanoglu S, Ainsworth P. Effect of fermented/germinated cowpea flour addition on the rheological and baking properties of wheat flour. Journal of Food Engineering 63 (2004): 177-184.
- Ndife J, Abdulraheem LO, Zakari UM. Evaluation of the nutritional and sensory quality of functional breads produced from whole wheat and soya bean flour blends. African Journal of Food Science 5 (2011): 466 -472.
- Sharara AI, Daroub H, Georges C, et al. Sensory characteristics of bowel cleansing solutions. World Journal of Gastrointestinal Endoscopy 8 (2016): 508-516.
- Association of official Analytical Chemists. Official methods of Analysis of AOAC International. 18th Washington D.C (2010).
- Food energy-methods of analysis and conversion factors. FAO Food and Nutrition paper 77, Food and Agriculture Organisation of the United Nations, Rome (2003).
- Desai DA, Kulkarni SS, Sahoo AK, et al. Effect of Supplementation of Malted Ragi Flour on the Nutritional and Sensorial Quality Characteristics of Cake. Advanced Journal of Food Science and Technology 2 (2010): 67-71.
- Butnariu M. An analysis of sorghum halepense’s behaviour in presence of tropane alkaloids from Datura stramonium extracts. Chemistry Central Journal 6 (2012): 75.
- Asuk AA, Agiang AM, Kayode D, et al. The biomedical significance of phytochemical, proximate and mineral compositions of the leaf, stem bark and root of Jatropha curcas. Asian Pacific Journal of Tropical Biomedicine 5 (2015): 650-657.
- Xiong Y, Zhang P, Warner RD, et al. Sorghum grain: from genotype, nutrition, and phenolic profile to its health benefits and food applications. Comprehensive Reviews in Food Sciences and Food Security 18 (2019): 2025-2046.
- Louassini M, Foulquie MR, Benitez R, et al. Activity of Key Enzymes in glucose Catabolism during the growth and Metacytogenesis of Leismania Infatum Promastigotes. Journal of Parasitology 85 (1999): 595-602.
- Ogbonna AC, Abuajah CI, Ide EO, et al. Effect of Malting conditions on the Nutritional and Antinutritional Factors of Sorghum Grist. Food Technology 36 (2012): 64-72.
- Singh E, Jain PK, Sharma S. Effect of different house hold processing on nutritional and antinutritional factors in Vigna aconitifolia and Sorghum bicolour (L.) Moech seeds and their products. Journal of Medical Nutrition and Nutraceuticals 4 (2015): 95-100.
- Aluge OO, Akinola SA, Osundahunsi OF. Effect of Malted Sorghum on Quality Characteristics of Wheat-Sorghum-Soybean Flour for Potential Use in Confectionaries. Food and Nutrition Sciences 7 (2016): 1241-1252.
- Homaei AA, Sariri R, Vianello F, et al. Enzyme immobilization: an update. Journal of Chemical Biology 6 (2013): 185-205.
- Benincasa P, Falcinelli B, Lutts S, et al. Sprouted grains: A comprehensive review. Nutrients 11 (2019): 421.
- Muyanja CMBK, Kikafunda JK, Narvhus JA, et al. Production methods and composition of Bushera: A Ugandan traditional fermented cereal beverage. African Journal of Food, Agriculture, Nutrition and Development 3 (2003): 10-19.
- Kondrashov FA, Koonin EV, Morgunov IG, et al. Evolution of glyoxylate cycle enzymes in Metazoa: evidence of multiple horizontal transfer events and pseudogene formation. Biology Direct 1 (2006): 31.
- Oluwole OB, Kosoko SB, Owolabi SO, et al. Development and Production of High Protein and Energy Density Beverage from Blends of Maize (Zea mays), Sorghum (Sorghum bicolour) and Soy beans (Glycine max) for school Aged Children: Effect of Malting period on Selected Proximate Parameters and Sensory qualities of Developed Beverages. International Journal of Applied Science and Technology 2 (2012): 285 -292.
- Abe I, Abe T, Wanibuchi K, et al. Enzymatic formation of quinolone alkaloids by plant type III polyketide synthase. Organic Letters 8 (2006): 6063-6065.
- Castillo-Gonzalez J, Ojeda-Barrios D, Hernanadez-Rodriguez A, et al. Zinc metalloenzymes in plants. Interciencia 43 (2018): 242-248.
- Elkhier MK, Hamid AO. Effect of malting on the chemical constituents, anti-nutritional factors and ash composition of two sorghum cultivars (Feteri and Tabati). Journal of Agriculture and Biological Sciences 4 (2008): 500-504.
- Afify AE, El-Beltagi HS, Omran AA. Biovailability of Iron, Zinc, Phytate and Phytate Activity during Soaking and Germination of White Sorghum Varieties. PLOS ONE 6 (2011): e25512.
- Lope HW, Leenhardt F, Coudray C, et al. Minerals and phytic acid interactions: Is it a real problem for human nutrition? International Journal of Food Science and Technology 37 (2002): 727-739.
- Nissar J, Ahad T, Naik HR, et al. Phytic acid: as antinutrient or nutraceutical. Journal of Pharmacognosy and. Phytochemistry 6 (2017): 1554-1560.
- Abdulwaliyu I, Arekemase SO, Adudu JA, et al. Investigation of the medicinal significance of phytic acid as an indispensable anti-nutrient in diseases. Clinical Nutrition Experimental 28 (2019): 42-61.
- Suma PF, Urooj A. Influence of germination on bioaccessible iron and calcium in pearl millet (Pennisetum typhoideum). Journal of Food Science and Technology 51 (2014): 976-981.
- Kayode AP, Hounhougigan JD, Nout MJ. Impact of brewing process operation on phytate, phenolic compounds and in vitro solubility of iron and zinc in opaque sorghum beer. LWT Food Science and Technology 40 (2007): 834-841.
- Rout GR, Sahoo S. Role of iron in plant growth and metabolism. Reviews in Agricultural Science 3 (2015): 1-24.
- Abbaspour N, Hurrell R, Kelishadi R. Review of iron and its importance for human health. Journal of Research in Medical Sciences 19 (2014): 164-174.
- Hepler PK. Calcium: Acentral Regulator of Plant growth and Development. American Society of Plant Biologists 17 (2005): 2142-2155.
- Chakraborty N, Ghosh R, Ghosh S, et al. Reduction of Oxalate Levels in Tomato Fruit and Consequent Metabolic Remodelling Following Over expression of a Fungal Oxalate Decarboxylase. Plant Physiology 162 (2013): 364-378.
- Gupta RK, Gangoliya SS, Singh NK. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. Journal of Food Science and Technology 52 (2015): 676-684.
- White PJ, Broadley MR. Calcium in plants. Annals of Botany 92 (2003): 487-503.
- Pravina P, Sayaji D, Avinash M. Calcium and its role in human body. International Journal of Research in Pharmaceutical and Biomedical Sciences 4 (2013): 659-668.
- Mousavi SR, Shahsavari M, Rezaei M. A general overview of manganese (Mn) importance for crops production. Australian Journal of Basic and Applied Sciences 5 (2011): 1799-1803.
- Avila DS, Puntel RL, Aschner M. Manganese in health and disease. Metal Ions of Life Sciences 13 (2013): 199-227.
- Wang Y, Hua X, Xu J, et al. Comparative genomics revealed the gene evolution and functional divergence of magnesium transport families in Saccharum. BMC Genomics 20 (2019): 83.
- Workinger JL, Doyle RP, Bortz J. Challenges in the diagnosis of magnesium status. Nutrients 10 (2018): 1202.


 Impact Factor: * 3.8
Impact Factor: * 3.8 Acceptance Rate: 77.96%
Acceptance Rate: 77.96%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 