Survival Benefits Associated with Surgery in Patients with Metastatic Breast Cancer by Breast Cancer Subtype
Hsueh-Han Tsai1, Jyh-Cherng Yu1, Mehra Golshan2, 3, Huan-Ming Hsu1, Chi-Hong Chu1, Zhi-Jie Hong1, Chun-Yu Fu1, Yu-Ching Chou4, Ming-Shen Dai5, Guo-Shiou Liao1
1Division of General Surgery, Department of Surgery, Tri-Services General Hospital, National Defense Medical Center, Taipei, Taiwan
2Division of Breast Surgery, Department of Surgery, Brigham and Women's Hospital, MA, USA
3Breast Oncology Program, Dana-Farber/Brigham and Women’s Cancer Center, MA, USA
4School of Public Health, National Defense Medical Center, Taipei, Taiwan
5Division of Hematology/Oncology, Tri-Service General Hospital, National Defense Medical Center, Taipei, Taiwan
*Corresponding Author: Guo-Shiou Liao, Division of General Surgery, Department of Surgery, Tri-Services General Hospital, National Defense Medical Center, No 325, Sec. 2, Cheng-Kung Rd, Nei-Hu 114, Taipei, Taiwan, Tel: +886-2-87927191; Fax: +886-2-87927273
Received: 08 August 2021; Accepted: 24 August 2021; Published: 06 September 2021
Article Information
Citation: Hsueh-Han, Tsai, Jyh-Cherng Yu, Mehra Golshan, Huan-Ming Hsu, Chi-Hong Chu, Zhi-Jie Hong, Chun-Yu Fu, Yu-Ching Chou, Ming-Shen Dai, Guo-Shiou Liao. Survival Benefits Associated with Surgery in Patients with Metastatic Breast Cancer by Breast Cancer Subtype. Journal of Cancer Science and Clinical Therapeutics 5 (2021): 414-425.
View / Download Pdf Share at FacebookAbstract
Background: Two randomized trials have examined the role of local therapy on survival in the stage IV setting of breast cancer, largely suggesting no benefit. However, the effect of surgery for stage IV disease amongst various breast cancer subtypes is not well characterized. In this study, we aimed to evaluate the survival benefit of surgery in patients with stage IV breast cancer based on molecular subtype.
Methods: Using an institutional database, we extracted 255 women with a clinicopathologic confirmed diagnosis of stage IV breast cancer who were treated between January 2006 and October 2016. We collected patient data including age, clinical T stage, clinical N stage, histological grade, estrogen receptor, progesterone receptor, human epidermal growth factor receptor 2, and the use of radiotherapy, chemotherapy, targeted therapy and/or endocrine therapy. Correlates of overall survival (OS) and breast cancer-specific survival (BCSS) in the surgical and non-surgical groups were identified on multivariate analysis.
Results: The median overall survival in the surgical group and the non-surgical group was 30.3 months and 20.7 months, respectively. There were significant differences between the two groups according to subtype in the distribution of OS and BCSS (P <0.001). On multivariate analysis for the OS and BCSS, using non-surgical group as a reference, there were significant improvement for the luminal B subtype (P=0.010).
Conclusion: This study suggest that surgery of the primary breast tumor may have a positive impact on OS and BCSS in patients with stage IV breast cancer, especially in the luminal B subtype.
Keywords
<p>Metastatic breast cancer; Breast cancer subtypes; Surgery; Overall survival; Breast cancer-specific survival</p>
Article Details
1. Introduction
Breast cancer is a significant health issue and is one of the most frequent types of cancers with increasing incidence rates in the world [1]. Approximately 6% of women with newly diagnosed breast cancer present with stage IV disease in the United States [1, 2]. According to the International Consensus Guidelines for Advanced Breast Cancer (ABC), the standard treatment for stage IV breast cancer patients is systemic therapy, including chemo-therapy, endocrine therapy, and/or targeted therapy [3, 4].
Traditionally, surgical removal of the primary breast tumor has been used only for palliation and relief of symptoms such as bleeding, inflammation, and skin ulceration. To date, the role of primary tumor excision in patients with stage IV breast cancer remains controversial, however randomized data published to date does not show a survival benefit. This uncertainty is illustrated by the National Comprehensive Cancer Network (NCCN) guideline, which states, “the role and timing of surgical removal of the primary (breast cancer) in patients presenting with de novo stage IV disease is the subject of ongoing investigations.” In clinical practice, the molecular nature of the breast cancer subtypes is a major determinant of the type of systemic therapy administered. Significant differences have been observed in response to treatment and survival outcome among the various subtypes. Therefore, accurate prognostication depends on multiple variables (such as ER, PR, HER2, grading, Ki67, age, lymph node involvement) that impact recurrence and survival [5-7]. The primary objective of this investigation was to evaluate the clinicopathologic characteristics of the surgical group and the non-surgical group in patients with stage IV breast cancer. The secondary objective was to investigate whether there is an association with surgical benefit and subtype with regard to the overall survival (OS) and breast cancer-specific survival (BCSS) rates.
2. Patients and Methods
2.1 Study population
From an institutional database, 255 women in Tri-Service General Hospital, Taipei, Taiwan, were consecutively selected from patients with confirmed histopathologic diagnosis with stage IV breast cancer treated between January 2006 and October 2016. Information recorded for patients included age at diagnosis, year of diagnosis, and date of death or last contact. The data also included death from breast cancer or other diseases/reasons ascertained from follow-up lasting until November 2018. Institutional review board approval was obtained through our institution’s human investigations committee.
2.2 Eligibility criteria
We collected data on the clinicopathologic characteristics, including clinical T stage, clinical N stage, histological grade (G), estrogen receptor status (ER), progesterone receptor status (PR), the HER2 expression along with treatment factors including surgery, radiotherapy, chemotherapy, targeted therapy and endocrine therapy. In our study, grading was used to capture proliferation activity as described earlier: luminal A (ER+ and/or PR+, HER2–, and G1/2), luminal B (ER+ and/or PR+, HER2–, and G3), luminal HER2+ (ER+ and/or PR+, HER2+, and any grade), HER2-type (ER–, PR–, and HER2+), and triple negative (TN) (ER–, PR–, and HER2–). ER/PR positivity was determined by immunohistochemical (IHC) analysis of the number of positively stained nuclei (>1%+). Tumors were considered as HER2+ when cells presented strong membrane staining (3+). Tumors exhibiting 0 or 1+ staining for HER2 protein overexpression were considered to be HER2−. In cases of equivocal membrane staining (score 2+) for HER2, fluorescence in situ hybridization (FISH) was used to evaluate gene amplification [8, 9]. Patients who did not complete the data of tumor characteristics or treatment were excluded.
2.3 Statistical analysis
Interrelationships between variables were assessed using contingency table analysis with the chi-square test for trend as appropriate. Univariate and multivariate survival analyses were performed using the Kaplan-Meier analysis and Cox proportional hazards model with a stepwise backward elimination to derive a final model of variables with a significant independent relationship with OS and BCSS. All statistical analyses were 2-sided with significance defined as a P value <0.05. Statistical analyses were performed using SPSS, version 22.0, statistical software (IBM, Armonk, NY).
3. Results
3.1 Clinicopathologic characteristics
The clinicopathologic characteristics and treatment of the 255 patients with stage IV breast cancer by surgical intervention (surgery vs. no surgery) are shown in Table 1. For the entire cohort, the median overall survival (OS) in surgical group and non-surgical group was 30.3 months and 20.7 months, respectively. There were significant differences between the surgical group and the non-surgical group with respect to clinical T stage (P <0.001), clinical N stage (p =0.021) and receipt of radiotherapy (p =0.011). In our series, 67 (57.8%) patients with clinical T4 stage did not undergo surgery compared with 21 (27.6%) patients who did undergo local therapy. In the surgical group, 52.6% of patients did not receive radiotherapy versus 69.2% of patients in the non-surgical group. There was also significant improvement in the OS (P <0.001) and BCSS (P <0.001) for the surgery group versus non-surgery group.
3.2 Survival outcomes
The OS and BCSS rates according to clinicopathologic characteristics and treatment are reported in Table 2. In the surgical group, there were significant differences in OS by subtype, receipt of chemotherapy, and endocrine therapy (P=0.012, 0.050, and <0.001, respectively); in the non-surgical group, there were significant differences in OS between receipt of chemotherapy and endocrine therapy (P=0.002 and 0.025, respectively). There were also significant differences in the BCSS according to the surgical group among the breast cancer subtypes, chemotherapy, and endocrine therapy (P=0.012, < 0.048 and <0.001, respectively). There were significant differences in BCSS according to the non-surgical group between chemotherapy, and endocrine therapy (P=0.004, 0.024, respectively). Kaplan-Meier plots were generated to compare the OS and BCSS rates according to the surgical group and the non-surgical group among the subtypes (P <0.001, and <0.001, respectively) (Figure 1). In terms of OS, the median OS in surgery vs non-surgery group was 30.3 months and 20.7 months. The median OS in luminal A of surgery vs non-surgery group was 83.2 and 46.9 months, which in luminal B was 50.5 and 16.7 months, and luminal HER2 was about 75.8 and 22.6 months, and HER2 subtype was 31.6 and 65 months, and TN breast cancer was 25.6 and 6.4 months. There were significant differences according to subtypes in luminal A, luminal B, and luminal HER2 (P=0.020, 0.001 and 0.016, respectively). In terms of BCSS, there were also significant differences according to the breast cancer subtypes in luminal A, luminal B, and luminal HER2 (P=0.029, 0.001 and 0.016, respectively). Multivariate Cox proportional hazard analysis was performed to identify risk factors that showed a significant association with survival outcomes in the surgical and non-surgical groups disaggregated by subtypes after controlling for age, clinical T, clinical N, radiotherapy, chemotherapy, and endocrine therapy (Table 3). We showed that surgery of the primary breast tumor has a positive impact on OS and BCSS in stage IV breast cancer patients among subtypes, especially in the luminal B subtype (P =0.003/ [ (HR), 12.411; 95 % (CI), 2.302–66.906]).
|
Variable |
Surgical group (n=96) |
Non-surgical group (n=159) |
P valueb |
|
Age |
0.163 |
||
|
<= 51 |
46 (47.9) |
62 (39.0) |
|
|
>51 |
50 (52.6) |
97 (61.0) |
|
|
Grade |
0.844 |
||
|
1 |
3 (3.3) |
4 (3.5) |
|
|
2 |
41 (45.6) |
58 (50.4) |
|
|
3 |
46 (51.1) |
53 (46.1) |
|
|
Clinical T stage |
<0.001* |
||
|
T1 |
5 (6.6) |
4 (3.4) |
|
|
T2 |
33 (43.4) |
22 (19.0) |
|
|
T3 |
17 (22.4) |
23 (19.8) |
|
|
T4 |
21 (27.6) |
67 (57.8) |
|
|
Clinical N stage |
0.021* |
||
|
N0 |
18 (24.0) |
21 (19.4) |
|
|
N1 |
32 (42.7) |
33 (30.6) |
|
|
N2 |
19 (25.3) |
27 (25.0) |
|
|
N3 |
6 (8.0) |
27 (25.0) |
|
|
Estrogen receptor |
0.320 |
||
|
postive |
63 (70.8) |
93 (64.1) |
|
|
Negative |
26 (29.2) |
52 (35.9) |
|
|
Progesterone receptor |
0.392 |
||
|
postive |
76 (84.4) |
114 (79.7) |
|
|
Negative |
14 (15.6) |
29 (20.3) |
|
|
HER2 |
0.254 |
||
|
postive |
19 (28.8) |
42 (37.8) |
|
|
Negative |
47 (71.2) |
69 (62.2) |
|
|
Radiotherapy |
0.011* |
||
|
Yes |
45 (47.4) |
49 (30.8) |
|
|
No |
50 (52.6) |
110 (69.2) |
|
|
Chemotherapy |
0.076 |
||
|
Yes |
92 (95.8) |
143 (89.9) |
|
|
No |
4 (4.2) |
16 (10.1) |
|
|
Endocrine therapy |
0.202 |
||
|
Yes |
55 (57.3) |
78 (49.1) |
|
|
No |
41 (42.7) |
81 (50.9) |
|
|
Overall survival |
<0.001* |
||
|
Alive |
46 (47.9) |
38 (23.9) |
|
|
Deceased |
50 (52.1) |
121 (76.1) |
|
|
Breast cancer-specific survival |
<0.001* |
||
|
Alive |
46 (48.4) |
38 (24.8) |
|
|
Deceased |
49 (51.6) |
115 (70.1) |
|
HER2=human epidermal growth factor receptor 2. a. The total number of cases and controls does not correspond because of missing data. b. P value from two-sided x2 test. *Statistically significant
Table 1: Clinicopathologic characteristics of all patients with stage IV breast cancer (n=255)a.
HER2=human epidermal growth factor receptor 2
- The total number of cases and controls does not correspond because of missing data.
- All P values from two-sided x2 test.
*Statistically significant
Table 2: Outcome of survival rate according to clinical characteristics and surgical groupsa.
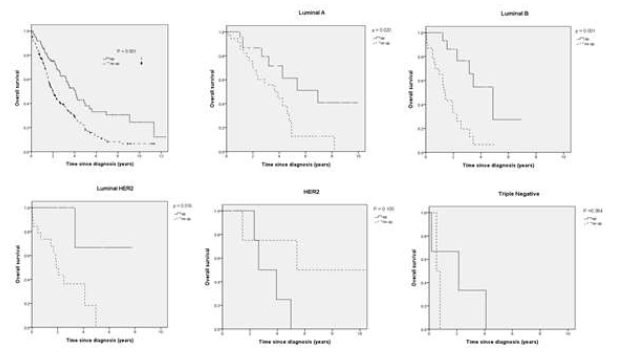
1 A) Overall Survival
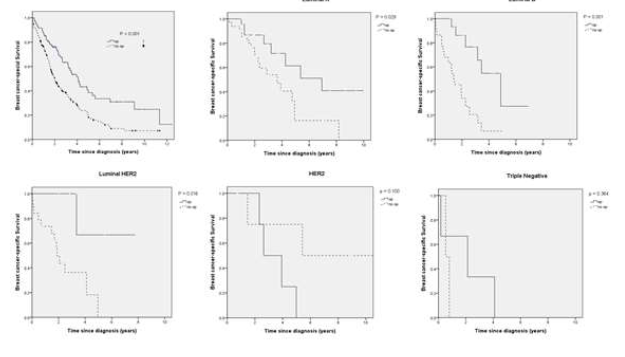
1 B) Breast cancer-specific Survival
Figure 1: A) Overall survival and B) Breast cancer-specific survival for surgery group and non-surgery group among breast cancer subtypes.
|
Luminal A |
Luminal B |
Luminal HER2 |
HER2 |
TN |
|
|
P value / HR (95%CI) |
|||||
|
Overall survival |
|||||
|
Surgical group |
1 |
1 |
1 |
1 |
1 |
|
Non-surgical group |
0.428 /1.969 (0.369-10.511) |
0.003?/12.411 (2.302-66.906) |
0.061/14.337 (0.889-231.355) |
NA |
NA |
|
Breast cancer-specific survival |
|||||
|
Surgical group |
1 |
1 |
1 |
1 |
1 |
|
Non-surgical group |
0.428 /1.969 (0.369-10.511) |
0.003?/12.411 (2.302-66.906) |
0.061/14.337 (0.889-231.355) |
NA |
NA |
HR=hazard ratio, CI=confidence interval.
- The model is adjusted for age, clinical T, clinical N, radiotherapy, chemotherapy and endocrine therapy.
*Statistically significant.
Table 3: Multivariate analysis of overall survival and breast cancer-specific survival rates according to the surgical group and non-surgical group among breast cancer subtypesa.
4. Discussion
Based on the International Consensus Guidelines for ABC, systemic therapy is the mainstream treatment for patients with stage IV breast cancer. The role of surgical excision of the primary tumor in stage IV breast cancer has been controversial due to conflicting outcomes from both retrospective and prospective studies. Some observational studies have shown that 35–60% of breast cancer patients with stage IV disease at diagnosis received surgical extirpation of the primary tumor and that this treatment was associated with a survival advantage [10-12]. A review of retrospective studies on the management of de novo metastatic breast cancer has largely shown mixed findings, with some studies reporting an improved OS with surgical intervention [2, 10, 13-16]. Other retrospective studies accounting for selection bias in the surgical group, mainly through matched pair analysis, show this survival benefit may no longer be apparent [17-19]. Two prospective randomized trials led by Soran et al. [20] (Turkey) and Badwe et al. [21] (India) evaluated the effect that removal of the primary tumor and axillary lymph nodes had on OS and progression-free survival. Soran et al. [20] presented the first randomized study to show a statistically significant improvement in median survival with surgery for patients with de novo stage IV breast cancer at the 5-year follow-up assessment. Notable limitations for this trial include that patient with HER2-positive disease were not treated with HER2-directed therapy, which would not be considered standard of care in most developed countries. In contrast, Badwe et al. [21] reported that locoregional resection of the primary tumor did not increase OS for the patients who had responded to frontline chemotherapy.
In the United States, it would be difficult not to initiate systemic therapy up front for patients with de novo metastatic breast cancer. One study, which addresses the role of systemic therapy followed by surgical intervention in this subgroup, is the United States-based Translational Breast Cancer Research Consortium (TBCRC) 013 trial, a prospective registry trial that enrolled 112 patients with an intact primary tumor between 2009 and 2012 at 14 institutions [22, 23]. After the patients had received first-line therapy by their treating providers, all the responders (94 patients, 85%) were considered for elective surgery. A multivariable analysis showed that surgery of the primary cancer did not improve overall survival among the responders with a median survival of 71 months versus 65 months for the patients without surgery (or 30-month survival rates of 77 and 76%, respectively; p=0.85). As such, we await the findings of prospective randomized trials (Eastern Cooperative Oncology Group [ECOG] 2108) to help add clarity to this controversy. In ECOG 2108, patients with disease who do not progress during initial systemic therapy and were stratified by molecular subtype are randomized to continued systemic therapy versus surgery with intention for negative surgical margins, either through breast-conserving therapy involving lumpectomy and radiation therapy or total mastectomy with or without radiation.
In many of the retrospective studies, women who received surgery tended to be younger, have smaller tumors, fewer comorbidities, lower burden of metastatic disease, were less likely to have visceral metastases, and more likely to have access to better care [10, 24]. The absolute survival benefit observed for women with small primary breast tumors is also consistent with a previous meta-analysis [24]. Thomas et al. [10] reported that women with larger tumors received surgery less often, which mirrors our findings. According to our database, only 39% of women who presented with stage IV breast cancer received surgery to the primary tumor. We found that patients who received surgery tended to have smaller tumors and fewer lymph node metastases. Breast cancer outcomes are steadily improving, and the paradigm shift of viewing stage IV disease as a chronic illness to be managed, rather than a terminal event, suggests that the role of surgery will be constantly evolving. There are several potential limitations to this study. First, the data used in this study are retrospective and not randomized. Second, the small sample size may limit our study’s findings especially when stratifying by molecular subtype. In conclusion, we found that surgery of the primary breast tumor has a positive impact on OS and BCSS in stage IV breast cancer patients among subtypes, especially in the luminal B subtype.
Acknowledgments
The authors would like to thank Enago (www.enago.tw) for the English language review.
References
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA: Cancer JClin 63 (2013): 11-30.
- Xie Y, Lv X, Luo C, et al. Surgery of the primary tumor improves survival in women with stage IV breast cancer in Southwest China: A retrospective analysis. Medicine 96 (2017): e7048.
- Untch M, Augustin D, Ettl J, et al. ABC3 consensus commented from the perspective of the German guidelines: Third International Consensus Conference for Advanced Breast Cancer (ABC3), Lisbon, 07. 11. 2015. Geburtshilfe and Frauenheilkunde 76 (2016): 156-163.
- Untch M, Wurstlein R, Marschner N, et al. 4th International Consensus Conference on Advanced Breast Cancer (ABC4), Lisbon, November 4, 2017: ABC4 Consensus: Assessment by a Panel of German Experts. Geburtshilfe und Frauenheilkunde 78 (2018): 469-480.
- Vasconcelos I, Hussainzada A, Berger S, et al. The St. Gallen surrogate classification for breast cancer subtypes successfully predicts tumor presenting features, nodal involvement, recurrence patterns and disease free survival. Breast 29 (2016): 181-185.
- Minicozzi P, Bella F, Toss A, et al. Relative and disease-free survival for breast cancer in relation to subtype: A population-based study. J Cancer Res Clin 139 (2013): 1569-1577.
- Xue C, Wang X, Peng R, et al. Distribution, clinicopathologic features and survival of breast cancer subtypes in Southern China. Cancer Sci 103 (2012): 1679-1687.
- Salhia B, Tapia C, Ishak EA, et al. Molecular subtype analysis determines the association of advanced breast cancer in Egypt with favorable biology. BMC Womens Health 11 (2011): 44.
- Puig-Vives M, Sanchez MJ, Sanchez-Cantalejo J, et al. Distribution and prognosis of molecular breast cancer subtypes defined by immunohistochemical biomarkers in a Spanish population-based study. Gynecol Oncol 130 (2013): 609-614.
- Thomas A, Khan SA, Chrischilles EA, et al. Initial surgery and survival in stage IV breast cancer in the United States, 1988-2011. JAMA Surg 151 (2016): 424-431.
- Lane WO, Thomas SM, Blitzblau RC, et al. Surgical resection of the primary tumor in women with De Novo stage IV breast cancer: Contemporary practice patterns and survival analysis. Ann Surg (2017).
- Warschkow R, Guller U, Tarantino I, et al. Improved survival after primary tumor surgery in metastatic breast cancer: A propensity-adjusted, population-based SEER trend analysis. Ann Surg 263 (2016): 1188-1198.
- Khan SA, Stewart AK, Morrow M. Does aggressive local therapy improve survival in metastatic breast cancer?. Surgery 132 (20): 620-626.
- Babiera GV, Rao R, Feng L, et al. Effect of primary tumor extirpation in breast cancer patients who present with stage IV disease and an intact primary tumor. Ann Surg Oncol 13 (2006): 776-782.
- Lang JE, Tereffe W, Mitchell MP, et al. Primary tumor extirpation in breast cancer patients who present with stage IV disease is associated with improved survival. Ann Surg Oncol 20 (2013): 1893-1899.
- Blanchard DK, Shetty PB, Hilsenbeck SG, et al. Association of surgery with improved survival in stage IV breast cancer patients. Ann Surg 247 (2008): 732-738.
- Ruiterkamp J, Voogd AC, Bosscha K, et al. Presence of symptoms and timing of surgery do not affect the prognosis of patients with primary metastatic breast cancer. EurJ Surg Oncol 37 (2011): 883-889.
- Dominici L, Najita J, Hughes M, et al. Surgery of the primary tumor does not improve survival in stage IV breast cancer. Breast Cancer Res Tr 129 (2011): 459-465.
- Cady B, Nathan NR, Michaelson JS, et al. Matched pair analyses of stage IV breast cancer with or without resection of primary breast site. Ann Surg Oncol 15 (2008): 3384-3395.
- Soran A, Ozmen V, Ozbas S, et al. Randomized trial comparing resection of primary tumor with no surgery in stage IV breast cancer at presentation: Protocol MF07-01. Ann Surg Oncol 25 (2018): 3141-3149.
- Badwe R, Parmar V, Hawaldar R, et al. Surgical removal of the primary breast tumor and axillary lymph nodes in women with metastatic breast cancer at first presentation: A randomized controlled trial. In: Presented at the 2013 San Antonio Breast Cancer symposium (2013).
- King TA, Lyman J, Gonen M, et al. A prospective analysis of surgery and survival in stage IV breast cancer (TBCRC 013). ASCO Annual Meeting (2016).
- Barbie TU, Golshan M. De Novo stage 4 metastatic breast cancer: A surgical disease?. Ann Surg Oncol 25 (2018): 3109-3111.
- Harris E, Barry M, Kell MR. Meta-analysis to determine if surgical resection of the primary tumour in the setting of stage IV breast cancer impacts on survival. Ann Surg Oncol 20 (2013): 2828-2834.


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 