Metformin Intake Suppresses the Degree of Liver Metastasis From Colorectal Cancer in Diabetic Patients but Does not Improve their Prognosis
Akira Saito1, Joji Kitayama1, Hisanaga Horie1, Koji Koinuma1, Hideyuki Ohzawa2, Hironori Yamaguchi2, Hiroshi Kawahira1, Toshiki Mimura1, Alan Kawarai Lefor1, Naohiro Sata1
1Department of Gastrointestinal Surgery, Jichi Medical University, Yakushiji 3311-1, Shimotsuke, Tochigi, 329-0498 Japan
2Department of Clinical Oncology, Jichi Medical University, Yakushiji 3311-1, Shimotsuke, Tochigi, 329-0498 Japan
*Corresponding author: Joji Kitayama, MD, Department of Gastrointestinal Surgery, Jichi Medical University, Yakushiji 3311-1, Shimotsuke, Tochigi, 329-0498 Japan
Received: 15 October 2021; Accepted: 18 November 2021; Published: 24 November 2021
Article Information
Citation: Akira Saito, Joji Kitayama, Hisanaga Horie, Koji Koinuma, Hideyuki Ohzawa, Hironori Yamaguchi, Hiroshi Kawahira, Toshiki Mimura, Alan Kawarai Lefor, Naohiro Sata. Journal of Surgery and Research 4 (2021): 687-693.
View / Download Pdf Share at FacebookAbstract
Background
Metformin reduces the risk of, and mortality from, colorectal cancer in patients with diabetes mellitus. However, the effect of metformin on patients with stage IV disease is unknown. In the present study we reviewed the clinical features and outcomes of patients with diabetes mellitus and stage IV colorectal cancer (M1, liver metastases) treated with or without metformin.
Methods
The 202 patients with colorectal cancer and macroscopic liver metastasis who were treated in the Department of Surgery or Department of Clinical Oncology at Jichi Medical University Hospital from January 2006 through June 2019 were surveyed treatment of diabetes, clinical and pathological factor and prognosis of these patients.
Results
We retrospectively examined the effect of metformin use on outcomes in 32 patients with liver metastases from colorectal cancer. Hepatic metastases were stage H1 in 8/8 patients taking metformin and stage H2-3 in 17/24 non-users. Of 22 patients who underwent colectomy, colorectal tumors were pT4 in 5 metformin users, and pT2-3 in 10/17 non-users. The mean survival of metformin users and non-users was equal (28.0 mo vs 29.3 mo, p>0.05). No significant difference was detected when survival was compared between 6 metformin users and 19 non-users who received systemic chemotherapy.
Conclusion
These results suggest that metformin has less potent anti-tumor effects in patients with advanced stage disease. Metformin for the treatment of patients with metastatic colorectal cancer requires further study.
Keywords
<p>Metformin, Colorectal cancer, Liver metastasis, Stage IV, Chenotherapy</p>
Article Details
1. Background
Type 2 diabetes mellitus (DM) is well known to increase the risk for development of various cancers, including colorectal cancer (CRC) [1-3]. Metformin, an oral anti-hyperglycemic agent, has been shown to reduce the incidence of cancer development among patients with type 2 DM, although the degree of reduction varies among different types of cancer [4-6]. Many epidemiologic studies have suggested that that metformin may also improve the outcome of patients with CRC [6-10], although a large population?based study did not support a significant association between metformin and cancer-specific mortality [11]. There is clear evidence that outcome of the diabetic patients who underwent curative surgery with stage II and III CRC is associated with better survival in metformin- users [9, 12-15]. However, survival benefit is less clear in patients with stage IV CRC. In fact, a retrospective study focused on the patients who received chemotherapy for stage IV CRC has shown that tumor response, change in target lesion size as well as patient outcome were not significantly different between the metformin treated and the non-treated patients [16]. In this study, therefore, we examined the clinical features and outcome of diabetic patients with satge IV CRC with liver metastases who were treated with or without metformin.
2. Methods
From January 2006 until June 2019, 202 CRC patients with liver macroscopic metastases were treated in the Department of Surgery or Department Clinical Oncology at Jichi Medical University Hospital. Among them, 32 patients (16%) suffered type 2 DM and received medical treatment at diagnosis. In these patients, data for gender, age, medical history, treatment method, laboratory and pathological data and outcome were extracted from an electronic database. The stage of metastatic hepatic tumors was classified into the three categories with the number of liver nodules and the size of the largest metastasis: H1, ≤4 lesions and ≤5 cm; H2, ≥5 lesions or >5 cm; and H3,
≥5 lesions and >5 cm, according to criteria of the Japanese Society for Cancer of the Colon and Rectum [17]. This study was approved by the ethics committee of the Jichi University Hospital (approval no. clinic19- 190) and was conducted in accordance with the guiding principles of the Declaration of Helsinki. Statistical differences in clinical and pathological factors were evaluated with Fisher’s exact test. Overall survival (OS) was calculated using the Kaplan-Meier method and differences were evaluated using the log- rank test. In all tests, the standard for a signi?cant difference was set at p < 0.05.
3. Results
As shown in Table 1, 8 patients (25%) were treated with medications including metformin, 500mg~1000mg daily from 1 to 23 year (median=7 year), while other 24 patients were not exposed to metformin at diagnosis. No differences were detected in age , gender, tumor location and metastases in other sites between metformin-users and non-users. However, the degree of liver metastasis was all H1 stage in 8 metformin-users, while H2~3 stage in more than half (13/24) of non-usres ( p<0.018). Surgery was performed in 22 patients (5 metformin-users and 17 metformin non-users). Curative surgery with colectomy including hepatectomy was performed in 2 metformin-users and 5 non-users, and other patients underwent only colectomy. Pathological examination revealed that the depths of invasion of resected colorectal tumors were pT4 in all the 5 metformin users while only 7 in 17 tumors (41%) were pT4 and others remained at pT2-3 satge in metformin non-users (p=0.024). The overall survival (OS) of the patients was shown in Figure 1. Mean survival time (MST) of the 8 metformin users and 24 non-users was equivalent (28.0 mo vs 29.3 mo, P=0.76). Systemic chemotherapy was performed in 6/8 (75%) metformin users. FOLFOX and FOLFOX+bevasizumab (Bmab) was used for 4 and 2 pateins, respectively, with metformin continuation. In 24 metformin-non-usres, 19 patients
(79%) received chemotherapy, 5 with FOLFOX, 6 with FOLFOX+ Bmab, 1 with FOLFOX+Cetuximab (Cmab), 4 with FOLFIRI and 1 with SOX and 2 with capecitabibe. No significant difference was detected even when overall survival was evaluated in patients who received chemotherapy (MST:42.8 mo vs 29.3 mo, p=0.96).
|
Clinical / pathological factor |
Taking Metformin |
P-value |
|
|
Yes (n=8) |
No (n=24) |
||
|
Age |
67 (53-78) |
65 (34-79) |
0.48 |
|
Gender (Male/Female) |
5/3 |
17/7 |
0.68 |
|
Tumor site (Right/Left) |
2/6 |
9/15 |
0.68 |
|
Surgery (Curative/Palliative/No) |
2/3/3 |
5/12/7 |
0.88 |
|
Other Metastases |
|||
|
Lung (Yes/No) |
2/6 |
6/18 |
1 |
|
Peritoneum (Yes/No) |
1/7 |
2/22 |
1 |
|
H stage (1/2/3) # |
8/0/0 |
11/11/2 |
0.018 |
|
pT stage (2/3/4) # |
0/0/5 |
1/9/7 |
0.024 |
|
pN stage(1/2/3) # |
2/2/1 |
5/8/4 |
1 |
|
Chemotherapy (Yes/No) $ |
2/6 |
5/19 |
1 |
|
Serum HbA1c |
7.1 (5.6-9.6) |
6.6 (5.2-10.0) |
0.81 |
#: Calculated from patients who underwent colectomy; $: Chemotherapeutic regimens were FOLFOX in 4 and FOLFOX+Bevasizumab in 2 of metformin users, and FOLFOX in 5, FOLFOX+Bevasizumab in 6, FOLFOX+ Cetuximab in 2, FOLFIRI in 4, SOX in 1 and Capecitabine in 2 of metformin non-users.
Table 1: Clinical and pathological features of patients taking and not taking metformin
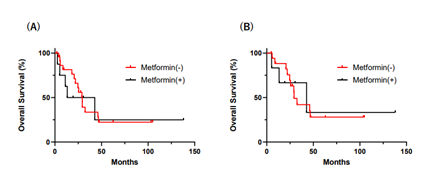
Figure 1: (A) Outcome of metformin users (n=8) or non-users (n=24) (B) Outcome of metformin users (n=6) or non- users (n=17) who received systemic chemotherapy Over all survivals (OS) were evaluated using the Kaplan-Meier method and p value was calculated with log-rank test.
4. Discussion
A growing body of evidence indicates that metformin have anti-tumor effects through various mechanisms including reduction of viability and proliferation of tumor cells, repression of epithelial-mesenchymal transition as well as modulation of tumor immuneenviromnet [18,19]. In this study, we found that grade of liver metastasis in CRC patients was limited in H1 stage even though pT stage of primary tumor was more advanced in all metformin-users. This is consistent with the results of previouos studies and supports the anti-metastatic effects of metformin. We previously reported that the number of nodal metastases was significantly lower in metformin- treated CRC patients with diabetes [15]. Taken together, these data susggest a possibility that metformin inhibits tumor metastasis not only at primary sites but also suppresses the growth of tumor cells in metastatic organs. Epidemiological studies have suggested that metformin not only reduces the risk of developing CRC but also improve the outcome of diabetic patients, especially in those who underwent
curative surgery with stage II and III CRC [6-10, 12- 15]. Interestingly, however, the impact of metformin intake on the outcome of the stage IV patients has not been clearly documented in previous literatures and and therapeutic effect of metformin in metastatic CRC still remains unclarified. In this study, we could not found apparent survival benefit of metformin intake in CRC patinets liver metastases, although H stage was less advanced in metformin-users. This is is consistent with a previous korean study [16], and suggest a possibility that anti-tumor effects of metformin is less prominent in advanced stage of CRC, although the sample sizes in both studies are not enough to draw a definite conclusion. In fact, a number of basic studies have shown metformin can sensitize tumor responses to different chemotherapeutic drugs through various molecular mechanisms [20]. Many clinical trials are currently active to examine the synergistic effect of metformin on chemotherapeutic agents for various cancers [21]. In CRC, metformin has been shown to enhance the effects of adjuvant chemotherapy [22,23]. In comparison, the effects of metformin on 5-Fu based
chemotherapy for refractory CRC was reported to be modest [24]. Extensive randomized control trials will be necessary to confirm the real therapeutic potential of metformin for stage IV CRC.
5. Conclusions
These results suggest that metformin has less potent anti-tumor effects in patients with advanced stage disease. Metformin for the treatment of patients with metastatic colorectal cancer requires further study.
List of abbreviations
DM: diabetes mellitus, CRC: colorectal cancer, OS: overall survival, MST: mean survival time
Declarations
Ethics approval and consent to participate
This study protocol was reviewed and approved by the Ethics Committee of the Jichi Medical University Hospital (approval no. clinicA20-110) and was conducted in accordance with the guiding principles of the declaration of Helsinki.
Consent for publication
Not applicable
Availability of data and materials
All data generated or analysed during this study are included in this published article.
Competing interests
The authors declare that they have no competing interests.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contributions
Joji Kitayama: Analyzed and interpreted the data. Akira Saito: Analyzed and interpreted the data and wrote the paper.
Hideyuki Ohzawa: Contributed analysis tools or data. Hironori Yamaguchi: Analyzed and interpreted the data.
Koji Koinuma, Hisanaga Horie, Hiroshi Kawahira, Toshiki Mimura, Alan Kawarai Lefor, Naohiro Sata: Contributed analysis tools or data.
Acknowledgement
We also thank Junko Shinohara, Hiromi Hatakeyama and Ikuko Nieda for technical and clerical work.
References
- Orgel E, Mittelman SD. The links between insulin resistance, diabetes, and cancer, Curr Diab Rep 13 (2013): 213-222.
- Peeters PJ, Bazelier MT, Leufkens HG, et al. The risk of colorectal cancer in patients with type 2 diabetes: associations with treatment stage and obesity, Diabetes Care 38 (2015): 495-502.
- Zhu B, Wu X, Wu B, et al. The relationship between diabetes and colorectal cancer prognosis: A meta-analysis based on the cohort studies, Plos One 12 (2017):
- Decensi A, Puntoni M, Goodwin P, et Metformin and cancer risk in diabetic patients: a systematic review and meta- analysis, Cancer Prev Res (Phila) 3 (2010): 1451-1461.
- Noto H, Goto A, Tsujimoto T, et Cancer risk in diabetic patients treated with metformin: a systematic review and meta- analysis. Plos One 7 (2012): e33411.
- Dulskas A, Patasius A, Linkeviciute- Ulinskiene D, et Metformin increases cancer specific survival in colorectal cancer patients-National cohort study, Cancer Epidemiol 62 (2019): 101587.
- Meng F, Song L, Wang W, et al. Metformin Improves Overall Survival of Colorectal Cancer Patients with Diabetes: A Meta- J Diabetes Res 20 (2017): 5063239.
- Ki YJ, Kim HJ, Kim MS, et al. Association between Metformin use and survival in nonmetastatic rectal cancer treated with a curative resection: A nationwide population Cancer Res Treat 49 (2017): 29-36.
- Du L, Wang M, Kang Y, et Prognostic role of metformin intake in diabetic patients with colorectal cancer: An updated qualitative evidence of cohort studies. Oncotarget 8 (2017): 26448-26459.
- Huang WK, Chang SH, Hsu HC, et Postdiagnostic metformin use and survival of patients with colorectal cancer: A Nationwide cohort study. Int J Cancer 147 (2020): 1904-1916.
- Mc Menamin UC, Murray LJ, Hughes CM, et Metformin use and survival after colorectal cancer: A population-based cohort study, Int J Cancer 138 (2016): 369-379.
- Garrett CR, Hassabo HM, Bhadkamkar NA, et al. Survival advantage observed with the use of metformin in patients with type II diabetes and colorectal cancer. Br J Cancer 106 (2012): 1374-1378.
- Lee JH, Kim TI, Jeon SM, et al. The effects of metformin on the survival of colorectal cancer patients with diabetes mellitus. Int J Cancer 131 (2012): 752-759.
- Fransgaard T, Thygesen LC, Gogenur Metformin Increases Overall Survival in Patients with Diabetes Undergoing Surgery for Colorectal Cancer. Ann Surg Oncol 23 (2016): 1569-1575.
- Saito A, Kitayama J, Horie H, et Metformin changes the immune microenvironment of colorectal cancer in patients with type 2 diabetes mellitus. Cancer Sci 111 (2020): 4012-4020.
- Lee DJ, Kim B, Lee JH, et al. The effect of metformin on responses to chemotherapy and survival in stage IV colorectal cancer with Korean J Gastroenterol 60 (2012): 355-361.
- Watanabe T, Itabashi M, Shimada Y, et al. Japanese Society for Cancer of the, Rectum, Japanese Society for Cancer of the Colon and Rectum Guidelines 2014 for treatment of colorectal cancer. Int J Clin Oncol 20 (2015): 207-239.
- Kamarudin MNA, Sarker MMR, Zhou JR, et Metformin in colorectal cancer: molecular mechanism, preclinical and clinical aspects. J Exp Clin Cancer Res 38 (2019): 491.
- Bahrambeigi S, Shafiei-Irannejad Immune-mediated anti-tumor effects of metformin; targeting metabolic reprogramming of T cells as a new possible mechanism for anti-cancer effects of metformin. Biochem Pharmacol 174 (2020): 113787.
- Tang Z, Tang N, Jiang S, et The Chemosensitizing Role of Metformin in Anti-Cancer Therapy. Anticancer Agents Med Chem 21 (2021): 949-962.
- Saraei P, Asadi I, Kakar MA, et The beneficial effects of metformin on cancer prevention and therapy: a comprehensive review of recent advances. Cancer Manag Res 11 (2019): 3295-3313.
- Singh PP, Shi Q, Foster NR, et Relationship between metformin use and recurrence and survival in patients with resected stage III colon cancer receiving adjuvant chemotherapy: Results from north central cancer treatment group N0147 (Alliance). Oncologist 21 (2016): 1509-1521.
- Coyle C, Cafferty FH, Vale C, et Metformin as an adjuvant treatment for cancer: a systematic review and meta- analysis. Ann Oncol 27 (2016): 2184-2195.
- Miranda VC, Braghiroli MI, Faria LD, et al. Phase 2 Trial of Metformin combined with 5- Fluorouracil in patients with refractory metastatic colorectal cancer. Clin Colorectal Cancer 15 (2016): 321-328.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 