Development and Management of Massive Subcutaneous Metastases with Unusual Clinical Course from Uterine Leiomyosarcoma
Filippini DM1, Nuvola G1*, Nannini M1, Saponara M1, Cammelli S2, De Paolis M3, Perrone AM4, De Iaco P4, De Leo A5, Astolfi A6, Pantaleo MA7
1Division of Medical Oncology, IRCCS University Hospital of Bologna, Bologna, Italy
2Radiotherapy Unit, University Hospital of Bologna, Bologna, Italy
3Orthopedic Unit, University Hospital of Bologna, Bologna, Italy
4Division of Oncologic Gynecology, IRCCS University Hospital of Bologna, Bologna, Italy
5Pathology Unit, University Hospital of Bologna, Bologna, Italy
6Department of Morphology, Surgery and Experimental Medicine, University of Ferrara, Ferrara, Italy
7Department of Specialized, Experimental and Diagnostic Medicine, University of Bologna, Bologna, Italy
*Corresponding Author: Nuvola Giacomo, Department of Specialized, Experimental and Diagnostic Medicine, Sant'Orsola-Malpighi Hospital, University of Bologna, Bologna, Italy
Received: 31 May 2021; Accepted: 01 July 2021; Published: 08 November 2021
Article Information
Citation: Filippini DM, Nuvola G, Nannini M, Saponara M, Cammelli S, De Paolis M, Perrone AM, De Iaco P, De Leo A, Astolfi A, Pantaleo MA. Development and Management of Massive Subcutaneous Metastases with Unusual Clinical Course from Uterine Leiomyosarcoma. Journal of Cancer Science and Clinical Therapeutics 5 (2021): 419-427.
View / Download Pdf Share at FacebookAbstract
Uterine leiomyosarcoma (U-LMS) is the most common histotype among all primary uterine sarcomas. The lung, peritoneum, pelvic or para-aortic lymph nodes, bone and liver are the most common metastatic sites involved. However, some case series of atypical sites of metastasis such as heart, brain and different endocrine glands have been described. Here we report the clinical presentation and therapeutic management of two patients affected by U-LMS with a very atypical behavior, characterized by soft tissue metastases with an impressive and massive enlargement during chemotherapy despite a good control of the other sites of disease.
Keywords
<p>Uterine leiomyosarcoma; Subcutaneous metastases; Unusual course; Chemotherapy; Radiotherapy</p>
Article Details
1. Introduction
Uterine leiomyosarcoma (U-LMS) is a rare tumor, accounting for about 1% of all uterine malignancies, but it represents the most common malignant gynecological mesenchymal tumor [1, 2]. For patients with localized disease, the standard of treatment is en bloc total hysterectomy [3]. Adjuvant radiotherapy or chemotherapy is not, so far, recommended, but it could be evaluated in selected cases [4, 5]. For women with advanced unresectable U-LMS, the administration of chemotherapy is the first choice of treatment. The systemic therapeutic regimens include doxorubicin associated or not to dacarbazine as first line. Other active agents are gemcitabine and docetaxel (alone or combined), trabectedin, dacarbazine, pazopanib and, rarely, endocrine therapy [6, 7].
Common sites of metastases from U-LMS are lungs, pelvic or para-aortic lymph nodes, bone, skin and soft tissues [8, 9]. Case reports of atypical site of metastasis such as heart, brain and different endocrine glands have been described [10]. Prompt detection and histological diagnosis of skin and soft tissue metastases from U-LMS are particularly important, as these lesions could be often misdiagnosed as de novo primary tumors compromising the appropriate management and the prognosis. Here, we describe the clinical manifestation and therapeutic management of isolated subcutaneous soft tissue metastatic lesions from U-LMS with atypical and exceptional behavior characterized by an impressive and massive enlargement during chemotherapy although all the other metastases were stable or only mild progressing. Our case series highlights the importance of a prompt and correct diagnosis of soft tissues metastatic lesions, emphasizes the role of locoregional treatment in metastatic U-LMS and could guide further investigation in this context.
2. Case Report
2.1 Case presentation 1
In September 2017, after multiple recurrent methrorragic events, a 53-year-old woman underwent laparotomy with total bilateral hysteroannessiectomy and multiple lymph nodes sampling. The histology report showed a U-LMS, grade 3 according to Fédération Nationale des Centres de Lutte Contre le Cancer (FNCLCC) grading system, with vascular, lymphatic invasion and massive necrosis. Positivity for desmin, p53 and negativity for CD10 and Bcl-2 were revealed by immunohistochemistry (Figure 1A). The stage was pT1 pN0 M0 using the International Federation of Gynecology and Obstetrics (FIGO) system. After surgery, the patient suffered from CMV-related enterocolitis and therefore, she did not receive adjuvant chemotherapy. In July 2018, she presented a sudden onset of a large right leg painful subcutaneous lesion involving the front of the leg, a biopsy confirmed the leiomyosarcoma origin and the computed tomography (CT) scan showed a large subcutaneous mass (Figure 1B, Figure 2A). A CT scan showed also the appearance of multiple pulmonary lesions (Figure 3A).
In August 2018, she therefore started a first line therapy with doxorubicin and olaratumab until February 2019 when, after pulmonary progressive disease (PD), she started a new line of therapy with doxorubicin (the last two courses before the total cumulative dose) and dacarbazine. In June 2019, a CT scan showed a massive PD of the right leg soft tissue metastasis measuring 17 cm, but stable disease (SD) in the lung (Figure 2B and 3B). Physical examination revealed a very painful right leg mass of about 20x8 cm. The patient underwent radiotherapy of the right leg (40 Gy in 4 fractions on the central area and 20 Gy in 4 fractions peripherally) with clinical benefit reducing the pain, then followed by local excision. The histological examination showed metastases from primary U-LMS, with more than 60% of necrotic component. The patient is currently alive, without evidence of soft tissue disease in the leg, but due to lung metastases, she is still receiving chemotherapy with gemcitabine and docetaxel.
2.2 Case presentation 2
In September 2016, after vaginal bleeding, a 55-year-old woman performed a transvaginal ultrasound showing the presence of an intrauterine mass. A biopsy was consistent with the diagnosis of U-LMS with positivity for desmin and p53 and negativity for CD10 and Bcl2 at immunohistochemistry (Figure 4 A). A staging CT scan showed pulmonary, hepatic, soft tissue and bone lesions. The stage of the disease was IV (M1). She started a first line treatment with Doxorubicin and Dacarbazine and after 3 months, due to persistent massive vaginal bleeding, she underwent a total laparotomy bilateral hysterectomy, bilateral adnexectomy, omentectomy, removal of peritoneal nodules and multiple peritoneal sampling. The histological examination revealed U-LMS, grade 3 according to FNCLCC, with massive necrosis, without vascular invasion. All the peritoneal sampling were negative for malignant cells (Figure 4 B). After surgery, chemotherapy with doxorubicin and dacarbazine followed by dacarbazine alone was continued until November 2017. A subsequent CT scan showed the appearance of peritoneal nodules and a new secondary bone lesion at the vertebral column. The patient started chemotherapy with gemcitabine and docetaxel, followed by gemcitabine alone for hematological toxicity.
In October 2018, the patient presented to the emergency department with lower limb weakness and dysesthesia and she was diagnosed with medullary compression due to the expansive bone lesion. She subsequently underwent medullary decompression and surgical stabilization. In March 2019, because of further PD, she started pazopanib but, after 3 months of treatment, a CT scan showed a mild pulmonary and hepatic PD, in contrast with the appearance of a rapidly increasing left axillary lesion measuring 13 cm (compared with the measure of 2 cm at the previous imaging 6 months before) which was resistant to standard pain care (Figure 5 A and B). After multidisciplinary evaluation, radiotherapy and then surgery were proposed for the high risk of bleeding but due to worsening of the patient's clinical conditions, the best supportive care was recommended.
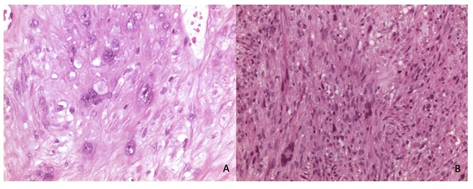
Figure 1A and B: Histologic appearance (hematoxylin and eosin, H&E) of leiomyosarcoma on hysterectomy (B, x400) and on metastasis biopsy (A, x100).
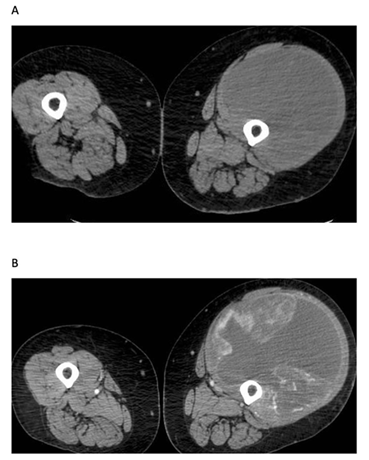
Figure 2A and B: CT scan showing a right leg subcutaneous lesion in July 2018 (A) and in June 2019 (B).
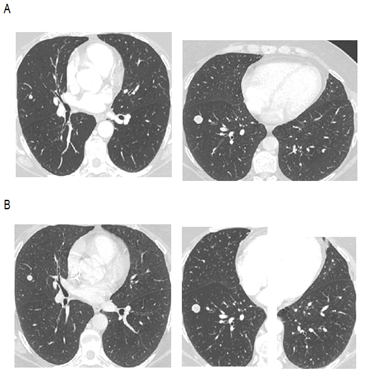
Figure 3A and B: CT scan showing a pulmonary metastasis in July 2018 (A) and in June 2019 (B).
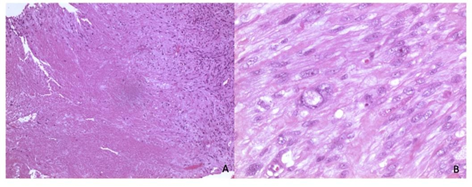
Fig 4A and B: Histologic appearance (hematoxylin and eosin, H&E) of leyiomiosarcoma on pre- operative biopsy (A, x40) and hysterectomy (B, x400).
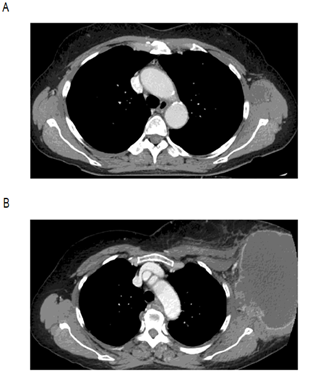
Figure 5A and B: CT scan showing a left axillary lesion measuring 2 cm (A) rapidly enlarging in 6 months measuring 13 cm (B).
3. Discussion
Here we describe two cases of patients with documented soft tissue metastases from U-LMS with an atypical clinical course, both characterized by the rapid appearance of enlarged and painful soft tissue lesions, seriously affecting their quality of life. Given the rarity of the disease, the unusual clinical presentation and therapeutic management deserve to be discussed. Leiomyosarcomas are rare malignant tumors that could arise in any location, although most of them are localized in retroperitoneal/intra-abdominal and pelvic sites, mainly in the uterus. Compared with other types of uterine cancers, LMS is associated with a high risk of recurrence and death, regardless of stage at presentation and it represents about 30-40% of all uterine sarcomas [1].
Although hysterectomy is the primary treatment for U-LMS, approximately 70% of patients will develop a recurrence within an average of 8 to 16 months after the initial diagnosis. Recurrent U-LMS is still difficult to manage and the prognosis is still poor. Therapeutic options include surgery, radiation therapy and chemotherapy encompassing doxorubicin and dacarbazine, gemcitabine-docetaxel, ifosfamide, trabectedin, pazopanib and temozo-lomide [6, 7, 11-17]. Uterine-LMS most commonly meta-stasizes to the lung (67.7%), cranial/intracranial sites (16.2%), skin/soft tissues (15.3%), and bone (13.8%). Other very rare sites include thyroid, salivary glands, heart, liver, pancreas, adrenal gland, bowel, and breast. When the lung is the only metastatic site, patients could be treated with metastasectomy obtaining a significantly better survival than those who did not undergo metastasectomy (p=0.003). There is also a wide range of time intervals to first metastases, highlighting the need of long-term follow-up, and appropriate diagnostic imaging. Lung and upper peritoneum metastases were often diagnosed earlier in the progression of U-LMS. Skin and soft-tissue metastases in U-LMS account for about 15% of cases and are particularly important to be accurately diagnosed, as they are often misinterpreted initially as de novo primary tumors [9, 18, 19]. Bartosch et al. described 20 cases (15.3%) of 130 metastatic U-LMS patients with skin/soft tissue metastases and 13 of them showed soft tissue metastases as first sites of metastases; nine patients out of 20 underwent metasta-sectomy. The authors found no significant differences in time to metastases development (p=0.06) [9]. In this retrospective study, lung is the most common metastatic site, followed by bone, cranial/intracranial and skin/soft- tissue metastasis.
A physical examination, such as skin, soft tissue, head and neck should be considered in patients with U-LMS to detect some of these metastatic sites. To the best of our knowledge there is no description of enlarging and rapidly progressing soft tissue metastases with impressive quality of life deterioration. In our reports, the masses originating from the soft tissues have exploded in a short time, presenting huge, hard, protruding towards the outside, very painful and giving functional limitations. Curiously, the masses responded to systemic treatment less than the other metastatic sites. In fact, in both cases, while the other metastatic sites were stable under chemotherapy, the soft tissue lesions rapidly progressed. While the second patient was not suitable to local treatment on the axillary lesion due to worsening clinical conditions, the first patient was treated with palliative radiotherapy obtaining a clinical and radiological response and benefit for pain reduction and better quality of life. Therefore, in the management of this uncommon clinical presentation involving a multidisc-ciplinary team (surgeons, radiotherapists, pain therapies supporters, medical oncologists) is fundamental to integrate loco-regional treatments with the systemic chemotherapy for metastatic sites that could be critical. Moreover, it’s advisable to perform, at reference centers, the histological assessment of soft tissues metastatic lesions with an atypical behavior to avoid misdiagnosis and promptly start the most correct therapeutic management.
4. Conclusion
In summary, soft tissue metastases from U-LMS need to be rapidly recognized and diagnosed through accurate physical, imaging and histopathological exams to be correctly managed. Moreover, they may present an unpredictable clinical course compared to the other metastatic sites. In fact, they could increase rapidly despite the systemic treatments, and a prompt, correct and multidisciplinary evaluation that can lead to a rapid loco-regional control could strongly benefit the patients’ quality of life avoiding serious complications and probably adding a survival advantage.
Conflicts of Interest
The authors declare no conflict of interest.
Declaration
The authors have no support or funding.
References
- D'Angelo E, Prat J. Uterine sarcomas: a review. GynecolOncol 116 (2010): 131-139.
- Abeler VM, Røyne O, Thoresen S, et al. Uterine sarcomas in Norway. A histopathological and prognostic survey of a total population from 1970 to 2000 including 419 patients. Histopathology 54 (2009): 355-364.
- Giuntoli RL, Metzinger DS, Di Marco CS, et al. Retrospective review of 208 patients with leiomyosarcoma of the uterus: prognostic indica-tors, surgical management, and adjuvant therapy. Gynecol Oncol 89 (2003): 460-469.
- Reed NS, Mangioni C, Malmström H, et al. Phase III randomised study to evaluate the role of adjuvant pelvic radiotherapy in the treatment of uterine sar-comas stages I and II: an European Organisation for Research and Treatment of Cancer Gynaecological Cancer Group Study. Eur J Cancer 44 (2008): 808-818.
- Hensley ML, Enserro D, Hatcher H, et al. Adjuvant Gemcitabine Plus Docetaxel Followed by Doxorubicin Versus Observation for High-Grade Uterine Leiomyosarcoma: A Phase III NRG Oncology/Gynecologic Oncology Group Study. J Clin Oncol 36 (2018): JCO1800454.
- Hensley ML, Blessing JA, Mannel R, et al. Fixed-dose rate gemcitabine plus docetaxel as first-line therapy for metastatic uterine leiomyosarcoma: a Gynecologic Oncology Group phase II trial. Gynecol Oncol 109 (2008): 329-334.
- Muss HB, Bundy B, DiSaia PJ, et al. Treatment of recurrent or advanced uterine sarcoma. A randomized trial of doxorubicin versus doxorubicin and cyclophosphamide (a phase III trial of the Gynecologic Oncology Group). Cancer 55 (1985): 1648-1653.
- Moskovic E, MacSweeney E, Law M, et al. Survival, patterns of spread and prognostic factors in uterine sarcoma: a study of 76 patients. Br J Radiol 66 (1993): 1009-1015.
- Bartosch C, Afonso M, Pires-Luís AS, et al. Distant Metastases in Uterine Leiomyosarcomas: The Wide Variety of Body Sites and Time Intervals to Metastatic Relapse. Int J GynecolPathol 36 (2017): 31-41.
- Artioli G, Borgato L, Calamelli S, et al. Unusual cardiac metastasis of uterine leiomyosarcoma: case report and literature review. Tumori 102 (2016): 10.5301/tj.5000498.
- Miller BE, Blessing JA, Stehman FB, et al. A phase II evaluation of weekly gemcitabine and docetaxel for second-line treatment of recurrent carcino-sarcoma of the uterus: a gynecologic oncology group study. Gynecol Oncol 118 (2010): 139-144.
- Sutton G, Blessing J, Hanjani P, et al. Gynecologic Oncology Group. Phase II evaluation of liposomal doxorubicin (Doxil) in recurrent or advanced leiomyosarcoma of the uterus: a Gynecologic Oncology Group study. Gynecol Oncol 96 (2005): 749-752.
- Monk BJ, Blessing JA, Street DG, et al. A phase II evaluation of trabectedin in the treatment of advanced, persistent, or recurrent uterine leiomyo-sarcoma: a gynecologic oncology group study. Gynecol Oncol 124 (2012): 48-52.
- Sutton G, Blessing JA, Malfetano JH. Ifosfamide and doxorubicin in the treatment of advanced leiomyosarcomas of the uterus: a Gynecologic Oncology Group study. Gynecol Oncol 62 (1996): 226-229.
- Van der Graaf WT, Blay JY, Chawla SP, et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet 379 (2012): 1879-1886.
- Demetri GD, von Mehren M, Jones RL, et al. Efficacy and Safety of Trabectedin or Dacarbazine for Metastatic Liposarcoma or Leiomyosarcoma After Failure of Conventional Chemotherapy: Results of a Phase III Randomized Multicenter Clinical Trial. J Clin Oncol 34 (2016): 786-793.
- Miller DS, Blessing JA, Kilgore LC, et al. Phase II trial of topotecan in patients with advanced, persistent, or recurrent uterine leiomyosarcomas: a Gynecologic Oncology Group Study. Am J Clin Oncol 23 (2000): 355-357.
- Giuntoli RL, Gostout BS, DiMarco CS, et al. Diagnostic criteria for uterine smooth muscle tumors: leiomyoma variants associated with malig-nant behavior. J Reprod Med 52 (2007): 1001-1010.
- Lee CH, Turbin DA, Sung YC, et al. A panel of antibodies to determine site of origin and malignancy in smooth muscle tumors. Mod Pathol 22 (2009): 1519-1531.


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 