Total Endoscopic Surgical Treatment of a Gigantic Coronary Arteriovenous Fistula between Circumflex Artery and Superior Vena Cava through Right Anterior Mini-Thoracotomy
Saad Salamate*, Ali Bayram, Ali Hojeij, Miriam Silaschi, Ömür Akhavuz, Ali El-Sayed Ahmad, Farhad Bakhtiary
Department of Cardiac Surgery, University Hospital Bonn, Sigmund-Freud-Str. 25, 53127 Bonn Germany
*Corresponding Author: Saad Salamate, Department of Cardiac Surgery, University Hospital Bonn, Sigmund-Freud-Str. 25, 53127 Bonn Germany
Received: 26 September 2023; Accepted: 05 October 2023; Published: 22 December 2023
Article Information
Citation: Saad Salamate, Ali Bayram, Ali Hojeij, Miriam Silaschi, Ömür Akhavuz, Ali El-Sayed Ahmad, Farhad Bakhtiary. Total Endoscopic Surgical Treatment of a Gigantic Coronary Arteriovenous Fistula between Circumflex Artery and Superior Vena Cava through Right Anterior Mini-Thoracotomy. Journal of Surgery and Research. 6 (2023): 448-452.
View / Download Pdf Share at FacebookAbstract
Coronary arteriovenous fistula (CAF) is a rare congenital anomaly that occurs when there is an abnormal connection between the coronary artery and cardiac chamber or a great vessel. We report an unusual case of a gigantic CAF between the circumflex branch of the left coronary artery (LCx) and the superior vena cava (SVC) in a 54-year-old patient treated by totally endoscopic surgical closure through right anterior mini-thoracotomy (RAMT). The excellent clinical and cosmetic results confirm the safety and effectiveness of this technique even in certain rare congenital cardiovascular anomalies.
Keywords
<p>Coronary arteriovenous fistula, Minimally invasive cardiac surgery, Right anterior mini thoracotomy, Congenital cardiovascular anomalies</p>
Article Details
Abbreviations
|
AF |
atrial fibrillation |
|
CAF |
coronary arteriovenous fistula |
|
CPB |
cardiopulmonary bypass |
|
CTA |
computer tomography angiogram |
|
ICS |
intercostal space |
|
LCx |
left circumflex coronary artery |
|
RAMT |
right anterior mini-thoracotomy |
|
SVC |
superior vena cava |
|
TEE |
transesophageal echocardiography |
Introduction
First described by Krause in 1865, coronary arteriovenous fistulas (CAF) represent a coronary abnormality where one or more coronary artery communicates with a cardiac chamber, coronary sinus, or a major adjacent vessel. These fistulas are most commonly congenital in nature, but can be acquired as well [1,2]. These anomalies are usually asymptomatic and discovered incidentally, but can however be associated with major cardiac manifestations and complications through different and multiple pathophysiological processes, including a left-to-right shunt, coronary “steal” phenomenon, and anatomical consequences associated with large fistulas and aneurysmal changes such as compression of nearby structures and/or ruptures [2,3]. Due to the apparent rarity of CAF and the subsequent scarcity of clinical data in the current literature, there is still debate regarding the treatment and management of this condition, where the current consensus is to intervene in cases of large, complicated or symptomatic CAF, either via endovascular or surgical closure. [6]. We report a case of a massive and aneurysmal CAF between the circumflex branch of the left coronary artery (LCx) and the superior Vena cava (SVC) that was surgically treated via a totally endoscopic approach through right anterior mini-thoracotomy (RAMT) in “on-pump beating-heart” approach.
Case Description
A 54-year-old man diagnosed with a massive CAF between the circumflex branch of the left coronary artery and the superior vena cava was referred at our institution for surgical treatment. The anomaly was discovered incidentally following extensive investigations of atrial fibrillation (AF), until percutaneous coronary angiography showed the gigantic aneurysmal CAF (Figure 1). Preoperative computed tomography angiography (CTA) confirmed the findings and demonstrated the course of the CAF running behind the aorta forming an aneurysm with a maximum diameter of 25 mm just before its confluence with the SVC. An attempt to close the tract percutaneously by coiling had previously failed due to the diameter and tortuosity of the CAF.
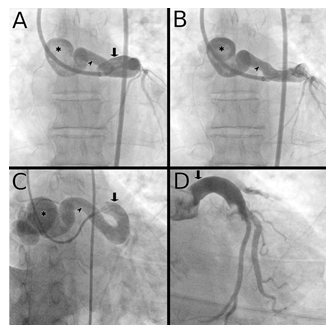
Figure 1: Pre-operative and post-operative left coronary angiography stills
Panels A and B show a pre-operative antero-postal view of the left coronary angiography with early left main coronary contrast filling in panel A, and late aneurysmal contrast filling in panel B. Panel C shows a pre-operative RAO view of the left coronary tree. Panel D is a still from the post-operative coronary angiography showing the left coronary tree with the absence of the coronary arteriovenous fistula and its aneurysmal formation. Legend: Asterisks denote the large aneurysm. Arrow heads show the fistula tract. Downward arrows point at the dilated left main coronary artery.
Surgical technique
The surgical technique is documented in a step-by-step manner in video 1. The patient was placed in a supine position on the operating table. After induction of general anesthesia and intubation, the right femoral vessels were cannulated percutaneously under transesophageal echocardiographic (TEE) control and cardiopulmonary bypass (CPB) was started.
Right anterior mini-thoracotomy (RAMT) was then performed via a 3 cm skin incision at the second right intercostal space (ICS) (Figure 2). Intercostal tissue was divided and a soft tissue retractor (Valve Gate™ Soft Tissue Protector, Geister, Germany) was placed followed by the introduction of the 3D camera port (Aesculap Einstein Vision, Tuttlingen, Germany) into the chest cavity via a separate stab incision above the main thoracotomy at the level of the first ICS.
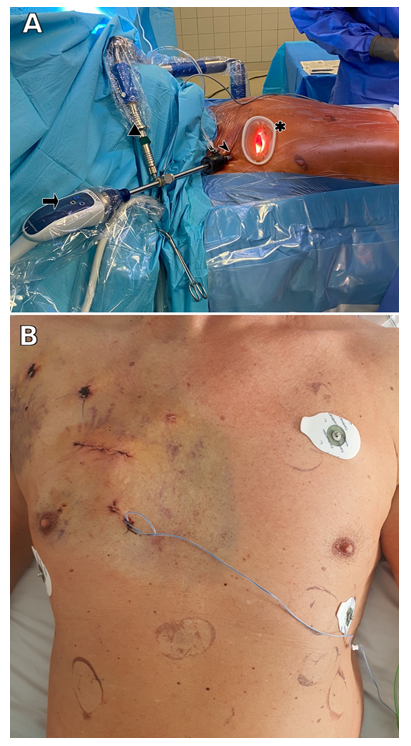
Figure 2: Endoscopic minimally invasive operative access through right anterior mini-thoracotomy.
Panel A shows the patient laying supine on the operating table. Right anterior mini-thoracotomy is performed at the level of the second intercostal space through which is installed the soft tissue retractor (Valve Gate™ Soft Tissue Protector, Geister, Germany) (Asterisk). 3D Camera port (Aesculap Einstein Vision, Tuttlingen, Germany) (Right facing arrow) in inserted via a stab incision at the level of the first intercostal space (Arrow head) and is held in place with the support arm (triangle). Panel B depicts the excellent cosmetic results of RAMT at the 4th post-operative day.
After CO2 in situ infusion through the camera port and good visualisation of the heart, pericardial fat was dissected until 2 cm above the phrenicus nerve before opening the pericardium transversely. Using a Prolene 4/0 stay suture, the aorta was then pulled anteriorly in order to obtain a better exposure of the aneurysmal CAF which started from the proximal LCx, running between the ascending aorta and the pulmonary artery, and finally drained into the SVC (Figure 3).
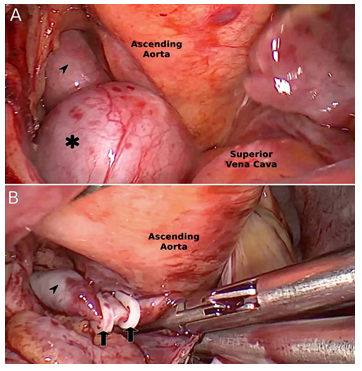
Figure 3: Intra-operative endoscopic view of the coronary arteriovenous fistula.
Starting with the distal end of the CAF at the junction between the aneurysmal formation and the SVC, clipping of the CAF was achieved using WeckÒ Hem-o-lokÒ XL Polymer Ligating clips (TeleflexÒ, Morrisville, NC 27560) (Figure 3), followed by ligation with 3/0 Prolene sutures. Several clips were then placed proximally near the ingress of the fistula behind the ascending aorta before performing the transection of the tract at the level of the aneurysm. Both open ends of the CAF were then sutured shut using 3/0 Prolene.
After control for any remaining leak or bleeding, the patient was weaned from CPB after the placement of a right ventricular pacing wire. Following the reduction of the extracorporeal circulation and the verification of intra-pericardial hemostasis, the camera port was removed and a pericardial as well as a right pleural drains were inserted.
Femoral decannulation was then performed. Venous hemostasis was achieved by slowly removing the cannula followed by manual compression for 10 minutes. The MANTA® vascular closure device (Essential Medical Inc., Malvern, Pennsylvania, PA, USA) was used for femoral artery closure. Steps for the percutaneous femoral decannulations and the use the MANTA® system were previously published in detail [7].
After hemostatic control, the ribs were secured with two FiberWire (Arthrex; Naples, FL, USA). The thoracotomy was closed in layers and the remaining auxiliary incisions were closed with single-button sutures. Finally, a compression bandage was applied to the right groin.
The patient was then extubated on the operating table. Our patient had an uneventful recovery after the surgery. A post-operative coronary angiography confirmed the closure of the fistula and an excellent coronary artery perfusion (Figure 1). Our patient was discharged home on the 6th postoperative day with satisfactory early results and favorable cosmesis (Figure 2).
Discussion
CAF, and especially when associated with a large aneurysm, is a rare anomaly that is usually diagnosed incidentally. In all patients undergoing selective coronary artery angiography, the incidence of CAF is estimated to be between 0.1% and 0.2% with the majority of fistulas being asymptomatic with no hemodynamic or ischemic impact [8,9]. Through various mechanisms, CAF can however have significant hemodynamic impacts such as left-to-right shunting and the diversion of blood flow away from the coronary circulation distal to the origin of the fistula, which is proposed as the pathophysiology of the coronary “steal phenomenon”. These hemodynamic changes may potentially cause cardiac ischemia, symptoms of congestive heart failure, arrhythmias, atrial fibrillation, and myocardial infarction, among others [2]. Additionally, hemopericadium and cardiac tamponnade can occur following the rupture of an accompanying large aneurysm of the fistula [10]. In addition to the potential rupture and bleeding, a large associated aneurysm has also been proposed to be a cause of cardiac ischemia by way of extrinsic stenosis of adjacent coronary arteries instead of stealing distal coronary supply [11].
Although the current literature lacks in large enough data sets enabling the establishment of clear therapeutic strategies for the treatment of CAF, it has been recommended to approach each patient with CAF on a case by case basis, and to percutaneously or surgically treat patients presenting with symptomatic CAF, complications thereof, or with a significant shunt [6].
Apart from the 25 mm large aneurysm complicating the CAF, our patient presented with atrial fibrillation, the investigations of which revealed the presence of the fistula. Although a causal link has not been established between idiopathic AF and CAF, some authors have theorized a relation between those two conditions [12]. With the risk of aneurysmal rupture, the large size of the fistula, and the presence of arrhythmia, our patient was deferred to surgical treatment following a failed attempt at endovascular closure. Failure of percutaneous treatment was due the large diameter of the fistula and its tortuosity as those factors have been reported to be the most important in limiting procedural success [13].
The choice of a minimally invasive and totally endoscopic approach to the surgical treatment via RAMT stems from our extensive experience in this technique [14]. Minimally invasive approaches to valvular surgery have already been shown to offer several advantages over classical sternotomy, including a better pain profile, less requirements for blood transfusion, shorter intensive care stay and satisfactory cosmetic results by sparing the surgical trauma of the classical approach [7,15]. Although RAMT is already established as a safe and effective approach to valvular surgery, the current body of literature reporting this techniques being used in the surgical treatment of such rare anatomical anomalies as CAF is very slim. Successful ligation of CAF through RAMT and other minimally invasive approaches such as partial sternotomy or sub-xyphoidal thoracotomy has indeed been described [16-18]. However, to our knowledge, the current literature lacks in reports of a successful surgical treatment by clipping, ligation, and excision of a large aneurysmal CAF using a totally endoscopic “on-pump beating-heart” approach through RAMT.
Conclusion
Totally endoscopic minimally invasive surgery through right anterior mini-thoracotomy in a patient with a large aneurysmal coronary arteriovenous fistula from the LCx to the SVC was performed safely with excellent clinical and cosmetic results.
Conflicts of interest
Farhad Bakhtiary discloses speakers’ honoraria and/or consulting fees from Edwards Lifesciences, LIS and Abbott. All other authors have nothing to disclose with regard to commercial support.
References
- Krause W. Über den ursprung einer akzessorischen a. Coronaria aus der a. pulmonalis. Z Ratl Med 1865: 225.
- Challoumas D, Pericleous A, Dimitrakaki IA, Danelatos C, Dimitrakakis G. Coronary arteriovenous fistulae: A review. Int J Angiol 23 (2014): 1-10.
- Said SAM, Lam J, Werf T. Solitary coronary artery fistulas: A congenital anomaly in children and adults. A contemporary review. Congenit Heart Dis 1 (2006): 63-76.
- Warnes CA, Williams RG, Bashore TM, et al. ACC/AHA 2008 guidelines for the management of adults with congenital heart disease: Executive summary: A report of the american college of cardiology/american heart association task force on practice guidelines (writing committee to develop guidelines for the management of adults with congenital heart disease). Circulation 118 (2008): 2395-2451.
- Stout KK, Daniels CJ, Aboulhosn JA, et al. 2018 AHA/ACC guideline for the management of adults with congenital heart disease: A report of the american college of cardiology/american heart association task force on clinical practice guidelines. J Am Coll Cardiol 73 (2019): e81-192.
- Baumgartner H, De Backer J, Babu-Narayan SV, et al. 2020 ESC guidelines for the management of adult congenital heart disease. Eur Heart J 42 (2021): 563-645.
- El-Sayed Ahmad A, Bayram A, Salamate S, et al. Percutaneous versus surgical femoral access in minimally invasive cardiac operations. Eur J Cardiothorac Surg 61 (2022): 1348-1354.
- Vavuranakis M, Bush CA, Boudoulas H. Coronary artery fistulas in adults: Incidence, angiographic characteristics, natural history. Cathet Cardiovasc Diagn 35 (1995): 116-120.
- Yildiz A, Okcun B, Peker T, et al. Prevalence of coronary artery anomalies in 12,457 adult patients who underwent coronary angiography. Clin Cardiol 33 (2010): E60-64.
- Bauer HH, Allmendinger PD, Flaherty J, et al. Congenital coronary arteriovenous fistula: Spontaneous rupture and cardiac tamponade. Ann Thorac Surg 62 (1996): 1521-1523.
- Bertolin Boronat J, Faga V, Aguar CP, et al. Left anterior descending coronary artery compressed by a giant coronary fistula aneurysm: A case report. Eur Heart J Case Rep 3 (2019): 1-5.
- Jamali HK, Raza U, Waqar F. Idiopathic atrial fibrillation and coronary arteriovenous fistulae: Is there a link. Cardiology 134 (2016): 433-435.
- Collins N, Mehta R, Benson L, et al. Percutaneous coronary artery fistula closure in adults: Technical and procedural aspects. Catheter Cardiovasc Interv 69 (2007): 872-880.
- El-Sayed AA, Salamate S, Bakhtiary F. Lessons learned from 10 years of experience with minimally invasive cardiac surgery. Front Cardiovasc Med 9 (2022): 1053572.
- Schmitto JD, Mokashi SA, Cohn LH. Minimally-invasive valve surgery. J Am Coll Cardiol 56 (2010): 455-462.
- Song L, Dong Y, Wang B, et al. The evolution of the minimally invasive approach and conventional median sternotomy for coronary artery fistula correction. Heart Lung Circ 28 (2019): 1277-1282.
- Liu H, Mei J, He Y, et al. Minimally invasive surgery for coronary-pulmonary artery fistula via parasternal minithoracotomy. Heart Lung Circ 28 (2019): e79-82.
- Kitahara H, Tanaka C, Nakanishi S, et al. Coronary-pulmonary artery fistula ligation and mitral valve repair through a mini-thoracotomy approach. Innovations (Phila) 14 (2019): 272-275.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 