Maxillary Radicular Cyst Excision and Reconstruction with Concentrated Bone Marrow Aspirate Cells Mixed with Xenograft - Case Report with 7-Year Follow-up
Radoslaw Jadach1, Karolina Osypko2*
Dental Salon, Wroclaw, Poland
Platinum Clinic, Wroclaw, Poland
*Corresponding author: Karolina Osypko, Platinum Clinic, Wroclaw, Poland
Received: September 19, 2022; Accepted: October 06, 2022; Published: October 19, 2022
Article Information
Citation: Radosław Jadach, Karolina Osypko. Maxillary Radicular Cyst Excision and Reconstruction with Concentrated Bone Marrow Aspirate Cells Mixed with Xenograft - Case Report with 7-Year Follow-up. Journal of Surgery and Research. 5 (2022): 569-575.
View / Download Pdf Share at FacebookAbstract
This article presents the case of a patient with a large maxillary radicular cyst. After apicoectomy of involved teeth and excision of the cyst, the bone defect was reconstructed with biomaterial made of concentrated bone marrow aspirate mixed with xenograft. Complete surgical procedure has been described and presented as well as steps of aspirating the bone marrow from anterior iliac crest and subsequent processing of the aspirate. Authors also provide long term follow-up, which shows healing and remodeling of both soft and hard tissues. Results of this method are very promising although using bone marrow as a source of mesenchymal stem cells seems challenging in everyday practice. More research must be done to determine whether or not peripheral blood and PRF could be the accessible source of mesenchymal stem cells with competing clinical results.
Keywords
<p>Periapical cyst, BMA, Concentrated bone marrow aspirate cells, Xenograft, GBR, Apicoectomy, MSC, Case report</p>
Article Details
Introduction
Treating large bone defects present a challenge in choosing proper surgical technique and require an individual approach for the patient. In this case, concentrated bone marrow aspirate cells mixed with xenograft have been chosen as a way to incorporate autologous stem cells into the biomaterial and therefore enhance the process of healing the defect. Below authors present long term results of with intraoral photos and tomography screens up to 7 years post operation.
Materials and Methods
Case
A 23-year-old female patient has been referred to our clinic in 2012 with a suspicion of cyst or tumor in the front region of maxilla. CBCT unveiled an ovoid radiolucency with narrow opaque margin around apices of six teeth no. 11-25. Lesion was approximately 25 x 21 x 16 mm (Figures 1 and 2).
Palatal wall seemed to be completely destroyed by the lesion, while the nasal and atrial wall had less than 1 mm of thickness. Tooth 11 underwent apicoectomy about 2 years prior, although the patient has not attended follow- up appointments, due to moving to another city. All teeth within the lesion were vivid, except for tooth 11, which was the cause of the lesion. Features such as size of the lesion and crossing the midline enforced the decision of combined surgical and endodontic approach instead of solely endodontic treatment. According to the rule excision & reconstruction, initial treatment plan assumed: 1.endodontic treatment of all teeth with apices within the lesion; 2. excision of the whole lesion with histopathological examination; 3. resection of the apices; 4.reconstruction of the bone cavity with xenograft mixed with stem cells obtained from iliac bone marrow.
Pre-surgery treatment
Referring dentist has been asked to stabilize using a composite wire splint all the teeth which apices were located within the lumen of the lesion, although by mistake teeth 15-23 have been wired and in consequence endodontically treated. Fortunately this overtreatment was discovered before the beginning of surgical intervention and remaining teeth 24-25 were endodontically treated. Canals were filled with MTA at least a few millimeters coronally from the level of apicoectomy, which is the floor of the defect, so that after cutting off the apices canals still will be properly closed with MTA. Fixation of the teeth is needed in this type of cases as it limits mobility of the teeth and helps keeping the xenograft still, therefore lowers the risk of improper healing.
Excision
The surgical procedure was performed in analgosedation with antibiotic i.v. Cefuroxime 1,5g. Due to better visibility of the operating field the buccal approach has been chosen. Wide, full thickness mucoperiosteal flap was designed with sulcular incision by teeth 13-25 and one vertical releasing incision distally from the tooth 25. After elevating the flap, an osteotomy window to the lesion’s cavity has been opened with a piezotome (Figure 3).
Cavity’s lining was gently separated from the fine bone walls and periosteum of palatal flap (Figures 4 and 5). The surgeon's finger pressing from the palatal side was a security measure preventing flap perforation. All collected material was preserved in 10% formalin and underwent histopathological examination, which later revealed periapical cyst. All apices within the lesion were cut with a fine diamond bur at the level of the cavity's floor. No backfilling of the canals was needed as they all were already filled with MTA.
Harvesting Bone Marrow Aspirate
As for the reconstruction part, the bone cavity was filled with xenograft derived from bovine bone (InterOss, SigmaGraft Biomaterials) mixed with centrifuged aspirate received from iliac bone marrow (BioCUE® BMA Bone Marrow Aspirate, Zimmer Biomet) (Figure 6). Step-by-step guidelines provided by the producer lead through preparing the patient, rinsing and preparing syringes and needles, positioning the needle at the anterior iliac crest, advancing and aspirating the bone marrow to 30 ml syringes (Figure 7). Afterwards, the BioCUE BMA Tube is filled with apirate (Figure 8) and placed in the centrifuge. After 15 minutes
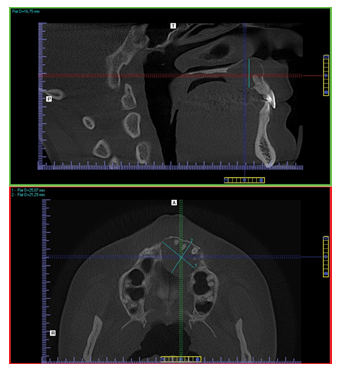
Figure 1: Dimentions of the cyst.
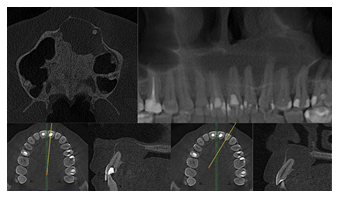
Figure 2: CBCT scans of the cyst.
of centrifugation at a speed of 3200 RPM aspirate should be separated in three distinct layers. From one port of the tube it is possible to collect plasma and from another cBMA cells (concentrated Bone Marrow Aspirate cells) (Figure 9). Mixing these two with xenograft results in material filled with autogenous cells. The skin wound above the donor site did not require suturing or any other type of closure.
Just before filling the cavity with the material, an acrylic plate was placed on the palate, hence material could be perfectly adjusted to the shape of the palate, symmetric to the opposite side. The bone cavity was filled with prepared material, gently compacted. Collagen membrane was placed on the top of the bone window, although nowadays authors do not see the necessity for placing the membrane. Wound was closed with nylon 5-0 sutures to obtain primary closure of the wound (Figure 10).
After the procedure, the patient was instructed that she could not take out the acrylic plate for 14 days, except for brief 1-minute moments for cleaning purposes. The plate’s role was to prevent touching and moving material by the tongue, as the bone from the palatal site was destroyed in about 95% and only soft tissues kept the material in place. Any mobility of the graft could compromise vascularisation and overall survival rating. Patient was prescribed Amoxicillin + clavulanic acid 875 + 125 mg (every 8 hours for 9 days) + probiotic, nimesulide 100 mg and ibuprofenum 400 mg per 8 hours in case of pain.
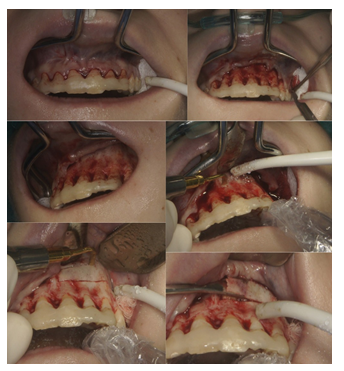
Figure 3: Mucoperiosteal flap elevation and opening a bone window with piezotome.
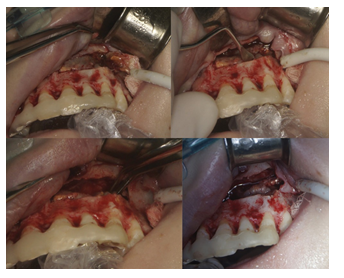
Figure 4: Removing the cyst.
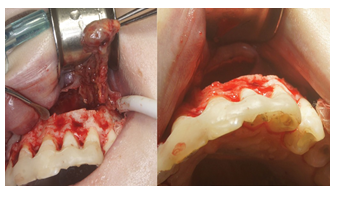
Figure 5: Removing the cyst.
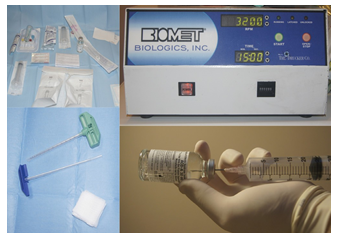
Figure 6: Preparing the BioCUE BMA set.
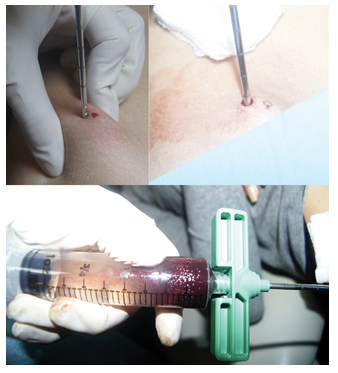
Figure 7: Aspirating.
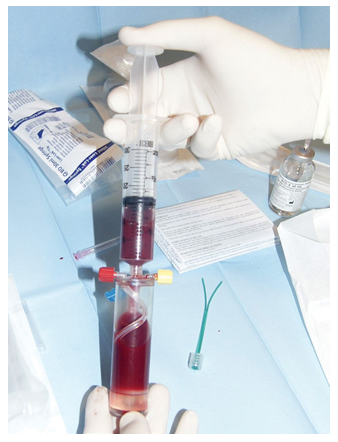
Figure 8: Filling the BMA Tube.
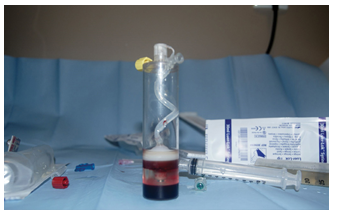
Figure 9: Centifuged tube after collecting plasma and cBMA cells.
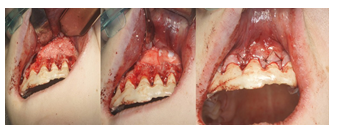
Figure 10: Placing the biomaterial, the membrane and suturing (nylon 5-0).
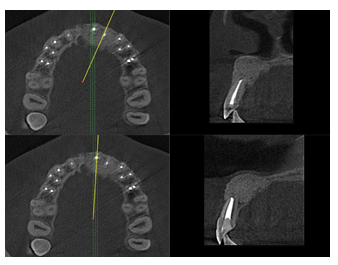
Figure 11: CBCT 12 months post op.
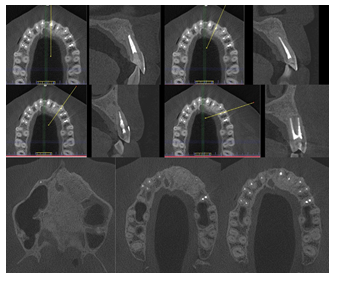
Figure 12: CBCT 4 years post op.
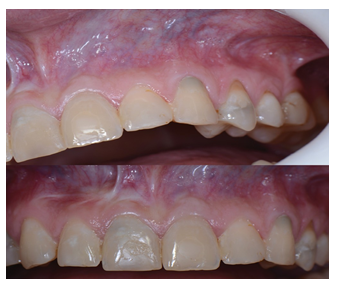
Figure 13: Soft tissues 4 years post op.
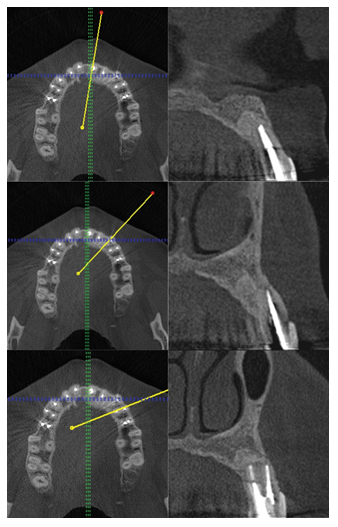
Figure 14: CBCT 7 years post op.
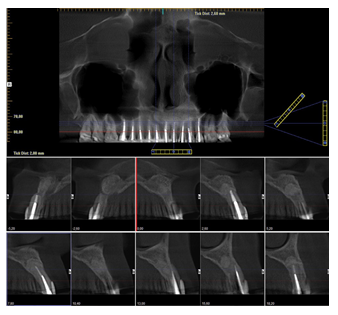
Figure 15: CBCT with an ortopantomographic view 7 years post op.
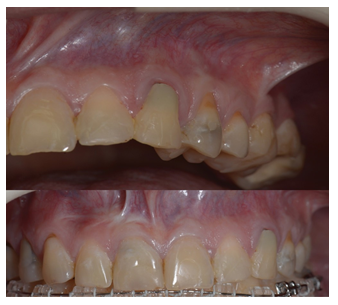
Figure 16: Soft tissues 7 years post op.
Results
Post procedure CBCT showed radiopaque material, cut apices and an osteotomy window. Patient attended follow-up appointments 7 days, 14 days, 3, 6, 12 months, 2, 4 and 7 years post op. Beneath authors present intraoral photos of soft tissues healing and CBCT preview after 12 months (Figure 11), 4 years (Figures 12,13) and 7 years post-op (Figures 14-16). Intraoral photos show no scars nor interdental papilla loss after the conducted procedure due to sulcular incision which left the whole superficial vasculature intact. Only visible scar is the one remaining after the apicoectomy of tooth 11.
Discussion
Following the flow of the treatment, one of the first questions that may arise is whether it is truly obligatory to treat all of the teeth within the cyst endodontically, even if they are perfectly vivid? While removing the cyst surgeon will presumably damage the neurovascular bundle of vivid teeth, hence they will require endodontic treatment nevertheless. Although it is possible to keep the adjacent teeth vivid as presented by Elhakim, [1] in that study only PRF was utilized to fill the bone cavity. Here in the present case, placing biomaterial into the defect created a greater risk of bacterial infection from the non-treated teeth and subsequently superinfection of the whole biomaterial.
Another aspect is filling the cavity with biomaterial if the diagnosis may be malignant. During honest conversation with the patient it was explained that untill the moment of histopathological results it is impossible to make a certain diagnosis. Nevertheless, the lesion needs to be excised and this is the perfect moment for bone augmentation. After
receiving the histopathological results we have two options. If it occurs to be neoplasmatic process, maxillectomy and removing all of the material with a margin of healthy tissues will be required. In this case the good news is that we detected the disease before it was too late in order to save the patient’s life. Bad news is that all the effort, especially financial effort, might be worthless. On the other hand, if the lesion turns out to be benign, the treatment is already finished. Patient does not require any other surgery and in case of losing any tooth, there are already suitable conditions for implantation.
If the patient has finansal limitations and could not afford xenograft with BMAC, the bone defect may be as well filled with PRF [2].
Authors advise against backfilling the canals after resection of the apices, as it extends the duration of the surgical procedure. Prolonged wound opening results in lesser healing outcomes [3] which may be avoided by having the canals already filled with MTA. Moreover, using piezotome for cutting the apex is not recommended, as micro vibrations of the tip may cause cracking within the MTA and therefore leakiness.
Surgical access issue
Authors consider palatal access as an alternative while buccal one is considered default and recommended. Palatal access may be deployed if:
- patient opens their mouth widely;
- lesion involve no more than one tooth;
- there is bone at the CEJ level from the palatal
The last point is probably the most important, as this tiny amount of bone determines whether we will result in good flap adherence and no bacterial leakage between the biomaterial and oral cavity.
Type of the material
Autogenous bone is considered the gold standard for its unique osteogenic properties. Unfortunately, acquiring such an amount of bone to fill the whole bone cavity would be problematic. Moreover, using solely xenograft in this size of defect might result in initial insufficient nutrition and oxygen supply. Another aspect is poor osteoblast reservoir as there is almost no cancellated bone adjacent to the defect. These issues pushed into creating material that will contain autologous stem cells - MSCs (Mesenchymal Stem Cells) and could be easily acquired from bone marrow. According to the producer of BMA kit, from the 50 ml of the bone marrow aspirate could be collected 6 ml of concentrated bone marrow aspirate cells and 25-35 ml of plasma. Nucleated Cell Recovery is 78,7% [4]
Bone Marrow Aspirate in dentistry may be applied in particularly difficult cases. Study by Sauerbier et al. showed similar results between cases employing autogenous bone mixed with xenograft (bovine bone mineral) and cases with bone marrow aspirate concentrate with xenograft [5]. Similar findings showed Sanz et al. as combining cells from BMA with biomaterial scaffolds gave comparable results as autogenous grafts [6]. Limiting the need for harvesting autogenous bone reduces morbidity, makes surgical procedures not only faster but also more effective due to osteogenic properties [7]. BMAC (Bone Marrow Aspirate Concentrate) positively influenced the amount of newly-formed bone in Bio-Oss® [8]. Moreover, that study proved that leucocyte and platelet- rich fibrin (L-PRF) and Emdogain® (Straumann) do not show similar properties as BMAC [8]. Histomorphometric analysis stated that BMAC led to higher amounts of vital mineralized tissue, higher level of Bio-Oss resorption, although no statistical difference of non mineralized tissue [9]. When applied to fresh extraction sockets, BMAC caused higher levels of mineralised tissue, higher osteocalcin expression and low expression of RUNX-2 (factor connected with osteoblast differentiation), while in control group RUNX- 2 was absent [10]. Despite these promising merits, initially there is higher volume loss in cases treated with BMAC, suggesting faster remodeling process. Nevertheless, in the final volume CBCT showed no significant differences in volume [11]. Study by Lavareda Corrêa et al. noted that none of the typical complications associated with allografts, such as late exposure, infections, and partial or total graft loss has occurred, although follow-up period was only 6 months [11]. On the other hand, a study by Wildburger et al. showed no significant difference between pure bovine bone material and the same material mixed with BMAC in sinus lift cases [12]. Similarly, presence of BMAC did not have such a explicit impact on new bone formation in sinus lift cases by Ting et al [13]. As a main advantage of BMAC is the presence of mesenchymal stem cells, which could be as well found in blood or other tissues [14] the question arises whether it is really necessary to use BMAC instead of much simpler PRF from peripheral blood. PRF with presence of MSC, slow cytokines release, leukocyte activation [15,16] affects vascularisation, tissue remodeling and healing [17] and could has similar effect as BMAC, although such hipotesis needs to proven or rejected with more advanced research. Supposing the acquired results are similar in both techniques, there would be no need for fairly complicated and challenging acquiring cells from the bone marrow.
Conclusion - take away lessons
Most important factors that influence the final result of treating radicular cyst:
- Leak tightness of the canals after resection of the apices
- Stabilization of the remaining tooth
- No movement from the palatal site, thanks to the acrylic plate - in regard of cases with palatal wall of the cyst destroyed
- Antibiotic therapy before and after the surgery
- Proper, gentle compacting the biomaterial within all recesses of the defect.
- Osteogenic properties of biomaterial - crucial in cases of massive cysts, such as the one
- As MSCs are present in both peripheral blood and bone marrow, more research needs to be conducted to compare results of using CBMA and
Acknowledgements
We would like to thank our patient, as she agreed for whole photographic documentation of the conducted procedures, publishing them in a scientific journal and that she regularly attended follow-up appointments.
Conflicts of interest
The authors declare no conflicts of interest.
Declarations
The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article.
References
- Elhakim A, Kim S, Kim E, et al. Preserving the vitality of teeth adjacent to a large radicular cyst in periapical microsurgery: a case report with 4-year follow-up. BMC Oral Health 21 (2021):
- Manjushree R and Prasad K. Application of cone-beam computed tomography in the management of dilacerated maxillary central incisor associated with radicular cyst and external root resorption - A case report. J. Conserv. Dent 24 (2021): 399-403.
- Waldorf H and Fewkes Wound healing. Adv. Dermatol 10 (1995): 77-96.
- Dregalla RC, Herrera JA, Koldewyn LS, et The choice of anticoagulant influences the characteristics of bone marrow aspirate concentrate and mesenchymal stem cell bioactivity in vitro. Stem Cells Int. 11 (2022): 8259888.
- Sauerbier S. Bone marrow concentrate and bovine bone mineral for sinus floor augmentation: a controlled, randomized, single-blinded clinical and histological trial-per-protocol analysis. Tissue Eng. Part A 17 (2011): 2187-2197.
- Sanz M. Biomaterials and regenerative technologies used in bone regeneration in the craniomaxillofacial region: Consensus report of group 2 of the 15th European Workshop on Periodontology on Bone Regeneration. J. Clin. Periodontol 46 (2019): 82-91.
- Melville JC, Tran HQ, Bhatti AK, et al. Is reconstruction of large mandibular defects using bioengineering materials effective?. J. Oral Maxillofac. Surg 78 (2020):
- Canellas JVDS, Drugos L, Ritto FG, et al Xenograft materials in maxillary sinus floor elevation surgery: a systematic review with network meta-analyses. Br. J. Oral Maxillofac. Surg 59 (2021): 742-751.
- Pasquali PJ, Teixeira ML, De Oliveira TA, et Maxillary Sinus Augmentation Combining Bio-Oss with the Bone Marrow Aspirate Concentrate: A Histomorphometric Study in Humans. Int. J. Biomater 11 (2015): 121286.
- Fontes Martins LC. Bone marrow aspirate concentrate and platelet-rich fibrin in fresh extraction sockets: A histomorphometric and immunohistochemical study in humans. J Craniomaxillofac Surg 49 (2021): 104-109.
- Lavareda CSC. Use of bone allograft with or without bone marrow aspirate concentrate in appositional reconstructions: A tomographic and histomorphometric study. Implant Dent 26 (2017): 915-921.
- Wildburger A, Payer M, Jakse N, et al. Impact of autogenous concentrated bone marrow aspirate on bone regeneration after sinus floor augmentation with a bovine bone substitute a split-mouth pilot study. Clin. Oral Implants Res 25 (2014): 1175-1181.
- Ting M, Afshar P, Adhami A, et al. Maxillary sinus augmentation using chairside bone marrow aspirate concentrates for implant site development: a systematic review of histomorphometric studies. Int. J. Implant Dent 4 (2018): 25.
- Ding DC, Shyu WC, Lin SZ. Mesenchymal stem cells. Cell Transplant 20 (2011): 5-14.
- Dohan DM. Platelet-rich fibrin (PRF): a second- generation platelet concentrate. Part II: platelet-related biologic features. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod 101 (2006): 45-50.
- Dohan Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part III: leucocyte activation: a new feature for platelet concentrates?. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod 101 (2006): e51-55.
- Choukroun Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part IV: clinical effects on tissue healing. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod 101 (2006): 56-60.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 