Effect of Orthotopic Liver Transplantation on the Health-Related Quality of Life in Indian Patients with End-Stage Liver Disease: A Prospective Study
Vinayak Nikam*, Vasudevan Ramaswamy, Abhideep Chaudhary, Suresh Singhvi
Department of Surgical Gastroenterology and Liver Transplantation, Sir Ganga Ram Hospital, New Delhi, India
*Corresponding Author: Vinayak Nikam, Department of Surgical Gastroenterology and Liver Transplantation, Sir Ganga Ram Hospital, Room no- 1469, Casualty Block, Old Rajinder Nagar, New Delhi, 110060, India
Received: 16 June 2022; Accepted: 24 June 2022; Published: 08 July 2022
Article Information
Citation:
Vinayak Nikam, Vasudevan Ramaswamy, Abhideep Chaudhary, Suresh Singhvi. Effect of Orthotopic Liver Transplantation on the Health-Related Quality of Life in Indian Patients with End-Stage Liver Disease: A Prospective Study. Journal of Surgery and Research 5 (2022): 435-440.
View / Download Pdf Share at FacebookAbstract
Background: End-stage liver disease severely affects all the aspects of a patient’s life including mental and physical health. Orthotopic liver transplantation (OLT) provides a definitive therapeutic measure for patients with end-stage liver disease. Improvements in patient survival have led to a shift in focus from acute management to long-term outcomes after liver transplantation. Health-related quality of life (HRQoL) is an important aspect of long-term outcomes as it incorporates the physical, social, and emotional well being of the patient. We report our experience regarding the HRQoL pre and post-liver transplant using Standard SF 36 questionnaire in living donor liver transplant (LDLT) recipients from India.
Methods: The study was designed as a single-center, observational, prospective study. We assessed the HRQoL using the SF 36 health questionnaire before transplant and three months post-liver transplant in sixty liver recipients from June 2013 to March 2015. Statistical analysis was performed using SPSS statistical software, version 17.0. Continuous and categorical variables were analyzed using paired t-test and non-parametric sign tests respectively. Pearson correlation test was used to analyze the relationships between HRQoL and pre-transplantation severity of the liver disease.
Results: Sixty patients underwent LDLT with 83.3% of them being male with a median age of 44.7 years. We found significant improvement in all the eight dimensions of SF 36 health score in recipients at 3 months post-liver transplant (p<0.001) compared to pre-transplant. Post-transplant HRQoL in domains of vitality, physical functioning, and mental component summary scores significantly correlated with liver disease severity, which measured by Model for End-stage liver disease (MELD) score and Child-Turcotte-Pugh (CTP)
Keywords
<p style="text-align:justify">Health-Related Quality of Life, Model for End-Stage Liver Disease, Child-Turcotte-Pugh Score, Orthotopic Liver transplantation, SF-36 questionnaire</p>
Article Details
1. Introduction
End-stage liver disease is a progressive illness with liver transplantation (LT) as an established form of treatment [1]. In India with a population of 1.37 billion till March 2019, the end-stage liver disease is becoming increasingly prevalent with an estimated annual requirement of LT between 30,000 and 32,000. Currently, only about 2200 to 2500 liver transplants operations are being performed due to limited cadaveric donor pool [2,3] and the approved number of government recognized centers [4]. Liver transplantation is the gold standard therapeutic option for end-stage liver disease [5]. In the last few decades, there have been progressive improvements in graft and patient survival [6]. The1, 5- and 10-years survival has been reported to be 92%, 83%, and 79%, respectively [7]. Etiological categorization of end-stage liver disease revealed the hepatitis C virus (HCV), hepatitis B virus (HBV), ethanol-related, and cryptogenic cirrhosis as a major cause of liver damage [8]. Patients with the end-stage Liver disease have a negative impact on daily health-related quality of life (HRQoL). As life expectancy after liver transplantation continuously improving, there has been a transit from the medical management of LT recipients to a more long-term approach in form of HRQoL, which includes not only the physical well-being of recipients but also their social and emotional well-being. The quality of life of recipients evaluated by several studies and has determined important elements such as medical complications, physical function, social function, fatigue, employment status, emotional function, and sexual dysfunction [9]. These studies have shown that the quality of life improves after LT; this is demonstrated by improvements in a recipient's functional capacity, perception of health condition, and self-esteem and these parameters continue to revamp over time. Short form-36(SF-36) is a validated questionnaire derived from a larger set of questions and provides a composite scale of mental and physical health for patients. Studies published outside India reported positive outcomes in HRQoL after LT [10-12]. In Indian scenario two studies have been reported with one determining HRQoL using SF-36 in liver cirrhosis patients compared with the same age and gender healthy controls, the severity of liver disease in this study was assessed by MELD and CTP scoring [13] while in another study Bidare et al [14] assed HRQoL using SF-36 in pre and post LT; hence this study will provide more insight on HRQoL in pre and post LT Indian patient's, Also it will give information on the correlation between the severity of liver disease (MELD and CTP) and Diabetes mellitus (DM).
2. Material and methods
The study was designed as a prospective, observational, and single-center study. Sixty end-stage liver disease patients evaluated for LT at Sir Ganga Ram Hospital, New Delhi between June 2013 to March 2015 in the department of Surgical Gastroenterology and Liver Transplantation were enrolled in the study. This study is approved by the Institutional ethics committee board (EC/08/13/586). Informed and written consent for participation was obtained from all the recipients and they all completed the validated SF-36 questionnaire by interview or E-mail pre-LT as well as Post-LT follow-up at 3 months. The study was performed under a license approval certificate (License Number- QM029912, Optuminsight Life Sciences, Inc) for the use of the SF-36 software. The results from questionnaires completed by interview versus mail were equivalent. In this study chronic end-stage liver disease patients were included like HCV-related cirrhosis, alcoholic liver cirrhosis, HBV-related cirrhosis, Autoimmune, Budd Chiari Syndrome, and non-alcoholic fatty liver (NASH). The other inclusion criteria were age ≥18 years, a fully-conscious, ambulatory, liver transplant recipient. Patients with acute liver failure, multiple-organ transplant listed, and patients not willing to participate or complete the SF-36 questionnaire were excluded.
2.1 Post-transplant Follow-up protocol
Our institute developed its own post –LT follow-up protocol model to assess the physical and psychosocial recovery of our recipients. At the time of discharge, recipients and their family members were educated about healthy lifestyle and general daily life precautions. It was also suggested that patients should visit transplant physicians or surgeons monthly in the first 6 months, every 3 monthly in the second 6 months, and 6 monthly beyond 1 year. Recipients were instructed to follow up with our transplant physicians in the out-patient clinic or by mail or telephone if they had any health-related problems. Regularly, our institute staff also doing telephonic communication with the recipient to know general health. Patients were further evaluated with clinical and laboratory investigation stools, and intervention or even readmission, in case any abnormalities were identified. Furthermore, an annual meeting was held to update our recipients with information related to medication and daily care, and maintaining optimum HRQoL was emboldened at the annual meeting.
2.2 SF-36- Tool to evaluate HRQoL
The SF-36 questionnaire was used to evaluate HRQoL consisting 36 questions (items) measuring physical and mental health status with eight health parameters: physical functioning (PF), role limitations due to physical problems: role physical (PR), body pain (BP), general health perceptions (GH), vitality: energy/fatigue (VT), social function (SF), role limitations due to emotional problems: role emotional (ER), and general mental health: psychological distress/wellbeing (MH). The SF-36 questionnaire was given to the recipient's pre and post-liver transplantation by interview or mail-in language he/she understands (Hindi and English version). Responses to each of the SF-36 items were scored and summed according to a standardized scoring protocol and expressed as a score on a 0–100 scale for each of the eight health concepts. Higher scores represent better self-perceived health. The maximum of 100 on these bipolar scales, therefore, indicated not just the absence of disability, but the presence of a positive state of health. Two-component summary measures have been calculated from eight health parameters: The Physical Component Score (PCS) and Mental Component Score (MCS). The summary scores were aggregated measures of the physical health and mental health dimensions underlying the SF-36 questionnaire. The severity of liver disease was assessed before transplant with MELD and CTP scores, with the diabetes status of the patients. The correlation between the severity of disease (MELD, CTP) and DM status with HRQoL at 3 months post-transplant was also evaluated.
2.3 Statistical analysis
Statistical data were analyzed by using Statistical Package for the Social Sciences (SPSS), version 17. Data values were calculated using software provided by the SF36 health questionnaire and reported as mean ± SD. The Categorical numbers were expressed as percentages and frequencies. Nominal categorical variables between the groups were compared using a nonparametric sign test, while the Paired t-test was used to compare the normally distributed continuous variables pre-operative and post-operative 3 months. Spearman correlation analysis was used to analyze the relationships between HRQoL and pre-transplantation severity of liver disease and DM status, a p-value of <0.05 was considered statistically significant.
3. Results
3.1 Recipient Characteristics
The demographic characteristics of the study population are listed in table 1. Most recipient's average age was 45 years at the time of transplantation and male (83.3%) to female (16.7%) ratio was 5:1. Classification by primary liver disease at the time of referral for transplantation showed that the largest group had HCV related cirrhosis with alcoholic and cryptogenic liver disease forming the second and third largest groups respectively. Among the participants, 13.3% had Hepatocellular carcinoma (HCC), 26.7% had Spontaneous bacterial peritonitis (SBP), 36.7 % experienced Hepato-renal syndrome (HRS) and18.3 % had Diabetes mellitus (DM), while 16.7% reported that they experienced early postoperative sepsis. All recipients underwent living donor LT, all of whom were first or second-degree relatives of their donors.
|
Variable |
n=60 |
|
Age (Mean ± SD) |
45±10 |
|
Gender |
|
|
Male |
50 (83.3%) |
|
Female |
10 (16.7%) |
|
Child-Turcotte-Pugh Score |
|
|
A |
2 (3.3%) |
|
B |
9 (15%) |
|
C |
49 (81.7%) |
|
Model for End-stage Liver Disease |
|
|
< 20 |
13 (21.7%) |
|
≥20 |
47 (78.3%) |
|
Etiology |
|
|
Hepatitis C virus |
25 (41.7%) |
|
Alcoholic |
15 (25.0%) |
|
Cryptogenic |
9 (15%) |
|
Hepatitis B virus |
7(11.7%) |
|
Auto Immune Liver Disease |
2 (3.3%) |
|
Budd-Chiari Syndrome |
1 (1.7%) |
|
Non-alcoholic Steatohepatitis |
1 (1.7%) |
|
Hepatocellular carcinoma |
52 (86.7%) |
|
Spontaneous bacterial peritonitis |
16 (26.7%) |
|
Hepato-renal syndrome |
22 (36.7%) |
|
Diabetes mellitus |
11 (18.3%) |
|
Post-operative Sepsis |
10 (16.7%) |
Table 1: Demographics and clinical variables of the patients
3.2 HRQoL scores Pre and Post-transplant.
The HRQoL scores of pre and post-transplant are mentioned in table 2 and figure 1. There was a great difference (p<0.001) in scores of eight domains of the SF-36. Compared to the pre-transplantation, significant improvements were seen in the post-liver transplant values in terms of PF, PR, BP, GH, VT, SF, RE, and MH. Overall there was a significant change in Mental Component Summary and Physical Component Summary in post-transplant as compared to the pre-transplant.
|
SF-36 |
Pre-transplant |
Post-transplant |
p Value |
|
Physical Functioning (PF) |
29.83± 21.59 |
81.00 ± 17.09 |
p<0.001 |
|
Physical Role (PR) |
12.08 ± 30.36 |
80.00 ± 33.45 |
p<0.001 |
|
Bodily Pain (BP) |
41.25 ± 17.72 |
83.12 ± 18.80 |
p<0.001 |
|
General Health (GH) |
13.37 ± 15.01 |
75.28 ± 17.53 |
p<0.001 |
|
Vitality (VT) |
27.92 ± 17.18 |
76.25 ± 12.54 |
p<0.001 |
|
Social Functioning (SF) |
32.71 ± 17.99 |
76.85 ± 14.87 |
p<0.001 |
|
Emotional Role (ER) |
15.55 ± 34.43 |
86.67 ± 28.92 |
p<0.001 |
|
Mental Health (MH) |
36.13 ± 19.19 |
79.67 ± 11.41 |
p<0.001 |
|
Physical Component Summary (PCS) |
29.08 ± 7.29 |
50.00 ±7.44 |
p<0.001 |
|
Mental Component Summary (MCS) |
30.96 ± 9.09 |
52.89 ± 6.09 |
p<0.001 |
Table 2: Pre and post-transplant HRQoL scores (mean ± SD).
3.3 Correlations between pre-transplant severity of liver disease, diabetes status, and HRQoL post-transplant
The results of the Spearman correlation test for determination of correlations between pre-transplant severity of liver disease (MELD/CTP)/DM status and HRQoL (post-transplant) assessed with SF 36 are shown in table 3.There was a negative correlation between overall SF-36 scores and MELD/CTP scores. In MELD negative correlation was found in VT (r= -0.32, p<0.05) and MCS (r= -30, p<0.05), while in CTP negative correlation was observed in PF (r=-0.30, p<0.05), which were found to be significant. No significant correlation was observed between the presence of diabetes mellitus and HRQoL after OLT.
|
SF-36 |
MELD |
CTP |
DM |
|||
|
r |
p |
r |
p |
r |
p |
|
|
PF |
-0.25 |
0.06 |
-0.3 |
0.02* |
-0.089 |
0.5 |
|
PR |
-0.24 |
0.07 |
-0.08 |
0.54 |
-0.092 |
0.48 |
|
BP |
-0.21 |
0.11 |
-0.19 |
0.14 |
-0.19 |
0.15 |
|
GH |
-0.18 |
0.17 |
-0.01 |
0.93 |
-0.02 |
0.9 |
|
VT |
-0.32 |
0.01* |
-0.14 |
0.29 |
0.02 |
0.92 |
|
SF |
-0.16 |
0.2 |
-0.15 |
0.24 |
0.01 |
0.97 |
|
ER |
-0.25 |
0.05 |
-0.19 |
0.14 |
-0.059 |
0.66 |
|
MH |
-0.25 |
0.05 |
-0.08 |
0.52 |
-0.013 |
0.92 |
|
PCS |
-0.25 |
0.05 |
-0.16 |
0.23 |
-0.16 |
0.23 |
|
MCS |
-0.3 |
0.02* |
-0.15 |
0.25 |
-0.013 |
0.92 |
Abbreviations: PF, physical functioning; PR, role physical; BP, bodily pain; GH, general health; VT, vitality; SF, social functioning; ER, role emotional; MH, mental health; PCS, physical component score; MCS, mental component score, MELD: Model for End-stage Liver Disease, CTP: Child-Turcotte-Pugh, DM: Diabetes mellitus.
Correlation coefficient between SF-36 and MELD / CTP and DM, Data analyzed using pearson correlation test (*p<0.05).
Table 3: The p values (correlation coefficient) between SF-36 post OLT with MELD / CTP and DM status.
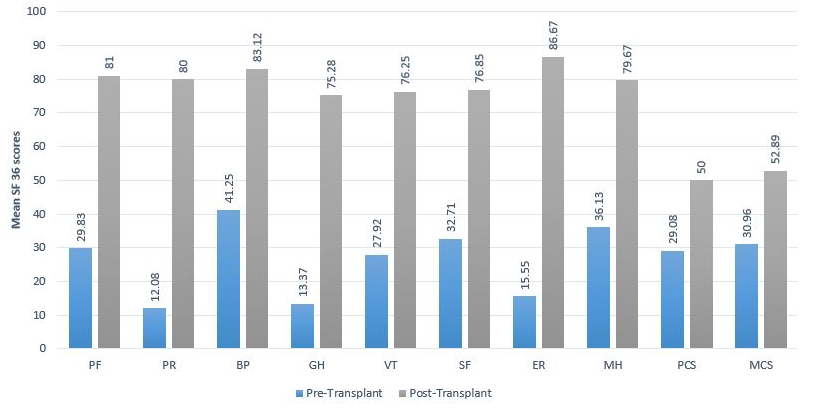
Figure 1: Mean SF-36 scores in the pre-transplant and post-transplant patient after 3 months
4. Discussion
The current study analyzed pre and post-liver transplant recipients HRQoL by using the SF-36 questionnaire, the pre-transplant patients were found to have low HRQoL scores indicating the altered quality of life. End-stage liver disease has a negative effect on all aspects of a patient's well-being and HRQoL [15]. Hence improving the patient's quality of life after liver transplantation takes on increasing importance [16]. The survival after LT is optimally increased hence healthcare providers and patients are more concerned about post-transplant HRQoL. HRQoL focuses on patient care, with the improvement in patient and recipient survival after LT [17]. In the present study, there was a significant improvement in all the domains of SF-36 related to HRQoL after 3 months of OLT. Telles-Correia et al found a significant increase in mental and physical component scores at 1 month after LT [18]. Radcliffe et al. [19] showed that at 3 months post-OLT, there was a statistically great improvement in HRQoL scores in all SF-36 domains except bodily pain. De Bona et al [20] reported an increase in the score for the first 6 months with a decrease in 13 to 24 months and improvement between 3 and 5 years. A study by Younoussi et al. [21] stated that an increase in all SF-36 HRQoL parameters within the first 6 months post-OLT. Likewise, Krasnoff et al. [22] studied post-LT HRQoL scores at 2, 6, 12, and 24 months after OLT, there was an enormous increase in SF-36 physical function, role function, bodily pain and general health domain scores at both 2 and 6 months. Thus significant improvement in HRQoL was observed in Indian patients after OLT from our study indicating positive outcomes in both mental and physical health status.
4.1 The severity of liver disease (CTP and MELD Score) and HRQoL
In our study MELD/CTP showed a negative correlation in all the SF-36 domains with a significant negative correlation in vitality (VT) and mental component score (MCS) in MELD and Physical functioning (PF) in CTP score. One might expect the liver disease severity negatively correlate with HRQoL scores. Saab et al. [23] attempted to correlate the severity of liver disease by the MELD and CTP scoring with pre-transplantation HRQoL scores and found that the MELD score was not predictive of HRQoL scores, with the SF-36 used as an assessment instrument. The presence of hepatic encephalopathy and/or ascites in ESLD is significantly associated with unfavorable quality of life. CTP score comprises of hepatic encephalopathy and ascites, due to this reason CTP more appropriately correlate with QOL. Estraviz et al25 found that the CTP score could be correlated with pre-transplantation HRQoL parameters at 6 months after transplant. Castaldo et al [26] reported that increased MELD score before grafting was associated with improved physical HRQoL in the first 12 months after LT and did not affect mental HRQoL. Rodrigue et al failed to show a correlation between CTP score and HRQoL, but they found a good correlation between MELD Score and HRQoL.
Kotarska et al [27] reported that liver severity analyzed with CTP and MELD scores before LT had a minimal effect on HRQoL after LT and exerts an insignificant effect on patients evaluated 1 year after LT. Few studies showed that there is no correlation between HRQoL and severity of the liver disease like Ortega et al [24] reported that MELD and CTP scores did not correlate with HRQoL either before or after transplantation.
4.2 Diabetes mellitus and HRQoL
In this study, the presence of diabetes mellitus did not have any significant correlation with HRQoL after OLT. Kotarska et al27 experienced that the recipients with DM seemed to have an unsatisfactory quality of life early after surgery.
Limitations of the study
Our study has a few important limitations. First, relatively few patients were enrolled in the study. Thus, it would be appropriate to extend it to include more number of patients. Second, the study had short term follow up and HRQoL assed once only after the first 3 months post LT, hence it is very difficult to assess long term HRQoL. Third, in this study pre LT patients were compared with post LT patients, however, in other studies post LT recipients were compared with the healthy general population. Lastly, there are many studies in which employment, income, sexual and reproductive parameters analyzed after transplant, but the present study didn't assed these. In conclusion, there was a significant increase in HRQoL at first quarter post-LT with no prediction between the severity of disease and diabetes status in Indian patients. However, HRQoL and correlation between severities of liver disease should be evaluated for a longer duration of time on larger Indian liver transplant population
Declarations
- Ethics approval Committee Name and consent to participate - 1. Institutional Ethics Committee, Sir Ganga Ram Hospital, New Delhi, India (EC/08/13/586). 2. All Recipients gave informed written consent for participation in study.
- Consent for publication – Not Applicable
- Availability of data and material- The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
- Competing interests- The authors declare that they have no competing interests.
- Funding- No grant support or any kind of assistance from other institute
- Authors' contributions
- A) Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work -VN, VR, AC, SS
- B) Drafting the work or revising it critically for important intellectual content- VN, VR, AC, SS
- C) Final approval of the version to be published- VN, VR, AC, SS
- D) Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved- VN, VR, AC, SS
Acknowledgments
The authors would like to thank Miss Anusha Nikam, Dr Leena Tayshete and Jay Hire for language editing help and technical support.
References
- Jamie Potosek, Michael Curry, Mary Buss, Eva Chittenden. Integration of Palliative Care in End-Stage Liver Disease and Liver Transplantation. Journal of Palliative Medicine 17 (2014): 11.
- Narasimhan G, Kota V, Rela M. Liver transplantation in India. Liver Transpl 22 (2016): 1019-1024.
- Viniyendra P, Deeplaxmi PB, Senthil K, et al. Deceased donor liver transplant: Experience from a public sector hospital in India. Indian J Gastroenterol 37 (2018): 18-24.
- Sanjay N, Aditya N, Aabha Nagral. Liver Transplantation in India: At the Crossroads. Journal of Clinical and Experimental Hepatology 5 (2015): 329-340.
- Neuberger J. What is the real gain after liver transplantation? Liver Transpl 15 (2009): S1-S5.
- Roberts MS, Angus DC, Bryce CL, et al. Survival after liver transplantation in the United States: A disease-specific analysis of the UNOS database. Liver Transpl 10 (2004): 886-897.
- AS Soin, S Thiagarajan. Liver transplant scene in India. MAMC Journal of Medical Sciences 2 (2016): 6-11.
- Nayak NC, Jain D, Vasdev N, et al. Etiologic types of end-stage chronic liver disease in adults: analysis of prevalence and their temporal changes from a study on native liver explants. Eur J Gastroenterol Hepatol 24 (2012): 1199-1208.
- Burra P. Sexual dysfunction after liver transplantation. Liver Transpl 15 (2009): S50-S56.
- Desai R, Jamieson NV, Gimson AE, et al. Quality of Life up to 30 years Following Liver Transplantation. Liver Transpl 14 (2008): 1473-1479.
- Gen-Shu Wang, Yang Yang, Hua Li, et al. Health-related quality of life after liver transplantation: the experience from a single Chinese center. HepatobiliaryPancreat Dis Int 11 (2012): 262-266.
- Pei-Xian Chen, Lu-Nan Yan, Wen-Tao Wang. Health-related quality of life of 256 recipients after liver transplantation. World J Gastroenterol 18 (2012): 5114-5121.
- Janani K, Joy V, Mayank J, et al. HRQOL using SF36 (generic specific) in liver cirrhosis. Indian Society of Gastroenterology 36 (2017): 313-317.
- Asha Bi, Kaiser R, Sonal A, et al. Effect of liver transplantation on the health-related quality of life (HRQoL) of a patient with end-stage liver failure. Indian Journal of Transplantation 8 (2014): 126-138.
- Younossi Z, Guyatt G. Quality of life assessments in chronic liver disease. Am J Gastroenterol 93 (1998): 1037-1041.
- Belle SH, Porayko MK, Hoofnagle JH, et al. Changes in quality of life after liver transplantation among adults. Liver Transpl Surg 3 (1997): 93-104.
- Bownik H, Saab S. Health-related quality of life after liver transplantation for adult recipients. Liver Transpl 15 (2009): 42-9.
- Telles-Correia D, Barbosa A, Mega I, et al. When does quality of life improve after liver transplantation? A longitudinal prospective study. Transpl Proc 41 (2009): 904-905.
- Ratcliffe J, Longworth L, Young T, et al. Cost-effectiveness of liver transplantation team assess health-related quality of life pre- and post-liver transplantation: a prospective multicenter study. Liver Transpl 8 (2002): 262-270.
- De Bona M, Ponton P, Ermani M, et al. The impact of liver disease and medical complications on quality of life and psychological distress before and after liver transplantation. J Hepatol 33 (2000): 609-615.
- Younoussi ZM, McCormick M, Price LL, et al. Impact of liver transplantation on health-related quality of life. Liver Transpl 6 (2000): 779-783.
- Krasnoff JB, Vintro AQ, Ascher NL, et al. Objective measures of health-related quality of life over 24 months post-liver transplantation. Clin Transplant 19 (2005): 1-9.
- Saab S, Ibrahim AB, Shpaner A, et al. MELD fails to measure the quality of life in liver transplant candidates. Liver Transpl 11 (2005): 218-223.
- Ortega T, Deulofeu R, Salamero P, et al. Impact of Health-Related Quality of Life in Catalonia Liver Transplant Patients. Transplantation Proceedings 41 (2009): 2187-2188.
- Estraviz B, Quintana JM, Valdivieso A, et al. Factor influencing change in health-related quality of life after liver transplantation. Clin Transplant 21 (2007): 481-490.
- Castaldo ET, Feurer ID, Russell RT, et al. Correlation of health-related quality of life after liver transplant with the Model for End-Stage Liver Disease score. Arch Surg 144 (2009): 167-172.
- Kotarska K, Raszeja-Wyszomirska J, Wunsch E, et al. Relationship Between Pre-transplantation Liver Status and Health-Related Quality of Life After Grafting: A Single-Center Prospective Study. Transplantation Proceedings 46 (2014): 2770-2773.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 