Impact of Different Prognostic Factors on the Survival of Patients with Malignant Granulosa Cell Tumors of the Ovary: An Institutional Based Study at the Egyptian National Cancer Institute
Ihab Samy Fayek MD1*, Alfred Elias Namour MD2, Nelly Alieldin MD3, Iman Gouda Farahat MD4
1Surgical Oncology Department, National Cancer Institute, Cairo University, Egypt
2Medical Oncology Department, National Cancer Institute, Cairo University, Egypt
3Epidemiology and statistics Department, National Cancer Institute, Cairo University, Egypt
4Surgical Pathology Department, National Cancer Institute, Cairo University, Egypt
*Corresponding Author: Dr. Ihab Samy Fayek, Surgical Oncology Department, National Cancer Institute, Cairo University, Egypt.
Received: 06 December 2019; Accepted: 17 December 2019; Published: 20 January 2020;
Article Information
Citation: Ihab Samy Fayek, Alfred Elias Namour, Nelly Alieldin, Iman Gouda Farahat. Impact of Different Prognostic Factors on the Survival of Patients with Malignant Granulosa Cell Tumors of the Ovary: An Institutional Based Study at the Egyptian National Cancer Institute. Journal of Cancer Science and Clinical Therapeutics 4 (2020): 049-055.
View / Download Pdf Share at FacebookAbstract
Introduction: Emphasizing and evaluating different prognostic factors including the age, size of the tumor, type of surgery, capsular infiltration and administration of adjuvant chemotherapy, which may affect the disease free survival (DFS) and overall survival (OS) in patients with malignant Granulosa cell tumors (MGCTs).
Patients and Methods: A retrospective study of 36 female patients diagnosed with MGCTs and managed at the National Cancer Institute – Cairo University-Egypt, in the period from May 2007 –October 2012. Follow –up reached up to 116 months (3-116 mo.) with a median of 72 months. Prognostic factors as the age, size of the tumor, type of surgery, capsular infiltration and administration of adjuvant chemotherapy with number of cycles in correlation with DFS and OS were statistically analyzed to detect any significant correlation.
Results: The median age was 44 years. The median tumor size was 12 cm. The median DFS was 41.9 months. The median OS was 45.0 months. The disease free survival (DFS) was not significantly affected by the size of the tumor (<10 cm/ ≥10 cm) nor the type of surgery (Panhysterectomy or salpingo-oophorectomy) nor administration of adjuvant chemotherapeutic agents where p-value=0.4487, 0.8471 and 0.0986 respectively. However, capsular invasion significantly affected the DFS (p-value=0.0076). The overall survival (OS) was not affected by the size of the tumor (<10 cm/ ≥10 cm) with a nearly significant correlation (p=0.0615). No statistical significance on OS regarding the type of surgery (Panhysterectomy or salpingo-oophorectomy) (p=0.7792), the capsular affection (p=0.0871), nor the adjuvant chemotherapeutic administration (p= 0.2848).
Conclusion: Capsular infiltration is by far the most independent prognostic factor affecting disease free survival (DFS) in patients with malignant granulosa cell tumors an
Keywords
<p>Granulosa cell tumor; Disease free survival; Overall survival; National cancer institute Egypt</p>
Article Details
1. Introduction
Granulosa cell tumors (GCT) of the ovary are classified as sex-cord stromal tumors and their occurrence is rare, represent about 5% of all ovarian tumors. Two distinct types of GCTs: adult (AGCT) and juvenile (JGCT) [1-3]. These tumors differ from other ovarian cancers with its hormonal behavior and low grade nature [4]. GCTO grow slowly and can recur late, after primary treatment The reported median age at the diagnosis is about 49 [5]. Surgery is the primary treatment of GCTs and includes hysterectomy, bilateral adnexectomy, and omentectomy as well as biopsies from peritoneal surfaces. The necessity for lymphadenectomy has varied over the decades and is not currently recommended [6-12]. The role of adjuvant therapy is unclear, although in advanced-stage disease, platinum-based chemotherapy is nowadays recommended [6, 13, 14], however, either adjuvant chemotherapy or radiotherapy could not obtain an evidence based consequence [12, 15]. Inspite the generally favorable prognosis of GCTs, 97% survive for 5-years, however poor prognosis up to 66.8% survival after 20 years and 30-35% mortality needing longer follow-up in early stages and for lifelong [7]. Recurrence rates were documented from as low as 5.6% after primary surgical treatment [16] up to 21% with a median time to relapse was 57.6 months (2-166 months) [17], and the longest reported times to recurrence were 40 years [18] and 25 years [19]. Indicating the need for prognostic factors alarming the possibility of recurrences even if late. The current study will address the National Cancer Institute experience along the management of granulosa cell tumors during the period from 2007-2012 emphasizing certain prognostic factors affecting DFS and OS.
2. Patients and Methods
A retrospective study of 36 female patients diagnosed with malignant GCTs and managed at the National Cancer Institute – Cairo University-Egypt, in the period from May 2007–October 2012. All patients were surgically managed either by Panhysterectomy or unilateral salpingo-oophorectomy with or without adjuvant chemotherapy. All surgical specimens were histopathologically examined regarding the size of the tumor, laterality and capsule infiltration. The malignant behavior of the tumor was confirmed. All GCTs with unpredictable biological behavior were excluded from the study. Adjuvant chemotherapy offered to our patients was documented regarding the regimen given and number of chemotherapy cycles administered. Follow –up reached up to 116 months (3-116 mo.) with a median of 72 months. During the period of follow-up recurrence was documented clinically and radiologically by CT and/or MRI of the pelvis. Chemotherapy regimens for recurrent tumors were documented. Prognostic factors as the age, size of the tumor, type of surgery, capsular infiltration and administration of adjuvant chemotherapy with number of cycles in correlation with DFS and OS were statistically analyzed to detect any significant correlation.
3. Statistical Method
Demographic data and survival outcome will be analyzed using SPSS statistical package version 20. Relations between qualitative data will be done using Chi-square test or Fisher’s exact test. Moreover, survival analyses will be done using Kaplan Meier’s method and Comparison between survival curves will be done using log-rank test. A p-value less than 0.05 will be considered significant.
4. Results
The median age was 44 years with the youngest patient, 19 years old and the eldest 73 years old. The median tumor size was 12 cm with the largest tumor 25 cm and the smallest one 3 cm. The median DFS was 41.9 months. The median OS was 45.0 months. The patients in the study were grouped according to age in three ways: ≤30/>30 years old, ≤40/>40 years old and ≤50/>50 years old where none of the age groups showed statistically significant affection of DFS (p=0.1597, 0.7379 and 0.1854 respectively) nor OS (p=0.4057, 0.8806 and 0.2975 respectively).
The tumor sizes in this study were grouped according to the longest diameter into: Tumor size < 10 cm. (14 patients, 38.9%) and tumor size ≥ 10 cm. (22 patients, 61.1%). Twenty four patients (66.7%) underwent panhysterectomy while 12 patients (33.3%) underwent salpingo-oophorectomy. Bilaterality was found in 3 patients (8.3%), left sided tumors in 19 patients (52.8%) while right sided tumors were found in 14 patients (38.9%). Thirty four patients (94.4%) had granulosa cell tumor, adult type while 2 patients had the juvenile type (5.6%). The capsular invasion was found in only 4 patients (11.1%). Eighteen patients (52.9 %) received adjuvant chemotherapy while 16 patients (47.1%) did not receive adjuvant chemotherapy. The most common regimen was the BEP (bleomycin 30 U on days 2, 9, and 16, etoposide 100mg/m2/day on days 1 - 5, and cisplatin 20 mg/m2/day on days 1 - 5) regimen given to 12 patients (66.7%) most of them received 2 to 3 cycles while Taxanes were given to 4 patients (22.3%) who received 6 cycles of this regimen Table 1.
Recurrence occurred in 6 patients (17.1%). By the end of follow up 26 patients (72.2%) were alive and 10 patients (27.8%) died. Two and four patients with recurrent disease received chemotherapy in the form of BEP (Bleomycin / Etoposide / Cisplatin) and Taxol/Carboplatin respectively Table 2. The disease free survival (DFS) was not significantly affected by the size of the tumor (<10 cm/ ≥10 cm) nor the type of surgery (Panhysterectomy or salpingo-oophorectomy) nor administration of adjuvant chemotherapeutic agents where p-value=0.4487, 0.8471 and 0.0986 respectively. However, capsular invasion significantly affected the DFS (p-value=0.0076) Figure 1.
The overall survival (OS) was not affected by the size of the tumor (<10 cm/ ≥10 cm) with a nearly significant correlation (p=0.0615). No statistical significance on OS regarding the type of surgery (Panhysterectomy or salpingo-oophorectomy) (p=0.7792), the capsular affection (p=0.0871) (Figure 2), nor the adjuvant chemotherapeutic administration (p= 0.2848).
|
Specification |
Count |
% |
|
|
Chemotherapy type |
BEP* |
12 |
66.7 |
|
CAP* |
1 |
5.6 |
|
|
PE* |
1 |
5.6 |
|
|
taxol/carboplatin |
3 |
16.7 |
|
|
taxol |
1 |
5.6 |
|
|
Group Total |
18 |
100 |
|
|
Number of chemotherapy cycles |
1 |
1 |
5.6 |
|
2 |
5 |
27.8 |
|
|
3 |
5 |
27.8 |
|
|
4 |
3 |
16.7 |
|
|
6 |
4 |
22.2 |
|
|
Group Total |
18 |
100 |
*BEP: Bleomycin / Etoposide / Cisplatin; *CAP: Cyclophosphamide / Doxorubicin / Cisplatin;
*PE: Cisplatin / Etoposide
Table 1: Adjuvant chemotherapeutic regimens and number of cycles administered.
|
Frequency |
% |
|
|
BEP |
2 |
33.3 |
|
taxol / carboplatin |
4 |
66.7 |
|
Total |
6 |
100 |
Table 2: Chemotherapeutic regimens administered for recurrent cases.
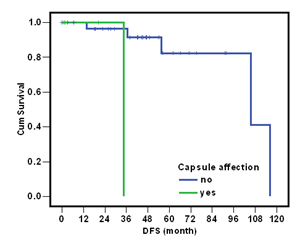
Figure 1: Correlation between DFS and Capsular invasion.
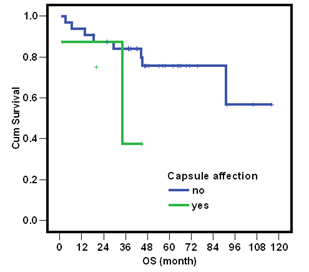
Figure 2: Correlation between OS and capsular invasion.
5. Discussion
Egyptian studies addressing granulosa cell tumors are few with limited number of patients due to the rarity of this type of tumors, requesting further studies to define the proper prognostic factors [9, 20]. Among 26 female patients diagnosed with ovarian cancer in Aswan – Egypt during the year 2008 only 3 patients (11.5%) were documented as GCTs [21]. This study, as far as our knowledge, is the largest one emphasizing the prognostic factors of malignant granulosa cell tumors and their impact on survival in Egyptian females, regarding the number of patients (36 patients) and period of follow-up (up to 116 months).
Sehouli et al. [22] reported The median age at diagnosis was 53 years, The reported median age at the diagnosis by Huang et al. [5] and Yesilyurt et al. [23] was about 49 while The mean age at diagnosis was 52.9 years reported by Bryk et al. [8]; in our study the median age was 44 indicating younger age at presentation in Egyptian females, and in concordance with Acharya et al. [4] where the mean age of the women was 42 years.
Sehouli et al. [22] stated that no difference significantly observed in survival rates between patients older and younger than 50 years old (p=0.45) [22]. This study subgrouped the patients according to age into 3 subgroups: ≤30/>30 years old, ≤40/>40 years old and ≤50/>50 years old where none of the age groups showed statistically significant affection of DFS (p=0.1597, 0.7379 and 0.1854 respectively) nor OS (p=0.4057, 0.8806 and 0.2975 respectively).
Sun et al. [17] concluded that tumor size > 13.5 cm. was significantly associated with recurrence, however, regarding our study the tumor size ≥ 10cm. did not significantly affect DFS (p=0.4487) but may affect OS (p=0.0615). Complete surgical excision is the key step in the management of patients with GCTs [9, 24]. However, surgical morbidity is affected negatively by Lymphadenectomy which does not improve survival in AGCTs [10-12, 25]. None of our study population underwent formal neither pelvic nor para-aortic lymphadenectomy.
Nasioudis et al. [26] concluded that chemotherapy does not affect survival after controlling several factors; similarly our study did not prove any significance between administration of adjuvant chemotherapy and DFS (p=0.0986) nor OS (p=0.2848).
Lee et al. [27] stated that several factors including capsule invasion, had no significant relation with recurrence, however, our study revealed the significant contribution of capsule invasion in the development of recurrence in such tumors, in agreement with a recent pathological study in Croatia emphasizing several other pathological prognostic factors such as LVSI, presence of necrosis and hemorrhage, diffuse growth pattern, and nuclear atypia [28].
6. Conclusion
Capsular infiltration is by far the most independent prognostic factor affecting disease free survival (DFS) in patients with malignant granulosa cell tumors and to a lesser extent overall survival (OS), Tumor size ≥10 cm is nearly affecting OS but not DFS. A Multi-institutional study with larger sample size and longer follow-up is needed to re-evaluate our results.
References
- Segal R, DePetrillo AD, Thomas G. Clinical Review of Adult Granulosa Cell Tumors of the Ovary. Gynecologic Oncology 56 (1995): 338-344.
- Khosla D, Dimri K, Pandey AK, et al. Ovarian Granulosa Cell Tumor: Clinical Features, Treatment, Outcome, and Prognostic Factors. North American Journal of MedicalSciences 6 (2014): 133-138.
- Karalök A, Ta?ç? T, Üreyen I et al. Juvenile Granulosa Cell Ovarian Tumor: Clinicopathological Evaluation of Ten Patients. Journal of the Turkish German Gynecological Association 16 (2015): 32-34.
- Acharya G, Premalatha TS, Kulkarni KA, et al. Granulosa Cell Tumour of Ovary: Review of Cases at Tertiary Care Centre. Journal of Cancer and Tumor International 8 (2018): 1-7.
- Huang BS, Sun HD, Hsu YM, et al. Clinical presentation and outcome of adult-type granulosa cell tumors: a retrospective study of 30 patients in a single institute. J Chin Med Assoc 77 (2014): 21-25.
- Park JY, Jin KL, Kim DY, et al. Surgical staging and adjuvant chemotherapy in the management of patients with adult granulosa cell tumors of the ovary. Gynecologic Oncology 125 (2012): 80-86.
- Mangili G, Ottolina J, Gadducci A, et al. Long-Term Follow-Up Is Crucial after Treatment for Granulosa Cell Tumors of the Ovary. British Journal of Cancer 109 (2013): 29-34.
- Bryk S, Färkkilä A, Bützow R, et al. Clinical Characteristics and Survival of Patients with an Adult-Type Ovarian Granulosa Cell Tumor: A 56-Year Single-Center Experience. International Journal of Gynecological Cancer 25 (2015): 33-41.
- Aziz H and Fathallah AE. Granulosa Cell Tumors of the Ovary: Retrospective Analysis of 17 Cases. Journal of Cancer Therapy 06 (2015): 1027-1033.
- Kuru O, Boyraz G, Uckan H et al. Retroperitoneal nodal metastasis in primary adult type granulosa cell tumor of the ovary: Can routine lymphadenectomy be omitted? Eur J Obstet Gynecol Reprod Biol 219 (2017): 70-73.
- Nasioudis D, Kanninen TT, Holcomb K, et al. Prevalence of lymph node metastasis and prognostic significance of lymphadenectomy in apparent early-stage malignant ovarian sex cord-stromal tumors. Oncol 145 (2017): 243-247.
- Erk?l?nç S, Tayla E, Karata?l V, et al. Does lymphadenectomy affect postoperative surgical morbidity and survival in patients with adult granulosa cell tumor of ovary? Journal of Obstetrics and Gynecology research 45 (2019): 1019-1025.
- Homesley HD, Bundy BN, Hurteau JA, et al. Bleomycin, etoposide, and cisplatin combination therapy of ovarian granulosa cell tumors and other stromal malignancies: A Gynecologic Oncology Group study. Gynecologic Oncology 72 (1999): 131-137.
- Pautier P, Gutierrez-Bonnaire M, Rey A, et al. Combination of bleomycin, etoposide, and cisplatin for the treatment of advanced ovarian granulosa cell tumors. Int J Gynecol Cancer 18 (2008): 446-452.
- Gurumurthy M, Bryant A, Shanbhag S. Effectiveness of different treatment modalities for the management of adult-onset granulosa cell tumours of the ovary (primary and recurrent). Cochrane Database System Rev 4 (2014): 6912.
- Ugianskiene A, Grove A, Soegaard-Andersen E. Adult Granulosa Cell Tumor of the Ovary: A Retrospective Study of 37 Cases. European Journal of Gynaecological Oncology 35 (2014): 621-624.
- Sun HD, Lin H, Jao MS, et al. A long-term follow-up study of 176 cases with adult-type ovarian granulosa cell tumors. Gynecologic Oncology 24 (2012): 244-249.
- East N, Alobaid A, Goffin F, et al. Granulosa cell tumour: a recurrence 40 years after initial diagnosis. J Obstet Gynaecol Cancer 27 (2005): 363-364.
- Fujita F, Eguchi S, Takatsuki M, et al. A recurrent granulosa cell tumor of the ovary 25 years after the initial diagnosis: A case report. Int J Surg Case Rep 12 (2015): 7-10.
- Al-Rayyan E, Maaita M, Alelwan O, et al. Clinicopathological outcome of ovarian granulosa cell tumors. Gulf Journal Oncology 28 (2018): 31-36.
- Ibrahim AS, Mikhail NN, Khaled H, et al. Cancer Profile in Aswan, Egypt. Methodology and Results Chart Book 2008. National Cancer Registry Program of Egypt (2010):
- Sehouli J, Drescher FS, Mustea A, et al. Granulosa Cell Tumor of the Ovary: 10 Years Follow-up Data of 65 Patients. Anticancer Research 24 (2004): 1223-1230.
- Yesilyurt H, Tokmak A, Guzel AI, et al. Parameters for Predicting Granulosa Cell Tumor of the Ovary: A Single Center Retrospective Comparative Study. Asian Pac J Cancer Prev 15 (2014): 8447-8450.
- Liu CH, Horng HC, Chang WH, et al. Granulosa cell tumor of ovary: Perspective of Taiwan. Taiwan J Obstet Gynecol 56 (2017): 711-712.
- Brown J, Sood AK, Deavers MT, et al. Patterns of metastasis in sex cord-stromal tumors of the ovary: Can routine staging lymphadenectomy be omitted? Gynecol Oncol 113 (2009): 86-90.
- Nasioudis D, Ko EM, Haggerty AF, et al. Role of adjuvant chemotherapy in the management of stage IC ovarian granulosa cell tumors. Gynecol Oncol Rep. 28 (2019): 145-148.
- Lee IH, Choi CH, Hong DG, et al. Clinicopathologic characteristics of granulosa cell tumors of the ovary: a multicenter retrospective study. J Gynecol Oncol. 22 (2011): 188-195.
- Babarovi? E, Franin I, Klari? M, et al. Adult Granulosa Cell Tumors of the Ovary: A Retrospective Study of 36 FIGO Stage I Cases with Emphasis on Prognostic Pathohistological Features. Analytical Cellular Pathology ID 9148124 (2018): 1-11.


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 