The Role of Percutaneous Nephrostomy for Uretric Obstruction due to Advanced Abdominopelvic Malignancy: A Retrospective Analysis
Rida Mansoor1, Muhammad Faisal2*, Shahan Raza3, Hannan Ali3, Osama Shakeel3, Zaeem Shahid3, Haroon Hafiz1
1Department of Palliative Medicine, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, Pakistan
2Shaukat Khanum Cancer Hospital, Peshawar, Pakistan
3Department of Surgical Oncology, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, Pakistan
*Corresponding Author: Muhammad Faisal, Shaukat Khanum Cancer Hospital, Peshawar, Pakistan
Received: 31 March 2022; Accepted: 29 April 2022; Published: 16 May 2022
Article Information
Citation: Rida Mansoor, Muhammad Faisal, Shahan Raza, Hannan Ali, Osama Shakeel, Zaeem Shahid, Haroon Hafiz. The Role of Percutaneous Nephrostomy for Uretric Obstruction due to Advanced Abdominopelvic Malignancy: A Retrospective Analysis. Journal of Cancer Science and Clinical Therapeutics 6 (2022): 214-221.
View / Download Pdf Share at FacebookAbstract
Introduction: In advanced or metastatic abdominopelvic malignancy, ureteric obstruction is a known complication. (PCN) is a diversion procedure for decompression in order to improve renal function. It is debatable whether PCN is an effective management to relieve ureteric obstruction in a stage IV abdominoperineal disease. The aim of the study is to determine the outcome of PCN tube insertion in a palliative care setting.
Methodology: The study was conducted at Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore (SKMCH&RC). Data was retrieved from the electronic Hospital Information System (HIS) of the hospital. Duration of the study was from January, 2018 to December, 2020. We included patients who underwent percutaneous nephrostomy under palliative setting.
Results: A total of 111 patients were included in the study. The median age at the time of nephrostomy was 4915.6 years. 67 patients (60.4%) were males, while 44 patients (39.6%) were females. Pre procedural median level of creatinine were was 3.3±3.36. Unilateral nephrostomy was performed in 71 patient (63.9%), while 40 patients (36.1%) underwent bilateral nephrostomy. Post-procedural mean creatinine level were 2.52 + 2.80. Relief of symptoms was observed among 68 patients (62.2%). 75 patients (67.6%) did not develop any complications after the procedure. The two most common complications of the procedure in our study were dislodgement (9%) and infection (9%).
Conclusion: PCN is good for symptomatic and biochemical relief even in stage IV cancer, however due to increased incidence of complications in these patients the benefits might outweigh the risks.
Keywords
<p>Abdominopelvic Tumors; Percutaneous Nephrostomy; Ureteric Obstruction</p>
Article Details
1. Introduction
In advanced pelvic malignancies, ureteric obstruction is one of the most common complication. It is due to invasion, direct compression by tumor or encasement by mesenteric lymph nodes [1]. With bilateral ureteric obstruction, patients may develop progressive and fatal renal failure. There are many ways to relieve the obstructive symptoms such as double-J stents or percutaneous nephrostomy. Retrograde stenting is frequently impossible because of anatomical deformities, ureteral compression, or bleeding. Percutaneous nephrostomy (PCN) is a diversion procedure for decom-pression of renal collecting system in order to improve renal function. In the past few decades, with the improvement of material of PCN and radiology techniques, PCN has become more safe and effective [2]. It results in early improvement of biochemical and laboratory para-meters of renal function test [2, 3].
The PCN does not have these technical difficulties however decompression by PCN of the urinary tract can be associated with prolonged morbidity with some patients spending their remaining limited lifetime in hospital(1). PCN provides better survival outcomes but it does not necessarily improves quality of life [4]. Many ethical, philosophical and emotional questions have been raised which make the indication of nephrostomy even more complex in patients with poor prognosis [5, 6]. The aim of the study is to determine the role of PCN tube insertion in a palliative care patient and its outcomes.
2. Methodology
It is a retrospective study with convenience sampling. The study was conducted at Shaukat Khanum Memorial Cancer Hospital and Research Centre (SKMCH & RC), Pakistan. The duration of study was from January, 2018 to December, 2020. All the patients who underwent PCN in advanced stage of the cancer under palliative setting were included in the study. The Variables recorded were age, gender, site and type of cancer, cause of obstruction, pre-procedural and post-procedural creatinine and survival outcomes. We included patients who had advanced pelvic malignancies requiring PCN with palliative intent at our hospital. Exclusion criteria include patients who underwent double-J stent or PCN for non-pelvic malignancies or curative intent was there. We also excluded patients with incomplete data.
Data was retrieved from electronic Hospital Information System (HIS) of SKMCH & RC. Calculations were performed with Statistical Package for the Social Sciences (SPSS Ver20) for Windows. Data was described using median with minimum and maximum value for skewly distributed quantitative variables. For categorical variables, number of observations and percentages were reported. The chi-square test was used to check for differences between proportions. Kaplan Meier Curve was used to estimate the survival outcomes.
3. Results
A total of 111 patients were included in the study. The median age in our study was 49 ± 15.6 years. Among our cohort, 67(60.4%) patients were males while 44(39.6%) patients were females. The male to female ratio was 1.5:1. The type of tumor in our study is illustrated in Figure 1. The mean pre-procedural renal function test was 3.3 ± 3.36. Pre-procedural indication of PCN was bilateral hydronephrosis in 60(54.1%) patients, left hydronephrosis in 29(26.1%) patients, and right hydronephrosis in 22(19.8%) patients. 71(64%) patients underwent unilateral PCN placement while 40(36%) patients had bilateral PCN placement. Out of 71 patients who underwent unilateral hydronephrosis, 36 (32.4%) patients had left PCN placement and 35(31.5%) patients had right PCN placement. Post-procedural mean creatinine level were 2.52 ± 2.80. Relief of symptoms was observed among 69(62.2%) patients. 75(67.6%) patients did not develop any complications after the procedure. The two most common complications of the procedure in our study were dislodgement and infection i.e. 10 patients each group (9%), followed by bleeding (n=6, 5.4%), blockage (n=5, 4.5%), leak (n=3, 2.7%), and renal failure (n=2, 1.8%). 22 patients (19.8%) had emergency department visits after the procedure related to PCN procedure. Eight patients (7.2%) were admitted post-procedure in the hospital for the management of the complications.
In our study, 66(59.5%) patients were dead at the time of study, 40(4.5%) patients were alive, and 40(36%) patients are lost to follow-up. The median survival of the patients in our study was 6 months ranging from 4.964 to 7.036 months (Figure 2). The median survival of prostate and urinary bladder cancers were 8 months respectively followed by colorectal malignancies with a median survival of 6 months (5.14-6.8 months). The median survival was 4 months (2.74-5.25 months) and 2 months (0.539-3.46 months) respectively for gynecological and germ cell tumors. The survival in patients who underwent unilateral PCN place-ment was 5 months (3.2-6.7 months) while those who underwent bilateral PCN placement was 6 months (4.83-7.16 months) (Figure 3 and Figure 4). The median survival in male and female patients in our study was 7 months (5.21-8.78 months) and 5 months (3.64-6.35 months) respectively.
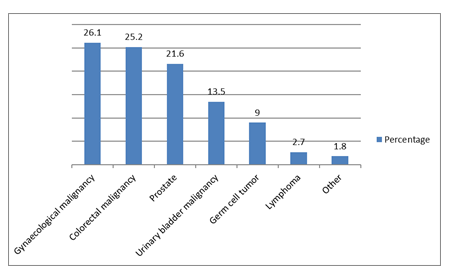
Figure1: Percentage of type of carcinoma.
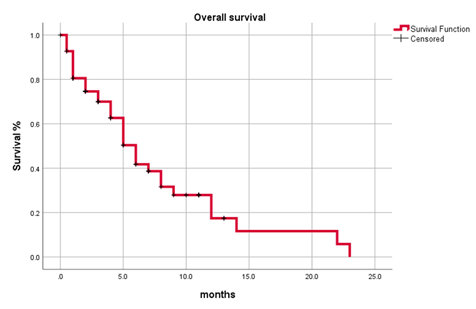
Figure 2: Overall survival.
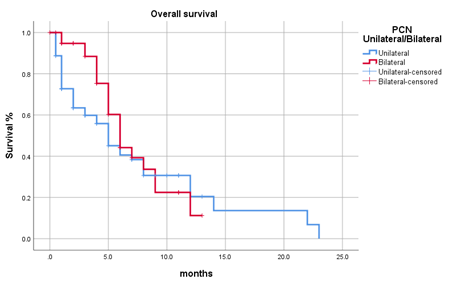
Figure 3: Survival based on laterality of placement.
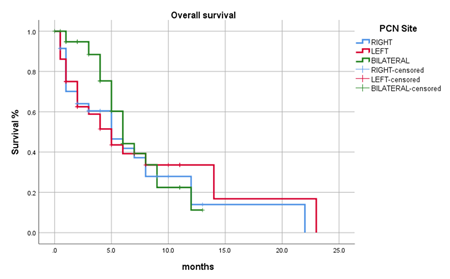
Figure 4: Survival based on side of placement P= 0.537.
4. Discussion
Ureteric obstruction is a troublesome sign in advanced malignancies with a poor outcome in the presence of available forms of treatment [7]. Due to advanced nature of disease, the functional status of the patients is usually poor. These interventions pose ethical issues whether these investigations and treatments are in the best interest of the patients. The judgment is to decide which patients would benefit the patients from decompression of the urinary tract. Such interventions need to be carefully addressed in view of ethical concerns and one has to weigh the benefits which could outweigh the drawbacks. There is a dire need to accurately predict and identify the patients who could benefit from decompression of urinary tract. Palliative decom-pression methods using either retrograde stenting or PCN do improve the outcomes but they are associated with significant morbidity [8]. However, retrograde stenting failure rates is upto 79% [9] especially in bladder and prostatic tumors [9, 10]. Domico and Dewolf reported a high post-operative failure rate after ureteric stents (46%). However, PCN provides a high success rate of up to 98% [11]. PCN was first described by Goodwin et al in 1955 for decompression of an obstructed urinary system [12]. Goodwin performed it for temporary purposes with the help of fluoroscopy. In 1965, Bartley first used seldinger technique for the PCN placement. PCN is an interventional radiology procedure in which a catheter is placed in the renal pelvis through the skin using fluoroscopic guided or ultrasound guided imaging. Demerit of fluoroscopy over ultrasound is the utilization of ionizing radiation.
Some studies report the incidence of complication rate of 10% with mortality rate of 0.05% to 0.3% [13, 14]. The complication rate after PCN insertion in our study was 32.4%. Due to the variable definition of complications, the rate of complications after this procedure is variable in the literature [15, 16]. Carrafiello et al. and Rafal et al. reported the incidence rate of minor complications of 14.4% and 6.1% respectively [17, 18]. However, some studies report the complication rates as high as 41% with 26% of the patients never leaving the hospital [19-21]. The high complications rate in our setting could be due to the patients with advanced pelvic malignancies with long standing urinary tract obstruction. The hospital admission rates after PCN insertion reported in the literature was 5-69% [22-24]. In our hospital, the post-procedural admission rate was 7.2% which is within the standard guidelines. In one of the studies, the hospital re-admission rate was 39% in the first 30-days [19-22]. As these patients suffer from advanced metastatic disease, the quality of life matters more than the quantity of life.
In our study, the median survival times after PCN insertion was 6 months. The median survival after this procedure in published literature was 5.2 months (range 2 to 7 months) [24-28]. Nariculam and colleagues in 2009, found that overall mean time to death after PCN was 7.5 months as compared to 8 months in our cohort. For bladder carcinoma, the reported survival rate was 4.9 months [29]. In our study, the reported survival rate was 8 months. We had improved survival contrary to reported literature which could be due to patient selection or the cancer behavior in our sets of patient. In terms of systemic complications, sepsis is the most common (15%) entity reported. Our cohort has shown dislodgment of nephrostomy tube to be the most common (10%) problem which is far less from what is found in published literature. Infection and bleeding are the next in the list of complications with an incidence rate of 9% and 6% respectively [8, 11, 19]. To the best of our knowledge this is the first study from Pakistan reporting the indications and complications associated with PCN insertion in a palliative setting. The limitation of the study is its retrospective nature and its sample size. Pain scores, emotional well-being and level of satisfaction could be the potential objectives in future studies.
5. Conclusion
PCN is found to be an effective treatment for biochemical relieve and alleviate obstructive symptoms in advanced abdominopelvic disease. But its utilization could not be transformed into survival benefit in the past studies. Decision on use of PCN must take into consideration clear symptomatic benefits, multidisciplinary decision making along with the involvement of patient himself and the family.
References
- Kouba E, Wallen EM, Pruthi RS. Management of ureteral obstruction due to advanced malignancy: optimizing therapeutic and palliative outcomes. The Journal of urology 180 (2008): 444-450.
- Soper J, Blaszczyk T, Oke E, et al. Percutaneous Nephrostomy in Gynecologic Oncology Patients. The Journal of Urology 141 (1989): 472-473.
- Hoe J, Tung K, Tan E. Re-evaluation of indications for percutaneous nephrostomy and interventional uroradiological procedures in pelvic malignancy. British journal of urology 71 (1993): 469-472.
- Kinn AC, Ohlsén H. Percutaneous nephrostomy--a retrospective study focused on palliative indications. APMIS Supplementum (2003): 66-70.
- Fallon B, Olney L, Culp D. Nephrostomy in cancer patients: to do or not to do?. British journal of urology 52 (1980): 237-242.
- Chiou RK, Chang WY, Horan JJ. Ureteral obstruction associated with prostate cancer: the outcome after percutaneous nephrostomy. The Journal of urology 143 (1990): 957-959.
- Wong L-M, Cleeve LK, Milner AD, et al. Malignant ureteral obstruction: outcomes after intervention. Have things changed? The Journal of urology 178 (2007): 178-183.
- Shekarriz B, Shekarriz H, Upadhyay J, et al. Outcome of palliative urinary diversion in the treatment of advanced malignancies. Cancer: Interdisciplinary International Journal of the American Cancer Society 85 (1999): 998-1003.
- Chitale S, Scott-Barrett S, Ho E, et al. The management of ureteric obstruction secondary to malignant pelvic disease. Clinical radiology 57 (2002): 1118-1121.
- Ganatra AM, Loughlin KR. The management of malignant ureteral obstruction treated with ureteral stents. The Journal of urology 174 (2005): 2125-2128.
- Ku JH, Lee SW, Jeon HG, et al. Percutaneous nephrostomy versus indwelling ureteral stents in the management of extrinsic ureteral obstruction in advanced malignancies: are there differences?. Urology 64 (2004): 895-899.
- Goodwin WE, Casey WC, Woolf W. Percutaneous trocar (needle) nephrostomy in hydronephrosis. Journal of the American Medical Association 157 (1955): 891-894.
- McGraw JK, Cardella J, Barr JD, et al. Society of Interventional Radiology quality improvement guidelines for percutaneous vertebroplasty. Journal of Vascular and Interventional Radiology 14 (2003): S311-S315.
- Zagoria RJ, Dyer RB. Do's and don't's of percutaneous nephrostomy. Academic radiology 6 (1999): 370-377.
- Wah T, Weston M, Irving H. Percutaneous nephronstomy insertion: outcome data from a propspective multioperator study at a UK training centre. Clinical radiology 59 (2004): 255-261.
- Kaskarelis IS, Papadaki MG, Malliaraki NE, et al. Complications of percutaneous nephrostomy, percutaneous insertion of ureteral endoprosthesis, and replacement procedures. Cardiovascular and interventional radiology 24 (2001): 224-228.
- Carrafiello G, Lagana D, Mangini M, et al. Complications of percutaneous nephrostomy in the treatment of malignant ureteral obstructions: Single–centre review. La radiologia medica 111 (2006): 562-571.
- Turo R, Horsu S, Broome J, et al. Complications of percutaneous nephrostomy in a district general hospital. Turkish Journal of Urology 44 (2018): 478.
- Ishioka J, Kageyama Y, Inoue M, et al. Prognostic model for predicting survival after palliative urinary diversion for ureteral obstruction: analysis of 140 cases. The Journal of urology 180 (2008): 618-621.
- Izumi K, Mizokami A, Maeda Y, et al. Current outcome of patients with ureteral stents for the management of malignant ureteral obstruction. The Journal of urology 185 (2011): 556-561.
- Monsky WL, Molloy C, Jin B, et al. Quality-of-life assessment after palliative interventions to manage malignant ureteral obstruction. Cardiovascular and interventional radiology 36 (2013): 1355-1363.
- Cordeiro MD, Coelho RF, Chade DC, et al. A prognostic model for survival after palliative urinary diversion for malignant ureteric obstruction: a prospective study of 208 patients. BJU international 117 (2016): 266-271.
- Wilson J, Urwin G, Stower M. The role of percutaneous nephrostomy in malignant ureteric obstruction. Annals of the Royal College of Surgeons of England 87 (2005): 21.
- Dienstmann R, da Silva Pinto C, Pereira MT, et al. Palliative percutaneous nephrostomy in recurrent cervical cancer: a retrospective analysis of 50 consecutive cases. Journal of pain and symptom management 36 (2008): 185-190.
- Jalbani MH, Deenari RA, Dholia KR, et al. Role of percutaneous nephrostomy (PCN) in malignant ureteral obstruction. JPMA The Journal of the Pakistan Medical Association 60 (2010): 280.
- Alawneh A, Tuqan We, Innabi A, et al. Clinical factors associated with a short survival time after percutaneous nephrostomy for ureteric obstruction in cancer patients: an updated model. Journal of Pain and Symptom Management 51 (2016): 255-261.
- Lienert A, Ing A, Mark S. Prognostic factors in malignant ureteric obstruction. BJU international 104 (2009): 938-941.
- Plesinac-Karapandzic V, Masulovic D, Markovic B, et al. Percutaneous nephrostomy in the management of advanced and terminal-stage gynecologic malignancies: outcome and complications. European journal of gynaecological oncology 31 (2010): 645-650.
- Ekici S, Sahin A, Özen H. Percutaneous nephrostomy in the management of malignant ureteral obstruction secondary to bladder cancer. Journal of endourology 15 (2001): 827-829.


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 