Chylothorax after Excision of The Right Broad Ligament Mass of The Uterus was Diagnosed Lymphangiomyomatosis A Case Report and Literature Review
Article Information
Liwan Dai, Tao Chen, Jinxing Wu*
Department of Respiratory and Critical Care Medicine, the First Affiliated Hospital of Chongqing Medical University, Chongqing, China
*Corresponding Author: Jinxing Wu, Department of Respiratory and Critical Care Medicine, the First Affiliated Hospital of Chongqing Medical University, Youyi Road 1, Yuzhong District, Chongqing, 400016, China
Received: 02 June 2022; Accepted: 09 June 2022; Published: 01 July 2022
Citation: Liwan Dai, Tao Chen, Jinxing Wu. Chylothorax after Excision of The Right Broad Ligament Mass of The Uterus was Diagnosed Lymphangiomyomatosis A Case Report and Literature Review. Journal of Surgery and Research 5 (2022): 400-406.
View / Download Pdf Share at FacebookAbstract
Lymphangioleiomyomatosis (LAM) is a rare disease frequently (often) occurs in women of childbearing age. It is a multi-systemic disorder characterized by the proliferation, metastasis, and infiltration of smooth muscle cells in the lung and other tissues. We will report a case of a patient diagnosed with LAM who presented with chylothorax after hysterectomy of broad ligament.
Background
Lymphangioleiomyomatosis (LAM) is a rare disease frequently occurs in women of childbearing age. It is a multi-systemic disorder characterized by the proliferation, metastasis, and infiltration of smooth muscle cells in the lung and other tissues. The exact etiology of this disease is not known, but due to the gender-specific nature of this disease, the female reproductive system is currently considered as the source of this cell. We will report a case of a patient diagnosed with LAM who presented with chylothorax after b road ligament resection. This phenomenon has not been reported in our literature study, which would provide new evidence for the mechanism of the disease.
Case presentation
A woman who was found to be occupying the right broad ligament developed dyspnea after surgical treatment. Chest CT found bilateral pleural effusion and multiple cystic lesions in both lungs. The pleural fluid Chyle test was positive, subsequent postoperative pathology confirmed LAM. The patient was treated by Sirolimus with Ideal therapeutic effect. However the presence of chylothorax after adnexal occupying resection has not been reported.
Conclusions
The pathogenesis of LAM is not completely clear, but the clinical manifestations of this patient seem to confirm the hypothesis that LAM originates
Keywords
Broad ligament, Chylothorax, Lymphangiomyomatosis, Hydrothorax
Broad ligament articles, Chylothorax articles, Lymphangiomyomatosis articles, Hydrothorax articles
Broad ligament articles Broad ligament Research articles Broad ligament review articles Broad ligament PubMed articles Broad ligament PubMed Central articles Broad ligament 2023 articles Broad ligament 2024 articles Broad ligament Scopus articles Broad ligament impact factor journals Broad ligament Scopus journals Broad ligament PubMed journals Broad ligament medical journals Broad ligament free journals Broad ligament best journals Broad ligament top journals Broad ligament free medical journals Broad ligament famous journals Broad ligament Google Scholar indexed journals Chylothorax articles Chylothorax Research articles Chylothorax review articles Chylothorax PubMed articles Chylothorax PubMed Central articles Chylothorax 2023 articles Chylothorax 2024 articles Chylothorax Scopus articles Chylothorax impact factor journals Chylothorax Scopus journals Chylothorax PubMed journals Chylothorax medical journals Chylothorax free journals Chylothorax best journals Chylothorax top journals Chylothorax free medical journals Chylothorax famous journals Chylothorax Google Scholar indexed journals Lymphangiomyomatosis articles Lymphangiomyomatosis Research articles Lymphangiomyomatosis review articles Lymphangiomyomatosis PubMed articles Lymphangiomyomatosis PubMed Central articles Lymphangiomyomatosis 2023 articles Lymphangiomyomatosis 2024 articles Lymphangiomyomatosis Scopus articles Lymphangiomyomatosis impact factor journals Lymphangiomyomatosis Scopus journals Lymphangiomyomatosis PubMed journals Lymphangiomyomatosis medical journals Lymphangiomyomatosis free journals Lymphangiomyomatosis best journals Lymphangiomyomatosis top journals Lymphangiomyomatosis free medical journals Lymphangiomyomatosis famous journals Lymphangiomyomatosis Google Scholar indexed journals Hydrothorax articles Hydrothorax Research articles Hydrothorax review articles Hydrothorax PubMed articles Hydrothorax PubMed Central articles Hydrothorax 2023 articles Hydrothorax 2024 articles Hydrothorax Scopus articles Hydrothorax impact factor journals Hydrothorax Scopus journals Hydrothorax PubMed journals Hydrothorax medical journals Hydrothorax free journals Hydrothorax best journals Hydrothorax top journals Hydrothorax free medical journals Hydrothorax famous journals Hydrothorax Google Scholar indexed journals postoperative recovery articles postoperative recovery Research articles postoperative recovery review articles postoperative recovery PubMed articles postoperative recovery PubMed Central articles postoperative recovery 2023 articles postoperative recovery 2024 articles postoperative recovery Scopus articles postoperative recovery impact factor journals postoperative recovery Scopus journals postoperative recovery PubMed journals postoperative recovery medical journals postoperative recovery free journals postoperative recovery best journals postoperative recovery top journals postoperative recovery free medical journals postoperative recovery famous journals postoperative recovery Google Scholar indexed journals lung infection articles lung infection Research articles lung infection review articles lung infection PubMed articles lung infection PubMed Central articles lung infection 2023 articles lung infection 2024 articles lung infection Scopus articles lung infection impact factor journals lung infection Scopus journals lung infection PubMed journals lung infection medical journals lung infection free journals lung infection best journals lung infection top journals lung infection free medical journals lung infection famous journals lung infection Google Scholar indexed journals lymphangiomyomatosis articles lymphangiomyomatosis Research articles lymphangiomyomatosis review articles lymphangiomyomatosis PubMed articles lymphangiomyomatosis PubMed Central articles lymphangiomyomatosis 2023 articles lymphangiomyomatosis 2024 articles lymphangiomyomatosis Scopus articles lymphangiomyomatosis impact factor journals lymphangiomyomatosis Scopus journals lymphangiomyomatosis PubMed journals lymphangiomyomatosis medical journals lymphangiomyomatosis free journals lymphangiomyomatosis best journals lymphangiomyomatosis top journals lymphangiomyomatosis free medical journals lymphangiomyomatosis famous journals lymphangiomyomatosis Google Scholar indexed journals renal angiolipomas articles renal angiolipomas Research articles renal angiolipomas review articles renal angiolipomas PubMed articles renal angiolipomas PubMed Central articles renal angiolipomas 2023 articles renal angiolipomas 2024 articles renal angiolipomas Scopus articles renal angiolipomas impact factor journals renal angiolipomas Scopus journals renal angiolipomas PubMed journals renal angiolipomas medical journals renal angiolipomas free journals renal angiolipomas best journals renal angiolipomas top journals renal angiolipomas free medical journals renal angiolipomas famous journals renal angiolipomas Google Scholar indexed journals abdominal hemorrhage articles abdominal hemorrhage Research articles abdominal hemorrhage review articles abdominal hemorrhage PubMed articles abdominal hemorrhage PubMed Central articles abdominal hemorrhage 2023 articles abdominal hemorrhage 2024 articles abdominal hemorrhage Scopus articles abdominal hemorrhage impact factor journals abdominal hemorrhage Scopus journals abdominal hemorrhage PubMed journals abdominal hemorrhage medical journals abdominal hemorrhage free journals abdominal hemorrhage best journals abdominal hemorrhage top journals abdominal hemorrhage free medical journals abdominal hemorrhage famous journals abdominal hemorrhage Google Scholar indexed journals
Article Details
1. Case Report
A 44-year-old woman was found a mass of the right broad ligament of the uterus by ultrasonography during physical examination. Two months ago she admitted to the gynecology and obstetrics department in a local hospital for treatment, the gynecologist advised her to do operation. One month ago her right broad ligament was removed, postoperative recovery was good. Two weeks ago she had a cough and persistent chest pain while she began to experience shortness of breath during routine activities after surgery. Ten days later she felt dyspnea, but without fever, hemoptysis. She had to re- admitted to the local hospital for treatment. Physical examination revealed body weight of 57.2 kg, pulse rate of 116 beats/min, respiratory rate of 26/min, blood pressure of 104/64mmHg, her respiratory sound were weak in both lower lungs. Ultrasound examination revealed a small amount of bilateral hydrothorax, but pleural puncture was not performed immediately. They thought she had bilateral pleural effusion because of lung infection by postoperative lying in bed after surgery, moxifloxacin was administered for six days without improvement. The patient’s dyspnea was deteriorating and she could not walk. She was admitted to our hospital. Before analyzing the patient's treatment, we did blood test, c-reactive protein, pleural fluid routine and spiral computed tomography (CT) (Figure 1). The blood tests showed white blood cells 9.73×109 /L, neutrophil 86%, C-reactive protein 61. The patient presented multiple cystic changes and bilateral pleural effusion in her chest CT scan. The preliminary diagnosis should consider with cystic lung disease, for example: CTD-ILD LAM (Lymphangioleiomyomatosis) PLCH (pulmonary Langerhans cell histiocytosis) LIP (lymfhoid interstitial pneumonia). Because of severe dyspnea, a lung biopsy could not be done on the patient, instead thoracocentesis became our preferred method. Surprisingly, the right pleural effusion was milky white, turbid, Rivalta test was positive. The total number of hydrothorax cells were 7000, mononuclear cells rate was 78%,multinucleated cells rate was 22%, total protein is 27g/L, albumin is 24g/L, lactate dehydrogenase is 144u/L, adenylic deaminase is 0.9u/L; and the left pleural effusion is also milky white, turbidity. Rivalta test is positive, total cellular score are 5000,mononuclear cells rate was 84%, multinucleated cells rate was 16%, total protein 26g/L, albumin 21g/L, lactate dehydrogenase 108u/L, and adenylic deaminase was 2.1u/L. The chyle test of bilateral pleural effusion were positive. At this moment, we highly suspected that this patient had LAM and acquired a post-operative pulmonary infection. We interrogated the patient again, she revealed she had an intermittent cough for 3 years. Because the symptoms were mild, she never got admitted to hospital for diagnosis or treatment. One year ago, the patient appeared anhelation after the activity and occurred irregular intermittent hemoptysis. Her chest X-ray showed increased lung texture. Unfortunately, chest CT and pulmonary function test were not performed on the patient. Based on the patient's history, the patient most probable diagnosis was lymphangiomyomatosis. At the same time, the patient's pathological report after surgery indicated: Lymphangiomyoma with mullerian epithelial hyperplasia and calcification (Figure 2). Taking into consideration the lungs cystic changes, chylothorax, and pathological report, we diagnosed that she had lymphangiomyomatosis with multiple system (damage). Sirolimus was used for her treatment. After 4 months of treatment with sirolimus, the patient's chylothorax and dyspnea got relieved, we were still looking at the patient dynamically.
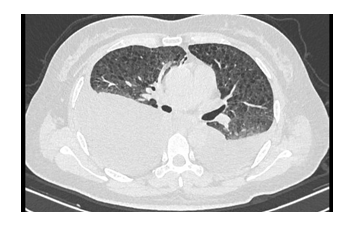
Figure 1: Presented multiple cystic changes and bilateral pleural effusion in her chest CT scan
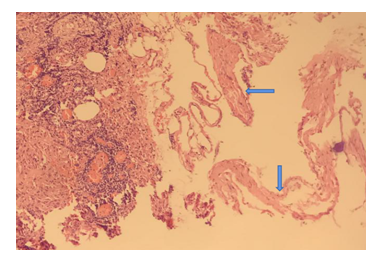
Figure 2: Arrow point Lymphangiomyoma with mullerian epithelial hyperplasia and calcification.
2. Discussion
Lymphangiomyomatosis (LAM) is a serious and rare disease that mainly affects women of childbearing age. The etiology and pathophysiology of the disease remains unclear. However, the overwhelmingly large proportion of female victims indicates an origin from the female reproductive system.The disease mainly affects smooth muscle cells (SMCs) [1,4] involving multiple organs, but the lungs are most often involved. Pulmonary cystic changes are characteristic, and pneumothorax (57%) and hemoptysis (32%) are the most common manifestations in the respiratory system. Furthermore, abdominal angiolipomas (32%), lymphangioleiomyoma (29%), pleural effusions (12%), and less commonly, chylothorax, chylous ascites, chyluria, chyloptysis, and abdominal hemorrhage caused by renal angiolipomas could appear in LAM [2].
Takuo Hayashi’s studies, based on An analysis of pathological features in 10 LAM patients by T. Hayashi et al. showed that the pathological features of LAM were present in the uterus in 9 out of the 10 patients, in the adnexa in 5 out of 8 patients, in the broad ligaments in 2 patients, and in the ovaries and the broad ligaments in 3 patients[5]. The study, despite its small sample size, proved the presence of LAM in female reproductive organs. It is of note that none of these patients had features of LAM in their lungs. The results from this study to some extent confirmed the hypothesis that LAM cells originated from the uterus [5].
A mass in the right broad ligament was found in our patient during routine physical examination, which was comfirmed to be lymphangiomyoma of the broad ligament by postoperative pathology. The result was consistent with the observation of T. Hayashi et al.As far as we know, lymphangiomyomatosis in the uterine broad ligament is a rare pathological finding. That our patient developed chylothorax after removal of the mass in the uterine broad ligament is even more intriguing.
If the female reproductive system is the source of LAM-specific selection, how did chylothorax develop after removel of the mass? Based on the conclusions of the above literature and review of history of the patient, we developed the following hypothesis. Extrapulmonary LAM mainly occurs in the mediastinum, the upper retroperitoneal areas close to the abdominal aorta, and the pelvic cavity [12]. LAM cell proliferation in lymph nodes through the direction of lymph reflux occurs most frequently in the retroperitoneum and pelvic cavity along the axial lymphatics, indicating that LAM cells may originate in this area or originate distal to this area in a direction toward the lymphatic stream [3].
As described above, the most plausible model for the development of pulmonary LAM is that LAM cells originating in the uterus invade through the lymphatic vessels, migrate through the pelvic nodes into the axial lymphatic system, reaching the thoracic duct [12], where the cells are transferred to the venous system at the jugulosubclavian angle and ultimately enter the pulmonary circulation [6]. Given their presence in the lymphatic flow, LAM cell clusters are transported through the lymphatic flow to the venous circulation and ultimately to the pulmonary microvasculature.
Thus, back to this patient, as the broad ligament of the uterus is a two-layered peritoneal fold containing blood vessels and lymphatic vessels, we speculate that the lymphatic vessels in the broad ligament of the patient were the origin of the LAM cells , where the LAM cells continued to proliferate and grow into a lesion. When the lesion was removed, the original proliferation and homing process of the LAM cells was affected in that the origin of the LAM cells was removed, and the remaining LAM cells in the lymphatic vessels flowed back to the pulmonary circulation along the lymphatic vessels, and their colonization in the lungs led to pulmonary lymphatic obstruction or pulmonary lymphatic damage to form new LAM cell attack areas, and therefore chylothorax was present. The absence of pleural effusion when the mass in the broad ligament was found and the development of pleural effusion after its resection were also in line with the hypothesized development model of lung LAM originating from the female reproductive system and migrating along the lymphatic vessels to the lungs.At the same time, our hypothesis has raised a question on whether there were reproductive system abnormalities at the time of diagnosis of pulmonary LAM for other female patients, which requires further study.
How do we treat this patient further? Researchers discovered that LAM tumor cells contain a very specific genetic defect: an inactivating mutation in tumor suppressor gene tuberous sclerosis (TSC)1 or TSC2 [7,8]. These proteins normally complex with Tre2-Bub2-Cdc16 1 domain family, member 7 (TBC1D7) [9] to inhibit Ras homolog-enriched in brain, an activator of mammalian target of rapamycin complex 1 (mTORC1) [10,11]. As a result, sirolimus is effective in the treatment of LAM. After oral administration of sirolimus, dyspnea was relieved and chylopleural fluid was controlled but the resolution was not complete, and the patient needed repeated drainage of pleural fluid in the outpatient clinic. Her activity was improved after treatment. The patient later underwent thoracic duct ligation because she was troubled by repeated thoracentesis, and the formation of chylothorax was relieved after the procedure. So far, the patient still visits regularly for follow-up observation and is still on oral sirolimus for maintenance treatment. No drug-related adverse effects have occurred so far. Pulmonary function tests showed that FEV1 and FVC have been stable.
3. Conclusion
Review of the diagnosis and treatment of this patient has led to new understanding of space occupying lesions of the female reproductive system. It is rare to find LAM in the female reproductive system. After surgical removal of the mass, there is a risk of chylothorax. A more comprehensive evaluation is required before surgery. Chest CT should be used as a routine preoperative examination to assess the lungs. The presence of multiple thin-walled cysts and pleural effusions on chest CT should raise an alert to the possibility of LAM.
List of abbreviations
LAM- Lymphangioleiomyomatosis
PEComas- Perivascular epithelioid cell tumor
AML- epithelioid angiomyolipoma
CCST- clear cell ‘sugar’ tumor
TSC- tuberous sclerosis complex
SMCs- smooth muscle cells
TBC1D7- Tre2-Bub2-Cdc16 1 domain family, member 7
mTORC1- mammalian target of rapamycin complex 1
Declarations
Ethics approval and consent to participate
Written informed consent was obtained from the patient for publication of this case report and any accompanying imagesConflict of Interest Statement.
Consent for publication
All of the authors read and approved the final version. If the manuscript passes review, all the authors agree to publish it.
Availability of data and materials
The images we used are available for download throughout the article
Competing interests
All author declare that they have no competing interests.
Funding
Not applicable
Authors' contributions
Jinxing,Wu conceived the whole article,Liwan,Dai are the managers of the patient, collect case data together and write. Tao Chen co-managed the patients and provided some part of data.
Acknowledgements
We would like to appreciate Zhu Xuyou for his help during the discussion.
References
- Fletcher CDM, Unni KK, Mertens F, et al. World Health Organization. Classification of Tumours Pathology and Genetics of Tumours of Soft Tissue and Bone. Lyon: IARC Press (2002): 221-222.
- Henske Elizabeth P, Józwiak Sergiusz,Kingswood J Christopher et al. Tuberous sclerosis complex.[J] .Nat Rev Dis Primers 2 (2016): 16035.
- Hayashi T, Kumasaka T, Mitani K, et al. Prevalence of uterine and adnexal involvement in pulmonary lymphangioleiomyomatosis. Am J Surg Pathol 35 (2011): 1776-1785.
- Qin W, Bajaj V, Malinowska I, et al. Angiomyolipoma have common mutations in TSC2 but no other common genetic events. PLoS One 6 (2011): e24919.
- Hayashi T, Kumasaka T, Mitani K et al. Prevalence of uterine and adnexal involvement in pulmonary lymphangioleiomyomatosis: a clinicopathologic study of 10 patients. Am. J. Surg. Pathol 35 (2011): 1776-1785.
- Henske EP, McCormack FX. Lymphangioleiomyomatosis- a wolf in sheep’s clothing. J Clin Invest 122 (2012): 3807-3816.
- Carsillo T, Astrinidis A, Henske EP. Mutations in the tuberous sclerosis complex gene TSC2 are a cause of sporadic pulmonary lymphangioleiomyomatosis. Proc Natl Acad SciUSA 97 (2000): 6085-6090.
- Smolarek TA, Wessner LL, McCormack FX, et al. Evidence that lymphangiomyomatosis is caused by TSC2 mutations: chromosome 16p13 loss of heterozygosity in angiomyolipomas and lymph nodes from women with lymphangiomyomatosis. Am J Hum Genet 62 (1998): 810-815.
- Dibble CC, Elis W, Menon S, et al. TBC1D7 is a third subunit of the TSC1-TSC2 complex upstream of mTORC1. Mol Cell 47 (2012): 535-546.
- Marygold SJ, Leevers SJ. Growth signaling: TSC takes its place. Curr Biol 12 (2002): 785-787.
- McManus EJ, Alessi DR. TSC1-TSC2: a complex tale of PKB-mediated S6K regulation. Nat Cell Biol 4 (2002): E214-216.Ando Hiroka, Ogawa Minori, Watanabe Yuko et al. Lymphangioleiomyoma of the Uterus and Pelvic Lymph Nodes: A Report of 3 Cases, Including the Potentially Earliest Manifestation of Extrapulmonary Lymphangioleiomyomatosis. Int J Gynecol Pathol 39 (2020): 227-232.


 Impact Factor: * 4.2
Impact Factor: * 4.2 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 