Use of Epigenetic Therapy Shortens Duration of Standard Chemotherapy for Ovarian Cancer with Minimal Toxicity to Normal Tissue
Leona Chang1, Krystal Hunter2, James Aikins1, Spencer Brown3, Olga Ostrovsky3*
1Division of Gynecologic Oncology, MD Anderson Cancer Center at Cooper, Cooper University Hospital, Camden, NJ, USA
2Cooper Research Institue – Biostatistics Group, Cooper University Hospital, Camden, NJ, USA
3Division of Surgical Research, Department of Surgery, Cooper, Cooper University Hospital, Camden, NJ, USA
*Corresponding Author: Dr. Olga Ostrovsky, Division of Surgical Research, Department of Surgery, Cooper, Cooper University Hospital, Camden, NJ, USA
Received: 27 March 2020; Accepted: 16 April 2020; Published: 27 April 2020
Article Information
Citation: Leona Chang, Krystal Hunter, James Aikins, Spencer Brown, Olga Ostrovsky. Use of Epigenetic Therapy Shortens Duration of Standard Chemotherapy for Ovarian Cancer with Minimal Toxicity to Normal Tissue. Journal of Cancer Science and Clinical Therapeutics 4 (2020): 100-114.
View / Download Pdf Share at FacebookAbstract
Objective: (1) Determine if sequential administration of standard chemotherapy (paclitaxel and cisplatin, P/C) with epigenetic drugs effectively targets ovarian cancer and limits toxicity to normal cells. (2) Define whether epigenetic treatment can shorten the exposure to P/C. Methods: Normal cells—adipocyte-derived stem cells (ASC), primary fibroblasts (PF), and human intestinal epithelial cells (HIEC-6)—were treated with 48 h IC50 values of P/C and epigenetic drugs, 5-azacytidine (AZA) and or suberoylanilide hydroxamic acid (SAHA), in combination and sequentially. The least toxic regimens to normal cells were administered to the ovarian cancer cell lines Caov-3, SKOV-3, OVCAR-3. Cell viability after treatments were assessed using the 3-(4,5-dimethylthiazol-2yl)-2,5-diphenyltetrazolium bromide (MTT) and cell count assays. Secretome analysis of conditioned medium collected from the treated ovarian cancer cells was performed using ELISA.
Results: P/C with AZA and SAHA targeted all ovarian cancer cell lines (82-99% cell death), but also caused significant normal cell death (66-100%). In contrast, P/C followed by AZA or SAHA is less toxic to ASC and PF (25-96% viability) when compared to a four-drug combination therapy (1% viability, p<0.0001). P/C followed by SAHA was least toxic to HIEC-6 (100% viability, p=0.0356). P/C followed by epigenetic drugs targets ovarian cancer cells more efficiently than two rounds of P/C (p<0.001). Levels of VEGF and IL-6 were downregulated after treatment with P/C followed by SAHA in SKOV-3 and OVCAR-3.
Conclusions: Sequential treatments of P/C with epigenetic drugs, specifically SAHA, preserves the viability of normal cells, efficiently targets ovarian cancer, and minimizes exposure to P/C.
Keywords
Ovarian cancer; Epigenetic therapy; Chemotherapy; 5-azacytidine; Suberoylanilide hydroxamic acid
Ovarian cancer articles, Epigenetic therapy articles, Chemotherapy articles, 5-azacytidine articles, Suberoylanilide hydroxamic acid articles
Ovarian cancer articles Ovarian cancer Research articles Ovarian cancer review articles Ovarian cancer PubMed articles Ovarian cancer PubMed Central articles Ovarian cancer 2023 articles Ovarian cancer 2024 articles Ovarian cancer Scopus articles Ovarian cancer impact factor journals Ovarian cancer Scopus journals Ovarian cancer PubMed journals Ovarian cancer medical journals Ovarian cancer free journals Ovarian cancer best journals Ovarian cancer top journals Ovarian cancer free medical journals Ovarian cancer famous journals Ovarian cancer Google Scholar indexed journals Epigenetic therapy articles Epigenetic therapy Research articles Epigenetic therapy review articles Epigenetic therapy PubMed articles Epigenetic therapy PubMed Central articles Epigenetic therapy 2023 articles Epigenetic therapy 2024 articles Epigenetic therapy Scopus articles Epigenetic therapy impact factor journals Epigenetic therapy Scopus journals Epigenetic therapy PubMed journals Epigenetic therapy medical journals Epigenetic therapy free journals Epigenetic therapy best journals Epigenetic therapy top journals Epigenetic therapy free medical journals Epigenetic therapy famous journals Epigenetic therapy Google Scholar indexed journals Chemotherapy articles Chemotherapy Research articles Chemotherapy review articles Chemotherapy PubMed articles Chemotherapy PubMed Central articles Chemotherapy 2023 articles Chemotherapy 2024 articles Chemotherapy Scopus articles Chemotherapy impact factor journals Chemotherapy Scopus journals Chemotherapy PubMed journals Chemotherapy medical journals Chemotherapy free journals Chemotherapy best journals Chemotherapy top journals Chemotherapy free medical journals Chemotherapy famous journals Chemotherapy Google Scholar indexed journals 5-azacytidine articles 5-azacytidine Research articles 5-azacytidine review articles 5-azacytidine PubMed articles 5-azacytidine PubMed Central articles 5-azacytidine 2023 articles 5-azacytidine 2024 articles 5-azacytidine Scopus articles 5-azacytidine impact factor journals 5-azacytidine Scopus journals 5-azacytidine PubMed journals 5-azacytidine medical journals 5-azacytidine free journals 5-azacytidine best journals 5-azacytidine top journals 5-azacytidine free medical journals 5-azacytidine famous journals 5-azacytidine Google Scholar indexed journals Suberoylanilide hydroxamic acid articles Suberoylanilide hydroxamic acid Research articles Suberoylanilide hydroxamic acid review articles Suberoylanilide hydroxamic acid PubMed articles Suberoylanilide hydroxamic acid PubMed Central articles Suberoylanilide hydroxamic acid 2023 articles Suberoylanilide hydroxamic acid 2024 articles Suberoylanilide hydroxamic acid Scopus articles Suberoylanilide hydroxamic acid impact factor journals Suberoylanilide hydroxamic acid Scopus journals Suberoylanilide hydroxamic acid PubMed journals Suberoylanilide hydroxamic acid medical journals Suberoylanilide hydroxamic acid free journals Suberoylanilide hydroxamic acid best journals Suberoylanilide hydroxamic acid top journals Suberoylanilide hydroxamic acid free medical journals Suberoylanilide hydroxamic acid famous journals Suberoylanilide hydroxamic acid Google Scholar indexed journals Malignant neoplasms articles Malignant neoplasms Research articles Malignant neoplasms review articles Malignant neoplasms PubMed articles Malignant neoplasms PubMed Central articles Malignant neoplasms 2023 articles Malignant neoplasms 2024 articles Malignant neoplasms Scopus articles Malignant neoplasms impact factor journals Malignant neoplasms Scopus journals Malignant neoplasms PubMed journals Malignant neoplasms medical journals Malignant neoplasms free journals Malignant neoplasms best journals Malignant neoplasms top journals Malignant neoplasms free medical journals Malignant neoplasms famous journals Malignant neoplasms Google Scholar indexed journals epigenetic treatment articles epigenetic treatment Research articles epigenetic treatment review articles epigenetic treatment PubMed articles epigenetic treatment PubMed Central articles epigenetic treatment 2023 articles epigenetic treatment 2024 articles epigenetic treatment Scopus articles epigenetic treatment impact factor journals epigenetic treatment Scopus journals epigenetic treatment PubMed journals epigenetic treatment medical journals epigenetic treatment free journals epigenetic treatment best journals epigenetic treatment top journals epigenetic treatment free medical journals epigenetic treatment famous journals epigenetic treatment Google Scholar indexed journals epithelial cells articles epithelial cells Research articles epithelial cells review articles epithelial cells PubMed articles epithelial cells PubMed Central articles epithelial cells 2023 articles epithelial cells 2024 articles epithelial cells Scopus articles epithelial cells impact factor journals epithelial cells Scopus journals epithelial cells PubMed journals epithelial cells medical journals epithelial cells free journals epithelial cells best journals epithelial cells top journals epithelial cells free medical journals epithelial cells famous journals epithelial cells Google Scholar indexed journals suberoylanilide hydroxamic acid articles suberoylanilide hydroxamic acid Research articles suberoylanilide hydroxamic acid review articles suberoylanilide hydroxamic acid PubMed articles suberoylanilide hydroxamic acid PubMed Central articles suberoylanilide hydroxamic acid 2023 articles suberoylanilide hydroxamic acid 2024 articles suberoylanilide hydroxamic acid Scopus articles suberoylanilide hydroxamic acid impact factor journals suberoylanilide hydroxamic acid Scopus journals suberoylanilide hydroxamic acid PubMed journals suberoylanilide hydroxamic acid medical journals suberoylanilide hydroxamic acid free journals suberoylanilide hydroxamic acid best journals suberoylanilide hydroxamic acid top journals suberoylanilide hydroxamic acid free medical journals suberoylanilide hydroxamic acid famous journals suberoylanilide hydroxamic acid Google Scholar indexed journals 5-azacytidine articles 5-azacytidine Research articles 5-azacytidine review articles 5-azacytidine PubMed articles 5-azacytidine PubMed Central articles 5-azacytidine 2023 articles 5-azacytidine 2024 articles 5-azacytidine Scopus articles 5-azacytidine impact factor journals 5-azacytidine Scopus journals 5-azacytidine PubMed journals 5-azacytidine medical journals 5-azacytidine free journals 5-azacytidine best journals 5-azacytidine top journals 5-azacytidine free medical journals 5-azacytidine famous journals 5-azacytidine Google Scholar indexed journals
Article Details
1. Introduction
Malignant neoplasms of the ovary account for more deaths than any other cancer of the female reproductive system [1]. In the United States in 2019, there are estimated to be 22,530 new cases of ovarian cancer with 13,980 deaths expected [2]. The 5-year survival rate for all stages combined is only 47.6% and 29.2% for distant metastasis [2]. The poor prognosis of ovarian cancer is primarily due to a lack of screening methods, nonspecific symptoms resulting in late stage of diagnosis, and recurrent disease that is resistant to conventional chemotherapy.
The standard of care for advanced ovarian cancer consists of primary debulking surgery (PDS) followed by platinum- and taxane- based chemotherapy [1]. For patients who are poor surgical candidates or have unresectable disease, neoadjuvant chemotherapy (NACT) should be considered [3]. The chemotherapeutic agents preferentially target rapidly dividing cells, including cancer cells, but even normal cells such as macrophages and fibroblasts involved in wound healing are susceptible to the toxic effects [4]. Hematologic toxicities, nausea, vomiting, fatigue, and neuropathy are other side effects commonly associated with chemotherapy. A balance between the treatment of ovarian cancer and minimizing toxic side effects is often a challenge. In addition, despite optimal cytoreduction and systemic chemotherapy, approximately 70% of patients’ disease will recur in the first 3 years [5]. Therefore, there has been a shift beyond conventional chemotherapy towards other treatment modalities such as targeted therapy, immunotherapy, and epigenetic drugs that can increase patient survival and minimize toxicities [6]. These new therapeutic agents also must take into consideration the timing of surgical intervention and wound healing, as the treatment of ovarian cancer is a complex, multi-modal approach.
Recent studies have shown cancer initiation and progression is not only affected by genetic alterations but also epigenetic modifications [7]. Epigenetics are heritable changes in gene expression that do not affect the DNA sequence itself. The two most notable epigenetic pathways are DNA methylation and histone acetylation, which are notably different in cancer [8, 9]. These modifications can contribute to cancer progression and metastases, but also contribute to chemoresistance in various cancers including ovarian [10, 11].
Several preclinical studies have shown epigenetic therapy, DNA methyltransferase inhibitors (DNMTi) or histone deacetylase inhibitors (HDAI), combined with standard chemotherapy to be effective for the treatment of ovarian cancer. Suberoylanilide hydroxamic acid (SAHA), via histone deactylation inhibition, has been shown to be cytotoxic to ovarian cancer cell lines when used in combination with a platinum or taxane compound [12, 13]. Usage of DNMTi 5-aza-2’-deoxycytidine (decitabine) combined with carboplatin increased sensitivity to carboplatin in mice xenografts [14]. A more recent in vitro study showed the combination of standard and epigenetic therapies exhibited high levels of toxicity against ovarian cancer cells and reversed effects of chemoresistance of metastatic omentum [15]. However, the few clinical translational studies demonstrated significant cytotoxic side effects of these combination therapies. In a Gynecologic Oncology Group sponsored phase II trial, SAHA or vorinostat as a single agent showed minimal activity in recurrent platinum-resistant or refractory ovarian cancer patients, as only two (7.4%) survived progression-free for longer than 6 months [16]. A phase II trial by Mendivil et al. assessed the combination of paclitaxel, carboplatin, and vorinostat as primary therapy for advanced stage ovarian cancer, was terminated early after 3 of 11 patients suffered gastrointestinal perforations [17]. Similarly, a phase I study by Matulonis et al. combined vorinostat with carboplatin and gemcitabine in recurrent platinum-sensitive patients and was terminated early due to unacceptable hematologic toxicity (grade 4 thrombocytopenia) [18].
Most of the experimental designs in these studies administered epigenetic and standard chemotherapy agents simultaneously, which resulted in significant adverse effects. Only three in vitro/in vivo studies explored the pharmacodynamics and effect of sequential treatment of epigenetic therapy with chemotherapy in ovarian cancer; their varied results led to the question of whether toxicity to normal tissues could be decreased by administrating the epigenetic therapy and standard chemotherapy in sequence [19-21]. The primary objective of this in vitro study was to determine the optimal sequence of standard chemotherapy use with epigenetic therapy that would maintain cytotoxic activity to ovarian cancer cell lines while limiting toxic side effects to normal tissues. The secondary objective was to establish whether the use of epigenetic treatment could allow for a shorter exposure time to standard chemotherapy, or to even take its place.
2. Materials and Methods
2.1. Cell cultures
Human ovarian adenocarcinoma cell lines Caov-3, SKOV-3, OVCAR-3 from the Type Culture Collection (ATCC, Manassas, VA, USA) were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM), McCoy’s 5A Medium, and RPMI-1640 (Gibco, Gaithersburg, MD) respectively, at 37°C in a humified atmosphere of 5% CO2. DMEM and McCoy’s 5A Medium were supplemented with 10% fetal bovine serum (FBS) and 1% penicillin and streptomycin. RPMI-1640 was supplemented with 20% FBS, 1% penicillin and streptomycin, and .01 mg/ml bovine insulin (Sigma-Aldrich, St. Louis, MO). Human intestinal epithelial cells (HIEC-6) were purchased from ATCC and cultured in OptiMEM 1 Reduced Serum Medium with 20 mM HEPES, 10 mM GlutaMAX, 10 ng/ml epidermal growth factor, and 4% FBS (all from Gibco, Gaithersburg, MD). Human primary dermal fibroblasts (PF) from ATCC were cultured with the aforementioned complete DMEM.
2.2. Human adipose-derived stem cells (ASCs) isolation and cell culture
Human adipose tissue was obtained with an approved Institutional Review Board for the Protection of Human Subjects protocol (IRB). Cells were washed with phosphate buffered saline (PBS) and incubated with 0.1% collagenase in a shaking water bath at 37°C for 1 hour. Next, 10 ml of M199 medium (Gibco, Gaithersburg, MD) with 10% FBS was added to inactivate the collagenase, and the cells were centrifuged at 1500 rpm for 10 min. After filtering with a 100-micron filter, the cell pellet obtained was suspended in 10 ml of M199 medium and centrifuged at 1200 rpm for 5 min. The supernatant was then aspirated and discarded, and the pellet was briefly resuspended in 1 ml sterile water to lyse the erythrocytes, followed by washing the pellet with 10 ml of M199 medium and centrifugation for a third time at 1200 rpm for 5 min. The cell pellet wasresuspended in 10 ml of M199 and filtered through a 40-micron filter. The stromal vascular fraction was plated at 1 x 106 cells per T75 flask, incubated overnight, and washed with PBS the following day to remove nonadherent cells. The retained ASCs were used between passages 1 and 5.
2.3. Drugs
Stock cisplatin, paclitaxel, 5-azacytidine (AZA), suberoylanilide hydroxamic acid (SAHA) were in powdered form stored at -20°C (Sigma-Aldrich, St. Louis, MO). Solutions of paclitaxel, AZA, and SAHA were prepared with 100% dimethyl sulfoxide (DMSO, Fisher Scientific, Hampton, NH); aliquots were stored in -20°C. Cisplatin solution was prepared using sterile normal saline and aliquots were stored in -4°C. Before each experiment, working solution for all drugs were prepared by diluting with fresh serum-free medium.
2.4. Treatment protocol
Ovarian cancer cells (Caov-3, SKOV-3, OVCAR-3) were plated at 5 × 105 cells/well and normal cells (HIEC, ASCs, and PF) at 5 × 104 cells/well in 96-well plates, incubated at 37°C for 24 h. The treatment protocol is depicted in Figure 1A.
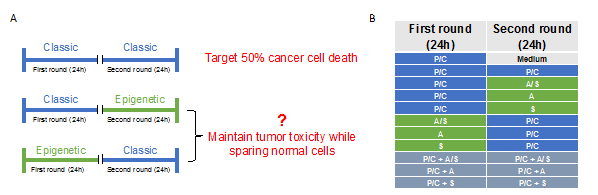
Figure 1: Schematic for each treatment strategy. (A) Sequence of treatment strategies. All drugs were administered at the same time point on each treatment day. (B) First and second rounds of treatment with P/C=paclitaxel + cisplatin, A/S=AZA + SAHA, A=AZA, S=SAHA, or combination of drugs as listed.
The medium was removed 24 h after plating, and each cell line was then treated with DMSO for control or one of the drugs listed in the first column in Figure 1B. The first round of drugs consisted of either standard chemotherapy alone (paclitaxel/cisplatin=P/C), epigenetic therapy alone (5-azacytidine=A, suberoylanilide hydroxamic acid=S, or 5-azacytidine and suberoylanilide hydroxamic acid=A/S), or a combination of standard and epigenetic therapy (P/C + A, P/C + S, P/C + A/S). Drugs were removed after the first round of 24 h treatment and cells were given a 72 h drug free period. The second round of drugs, consisting of the same treatment regimens (listed in Figure 1B), was then administrated at the same time for another 24 h. Drugs were then removed and replaced with fresh medium for 48-72 h. Analysis was conducted on triplicate samples, and each experiment was performed three times.
The drug concentrations used were 48 h IC50 values obtained from previous literature [13, 22-27]. The following are the 48h IC50 concentrations for Caov-3: 7.5 μM cisplatin, 5.4 nM paclitaxel, 11 μM AZA, 44 μM SAHA; SKOV-3: 51.7 μM cisplatin, 4.45 nM paclitaxel, 30 μM AZA, 9 μM SAHA; OVCAR-3: 152 μM cisplatin, 5.05 nM paclitaxel, 15 μM AZA, 2.1 μM SAHA. The highest IC50 of each drug was then used to treat the normal cells (HIEC, ASC, and PF): 152 μM cisplatin, 5.4nM paclitaxel, 30 μM AZA, 44 μM SAHA.
2.5. Cell proliferation assay
To count the cells, they were trypsinized in triplicates after completion of the aforementioned treatment protocol using the automated cell counter, NucleoCounter NC-200 (Chemometec, Bohemia, NY).
2.6. Cytotoxic assay
A 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) cell proliferation assay kit was utilized according to manufacturer’s experimental protocol (Thermo Fisher Scientific, Waltham, MA). The 96-well plates were placed in the SpectraMax M3 (Molecular Devices, San Jose, CA) to obtain optimal density readings at 570 nm.
Preparation of conditioned medium from ovarian cancer cell lines.
The ovarian cancer cells were grown to approximately 90% confluence in 100 mm cell culture dishes and or T-75 flasks. The cells were treated with the aforementioned treatment protocol, and after 24 h of the second round of drugs, cells were placed in serum-free medium and incubated at 37°C in a 5% CO2 atmosphere for 48-72 h. The conditioned medium was then collected and centrifuged for 10 min at 1700 × g to remove particulate matter. The supernatant was filtered through 0.22 μm filter and stored at -20ºC until tested.
2.7. Enzyme-linked immunosorbent assay (ELISA)
Interleukin-6 (IL-6) and vascular endothelial growth factor (VEGF) were quantified with their corresponding capture antibodies using sandwich enzyme-linked immunosorbent assay (ELISA, per manufacturer’s instructions R&D Systems, Minneapolis, MI). In brief, ELISA plates were coated with antibodies for IL-6 or VEGF and incubated with conditioned medium collected from each treated ovarian cancer cell line. Levels of bound IL-6 and VEGF were detected with a secondary antibody and developed with streptavidin-HRP (R&D systems). Standard curves generated with concentrations of IL-6 and VEGF in conditioned media of each ovarian cancer cell line were then used to convert optical density units to cytokine or growth factor concentrations.
2.8. Statistical analysis
Group comparisons were performed using independent T-test, multiple t test, chi square test, and one-way ANOVA. A p value of<0.05 was considered significant. Data analysis was performed using GraphPad Prism8 and SDS software.
3. Results
3.1. Effect of treatment regimens on normal cell and ovarian cancer cell survival
We began with combination therapies on all cell types to demonstrate the results reported in the clinical translation studies by Mendivil et al. and Matulonis et al. [17, 18]. As seen in Table 1, the treatment combination of P/C + A/S targeted three different cell lines of ovarian cancer (82-99% cell death), but also caused a high percentage of normal cell death (66-100%).
|
Normal Cells |
Ovarian cancer cells |
|||||
|
HIEC-6 |
ASC |
PF |
Caov-3 |
SKOV-3 |
OVCAR-3 |
|
|
% cell death after P/C + A/S |
100 |
65.9 |
93.1 |
85.3 |
99.1 |
81.7 |
|
p value |
0.0057 |
0.0101 |
0.0450 |
0.0014 |
0.006 |
0.0001 |
Table 1: Percent cell death after combination therapy with P/C +A/S (relative to control). Statistical significance was determined by multiple t test, n=3.
This high level of human intestinal epithelial cell (HIEC-6) death after treatment with standard and epigenetic therapy could explain the severe gastrointestinal toxicities observed in the study by Mendivil et al. As evident in Figure 2, simultaneous administration of standard and epigenetic therapies (P/C+A, P/C+S, P/C+A/S) dramatically decreases the viability of HIEC-6.

Figure 2: Brightfield images of human intestinal epithelial cells (HIEC-6) after treatment at 10 × magnification. Simultaneous administration of standard chemotherapy with epigenetic drugs has high level of toxicity to HIEC-6 while sequential treatments demonstrate higher cell viability.
In following experiments, standard and epigenetic drugs were then administered sequentially in attempt to decrease toxicity to normal cells. Surprisingly, our control—two rounds of standard chemotherapy (P/C)-was toxic to ASC and PF, but had more moderate effects on intestinal epithelial cells (Figure 3).
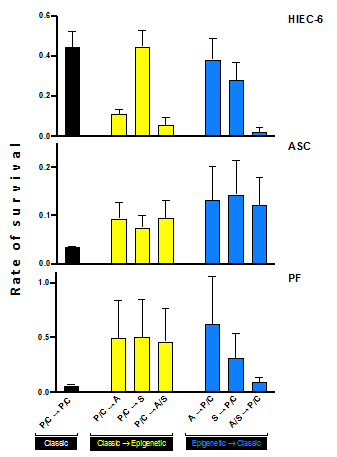
Figure 3: Sequential treatments in normals cells: human intestinal epithelial cells (HIEC-6), adipocyte-dervied stem cells (ASC), and primary fibroblasts (PF). Classic followed by epigenetic therapy had less toxic effects on ASC and PF. Specifically, P/C à S was the least toxic to HIEC-6. Epigenetic therapy (A/S à P/C) followed by standard chemotherapy was highly toxic to HIEC-6 and PF.
As shown in Figure 3, P/C followed by epigenetic treatment had less of an effect on ASC and PF (25-96% viability), while P/C à S was not toxic to HIEC (100% viability, p=0.0356) as compared to P/C à A (6% viability, p<0.0001) and P/C à A/S (1% viability, p<0.0001). Epigenetic followed by standard treatment (A/S à P/C) had high toxicities, 87% cell death of PF and 74% cell death of HIEC-6. Unexpectedly, P/C à A was significantly toxic to HIEC-6 with minimal negative effect to ASC and PF (Figures 2 and 3). Therefore, we concluded that standard followed by epigenetic therapy offered the least amount of toxicity to normal cells.
Next, the efficacy of the sequential treatment regimens less toxic to normal cells (P/C à A, P/C à S, P/C à A/S) were tested on three different ovarian serous adenocarcinoma cell lines. Caov-3 is platinum-sensitive, SKOV-3 is moderate-well differentiated and platinum-resistant, and OVCAR-3 is poorly differentiated and platinum resistant [28]. One round of standard chemotherapy followed by one round of epigenetic therapy was compared to the current standard treatment, which is two rounds of paclitaxel/cisplatin (P/C à P/C). One round of P/C followed by a round of epigenetic therapy was shown to be as effective or even more efficient in killing ovarian cancer cells as two rounds of standard chemotherapy (Figure 4).
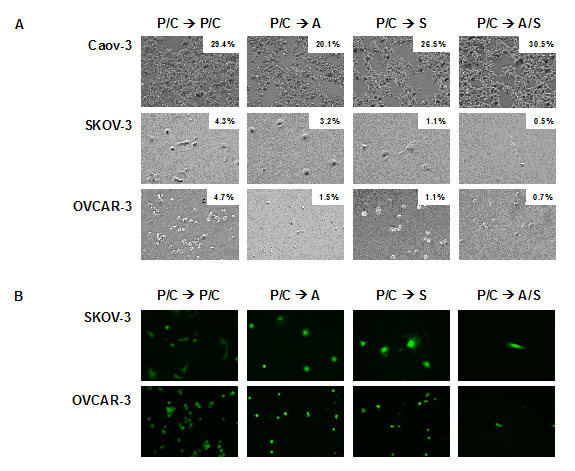
Figure 4: Response of ovarian cancer cells after treatment with standard chemotherapy followed by epigenetic therapy. (A) Percent cell survival shown in upper right hand corner of each image. (B) Representative images of LIVE/DEAD cell viability assay. Images at 10 x magnification. n=3.
Figure 5A shows in all three ovarian cancer cell lines, each standard followed by epigenetic therapy regimen had lower percentages of cell survival compared to P/C à P/C.
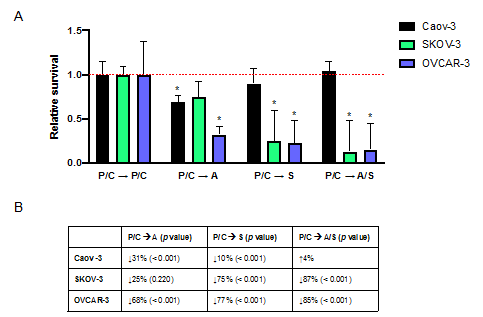
Figure 5: Ovarian cancer cell viability is decreased with standard chemotherapy followed by epigenetic therapy more efficienty than after two rounds of standard chemotherapy. (A) The relative survival of ovarian cancer cells after sequential treatments with epigenetic therapy was compared to standard chemotherapy (P/C à P/C). The asterisks indicate which treatment regimens had significantly lower cell survival than P/C à P/C. (B) Comparison of relative survival of ovarian cancer cells after sequential treatments with epignetic therapy as compared to two rounds of standard chemotherapy. Percentages show decrease or increase in relative cell survival after P/C à A, P/C à S, and P/C à A/S as compared to P/C à P/C. p<0.05 denotes statistical significance determined by chi square tests, n=3.
Caov-3 cells had 31% less survival after P/C à A than after P/C à P/C (p<0.001; Figure 5B). SKOV-3 cells had 75% less survival after P/C à S (p<0.001) and 87% less after P/C à A/S (p=0.004) when compared to P/C à P/C. All three regimens with epigenetic drugs (P/C à A, P/C à S, P/C à A/S) resulted in significantly lower percentages of cell viability after treatment than P/C à P/C (68%, p<0.001; 77%, p<0.001; 85%, p<0.001; Figure 5B). The only exception was for the treatment of Caov-3 cells with P/C à A/S. The addition of AZA or SAHA improved the cytotoxic activity on ovarian cancer cells, however P/C à S was the only regimen that greatly spared HIEC-6 as previously shown in Figure 3. Thus, P/Cà S demonstrates the least amount of toxicity to normal cells while maintaining efficacy of killing ovarian cancer cells as compared to two rounds of standard chemotherapy (P/C à P/C).
3.2. Effect of treatment regimens on secretomes of ovarian cancer cells
Cancer cells are known to secrete various growth factors and cytokines to support tumor growth and metastasis [29, 30]. Two of the major factors involved in tumor propagation are IL-6 and VEGF. We tested for secretion of these factors by ovarian cancer cells after administration of sequential treatments that were less toxic to normal cells via ELISA. The quantitative ELISA results are represented in Figure 6.
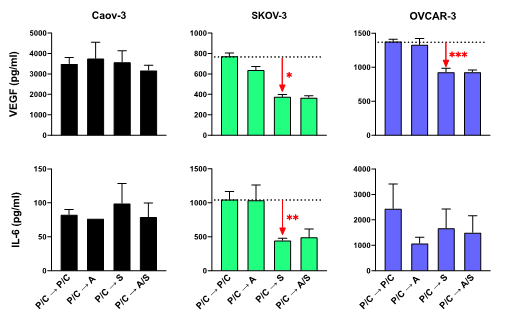
Figure 6: ELISA analysis of VEGF and IL-6 levels in each ovarian cancer cell line after treatment with standard chemotherapy followed by epigenetic therapy. The red arrows and asterisks denote where levels of VEGF and IL-6 were significantly downregulated after treatment with P/C à S. All results were normalized to equal amounts of total protein between samples. Independent T test, n=3.
For Caov-3 cells, levels of VEGF or IL-6 were not significantly different after treatment with two rounds of P/C or one round of P/C followed by epigenetic therapy. VEGF levels were significantly different when SKOV-3 cells were treated with one round of P/C followed by epigenetic therapy as compared to two rounds of P/C (P/C à A, p=0.012; P/C à S, p<0.001; P/C à A/S, p<0.001). VEGF was downregulated 20% after treatment with P/C à A versus P/C à P/C, and more than 50% after treatment with P/C à S. Similarly, IL-6 levels in SKOV-3 cells were not significantly different after two rounds of P/C as compared to one round of P/C followed by AZA (p=0.785), but P/C à S significantly decreased levels of IL-6 (p=0.006) as did treatment with P/C à A/S (p=0.002) when compared to P/C à P/C. In addition, AZA and SAHA were tested simultaneously after one round of P/C, and no benefit was observed in the reduction of VEGF (p=0.747) or IL-6 (p=0.863). Therefore, there is no benefit of using combined epignetic treatment (A/S) following P/C in the downregulation of VEGF or IL-6 in SKOV-3 cells.
For OVCAR-3 cells, there was no difference in VEGF levels after treatment with P/C à A when compared to P/C à P/C, however VEGF levels were significantly downregulated with treatments of P/C à S and P/C à A/S (p=0.008 and 0.004). However, similarly to SKOV-3 cells, there was no difference in VEGF levels between standard chemotherapy followed by SAHA or A/S (p=0.994). Treatment of OVCAR-3 cells with one round of P/C followed by epigenetic therapy resulted in lower levels of IL-6 than after treatment with two rounds of P/C. There was no statistically significant difference amongst the treatments in the levels of IL-6 (P/C à A, p=0.066; P/C à S, p=0.281; P/C à A/S, p=0.192).
Overall, the major epigenetic component that had the highest level of influence on the tumorigenic factors in ovarian cancer cells is SAHA. In summation from the ELISA results, there is no synergistic effect of AZA with SAHA in regulation of tumorigenic factors in ovarian cancer cell lines.
4. Discussion
In the last decade epigenetic drugs have been explored in different types of cancer in search of more effective alternate therapies. The major downfall observed from the clinical translational studies on simultaneous use of epigenetic therapy with classic chemotherapy (P/C) was the evidence of severe gastrointestinal and hematologic toxicities [17, 18]. This in vitro study is consistent with findings from the in vivo study by Mendivil et al. and partially explains the gastrointestinal toxicity seen in patients because simultaneous adminstration of standard chemotherapy with epigenetic therapy resulted in 100% cell death of human intestinal epithelial cells (HIEC-6, Table 1). Similarly, combination regimens were also toxic to human derived adipose stem cells and primary fibroblasts when administered simultaneously (Table 1 and Figure 3). The combined use of standard and epigenetic treatments are cytotoxic to ovarian cancer cells but also significantly harmful to normal tissues as seen in this current study (Table 1, Figures 2 and 3), and therefore should be minimized.
To explore our hypothesis that the sequential use of classic and epigenetic therapy can decrease toxicity to normal cells while maintaining cytotoxicity to ovarian cancer cells, we designed our experiment to mimic clinical administration of chemotherapy. Treatment typically occurs once every several weeks for patients, hence our drug free periods between each round of treatment (experimental design presented in Figure 1). We used three different commercially available ovarian cancer cell lines, Caov-3 (platinum-sensitive), SKOV-3 (moderate-well differentiated and platinum-resistant), and OVCAR-3 (poorly differentiated and platinum resistant), which exist in patients and are clinically relevant. We found that when epigenetic therapy is administered after P/C, cell viability is preserved in HIEC-6, contrasting with high toxicity observed from simultaneous administration (Figure 2). Similarly, the sequential treatments did not have adverse effects on cell populations critical for recovery and wound healing after surgery. ASC and PF were not significantly affected by sequential treatments of P/C followed by epigenetic drugs, as compared with higher toxicities after two rounds of P/C (Figure 3). Since aberrant epigenetic modifications are common in ovarian cancer [31], we tested next whether regimens of P/C followed by epigenetic drugs that were not toxic to normal cells can efficiently target ovarian cancer cells as compared to two rounds of P/C that nonspecifically targets all rapidly dividing cells. Results from our study showed in contrast to normal cells, the administration of epigenetic therapy after P/C not only effectively killed all three lines of ovarian cancer cells, but had more potent therapeutic effects against ovarian cancer cells as compared to two rounds of P/C (Figure 5). In esssence, the use of epigenetic drugs allows for a tumor directed effect while sparing normal cells likely because of its aberrant epigenetic modifications on cancer cells [31].
The use of epigenetic therapy after one round of P/C permits decreased exposure to standard chemotherapy. This treatment also is minimally toxic to normal cells while highly potent against cancer cell growth. This potential new strategy in the treatment of ovarian cancer can help improve survival of patients and minimize toxicities faced during treatment because epigenetic therapy maintains tumor efficacy similar to standard chemotherapy with less toxicity to normal cells.
Cancer cells survive not only through cell proliferation, but impact the miroenvironment by secreting specific proteins responsible for cross talk among cells for recruitment, propagation, and metastasis of the tumor [32]. VEGF is a key mediator of angiogenesis secreted by cancer cells, which impacts tumor development and growth [33]. This study strongly shows that sequential treatment of P/C followed by epigenetic therapy not only decreases certain ovarian cancer cell viability as or more efficiently as classic chemotherapy, but also inhibits changes to the cancer microenvironment by downregulating VEGF. Most interestingly, SKOV-3 and OVCAR-3 (which are two platinum resistant ovarian cancer cell lines) had greater downregulation of VEGF after P/C à S and P/C à A/S treatments as compared to the minor effects in Caov-3 (platinum sensitive; Figure 6). VEGF is important in promoting angiogenesis and inducing immunosuppressive micronevironments in tumors, thus is a noteworthy finding that it can be downregulated by SAHA in two of three investigated ovarian cancer cell lines.
Another factor which is important for cancer propagation is IL-6, which was shown to be secreted by all three ovarian cancer cell lines [30]. Watson et al. demonstrated Caov-3 and SKOV-3 cell lines produce moderate to high levels of IL-6 as compared to OVCAR-3 which produces low amounts of IL-6 [29]. Our study shows that this factor is significantly downregulated in the two platinum-resistant cell lines (SKOV-3, OVCAR-3) after exposure to epigenetic drugs, especially SAHA (Figure 6). In consideration of the ELISA results, the use of epigenetic therapy is beneficial in treatment of ovarian cancer as compared to P/C alone because of the modifications to secretion of growth factors and cytokines critical for cancer propagation and metastasis. To our knowledge, this is the first systematic study that investigates the influence of epigenetic agents SAHA and AZA at the level of critical tumorigenic factors, VEGF and IL-6.
This study reaffirmed the cytotoxic effects of simultaneous administration of classic with epigenetic therapy and demonstrated opportunities to spare normal cells through sequential treatments of standard chemotherapy with epigenetic drugs. In fact, clinical translational studies examining SAHA or AZA alone had minimal side effects on normal cells but also minimal cytotoxic effects on ovarian cancer cells [20, 29]. In our study, we were able to replace one round of P/C with an epigenetic drug which resulted in the same or more efficient killing of ovarian cancer cells with less toxicity to normal cells than P/C. The efficacy of each sequence of P/C followed by epigenetic therapy was also found to have a beneficial effect on proliferation of ovarian cancer cell lines but had a different effect on the secretomes of the same cell lines.
The ability to modify standard chemotherapy regimens could impact a patient’s treatment course significantly by shortening duration of treatments, decreasing cytotoxic dosages, and minimizing negative side effects. Limitations of this study include the in vitro model and limited secretrome analysis; the level of epigenetic alterations and its downstream effects were not explored. Further research in our lab will pursue these mechanisms. Additional research to determine treatment effects on other cell proteins such as the immune checkpoint protein, PDL-1, could provide new targeted approaches for the use of epigenetic therapy with other treatment modalities in ovarian cancer with far less toxicity than current regimens. There is a growing number of studies demonstrating that epigenetic exposure can augment the potency of the immune system [34, 35]. Currently, a phase II clinical trial on oral azacitidine and pembrolizumab in platinum-resistant ovarian cancer patients is underway [36].
5. Conclusions
In conclusion, in vitro results show sequential treatments of standard chemotherapy (P/C) with epigenetic drugs, specifically suberoylanilide hydroxamic acid (SAHA), could be a beneficial treatment option for ovarian cancer by decreasing cytotoxicity to normal tissue cells while maintaining tumor toxicity against ovarian cancer cells. Further studies in an in vivo animal model could substantiate the use of these sequential treatments.
Author Contributions
Leona Chang—conceptualization, data curation, formal analysis, investigation, methodology, visualization, writing original draft, review & editing. Krystal Hunter—data curation, formal analysis. James Aikins—review & editing. Spencer Brown—project administration, resources. Olga Ostrovsky—conceptualization, data curation, formal analysis, investigation, methodology, project administration, resources, supervision, visualization, writing original draft, review & editing.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Creasman WT, DiSaia PJ, Mannell R, et al. Clinical gynecologic oncology. 9th Ed. Philadelphia: Elsevier (2018): 253-289.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, CA: a cancer journal for clinicians 68 (2018): 7-30.
- Wright AA, Bohlke K, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology Clinical Practice Guideline. Gynecologic oncology 143 (2016): 3-15.
- Payne WG, Naidu DK, Wheeler CK, et al. Wound healing in patients with cancer. Eplasty 8 (2008): e9.
- Ledermann JA, Raja FA, Fotopoulou C, et al. Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology 6 (2013): vi24-32.
- Zamarin D. Novel therapeutics: response and resistance in ovarian cancer. Int J Gynecol Cancer 29 (2019): s16-s21.
- Kanwal R, Gupta S. Epigenetic modifications in cancer. Clinical genetics 81 (2012): 303-311.
- Xie S, Wang Z, Okano M, et al. Cloning, expression and chromosome locations of the human DNMT3 gene family. Gene 236 (1999): 87-95.
- Weichert W, Denkert C, Noske A, et al. Expression of class I histone deacetylases indicates poor prognosis in endometrioid subtypes of ovarian and endometrial carcinomas. Neoplasia (New York, NY) 10 (2008): 1021-1027.
- Chang X, Monitto CL, Demokan S, et al. Identification of hypermethylated genes associated with cisplatin resistance in human cancers. Cancer research 70 (2010): 2870-2879.
- Su HY, Lai HC, Lin YW, et al. Epigenetic silencing of SFRP5 is related to malignant phenotype and chemoresistance of ovarian cancer through Wnt signaling pathway. International journal of cancer 127 (2010): 555-567.
- Dietrich CS, Greenberg VL, DeSimone CP, et al. Suberoylanilide hydroxamic acid (SAHA) potentiates paclitaxel-induced apoptosis in ovarian cancer cell lines. Gynecologic oncology 116 (2010): 126-130.
- Ong PS, Wang XQ, Lin HS, et al. Synergistic effects of suberoylanilide hydroxamic acid combined with cisplatin causing cell cycle arrest independent apoptosis in platinum-resistant ovarian cancer cells. International journal of oncology 40 (2012): 1705-1713.
- Appleton K, Mackay HJ, Judson I, et al. Phase I and pharmacodynamic trial of the DNA methyltransferase inhibitor decitabine and carboplatin in solid tumors. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 25 (2007): 4603-4609.
- Sookram J, Zheng A, Linden KM, et al. Epigenetic therapy can inhibit growth of ovarian cancer cells and reverse chemoresistant properties acquired from metastatic omentum. International journal of gynaecology and obstetrics: the official organ of the International Federation of Gynaecology and Obstetrics 145 (2019): 225-232.
- Modesitt SC, Sill M, Hoffman JS, et al. A phase II study of vorinostat in the treatment of persistent or recurrent epithelial ovarian or primary peritoneal carcinoma: a Gynecologic Oncology Group study. Gynecologic oncology 109 (2008): 182-186.
- Mendivil AA, Micha JP, Brown JV, et al. Increased incidence of severe gastrointestinal events with first-line paclitaxel, carboplatin, and vorinostat chemotherapy for advanced-stage epithelial ovarian, primary peritoneal, and fallopian tube cancer. Int J Gynecol Cancer 23 (2013): 533-539.
- Matulonis U, Berlin S, Lee H, et al. Phase I study of combination of vorinostat, carboplatin, and gemcitabine in women with recurrent, platinum-sensitive epithelial ovarian, fallopian tube, or peritoneal cancer. Cancer Chemother Pharmacol 76 (2015): 417-423.
- Cooper AL, Greenberg VL, Lancaster PS, et al. In vitro and in vivo histone deacetylase inhibitor therapy with suberoylanilide hydroxamic acid (SAHA) and paclitaxel in ovarian cancer. Gynecologic oncology 104 (2007): 596-601.
- Modesitt SC, Parsons SJ. In vitro and in vivo histone deacetylase inhibitor therapy with vorinostat and paclitaxel in ovarian cancer models: does timing matter? Gynecologic oncology 119 (2010): 351-357.
- Meng F, Sun G, Zhong M, et al. Anticancer efficacy of cisplatin and trichostatin A or 5-aza-2'-deoxycytidine on ovarian cancer. British journal of cancer 108 (2013): 579-586.
- Sun LM, Liu YC, Li W, et al. Nivolumab effectively inhibit platinum-resistant ovarian cancer cells via induction of cell apoptosis and inhibition of ADAM17 expression. Eur Rev Med Pharmacol Sci 21 (2017): 1198-1205.
- Rahman MA, Ramli F, Karimian H, et al. Artonin E Induces Apoptosis via Mitochondrial Dysregulation in SKOV-3 Ovarian Cancer Cells. PloS one 11 (2016): e0151466.
- Cao D, Li D, Huang Y, et al. 5-Azacytidine promotes invadopodia formation and tumor metastasis through the upregulation of PI3K in ovarian cancer cells. Oncotarget 8 (2017): 60173-60187.
- Sukovas A, Cesna V, Jasukaitiene A, et al. Response of OVCAR-3 cells to cisplatin and hyperthermia: does hyperthermia really matter? Anticancer Res 37 (2017): 5011-5018.
- Chung WM, Ho YP, Chang WC, et al. Increase paclitaxel sensitivity to better suppress serous epithelial ovarian cancer via ablating androgen receptor/aryl hydrocarbon receptor-ABCG2 axis. Cancers 11 (2019): 1-22.
- Li Y, Hu W, Shen DY, et al. Azacitidine enhances sensitivity of platinum-resistant ovarian cancer cells to carboplatin through induction of apoptosis. American Journal of Obstetrics and Gynecology 200 (2009): 171.e1-177.e9.
- Beaufort CM, Helmijr JC, Piskorz AM, et al. Ovarian cancer cell line panel (OCCP): clinical importance of in vitro morphological subtypes. PloS one 9 (2014): e103988.
- Nervi C, De Marinis E, Codacci-Pisanelli G. Epigenetic treatment of solid tumours: a review of clinical trials. Clin Epigenetics 7 (2015): 127.
- Watson JM, Sensintaffar JL, Berek JS, et al. Constitutive production of interleukin 6 by ovarian cancer cell lines and by primary ovarian tumor cultures. Cancer Res 50 (1990): 6959-6965.
- Fardi M, Solali S, Hagh MF. Epigenetic mechanisms as a new approach in cancer treatment: An updated review. Genes & Diseases 5 (2018): 304-311.
- Makridakis M, Vlahou A. Secretome proteomics for discovery of cancer biomarkers. Journal of proteomics 73 (2012): 2291-230.
- Carmeliet P. VEGF as a key mediator of angiogenesis in cancer. Oncology 69 (2005): 4-10.
- Woods DM, Sodre AL, Villagra A, et al. HDAC inhibition upregulates PD-1 ligands in melanoma and augments immunotherapy with PD-1 blockade. Cancer Immunol Res 3(2015): 1375-1385.
- Hicks KC, Fantini M, Donahue RN, et al. Epigenetic priming of both tumor and NK cells augments antibody-dependent cellular cytotoxicity elicited by the anti-PD-L1 antibody avelumab against multiple carcinoma cell types. Oncoimmunology 7 (2018): e1466018.
- Glaspy JA. Study of pembrolizumab with or without CC-486 in patients with platinum-resistant ovarian cancer. Translational Research in Oncology. Identification No. NCT02900560.


 Impact Factor: * 4.1
Impact Factor: * 4.1 Acceptance Rate: 74.74%
Acceptance Rate: 74.74%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 