Effects of Granulocyte Colony-Stimulating Factor Administration on Liver Hypertrophy After Portal Vein Embolization in a Rabbit Model
Kohei Nishio1, Kenjiro Kimura1*, Akira Yamamoto2, Ryosuke Amano1, Go Ohira1, Ken Kageyama2, Kotaro Miura3, Naoki Kametani4, Takeaki Ishizawa1
1Department of Hepato-Biliary-Pancreatic Surgery, Osaka Metropolitan University Graduate School of Medicine, 1-4-3 Asahimachi, Abeno-ku, Osaka, 545-8585, Japan.
2Department of Diagnostic and Interventional Radiology, Osaka Metropolitan University Graduate School of Medicine, 1-4-3 Asahimachi, Abeno-ku, Osaka 545-8585, Japan.
3Department of Gastrointestinal Surgery, Izumi City General Hospital, 4-5-1 Wakecho, Izumi City, Osaka 594-0073, Japan.
4Department of Gastrointestinal Surgery, Sumitomo Hospital, 5-3-20 Nakanoshima, Kita-ku, Osaka 530-0005, Japan.
*Corresponding Author: Kenjiro Kimura, M.D., Ph.D., Department of Hepato-Biliary-Pancreatic Surgery, Osaka Metropolitan University Graduate School of Medicine, 1-4-3 Asahimachi, Abeno-ku, Osaka, 545-8585, Japan.
Received: 24 August 2022; Accepted: 30 August 2022; Published: 07 September 2022
Article Information
Citation: Kohei Nishio, Kenjiro Kimura, Akira Yamamoto, Ryosuke Amano, Go Ohira, Ken Kageyama, Kotaro Miura, Naoki Kametani, Takeaki Ishizawa. Effects of Granulocyte Colony-Stimulating Factor Administration on Liver Hypertrophy After Portal Vein Embolization in a Rabbit Model. Journal of Surgery and Research 5 (2022): 494-499.
View / Download Pdf Share at FacebookAbstract
Background: Postoperative liver failure is one of the most frequent causes of perioperative mortality in hepatectomy patients, even with preoperative portal vein embolization (PVE). However, recent research has found that administration of granulocyte colony-stimulating factor (G-CSF) improves liver function and increases the survival rate of patients with decompensated liver cirrhosis. This study aimed to determine the effects of G-CSF administration on liver hypertrophy after PVE in a rabbit model.
Methods: Eight rabbits were divided into an embolization only (PVE) group (n = 4) and an embolization with G-CSF administration (G-CSF) group (n = 4). The degree of nonembolized liver volume hypertrophy (DLV) and the immunohistochemistry for Ki67, RAM11, and CD34 levels were compared between the two groups to quantify macrophage and cell proliferation and the presence of CD34-positive cells in the liver.
Results: The median DLV in the PVE group was 14.7%, compared to 18.8% in the G-CSF group. This was a significant difference (p = 0.042). The expression of both Ki67 and RAM11 in the nonembolized parts of the livers of the G-CSF group was significantly greater than in the nonembolized livers of the PVE group (p = 0.0003). There was no significant difference in CD34 expression in the nonembolized livers of the rabbits in the two groups.
Conclusions: In our rabbit model, the DLV and cell proliferation in the G-CSF group were significantly greater than in the PVE group. This suggests that G-CSF administration with PVE prompts the proliferation of liver cells.
Keywords
<p>CD34, Granulocyte colony-stimulating factor, Liver hypertrophy, Macrophages, Portal vein embolization, Rabbit model</p>
Article Details
Abbreviations
ACLF Acute-on-chronic liver failure
CLV Caudal liver volume
CT Computed tomography
DLV Degree of nonembolized liver volume
FRL Future remnant liver
PVE Portal vein embolization
TLV Total liver volume
Introduction
Currently, liver resection is the only curative therapy for primary or secondary hepatic tumors and major resection is essential in over 50% of patients. To minimize the risk of liver failure after major hepatectomy, a remnant liver volume of at least 25% is needed in patients with normal livers and at least 40% in those with chronic liver disease [1]. Portal vein embolization (PVE) is an effective means of preventing postoperative liver failure. This is achieved by its induction of hypertrophy in the future remnant liver (FRL). PVE is the optimal treatment before major hepatectomy for hepatocellular carcinoma or colorectal liver metastases. However, up to one-third of patients fail to achieve a sufficient increase in FRL size after PVE [2]. Granulocyte colony-stimulating factor (G-CSF) is a growth factor that stimulates the bone marrow to produce granulocytes and stem cells and release them into the circulation [3]. G-CSF treatment has been shown to mobilize hematopoietic stem cells and improve hepatic progenitor cell proliferation. G-CSF therapy has been shown to improve survival and speed clinical recovery in liver failure and acute-on-chronic hepatitis [4-6]. This study aimed to determine the effects of G-CSF administration on liver hypertrophy after PVE in a rabbit model.
Materials and Methods
Animals
The experimental procedures were approved by Instituional Review Board of Osaka City University (approval number: 4376) and Institutional Animal Care and Use Committee of Osaka City University (approval number: 17027). Female New Zealand white rabbits (n = 8) were acclimatized for 2 weeks in standardized laboratory conditions with a 12-hour light-dark cycle and with water and standard rabbit food available ad libitum.
Study design
The rabbits were divided into an embolization only (PVE) group (n = 4) and an embolization with G-CSF administration (G-CSF) group (n = 4). Before PVE, all rabbits underwent enhanced computed tomography (CT). The portal vein branch to the cranial liver lobes which contained left lateral lobe, left median lobe and right lobe was embolized using polyvinyl alcohol particles, leaving the caudal liver lobe free. G-CSF was administered subcutaneously at a dose of 50 μg/kg every 24 h for five consecutive days after embolization to the G-CSF group. Enhanced CT volumetry was performed 7 days before PVE and 7 days after PVE. All rabbits were humanely killed 7 days after PVE to collect histological specimens.
Computed tomography volumetry
The rabbits were anesthetized by subcutaneous injection (2 ml) of ketamine and placed in a supine position. Contrast solution (6 ml) (Iomelon, Eisai, Tokyo, Japan) was injected through a 24G catheter via the ear vein followed by a flush with 3 ml of sterile physiological saline. A contrast-enhanced scan of 2 mm axial slices was performed 45 seconds (venous phase) after injection of the contrast solution. The caudal liver lobe and the cranial liver lobes were manually delineated and the total liver volume (TLV) and caudal liver volume (CLV) were calculated. We divided the preoperative nonembolized caudal liver lobe volume (CLV pre-PVE) by the preoperative total liver lobe volume (TLV pre-PVE) to provide the percentage of the preoperative nonembolized caudal liver lobe (%CLV pre-PVE). We divided the postoperative nonembolized caudal liver lobe volume (CLV post-PVE) by the postoperative total liver lobe volume (TLV post-PVE) to provide the percentage of the postoperative nonembolized caudal liver lobe (%CLV post-PVE). The degree of nonembolized liver volume hypertrophy (DLV) was calculated by subtracting the %CLV pre-PVE from the %CLV post-PVE.
PVE procedure
All of the rabbits were anesthetized by subcutaneous injection (4 ml) of ketamine. The animals were placed in the supine position and a midline laparotomy was performed after the injection of 1 ml of 1% xylocaine mixed with 9 ml sterile physiological saline (total 10 ml). A branch of the ileocolic vein was cannulated with a 24-gage catheter (Surflo V3, Terumo, Tokyo, Japan). A guidewire (0.018 in) was inserted and a microcatheter was introduced into the portal vein. Portography was performed and the microcatheter was positioned in the main portal branch supplying the cranial liver lobes. The portal branches were embolized by transcatheter infusion of polyvinyl alcohol particles (90–180 μm). After portographic confirmation of successful PVE, the catheter was removed and the mesenteric vein was closed with a ligature. The abdomen was closed in two layers. The PVE procedure is shown in figure 1.
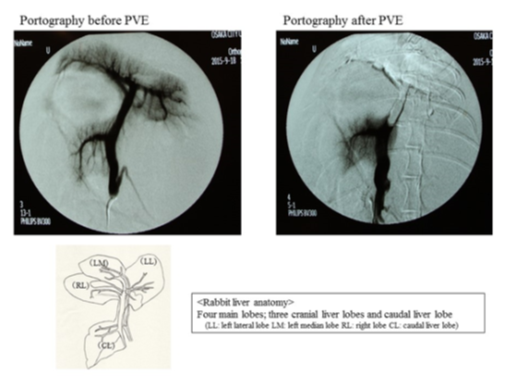
Figure 1: Rabbit liver anatomy and Portal vein embolization
Histological examination
Immediately after the animal was sacrificed, the liver of each rabbit was excised and split into the caudal and cranial lobes. Each liver lobe was fixed in 10% formaldehyde, dehydrated in graded steps with ethanol and xylene, embedded in paraffin, and cut into 4-μm sections. The sections were stained with hematoxylin-eosin. They were also immunostained with monoclonal mouse Ki-67 antibodies (1:100 dilution) (Dako Cytomation, Glostrup, Denmark), monoclonal mouse antirabbit macrophages (1:100 dilution) (Dako Cytomation), and monoclonal mouse antirabbit CD34 (1:100 dilution) (Santa Cruz Biotechnology, California, US). The immunostained sections were then counterstained with hematoxylin-eosin. The proliferation index was defined as the percentage of Ki-67 positive hepatocytes per total hepatocytes in the field of view. Ki-67 positive hepatocytes were counted in five fields of view per section (at ×40 magnification). The percentage of area positive for RAM11 and CD34 area were determined in five fields of view per section (at ×40 magnification).
Statistical analysis
Continuous variables were expressed as the mean ± standard deviation and compared using Mann–Whitney U tests. A p-value of <0.05 was considered statistically significant. All statistical analyses were performed using JMP® version 12 (SAS Institute, Cary, NC, USA).
Results
Comparison of the degree of nonembolized liver volume hypertrophy between the PVE and G-CSF groups
In the PVE group, the median %CLV pre-PVE was 18.1% (16.5%–21.7%) and the median %CLV post-PVE was 33.8% (28.7%–36.4%). In the G-CSF group, the median %CLV pre-PVE was 17.2% (13.2%–28.7%) and the median %CLV post-PVE (median) was 36.0% (30.9%–47.6%). The median DLV in the G-CSF group (18.8%) was significantly greater than that of the PVE group (14.7%) (p = 0.042) (Table 1).
|
G-CSF group (n=4) |
PVE group (n=4) |
P value |
|
|
%CLV pre-PVE (median) |
13.2-28.7 (17.2) |
16.5-21.7 (18.1) |
0.042 |
|
%CLV post-PVE (median) |
30.9-47.6 (36.0) |
28.7-36.4 (33.8) |
|
|
DLV (median) |
17.7-18.9 (18.78) |
10.5-18.5 (14.7) |
The percentage of preoperative caudal nonembolized liver lobe (%CLVpre-PVE)
= preoperative caudal nonembolized liver lobe volume (CLVpre-PVE) / preoperative total liver lobe volume (TLVpre-PVE)×100%
The percentage of postoperative caudal nonembolized liver lobe (%CLVpost-PVE)
=postoperative caudal nonembolized liver lobe volume (CLVpost-PVE) / postoperative total liver lobe volume (TLVpost-PVE)×100%
The degree of nonembolized liver volume hypertrophy (DLV) = (%CLVpost-PVE) - (%CLVpre-PVE)
Table 1: The comparison of the degree of nonembolized liver volume hypertrophy between PVE group and G-CSF group in a rabbit model
Comparison of Ki67 expression between the PVE and G-CSF groups
The Ki67 expression of the nonembolized livers in the G-CSF group was significantly greater than that of the nonembolized livers of the rabbits in the PVE group (p < 0.001). The Ki67 expression of the nonembolized livers in the G-CSF group was also significantly greater than that of the embolized livers in both the G-CSF and the PVE groups (p < 0.001) (Figure 2).
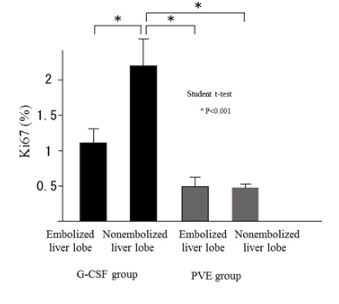
Figure 2: The comparison of the Ki 67 expression between PVE group and G-CSF group in a rabbit model. The Ki67 expression of nonembolized liver in G-CSF group was significantly more than that of nonembolized liver in PVE group (p<0.001). And that of nonembolized liver in G-CSF group was significantly more than that of embolized liver in G-CSF group and in PVE group (p<0.001)
Comparison of RAM11 expression between the PVE and G-CSF groups
The total liver RAM11 expression in the G-CSF group was significantly greater than that in the PVE group (p = 0.0035) (Figure 3). Furthermore, the RAM11 expression in the nonembolized livers of the rabbits in the G-CSF group was significantly greater than that in the nonembolized livers of the rabbits in the PVE group (p = 0.003) (Figure 4).
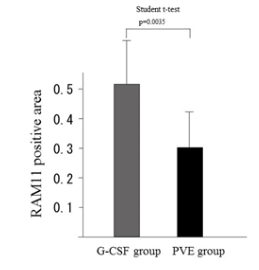
Figure 3: Comparison of the RAM 11 expression between PVE group and G-CSF group in a rabbit model.
The RAM 11 expression of total liver in G-CSF group was significantly more than that of total liver in PVE group (P=0.0035)
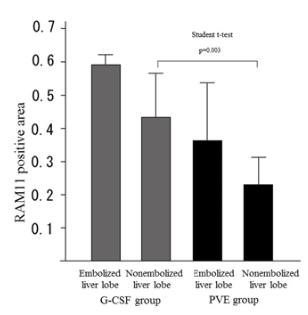
Figure 4: Comparison of the RAM 11 expression between PVE group and G-CSF group in a rabbit model.
The RAM 11 expression of nonembolized liver in G-CSF group was significantly more than that of nonembolized liver in PVE group (P=0.003)
Comparison of CD34 expression between the PVE and G-CSF groups
There was no significant difference between the two groups in the CD34 expression of the nonembolized liver (p = 0.068) (Figure 5).
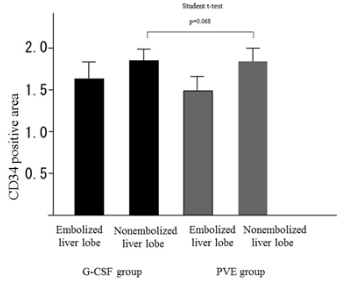
Figure 5: Comparison of the CD34 expression between PVE group and G-CSF group in a rabbit model.
There is no significant difference between the CD34 expression of nonembolized liver in G-CSF group and that of nonembolized liver in PVE group (P=0.068)
Discussion
The present study found both the DLV and the amount of cell proliferation to be significantly greater in the G-CSF group than in the PVE group in a rabbit model. Moreover, the total liver RAM11 expression in the G-CSF group was significantly higher than that in the PVE group. This suggests that the administration of G-CSF after PVE induces macrophages to produce growth cytokines in the liver, prompting the proliferation of liver cells. De Graaf et al. successfully devised a rabbit model for PVE that resembles the human clinical situation [7]. Rabbits provide a useful means of evaluating liver volume increase and liver function after PVE because they are amenable to the application of CT volumetry [8], the cranial liver lobes can be readily embolized, and the caudal lobe can be easily delineated and measured because of the clear separation between the caudal lobe and cranial lobes of the rabbit liver. Previous research suggests that PVE induces more liver regeneration than portal vein ligation in a standardized rabbit model and that permanent embolization materials such as polyvinyl alcohol particles result in significantly more hypertrophy than absorbable materials [9,10]. Therefore, in the present study, we performed PVE using polyvinyl alcohol particles in our rabbit model. The volume increase of the FRL weight is greatest in the first 7 days after PVE, with volume increases ranging from 33.6%-80% on day 7 in rabbit models [8]. In our study, the volume of the nonembolized lobe in the rabbits of our PVE group had increased by 42%-89% on day 7. Engelmann et al. found G-CSF to have no significant beneficial effects on patients with acute-on-chronic liver failure (ACLF) [11]. However, other studies have reported that G-CSF mobilizes hematopoietic bone marrow stem cells to aid liver regeneration and improve survival in patients with severe alcoholic hepatitis or liver failure [4-6,12,13]. Theocharis et al. found that G-CSF not only ameliorates histological evidence of liver injury in a statistically significant manner but also enhances the proliferative capacity of hepatocytes in an animal model [14]. Hence, the current study compared the degree of liver volume hypertrophy after PVE between a G-CSF-treated group and a non-G-CSF-treated group. Spahr et al. have suggested that the mechanism by which G-CSF administration induces liver hypertrophy is the mobilization of CD34+ cells. This increases hepatocyte growth factor and causes hepatic progenitor cells to proliferate [15]. Hepatic histology has shown that the use of G-CSF in patients with ACLF increases the recruitment of CD34+ cells [5]. Another study found that G-CSF administration increases neutrophil and CD34+ cell counts in the peripheral circulation of patients with hepatitis B-related ACLF [12]. We found that the CD34 expression of the nonembolized liver in the G-CSF group tended to be higher than that in the PVE group, but not significantly so. We also examined the expression of macrophages in liver tissue. Thomas et al. have shown that macrophage therapy for murine liver fibrosis recruits host effector cells, reducing fibrosis increasing regeneration, and improving function [16]. Macrophages support the cellular repair and regeneration of tissue. Both Kupffer cells and monocyte-derived macrophages are involved in liver regeneration [17] Kupffer cells, the resident macrophages of the liver, produce IL-6 and TNF-α which are important mediators of liver regeneration [18]. In the current study, the RAM11 expression of the total liver in the G-CSF group was significantly greater than that in the PVE group. The neutrophils mobilized by G-CSF promote macrophage production and these orchestrate the repair of the liver [19]. Our results suggest that the administration of G-CSF increases the number of macrophages in the liver and these macrophages produce growth cytokines such as IL-6 and TNF-α, which induce liver hypertrophy. Recently, it has been reported that associated liver partition and portal vein ligation for staged hepatectomy leads to acceleration of the increase in remnant liver volume but also has higher morbidity rates [20]. Other research suggests that the addition of hepatic vein embolization to PVE induces greater hypertrophy of the remnant liver with no greater risk than that from PVE alone [21]. There are currently two ongoing randomized controlled trials aimed at verifying this [21,22]. This study is the first to report on combined preoperative treatment with PVE and G-CSF administration as a means of promoting increased remnant liver regeneration following hepatic resection. However, the study had some limitations. Our research was conducted at a single institution with a small sample. An investigation with a larger sample size is needed to confirm our findings.
Conclusions
In a rabbit model, the DLV in a group administered G-CSF after PVE was significantly greater than that of a group that received PVE only. Our study suggests that the additional administration of G-CSF following PVE induces macrophages to produce growth cytokines in the liver, which prompts the proliferation of liver cells.
Acknowledgments
We would like to give special thanks to the Laboratory Animal Center of Osaka Metropolitan University Graduate School of Medicine for taking care of the rabbits used in this study.
Conflict of Interest Statement
The authors declare that they have no competing interests.
Funding
There is no funding sources.
Data Availability
No additional data are available.
Author Contributions
Nishio K designed the study and wrote the draft of the article. Kimura K and Ishizawa T contributed to interpretation of the data and the critical revision of the article content. All the other authors (Yamamoto A, Amano R, Ohira G, Kageyama K, Miura K, Kametani N) contributed the data collection and interpretation and critical reviewed the article. All the authors have read and agreed to the article.
References
- Zou RH, Li AH, Han F, et al. Liver hypertrophy and accelerated growth of implanted tumors in nonembolized liver of rabbit after left portal vein embolization. J Surg Res 178 (2012): 255-253.
- Treska V, Skalicky T, Sutnar A, et al. Prognostic importance of some clinical and therapeutic factors for the effect of portal vein embolization in patients with primarily inoperable colorectal liver metastases. Arch Med Sci 9 (2013): 47-54.
- Gianni AM, Siena S, Bregni M, et al. Granulocyte-macrophage colony-stimulating factor to harvest circulating haemopoietic stem cells for autotransplantation. Lancet 2 (1989): 580-585.
- Yang Q, Yang Y, Shi Y, et al. Effects of Granulocyte Colony-Stimulating Factor on Patients with Liver Failure: a Meta-Analysis. J Clin Transl Hepatol 4 (2016): 90-96.
- Garg V, Garg H, Khan A, et al. Granulocyte colony-stimulating factor mobilizes CD34(+) cells and improves survival of patients with acute-on-chronic liver failure. Gastroenterology 142 (2012): 505-512.
- Saha BK, Mahtab MA, Akbar SMF, et al. Therapeutic implications of granulocyte colony stimulating factor in patients with acute-on-chronic liver failure: increased survival and containment of liver damage. Hepatol Int 2017;11:540-546.
- De Graaf W, Van den Esschert JW, Van Lienden KP, et al. A rabbit model for selective portal vein embolization. J Surg Res 171 (2011): 486-494.
- Huisman F, Van Lienden KP, Damude S, et al. A review of animal models for portal vein embolization. J Surg Res 191 (2014): 179-188.
- Van den Esschert JW, Van Lienden KP, De Graaf W, et al. Portal vein embolization induces more liver regeneration than portal vein ligation in a standardized rabbit model. Surgery 149 (2011): 378-385.
- Van den Esschert JW, Van Lienden KP, Alles LK, et al. Liver regeneration after portal vein embolization using absorbable and permanent embolization materials in a rabbit model. Ann Surg 255 (2012): 311-318.
- Engelmann C, Herber A, Franke A, et al. Granulocyte-colony stimulating factor (G-CSF) to treat acute-on-chronic liver failure: A multicenter randomized trial (GRAFT study). J Hepatol 75 (2021): 1346-1354.
- Duan XZ, Liu FF, Tong JJ, et al. Granulocyte-colony stimulating factor therapy improves survival in patients with hepatitis B virus-associated acute-on-chronic liver failure. World J Gastroenterol 19 (2013): 1104-1110.
- Singh V, Keisham A, Bhalla A, et al. Efficacy of Granulocyte Colony-Stimulating Factor and N-Acetylcysteine Therapies in Patients With Severe Alcoholic Hepatitis. Clin Gastroenterol Hepatol 16 (2018): 1650-1656.
- Theocharis SE, Papadimitriou LJ, Retsou ZP, et al. Granulocyte-colony stimulating factor administration ameliorates liver regeneration in animal model of fulminant hepatic failure and encephalopathy. Dig Dis Sci 48 (2003): 1797-1803.
- Spahr L, Lambert JF, Rubbia-Brandt L, et al. Granulocyte-colony stimulating factor induces proliferation of hepatic progenitors in alcoholic steatohepatitis: a randomized trial. Hepatology 48 (2008): 221-229.
- Thomas JA, Pope C, Wojtacha D, et al. Macrophage therapy for murine liver fibrosis recruits host effector cells improving fibrosis, regeneration, and function. Hepatology 53 (2011):2003-2015.
- Chazaud B. Macrophages: supportive cells for tissue repair and regeneration. Immunobiology 219 (2014): 172-178.
- Taub R. Liver regeneration: from myth to mechanism. Nat Rev Mol Cell Biol 5 (2004): 836-847.
- Yang W, Tao Y, Wu Y, et al. Neutrophils promote the development of reparative macrophages mediated by ROS to orchestrate liver repair. Nat Commun 10 (2019): 1076.
- Chan KS, Low JK, Shelat VG. Associated liver partition and portal vein ligation for staged hepatectomy: a review. Transl Gastroenterol Hepatol 5 (2020): 37.
- Heil J, Schadde E. Simultaneous portal and hepatic vein embolization before major liver resection. Langenbecks Arch Surg 406 (2021): 1295-1305.
- Chebaro A, Buc E, Durin T, et al. Liver Venous Deprivation or Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy?: A Retrospective Multicentric Study. Ann Surg 274 (2021): 874-880.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 