Hair Growth and Restoration by Transcriptional Control of Tissue Regeneration in Cancer Recovery Process by Huaier
Manami Tanaka1*, Tomoo Tanaka1, Xiaolong Zhu2, Fei Teng2*, Hong Lin2, Zhu Luo3, Ying Pan2,
Sotaro Sadahiro4, Toshiyuki Suzuki5, Yuji Maeda6, Ding Wei7 and Zhengxin Lu8
1Bradeion Institute of Medical Sciences, Co., Ltd., Itado 433-1, Isehara, Kanagawa 259-1145, Japan
2BGI-Shenzhen, Building NO.7, BGI Park, No.21 Hongan 3rd Street, Yantian District, Shenzhen 518083, China
3BGI-Japan, KIMEC Center BLDG. 8F 1-5-2 Minatojima-minamimachi, Chuo-ku, Kobe 650-0047 Japan
4Department of Surgery, Kameda-Morinosato Hospital, 3-1-1 Morinosato, Atsugi, Kanagawa 243-0122, Japan
5Department of Surgery, Oiso Hospital attached to Tokai University School of Medicine, 21-1 Gakkyo, Oiso, Naka-District, Kanagawa 259-0198, Japan
6Department of Surgery, Kanagawa National Hospital, National Hospital Organization, 666-1 Ochiai, Hadano, Kanagawa 257-8585, Japan
7Japan Kampo NewMedicine, Co. Ltd., 2-8-10 Kayaba-Cho, Chuo-Ku, Tokyo 103−0025, Japan
8QiDong Gaitianli Medicines Co. Ltd., Jiangsu Province, China
*Corresponding Author: Manami Tanaka, Bradeion Institute of Medical Sciences, Co., Ltd., Itado 433-1, Isehara, Kanagawa 259-1145, Japan.
Received: 03 August 2022; Accepted: 08 August 2022; Published: 12 August 2022
Article Information
Citation: Manami Tanaka, Tomoo Tanaka, Xiaolong Zhu, Fei Teng, Hong Lin, Zhu Luo, Ying Pan, Sotaro Sadahiro, Toshiyuki Suzuki, Yuji Maeda, Ding Wei and Zhengxin Lu. Hair Growth and Restoration by Transcriptional Control of Tissue Regeneration in Cancer Recovery Process by Huaier. Journal of Cancer Science and Clinical Therapeutics. 6 (2022): 288-316.
View / Download Pdf Share at FacebookAbstract
Objectives: In the process of caner recovery, Huaier simultaneously promotes significant hair growth and restoration as a typical evidence of its efficacy on induced tissue regeneration and repair function. As a series of our clinical research, the molecular basis of hair growth was investigated.
Methods: The blood samples from volunteer cancer patients and normal healthy controls were collected and produced by next generation sequencing, and searched for significant alterations in genetic level specifically focused on all the signaling networks related to hair growth and restoration, and also related to Huaier effects on tissue regeneration and repair. The results were compared between cancer patients with or without conventional chemotherapy, and normal control group with or without Huaier treatment.
Results: We identified Hair growth after conventional chemotherapy was a first and significant observation, and that in dose-dependent manner. The molecular basis of hair growth was identified as the activation of Hedgehog signaling pathway, which initiates systematic tissue repair not only in cancer lesions together with the cure of skin problems and perturbed inter/ intra neural signal transfer. Huaier efficacy on the activation of Hedgehog signaling pathway was followed by the subsequent inhibition at 3-6 months interval. This inhibitory effects seemed to contribute to the prevention of possible complications in the process of cancer recovery, such as recurrence and metastasis. Small nuclear RNA analysis revealed that the regulation of tissue regeneration, including hair growth were based on the miRNA-mediated transcriptional control of the pluripotency of stem cells.
Conclusion: The present study thus provides a simple and clear proof of Huaier effects on tis
Keywords
<p>Huaier (Trametes Robiniophila Murr); Hedgehog Signaling Pathway; miRNA-Medicated Transcription Control; Tissue Regeneration and Repair; Cancer Stem Cell</p>
Article Details
1. Introduction
In the long process of cancer recovery, it is as much important to recognize the improvement by each individual patient day by day, together with the results obtained by clinical examinations at the hospitals and clinics. Each time of looking into the mirror to examine physical appearance, checking body weight by scale, shampooing the hair at bath or by hair dressers, the self-confirmation of recovery none the less provides positive supportive effects mentally and physically. Conventional chemotherapy usually causses hard side effects such as nausea, vomiting, insomnia, and also easily leads to depression by the perturbed intra/inter neural signal transfer.
Administration of kinase regulator Huaier (Trametes robiniophila murr) has been reported significantly effective on the broad range of cancers (Chinese administration license No. Z-20000109) [1-12]. However, the recovery process requires years of time to confirm the consequence without recurrence and/or metastasis.
The advantage of Huaier therapy is that: 1) no side effects or toxicity, and 2) each patient can confirm the recovery day by day as written above. These signs are also easily recognized by family, friends, and colleagues, which mentally encourages patients toward the complete remission, so to be the real cure from the disease.
Although medical doctors usually pursuit and monitor the results of clinical blood tests and CT or MRI image analysis, but the individual recognition of recovery process in daily life has as much impact. Huaier effects on tissue regeneration and repair has been emphasized in the previous reports through a series of our clinical research [1-12], which seemed to contribute more than the effects on cancer cell-specific cell death. The inhibition of cancer progression observed in Huaier therapy depends on the transcriptional regulation of pluripotency of stem cell [1, 4, 11].
Through our clinical research from 2018, we have reported sequentially about the molecular basis of anti-cancer effects of Huaier, and that the results on the minimum essential rescue of disruption and dysfunctions in the integrated signaling pathway networks by miRNA-mediated transcriptional control [1-12]. Hair growth and restoration is a typical proof of tissue regeneration. We have had enough molecular information by total RNA sequencing and subsequent thorough analysis of translation and transcription processes to identify the molecular basis on hair growth [13-17]. With the results from the previous publications [18-21], we investigated KEGG characterization analysis on Hedgehog signaling pathway [17]. Together with steady process of cancer recovery, the significant up-regulation of responsible elements in Hedgehog signaling pathway were observed among the cancer patients who had massive hair growth, and more importantly, followed by the down-regulation after 3-6 month interval to reach to the normal level. The activation in Hedgehog signaling pathway was only one part of drastic and extensive up-regulations identified in transcriptomes (DEGs; differentially expressed genes), resulted from the drastic genomic flexibility and plasticity of each patient induced by Huaier administration [1, 11]. Thus, Huaier functioned as a regulator, not an activator or inhibitor.
The present study successfully identified the Huaier effects on regulating Hedgehog signaling pathway functions [22, 23] within 1-3 months after administration, and that in a dose dependent manner. Huaier administration initiated massive hair growth with an increase of body weights t over 5 Kg /month, with healthy appetite, and totally cure of skin lesions as shown in the previous report [12]. Although Huaier has an effect to rewind the life time (anti-ageing) [1-12] as an effect as (tyrosine) kinase regulator [2, 8], it is noteworthy to observe the hair restoration with black hair (typical for Japanese population) even over the age of 70’s.
We have demonstrated the relationship of hair growth and restoration and Hedgehog signaling pathway cascade in prostate cancer [1, 10], basal cell carcinoma[ 1, 11], lung cancer [1,2], lung metastasis [1, 7, 9 ], and meningioma [1, 8, 11]. There has been a long history to investigate Hedgehog signaling pathway [18-33], especially aimed for development of its controlling agents [25-33]. Recently it has been reported that inhibition of Hedgehog signaling pathway function was the cause hair loss [21], and the present study provided a solution and rescue to this problem in cancer patients.
However, normal healthy individual did not have any significant alterations or up/down-regulation in any responsible elements in Hedgehog signaling pathway, which seems that Huaier did not affect to normal healthy conditions, with no requirement for functional alterations. Up-regulation of SMO was simultaneously observed in many patients with massive hair growth.
Presumably Huaier compensation to Hedgehog signaling pathway and resulted hair growth will be most beneficial to the pre-cancer stage patients, such as prostate cancer in situ, or the patients with unexpected multiple cysts production in liver and/or pancreas by CT or MRI image analysis.
2. Materials and Methods
2.1 Sample Collection and Medical Ethics Policy
Blood samples were taken from 8 patients, two of them were healthy volunteers as controls. The clinical characteristics of patients like sex/age/cancer origin, complications, and additional therapy to Huaier administration, are summarized in table 1. The present study was strictly conducted according to the guidelines of the Declaration of Helsinki and the principles of good clinical practice. Written informed consent was obtained from the patients. This clinical research was applied according to the Consolidated Standards of Clinical Research Trials guidelines and was registered on the Japanese Medical Association (ID: JMA-IIA00335, 1st February 2018). The project has been strictly conducted with a review by the ethics committee consisted by the experts on Medicine, Nursing, Laws, Pharmaceutics and Business Community (first committee held on 9th February, 2018) [1-12].
2.2 Total RNA Sample QC
The Agilent 2100 Bio analyzer (Agilent RNA 6000 Nano Kit) were used to do the total RNA sample QC. The QC Item contains the RNA concentration, RIN value, 28S/18S and the fragment length distribution [13].
2.3 Library and Sequencing
The obtained peripheral blood samples (28 samples from 8 cancer patients) were designated to be further sequencing. First purifying the poly-A containing mRNA molecules using poly-T oligo-attached magnetic beads. Following purification, the mRNA is fragmented into small pieces using divalent cations under elevated temperature. After removal of rRNA, the RNA was converted into cDNA using reverse transcriptase and random primers. This is followed by second strand cDNA synthesis using DNA Polymerase I and RNase H. These cDNA fragments then have the addition process by A-tailing and the cDNA was amplified. To generate libraries, the amplified products were separated into single strand DNA and cyclized. The RNA-seq libraries were subjected and sequenced with the pair-end option using BGISEQ-500 system at Beijing Genomics Institute (BGI), China [1-13].
We have also performed total non-coding small RNA sequencing on the same platform. First, separate 18-30nt RNA segment by PAGE gel. And add ligation of corresponding adaptors to the RNA 5′ and 3′ ends, the adapter-ligated small RNAs were subsequently transcribed into cDNA, and amplified the product by several cycles. Then reverse extend the RT primer in next step to synthetize strand cDNA and use high-ping polymerase to amplify cDNA, enrich cDNA with both 3' and 5' adaptor. Purified DNA was used for cluster generation and sequencing analysis using the BGISEQ-500 platform.
Total RNA was isolated from the serum samples and was pooled together to construct cDNA Libraries. These libraries were subsequently sequenced throw Illumina HiSeq sequencing with pair-end reads of length 2*100bp according to manufacturer’s instructions as previously described [1-13].
2.4 Data Process and Expression Analysis of RNAseq
The sequencing data were analyzed and filtered using software soapnuke [34]. Clean reads were mapped to the human reference genome GRCh38 with software bowtie2. Then, the gene expression level of each sample was calculated using RSEM [35]. The expressed genes were further analyzed between the groups were detected by software DESeq2 [36], the significant differentially expressed genes (DEGs) were defined with fold-change lager than 2 and p-value smaller than 0.05.
2.5 Data Process and Expression Analysis of miRNA
Low quality sequencing data were removed at first, and align the reads to miRbase database with bowtie2 [37] and calculated the expression level of miRNAs which standardized by TPM. Differentially expressed miRNAs between two samples were screened out strictly based on Poisson distribution. Then, we make multiple hypothesis test correction for the p value of difference test, and to judge the significance of gene expression difference, FDR≤ 0.001 and the absolute value of Log2Ratio≥1 is set as the default threshold. Software miRanda [38] and TargetScan [39] were used to get the target gene of differential expressed miRNA and extract intersection or union of target gene as final prediction result.
2.5 Functional Analysis of Target Genes and DEGs
And to determine the metabolic and signal transduction pathways and their biological functions. Pathway enrichment and GO significance enrichment analysis was performed on the target genes of differentially expressed miRNAs or DEGs by using R package ‘phyper’. For each P-value, we correct for multiple comparisons by controlling the False Discovery Ratio. And the terms for which the FDR was not greater than 0.01 were defined as statistically significantly enriched.
3. Results
3.1 Patient Characterization
The medical characteristics of total 15 volunteers with massive hair growth, out of over 50 patients examined in our clinical research, including 5 healthy controls were summarized in Table 1. All these patients have been introduced in the previous reports [1-12] with their thorough molecular information based on MEGA-data analysis from total RNA sequencing [13-16]. The present study picked up 15 patients by identification of significant hair growth and restoration, first recognized by the patient themselves, together with the improvement of skin lesion, and gain of body weights. The hair growth and restoration were also recognized by their family members, colleagues, and by hair dressers. The improvement of skin lesion and skin conditions was continuously monitored by walk-in clinic (dermatologists’). The most significant hair growth was observed among the patient Nos. 7-9, with or after conventional chemotherapy with platinum (II) complex such as FOLFOX or FOLFIRINOX [1, 2, 9], who were completely hair loss before Huaier administration. Interestingly, the results demonstrated not only regeneration of hair, but also hair with a color of brownish to back hair (typical to Japanese population).
Pathogenic skin lesion seemed to be observed among highly stressed persons, in various part of the body [1, 9-12]. The loss of hair was accompanied in the Patient No. 13 [11], together with multiple cysts production in the liver detected by CT image analysis.
The cancer origin of the patients was meningioma [1, 7, 8, 11], prostate [1, 7, 10 11], lung [1, 2], pancreas [1, 2, 7, 9,11], breast [1, 11], or basal cell carcinoma [1, 11] in face. Huaier treatment on lung metastasis [1, 2, 5-7, 11] resulted in the similar increase of hair growth and restoration, too (patient Nos. 8 and 9). The quantity of Huaier administration was basically 20g per day for 3 months, but for the most severely advanced-stage cancer patients (patient Nos. 8 and 9), we chose 60g Huaier per day were applied as generally used in Chinese hospitals, based on a dose-dependent efficacy of Huaier.
The characteristics in activation of intra/inter neural signal transfer [5], tissue regeneration [6], transcriptional regulations of pluripotency of stem cells [7] were described previously with further information of qualitative and quantitative alterations of responsible molecules, such as KIT, Myc, OCT 3/4, SOX2, LIN28A, and NANOG, according to the time course of Huaier treatment [1-5, 7-11]. Consequently, there found variety of combination of up/down regulation in these molecules, which demonstrated no specific alterations in any factor in any of the cancer patients [1, 7, 8, 11].
3.2 KEGG Pathway Classification on the Hedgehog Signaling Pathway and Related Molecules
We have already reported basic molecular characterization of the patients appeared in the present study, such as summary of sequence events, RNA Editing events, comparison of differentially expressed genes (up-regulated as well as down-regulated ones) before and after Huaier administration, including analysis of differentially expressed small RNAs (DSGs) with abundant identification of novel siRNA, piRNA, and miRNA [1-11].
In those reports, we paid special attention to DEG- TF (transcription factor) network analysis, which remarkably indicates the genomic potential of each patients [1-11]. The patients in the present study showed the largest quantitative potential in genetic and genomic flexibility and capability after Huaier administration, from 25% (patient No. 5) up to 85% alterations in gene expression level (patient No.6) [1, 11].
We focused so intensively on the recovery process of those cancer patients, but did not pay enough attention to the face, skin, and the external appearance. The personal declaration on skin color (bright), gain of body weights (3-5 Kg/month), and especially, gross increase and restoration of hair, which significantly improved mental and psychological situation in daily Quality of Life (QoL). Of course, the patient lost body weights by 10 Kg or more before clinical treatment of the original disease, the total sum of body weights was ca. 5 Kg increase after 3 months of Huaier administration.
With quantitative analysis of DEGs, we performed KEGG pathway classification (https://www.genome.jp/kegg/) to define the detailed molecular mechanism of Huaier effects on hair growth and improvement of skin problems [17]. The cancer origins shown in the present study have been reported to have a close relationship to the functional disruption in hedgehog signaling pathway [22, 23].
Fig. 1 demonstrated the significant increase of responsible elements in Hedgehog signaling pathway by the time course of Huaier administration. The comparison of the up/down regulation of SMO (smoothhead) were also indicated by red arrow in the panels [29, 30]. Huaier effects on the other patients were shown in Supplementary extended Fig. 1 to show all the patients’ data appeared in Table 1.
It was a simple and clear proof to show that Huaier promotes hair growth and restoration by activation of Hedgehog signaling pathway, followed by inhibition to be normal level after 3-6 months’ interval. The effect seemed to be as a result of promoting tissue regeneration by using iPS/ES production and normal specifications [1, 5-7], which might have a close relationship to the recovery of signal transfer inter/intra neurons in central and peripheral nervous systems [5]. In addition, Huaier functioned both as activator and as inhibitor in the recovery process, as shown in the kinase function rescue 2. Hedgehog signaling pathway were finally settled in stable normal condition over 1 year of Huaier treatment unless the patients had another problems such as opportunistic infections [1, 11].
As shown in Fig. 1 Panel b, in the patient No.5, strong steroid therapy on idiopathic dysacousis resulted in complete inhibition and down-regulated responsible molecules in Hedgehog signaling pathway [1, 11]. Basal cell carcinoma progression seemed to be caused as a result of the massive and strong down-regulation of almost all the elements involved in Hedgehog signaling pathway by steroid therapy, but following Huaier treatment by 20g per day after surgical dissection of pathogenic lesion brought successful recovery by reverse massive activation.
The down-regulation of SMO recovered to normal levels after 6 months of Huaier treatment. The patients then defined as the complete remission, and medical examinations did not demonstrate any signs of recurrence or metastasis over 3 years.
In contrast, patient No.6 (meningioma) took 9 months to activate Hedgehog signaling pathway, even without any additional treatment to Huaier after the surgical dissection, and required another 9 months to be back to normal condition (Fig. 1 Panel b). Compared with the results in the Patient No. 5, SMO exhibits repeated down-regulation followed by up-regulation until the retrieval of healthy condition, which took 1.5 years.
Red arrow in Panels indicates up-regulation of SMO, blue arrow; down-regulation, and black arrow; no change in expression level. It is surprising that there were no genetic alterations (written in red letters) in any of responsible elements in any Panels. Conventional chemotherapy using with platinum (II) complex (FOLFOX or FOLFIRINOX) surpassed DEG alterations nor expression level of SMO. The difference between patient’s No. 8 and 9 were the time of Huaier administration, the Patient No.8 took FOLFIRINOX at the same time of Huaier administration, and No.9 had 6 months after FOLFOX treatment.
Massive up-regulation of SMO was detected only in the cases of prostate cancer (Fig. 1 Panel a) and at the time of cure of skin lesions and hair loss in the normal control (Patient No. 13, Fig. 1 Panel d). But prostate cancer patients did not show the identical up-down-regulation in SMO.
Normal healthy controls did not demonstrate the significant alterations in Hedgehog signaling pathway, nor in any pathways without any disruption or dysfunction [1, 11]. Interestingly, hypertension in the Patient No.15 became within normal range by 20g Huaier administration per day for 3 months.
Transcriptional control of tissue regeneration in those patients by production of iPS/ES cells has been already reported with the comparison of alterations in related genes, such as kit, Myc, OCT 3/4, SOX2, LIN28A, and NANOG, according to the time course of Huaier treatment [1-5, 7-11]. Consequently, no specific alterations were demonstrated in any factors as reported in any of the cancer patients [1, 7, 8, 11].
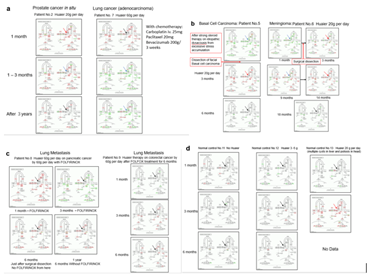
Figure 1: KEGG pathway analysis on hedgehog signaling pathway of the cancer patient according to Huaier administration. Panel a) Prostate cancer in situ and Lung cancer (adenocarcinoma); b) Basal cell carcinoma and Meningioma long treated by 20g Huaier per day; c) Lung metastasis from pancreatic cancer and from colorectal cancer treated by 60g Huaier per day; d) Normal healthy controls without Huaier administration, with 3-5g Huaier administration per day, and with skin lesions with 20g Huaier treatment per day. Red box indicates significant up-regulation of the transcriptomes, and blue box indicates significant decrease of the transcriptomes. Red arrow in Panels indicates up-regulation of SMO, blue arrow; down-regulation, and black arrow; no change in expression level. Black letters in the box indicate no genetic alterations.
Table 1: Clinical features of cancer patients and normal controls.
3.3 Quantitative and Qualitative Alterations in miRNAs
Tissue regeneration promoted by Huaier administration was initiated by miRNA-mediated transcriptional control [1, 5-11]. There have been reported the regulation by specific miRNAs for controlling Hedgehog signaling pathway [24, 25], we have accomplished the complete analysis of any up/down regulation of miRNAs throughout the observation period in each patient. However, over 1,500 numbers of any miRNA with up/down-regulation of miRNA had no match to those miRNAs previously reported [24, 25]. The representative top 10 alterations in miRNAs were summarized in Fig. 2. There were some miRNAs repeatedly appeared in different cancer origin (indicated by * in the Figure), however, all of these were novel sequences. Specific or general characterization of those novel molecules remained unknown.
Massive up-regulation followed by down-regulation miRNA DSGs were observed both in the cancer patients as well as normal healthy control with Huaier administration [1, 5-11]. It was unlikely that miRNA alterations were not dose-dependent, more relationship to the severity and required transcriptional factor alterations [1-11]. In normal control No.11, highest numbers of down-regulation of DSGs were observed, whereas highest numbers of up-regulated DEGs contributed to accelerate cancer recovery in the patient No.7 [2]. The similar analysis in the Patient No. 6 have been reported before [1. 8], to focus on Huaier effects on mutated EGFR and other receptor tyrosine kinase case [2,8]. The novel miRNA information has been together deposited to the NCBI GEO (GSE157086).
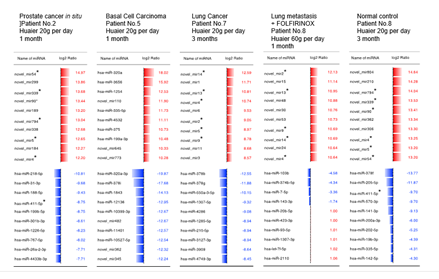
Figure 2: The up/down regulation of quantitatively regulated miRNAs. The quantitative changes of miRNAs were indicated by log2 transformed fold change calculation by the red bars (up-regulated molecules) and blue bars (down-regulated ones). Asterisk * indicates the repeated appearance of up-regulated miRNA, only one in down-regulation, in different cancer origin.
4. Discussion
The present study provides typical and additional effects of Huaier, easy to be recognized, on tissue repair and regeneration system in the process of cancer recovery. This tissue repair system was essential for cancer recovery, after cancer -specific cell death by Huaier, and derived with a support of transcriptional regulations of pluripotency of stem cells as previously reported [1-11]. Hair growth and restoration were often observed with the cure of skin lesions (idiopathic, atopic, and/or drug-induced) [1, 12], with the improvement of hypertension (over 20g Huaier per day). Essential hypertension is common to Japanese population to take a life-long medication. Huaier administration over 20g per day contributed to do without excessive medication related to this disorder (normal control No.15).
Interestingly, the severely damaged pneumonia with massive fibrosis in both lungs caused by SARS-CoV-2 infection has been also successfully recovered by judging from the CT image analysis with 3 months of 30g Huaier administration per day. In these patients, Huaier efficacy on tissue regeneration seemed to influence at most. The detailed molecular mechanism of this recovery and repair system is now being under investigation.
As noted before [1, 12], Huaier effects appear only on the disrupted functions, and never demonstrate unnecessary alterations to normal healthy control (Fig. 1 Panel d). In contrast, we should be cautious to possibility of pathogenic lesions even in individuals without any symptoms or disorders, when we identify significant genetic alterations in our clinical research.
It is well investigated about the functions of, and targeting strategy against Hedgehog signaling pathway [24-33]. Hedgehog signaling pathway functions in the process of cell proliferation, apoptosis, angiogenesis, epithelial-mesenchymal transition and self-renewal and repair system by utilizing stem cells [22, 23]. Dysregulation of those processes has been linked to carcinogenesis, in the development of cancers in various organs, including brain, lung, mammary gland, prostate and skin. Basal cell carcinoma, the most common form of cancerous malignancy, has the closest association with hedgehog signaling [29]. On the contrary to the previous reports, there were no loss-of-function mutations either activating mutations in SMO (smoothened) have been identified in the present study. SMO did not show any alterations on the process of either carcinogenesis or recovery in vivo. More importantly, the regulation of up- and down-regulation of SMO expression level, and that repeatedly, was observed in the present study, which would suggest that simple inhibition nor activation did not contribute practically in a long course of cancer recovery.
Aberrant hedgehog signaling has also been implicated in the maintenance of cancer stem cells. In breast cancer, inhibition of hedgehog signaling has been designated to reduce stem cell propagation and renewal. Shh (sonic hedgehog) signaling in cancer stem cells drives epithelial-mesenchymal transition and, ultimately, cancer metastasis [33]. The patient No. 8 and 9 provided valuable information on these points.
Patient No. 8 had conventional chemotherapy with FOLFIRINOX at the same time of 60g Huaier per day. On the other hand, in the Patient No. 9, administration of Huaier started from 6months after chemotherapy with FOLFOX2. The difference resulted in the drastic changes in clinical features in lung metastasis between those patients. In the Patient No.9, effective function of activated Hedgehog signal pathway continued and no more metastasis, nor recurrence were observed even after 2 years’ of treatment [1, 5-9]. Currently this patient only has Huaier, and no chemotherapy was necessary.
In contrast, Patient No. 9, after 1.5 years of Huaier treatment, was detected gradually increased CEA titers and size of lung coin lesions (no more new lesions, though). This patient has been detected to have mutations in those transcriptomes as written below
- Oncogenes; β-catenin, K-Ras (p21ras), c-myc, TERT, receptor tyrosine kinase c-MET, PIK3CA
- Tumor suppressor genes; APC, DCC. TGFβ, Smad2. Mad4, Bax, p53, RB, PTEN
- DNA mismatch repair gene: hMLH1, hMSH2, hMSH3, hMSH6
With those mutations, FOLFOX treatment for 6 months seemed to initiate the production and maintenance of cancer stem cells (CSC) [25, 33]. After 1.5 years, CSC exhibits increased potential for self-renewal, differentiation, and for starting secondary tumors in metastatic lesions. CSC also exhibited drug resistance to chemotherapies within 6 months’ of treatment period. As a result, while FOLFOX was successful in eliminating the bulk of the tumor at first (which consists of non-CSC), CSC left behind can lead to tumor recurrence now [33]. Thus, via its role in CSC maintenance, hedgehog signaling contributes towards the failure of anti-cancer therapies.
Targeting and inhibition strategy of SMO intended to prevent the induction of glioma-associated transcriptional activity upon exposure of cancer cells to Shh ligands. Therefore, SMO has been reported to become a primary target in the development of Hedgehog pathway inhibitors26-33. Not only SMO, but the other ligands have been targeted for efficient development of targeting-agents against hedgehog signaling pathway [26-33].
However, as shown in the present study, SMO was not specifically associated with the disruption or dysfunction of hedgehog signaling pathway in the process of cancer recovery with Huaier treatment. Periodical regulation in both ways to activate and inhibit the Hedgehog signaling pathway function is required. Effective regulator, with no toxicity or side effects like Huaier, seemed to be the ideal for the cancer strategy. We can search for the natural herb therapy to identify similar, would be more effective than simple inhibitor or activator. The advantage of Huaier exists in the function as (tyrosine) kinase regulator as reported before2, by typical regulation in PI3K/AKT signaling pathway, which appears in CSC theory, too [40, 41].
The CSC theory written above was proved in the process of carcinogenesis, but not necessarily reproduced or demonstrated on the way of recovery process in cancer patients. Our clinical research provided many aspects to skip the process or key points found in carcinogenesis were lost on the way back to recovery toward normal healthy conditions.
Thus, the present study provides simple evidence by demonstrating Huaier effects on hedgehog signaling pathway, as one example of tissue repair and regeneration system essential to cancer recovery.
Acknowledgments
The authors wish to thank cancer patient volunteers and many healthy volunteers kindly collaborated with the present study.
Data Availability
The complete sequencing data of the cases have been deposited to NCBI database, and the clinical outcomes of these cases are not publicly available for data privacy but are available from Dr. Manami Tanaka (e-mail: tubu0125@gmail.com, manami-tanaka@bradeion.com) and Dr. Fei Teng (e-mail: Sky.teng@bgi.com)upon request for research collaboration. The timeframe for response to data access requests is 30 days. There are no restrictions on the reuse of data. In addition, the raw data of the longitudinal cohort and healthy individuals analyzed in this study were available at GEO with identifiers of NCBI GEO (GSE157086).
Author Contributions
T., T. T., F.T., designed the study from the clinical observation of the cancer patients with Huaier treatment (as a complementally therapy), and managed the sampling and clinical assessment of the patient volunteers, statistically analyzed the data, and drafted the manuscript. F. T., X. Z., H. L. Z., L., Y.P., managed total RNA and small nuclear RNA sequencing and conducted systematic analysis of the data. S. S., T. S., and Y. M., contributed clinical diagnosis and treatment of the patients, together with the assessment of QOL and the effects of Huaier administration, Z. L., D. W., contributed to the provision of Huaier granules and clinical evaluation of the data, especially focused on Immunological evaluation.
Funding
The present study was grant-in-aid from QiDong Gaitianli Medicines Co., Ltd. and Japan Kampo New Medicine, Co., Ltd.
Competing Interests
The authors declare no competing interests.
References
- Tanaka M, Tanaka T. Huaier: Natural Herb Therapy for Cancer, Bradeion Institute of Medical Sciences Press (2022): 342.
- Tanaka M, Tanaka T, Teng F, et al. Huaier effects on functional compensation with destructive ribosomal RNA structure after anti-SARS-CoV-2 mRNA vaccination. Arch Clin Biomed Res 6 (2022): 553-574.
- Tanaka M, Tanaka T, Teng F, et al. Biomedicine in 21st century: The practical health maintenance and successful recovery from cancer. J Biomed Biosens 1 (2021): 33-56.
- Tanaka M, Tanaka T, Teng F, et al. Huaier therapy for successful recovery of cancer and health maintenance: Steady progress and the end of failed promise. Arch Clin Biomed Res 5 (2021): 457-483.
- Tanaka M, Tanaka T, Teng F, et al. Huaier compensates impaired signal transfer inter/intra neurons in central and peripheral nervous systems. Arch Clin Biomed Res 5 (2021): 484-518.
- Tanaka T, Tanaka M, Teng F, et al. Molecular basis of Huaier effects on immunomodulation, and natural selection of iPS cells with stable growth in vivo. J Pharm Res Dev (2021).
- Tanaka M, Tanaka T, Teng F, et al. Huaier inhibits cancer progression and induces tissue regeneration by transcriptional regulation of pluripotency of stem cells. J Altern Compl Integr Med 7 (2021): 162-172.
- Tanaka M, Tanaka T, Teng F, et al. Huaier inhibits cancer progression correlated with the mutated EGFR and other receptor tyrosine kinases (c-MET/erbB-2) by down-regulation of multiple signal transduction pathways. Arc Clin Biomed Res 5 (2021): 262-284.
- Tanaka M, Tanaka T, Teng F, et al. Complete remission of the severe advanced stage cancer by miRNA-mediated transcriptional control of Bcl-xL with Huaier therapy compared to the conventional chemotherapy with platinum (II) complex. Arch Clin Biomed Res 5 (2021): 230-261.
- Tanaka M, Tanaka T, Teng F, et al. Anti-cancer effects of Huaier on prostate cancer; miRNA-mediated transcription control induced both inhibition of active progression and prevention of relapse. J Altern Compl Integr Med 7 (2021): 146-155.
- Tanaka M, Tanaka T, Teng F, et al. Huaier Induces Cancer Recovery by Rescuing Impaired Function of Transcription Control Based on the Individual Genomic Potential. Arch Cli. Biomed Res 4 (2020): 817-855.
- Tanaka T, Suzuki T, Nakamura J, et al. Huaier regulates cell fate by the rescue of disrupted transcription control in the Hippo signaling pathway. Arch Clin Biomed Res 1 (2017): 179-199.
- Zhu X, wang Y, Nan J, et al. Comparative gene expression profiling and character development instable angina pectoris disease. J Altern Compl Integr Med 7 (2021): 192.
- Peng Z, Cheng Y, Tan BC, et al. Comprehensiveanalysis of RNA-Seq data reveals extensive RNA editing in a human transcriptome. Nat Biotechnol 30 (2012): 253-260.
- Jiang Y, Sun A, ZhaoY, et al. Proteomics identifies new therapeutic targets of early-stage hepatocellular carcinoma. Nature 567 (2019): 257-261.
- Song Y, Li L, Ou Y, et al. Identification of genomic alterations in oesophageal squamous cell cancer. Nature 509 (2014): 91-95.
- Kanehisa M, Araki M, Goto S, et al. KEGG for linking genomes to life and the environment. Nucleic Acids Res 36 (2008): 480-484.
- Paladini RD, Saleh J, Qian C, et al. Modulation of hair growth with small molecule agonists of the hedgehog signaling pathway. J Invest Dermatol 125 (2005): 638-646.
- Matsumura H, Mohri Y, Thanh Binh N, et al. Hair follicle aging is driven by transepidermal elimination of stem cells via COL17A1 proteolysis. Science 351 (2016): 575-589.
- Matsumura H, Liu N, Nanba D, et al. Distinct types of stem cell divisions determine organ regeneration and aging in hair follicles. Nature Aging 1 (2021): 190-204.
- Morinaga H, Mohri, Y, Grachtchouk M, et al. Obesity accelerates hair thinning by stem cell-centric converging mechanisms. Nature 595 (2021): 266-271,
- Varjosalo, Taipale J. Hedgehog: functions and mechanisms. Genes & development 22 (2008): 2454-2472.
- Scales SJ, de Sauvage FJ. Mechanisms of Hedgehog pathway activation in cancer and implications for therapy. Trends Pharmacol. Sci 30 (2009): 303-312.
- Fu J, Rodova M, Nanta R, et al. NPV-LDE-225 (Erismodegib) inhibits epithelial mesenchymal transition and self-renewal of glioblastoma initiating cells by regulating miR-21, miR-128, and miR-200. Neuro-oncology 15 (2013): 691-670.
- Nanta R, Kumar D, Meeker D, et al. NVP-LDE-225 (Erismodegib) inhibits epithelial–mesenchymal transition and human prostate cancer stem cell growth in NOD/SCID IL2Rγ null mice by regulating Bmi-1 and microRNA-128. Oncogenesis 2 (2013): e42.
- Wickström M, Dyberg C, Shimokawa T, et al. Targeting the hedgehog signal transduction pathway at the level of GLI inhibits neuroblastoma cell growth in vitro and in vivo. Int J Cancer 132 (2013):1516-1524.
- D’Amato C, Rosa R, Marciano R, et al. Inhibition of Hedgehog signaling by NVP-LDE225 (Erismodegib) interferes with growth and invasion of human renal cell carcinoma cells. Br J Cancer 111 (2014): 1168.
- Rudin CM, Hann CL, Laterra J, et al. Treatment of medulloblastoma with hedgehog pathway inhibitor GDC-0449. N Engl J Med 361 (2009): 1173-1178.
- Xie J, Murone M, Luoh SM, et al. Activating Smoothened mutations in sporadic basal-cell carcinoma. Nature 391 (1998): 90-92.
- Rimkus T, Carpenter R, Qasem S, et al. Targeting the sonic hedgehog signaling pathway: review of smoothead and GLI inhibitors. Cancers 8 (2016): 22.
- Wickström M, Dyberg C, Shimokawa T, et al. Targeting the hedgehog signal transduction pathway at the level of GLI inhibits neuroblastoma cell growth in vitro and in vivo. Int J Cancer 132 (2013): 1516-1524.
- Srivastava RK, Kaylani SZ, Edrees N, et al. GLI inhibitor GANT-61 diminishes embryonal and alveolar rhabdomyosarcoma growth by inhibiting Shh/AKT-mTOR axis. Oncotarget 5 (2014): 12151.
- Liu S, Dontu G, Mantle ID, et al. Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Res 66 (2006): 6063-6071.
- Chen Y, Chen Y, Shi C, et al. SOAPnuke: a MapReduce acceleration-supported software for integrated quality control and preprocessing of high-throughput sequencing data. GigaScience 7 (2018): 1-6.
- Li B, Dewey CL. RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform 12 (2011): 323.
- Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15 (2014): 1-21.
- Ben L, Salzberg SL. Fast gapped-read alignment with Bowtie 2. Nat Methods 9 (2012): 357-359.
- Enright AJ, John B, Gaul U, et al. MicroRNA targets in Drosophila. Genome Biol 5 (2003): R1.
- Agarwal V, Bell GW, Nam J, et al. Predicting effective microRNA target sites in mammalian mRNAs. eLife 4 (2015): e05005.
- Yan X, Lyu T, Jia N, et al. Huaier aqueous extract inhibits ovarian cancer cell motility via the AKT/GSK3beta/beta-catenin pathway. PLoS ONE 8 (2013): e63731.
- Wang X, Qi W, Li Y, et al. Huaier extract induces autophagic cell death by inhibiting the mTOR/S6K pathway in breast cancer cells. PLOS ONE 10 (2015): e0131771.
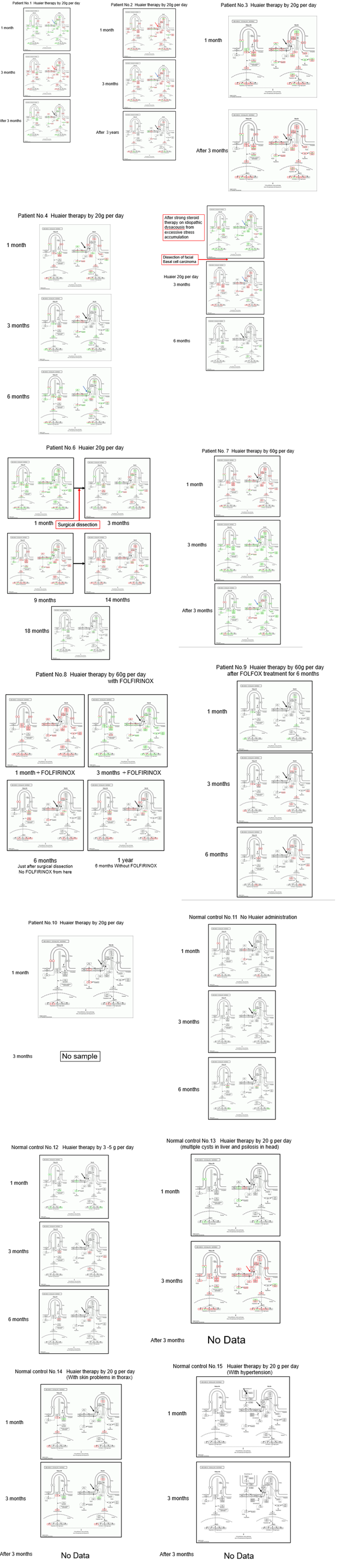
Supplement extended Figure 1: KEGG pathway analysis on hedgehog signaling pathway of the remained cancer patients who did not appear in Fig. 1. Red arrow in Panels indicates up-regulation of SMO, blue arrow; down-regulation, and black arrow; no change in expression level. Black letters in the box indicate no genetic alterations.


 Impact Factor:
* 4.1
Impact Factor:
* 4.1
 Acceptance Rate:
74.74%
Acceptance Rate:
74.74%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
