An Unusual Cause of Meckels Diverticulum Enterolithiasis Imitating a Pelvic Teratoma-What to do in Case of Unexpected Meckels Diverticulum?
Yusef Moulla1*, Romy Handzel1, Jens Einenkel1, Arne Dietrich2
1Centre for Women’s and Children’s Health, Department of Obstetrics and Gynecology, Leipzig University, Leipzig, Germany
2Clinic for Abdominal, Vascular, Thoracic and Transplant Surgery, Leipzig University, Leipzig, Germany
*Corresponding Author: Dr. Yusef Moulla, Clinic for Abdominal, Vascular, Thoracic and Transplantat Surgery, Leipzig University, Liebigstr 2004103 Leipzig, Germany
Received: 10 January 2019; Accepted: 21 January 2019; Published: 28 January 2019
Article Information
Citation: Yusef Moulla, Romy Handzel, Jens Einenkel, Boris Jansen-Winkeln, Ines Gockel, Arne Dietrich. An Unusual Cause of Meckel’s Diverticulum Enterolithiasis Imitating a Pelvic Teratoma-What to do in Case of Unexpected Meckel’s Diverticulum?. Journal of Surgery and Research 2 (2019): 10-14.
View / Download Pdf Share at FacebookAbstract
A 47-year-old woman presented with a history of 4 weeks of recurring pain in the right lower abdomen. Transvaginal ultrasound scan showed an irregular shaped solid mass of 45 × 26 × 45 mm in the right ovarian fossa with increased perfusion. CT-scan suspected an ovarian tumor with evidence of teeth in terms of a teratoma in the right adnexal area. Finally, our findings were suggestive of an ovarian teratoma and the decision for laparoscopy was made. Intraoperatively both ovaries were unsuspicious but a Meckel’s diverticulum with enterolithiasis was found in the right pelvis and removed. Meckel’s diverticulum is present in 1-2% of the population and here we will discuss general removel recommandations.
Keywords
Meckels diverticulum, Teratoma
Article Details
1. Introduction
Ovarian teratomas are encapsulated tumors developing from all three germ layers. They are of the mature or immature type, former can contain hair, teeth or other tissue e.g. lung or thyroid. Teratomas are mostly asymptomatic until they reach a certain size, causing symptoms such as pain due to a pressure effect, torsion or rupture [1]. The risk of malignant transformation is estimated to be between 0.17-2% and this most commonly occurs in postmenopausal women [2]. Mostly they are diagnosed via ultrasound or CT scans, presenting with signs of an atypical cyst and irregular tissue [2, 3]. If diagnostic imaging conceives suspicion of an ovarian teratoma, it should always be removed, depending on their size either laparoscopically or by laparotomy. The surgical treatment has a low operative morbidity [4-6]. In the gastrointestinal tract Meckel’s diverticulum is the most common prevalent congenital abnormality. It is present in 1 to 2% of the population [7]. It remains as the result of incomplete obliteration of the omphalomesenteric duct and is located usually about 45-90 cm proximal to the ileocecal valve. Variations on location, size and form are reported. Most of all Meckel’s diverticula remain livelong asymptomatic. The clinical manifestation can vary a lot, commonly it is diagnosed intraoperatively due to its mimic of appendicitis [8]. Here we report an unusual cause of Meckel’s diverticulum enterolithiasis imitating a pelvic teratoma and brings together gynecology, abdominal surgery and radiology.
2. Case Report
A 47-year-old woman presented with a history of 4 weeks of recurring abdominal complaints. Pain started in left upper abdomen/epigastrium and shifted to the right lower abdomen. The patient had reduced appetite, no nausea or vomiting, no diarrhoea, no fever. She also had a long history of mild constipation. On examination, she had a moderate tenderness on palpation of the right lower abdomen, no palpable mass, normal peristaltic sounds. The gynaecological examination found no abnormalities. Transvaginal ultrasound scan showed a tumor suspicious for malignancy: an irregular shaped solid mass of 45 × 26 × 45 mm in the right ovarian fossa with increased perfusion. CT-scan suspected a benign ovarian tumor with evidence of teeth in terms of a teratoma in the right pelvis, furthermore parailiacal and retroperitoneal lymphadenopathy (Figure 1). Liver, gallbladder, spleen and pancreas were inconspicuous. Serum tumor markers CA 125 and AFP were found within normal limits, CA 72-4 was elevated to 14.1 U/mL [normal <6.9]. Finally, our findings were suggestive of an ovarian teratoma and the decision for laparoscopy was made. Laparoscopy was performed with a 10 mm umbilical camera trokar, a 5 mm trokar in the right lower abdomen and a 15 mm trokar in the left lower abdomen. Intraoperatively both ovaries were unsuspicious but a Meckel’s diverticulum was found in the right pelvis instead (Figure 2). The decisicon for removel was made. The 15 mm trokar was removed and the Meckel´s diverticulum was externalised at the trokar site. With a linear cutter (blue load) the Divertivulum was divided at the base and the staplerline was oversewn with a running absorble suture.
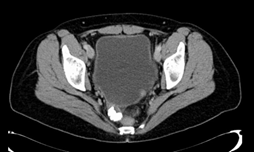
Figure 1: Pelvic CT-scan with a tumor at the right pelvic wall, suspicious for a teratoma.
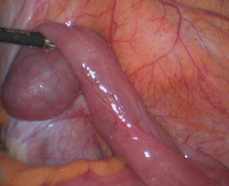
Figure 2: Introperative situs with a Meckel's diverticulum in the right lower abdomen.
Histological examination of the excised diverticel revealed a part of the intestinal wall with a sack-shaped/saccate eversion with a length of 4.5 cm and up to 3.3 cm in diameter. It has a pore-shaped transition to the intestinum of maximal 0.1 cm. Inside the eversion there were 17 concrements between 4 and 8 mm in diameter in terms of gallstones. Furthermore, an antrum type heterotopic mucosa was found. The following hospital stay was uneventful; the patient was discharged on day three postoperatively with primary wound healing.
3. Discussion
Meckel’s diverticulum is the most common prevalent congenital abnormality within the gastrointestinal tract. Variations on location, size and form are reported. Most of them remain livelong asymptomatic in affected individuals. However, in 4 to 6% they become symptomatic. The clinical manifestation can vary a lot, including gastrointestinal bleed, inflammation, perforation, tumor development and intestinal obstruction [9, 10]. Commonly it is diagnosed intraoperatively due to its mimic of appendicitis [8]. Gastrointestinal bleeding is the major clinical finding in children with a symptomatic Meckel's diverticulum. The bleeding incidence rate for this complication has been described as high as 50% [11]. Usually, ectopic tissue, mainly of gastric and pancreatic origin, is the bleeding reason. Gastric tissue was seen in 60-65% of all cases and can cause ileal ulcerations and bleedings due to acidic secretion (or alkaline secretions from pancreatic tissues) [12-16]. Meckel’s diverticulum related intestinal obstruction is the most common finding in adults and the second common in children [17]. Stone formation, (inflammatory) tumors or invagination (ileo-ileal intussusception) can result in intestinal obstruction [11, 18]. Also, Meckel’s diverticulum lithiasis, quite frequently seen, is likely to promote diverticulum inflammation [11]. Meckel’s diverticulitis, accounting for 20% of the complications is more seen in adults. Most of these patients are misdiagnosed as appendicitis preoperatively [19]. Tumors originated from Meckel’s diverticula are rarely to be found; benign tumors such as lipoma, neuromuscular tumors and hamartomas are mentioned as well as malignant tumors, the majority of the latter are carcinoids [8, 20]. Undoubtably, symptomatic Meckel’s diverticula or diverticula with any suspicious macroscopic findings have to be removed when diagnosed or found incidentally. However, what should be done with unsuspicious asymptomatic Meckel’s diverticula? Gynecologists, urologists and surgeons, whoever performs laparotomy or laparoscopy, will be faced with this question. On the one hand, the incidence of Meckel’s diverticulum in the general population has been estimated to be about 1-2 cases per 100 persons in the general population and there is a lifetime risk of 4 to 6% for Meckel’s diverticula to develop complications; on the other hand, following surgical removal of Meckel’s diverticulum there is a complication rate of approximately 1% [21-23]. Among surgeons the indication for the removel of asymptomatic diverticula it is still discussed with some controversy. Since there is a certain risk of an asymptomatic Meckel’s diverticulum becoming symptomatic the authors mostly recommend removel to avoid secondary complications [7, 24]. In summary, in any case of acute abdomen the entire abdomen should be explored, including a search for Meckel’s diverticulum. Whenever found and irrespective of symptoms, Meckel’s diverticulum should be removed to avoid later potential complications.
Conflict of Interest Statement
We declare that we have no conflict of interest.
Ethical Approval
For this type of study formal consent is not required.
Informed Consent
Does not apply
References
- Al-Harfoushi R, Abdulaziz el-H, Andrabi SI, et al. Ovarian teratoma presenting as small bowel obstruction in an elderly lady-A case report. Int J Surg Case Rep 2 (2011): 6-8.
- Hinshaw H, Smith A, Desouki M, et al. Malignant Transformation of a Mature Cystic Ovarian Teratoma into Thyroid Carcinoma, Mucinous Adenocarcinoma, and Strumal Carcinoid: A Case Report and Literature Review. Case Rep Obstet Gynecol (2012).
- Papakonstantinou E, Iavazzo C, Hasiakos D, et al. Extraovarian mature cystic teratoma of the mesentery. A case report and literature review. Clin Exp Obstet Gynecol 38 (2011): 291-293.
- Vicus D, Beiner ME, Clarke B, et al. Ovarian immature teratoma: treatment and outcome in a single institutional cohort. Gynecol Oncol 123 (2011): 50-53.
- Kim MJ, Kim NY, Lee DY, et al. Clinical characteristics of ovarian teratoma: Age-focused retrospective analysis of 580 cases. Am J Obstet Gynecol 205 (2006): 1-4.
- Szpakowski M, Nowinska A, Kufelnicka M, et al. Laparoscopy or laparotomy in the treatment of ovarian teratomas? Ginekol Pol 77 (2006): 502-509.
- Tauro LF, George C, Rao BS, et al. Asymptomatic Meckel’s diverticulum in adults: is diverticulectomy indicated? Saudi J Gastroenterol 16 (2010): 198-202.
- Uppal K, Tubbs RS, Matusz P, et al. Meckel’s diverticulum: A review. Clin Anat 24 (2011): 416-422.
- Tomikawa M, Taomoto J, Saku M, et al. A loop formation of Meckel’s diverticulum: A case with obstruction of the ileum. Ulus Travma Acil Cerrahi Derg 9 (2003): 134-136.
- Limas C, Seretis K, Soultanidis C, et al. Axial torsion and gangrene of a giant Meckel’s diverticulum. J Gastrointestin Liver Dis 15 (2006): 67-68.
- Sharma RK, Jain VK, Kamboj S, et al. Gangrenous Meckel's diverticulum causing acute intestinal obstruction in an adult. ANZ J Surg 78 (2008): 1046-1047.
- Giusti S, Iacconi C, Giusti P, et al. Ileal invaginated Meckel’s diverticulum: Imaging diagnosis. Eur Radiol 14 (2004): 2368-2371.
- Haubrich WS, Schaffner F, Berk JE. Gastroenterology. Philadelphia: Saunders (1995): 12-14.
- Koh HC, Page B, Black C, et al. Ectopic pancreatic-type malignancy presenting in a Meckel’s diverticulum: A case report and review of the literature. World J Surg Oncol 22 (2009).
- Dolezal J, Vizda J. Experiences with detection of the ectopic gastric mucosa by means of Tc-99m pertechnetate disodium scintigraphy in children with lower gastrointestinal bleeding. Eur J Pediatr Surg 18 (2008): 258-260.
- Mittal BR, Kashyap R, Bhattacharya A, et al. Meckel’s diverticulum in infants and children; technetium-99m pertechnetate scintigraphy and clinical findings. Hell J Nucl Med 11 (2008): 26-29.
- Garrigos G, Gomez SA, Peris N, et al. Intestinal obstruction due to Meckel’s diverticulum enterolithiasis. Cir Esp 90 (2011): 674-676.
- Kopacova M, Vykouril L, Vacek Z, et al. Inverted Meckel’s Diverticulum with Ectopic Pancreatic Tissue as a Source of Severe Gastrointestinal Bleeding. J Gastrointest Surg 14 (2010): 578-581.
- Matsagas MI. Incidence, complications, and management of Meckel’s diverticulum. Arch Surg 130 (2005): 143-146.
- Thirunavukarasu P, Sathaiah M, Sukumar S, et al. Meckel’s diverticulum-a high-risk region for malignancy in the ileum. Insights from a population-based epidemiological study and implications in surgical management. Ann Surg 253 (2011): 223-230.
- Michas CA, Cohen SE, Wolfman EF. Meckel’s diverticulum: Should it be excised incidentally at operation? Am J Surg 129 (1975): 682-685.
- Ciardo LF, Agresta F, Bedin N. Meckel’s diverticulum: A neglected (or deliberately ignored) entity. Chir Ital 56 (2004): 689-692.
- Malik AA, Shams-ul-Bari KA, Khaja AR. Meckel’s diverticulum-revisited. Saudi J Gastroenterol 16 (2010): 3-7.
- Cullen JJ, Keith K, Moir C, et al. Surgical management of Meckel’s diverticulum. Ann Surg 220 (1994): 564-569.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 