Percutaneous Pericardial Drainage: A Reliable and Efficient First-Line Treatment in Case of Non-Iatrogenic Pericardial Effusion
Delon C1, 2, Bouisset F2, Porterie J3, Biendel C1, 2, Blanco S2, Roncalli J2, Grunenwald E3, Carrié D2, Elbaz M2, Galinier M1, 2, Lairez O2, 4, Delmas C1, 2*
1Intensive Cardiac Care Unit, Rangueil University Hospital, Toulouse, France
2Department of Cardiology, Rangueil University Hospital, Toulouse, France
3Department of Cardiovascular Surgery, Rangueil University Hospital, Toulouse, France
4Cardiac Imaging Center, Rangueil University Hospital, Toulouse, France
*Corresponding Author: Clément Delmas, Intensive Cardiac Care Unit, Department of Cardiology, Toulouse University Hospital 1, Avenue Jean Poulhès, TSA 50032, 31059 Toulouse Cedex 9, France
Received: 04 September 2020; Accepted: 14 September 2020; Published: 10 November 2020
Article Information
Citation: Delon C, Bouisset F, Porterie J, Biendel C, Blanco S, Roncalli J, Grunenwald E, Carrié D, Elbaz M, Galinier M, Lairez O, Delmas C. Percutaneous Pericardial Drainage: A Reliable and Efficient First-Line Treatment in Case of Non-Iatrogenic Pericardial Effusion. Journal of Surgery and Research 3 (2020): 393-406.
View / Download Pdf Share at FacebookAbstract
Background: Pericardial effusion can be a life-threatening condition and urgent drainage is then life-saving but no consensus exists as to the preferential use of a percutaneous drainage (PCD) or a surgical drainage (SD) approach.
Method and Results: All patients admitted for symptomatic pericardial effusion in 2018 in our tertiary Intensive Cardiac Care Unit (Toulouse, France) were prospectively included. In- hospital complications, length of stay, 6-months mortality and recurrences have been collected and compared according to the procedure (PCD vs. SD). One hundred and twenty nine patients were admitted for pericardial effusion and 92 were drained: 54 (59%) and 38 (41%) by a surgical and percutaneous procedure, respectively. In- hospital complications were the same between the 2 procedures (7% for SD group and 15% for PCD; p = 0.39). However, duration of hospitalization was shorter for patients treated by PCD (5.9±4.4 versus 8.2±4.9 days; p = 0.02). Six-month mortality (22% for SD versus 37% for PCD; p = 0.12) or recurrences (11% for PCD versus 24% for SD; p = 0.11) were not different.
Conclusion: In case of symptomatic non-iatrogenic pericardial effusions, PCD is an efficient and reliable less invasive technique. Its possible and wide availability, and its shorter length of hospitalization justifies its use as first line therapy in trained team especially for cancer patients.
Keywords
<p>Pericardial effusion; Pericardiocentesis; Pericardiotomy; Mortality; Recurrence</p>
Article Details
Abbreviations
ICCU: intensive cardiac care unit; PE: pericardial effusion; PCD: percutaneous drainage; SD: surgical drainage
1. Introduction
Pericardial effusion (PE) can be a life-threatening condition when fluid accumulation in the pericardial space cause intra-pericardial pressure increase, exceeding the limit of pericardial stretch. Diastolic cardiac chambers pressure equalizes intra-pericardial pressure, which results in reducing myocardial diastolic compliance and then limiting cardiac inflow. This pathophysiology defined the concept of cardiac tamponade, which requires urgent management to prevent cardiogenic shock and death [1, 2]. Treatment consists in fluid drainage by surgical drainage (SD) or percutaneous drainage (PCD). The first surgical sub-xyphoid approach was described in 1829 [3] by Larrey et al. whereas the first percutaneous pericardiocentesis series was published in 1986 by Kopecky et al [4]. Surgical drainage remains the gold standard [5, 6] but the optimal approach remains uncertain [7-10]. Etiologies of PE are various [9-11]: mains causes are secondary to acute idiopathic pericarditis (20%), iatrogenic effusions (16%), cancer (13%) [12, 13], and chronic idiopathic pericardial effusion (9%). Other causes are secondary to infectious and inflammatory diseases, metabolic disturbance (uremic, hypothyroidism), thoracic trauma, or type A aortic dissection.
To date European guidelines suggest considering PE drainage in case of cardiac tamponade, or for symptomatic moderate to large effusion not responsive to medical therapy, and for suspicion of unknown etiology (class I level C) [5]. Same guidelines propose to use a consensual not evidence based triage system to guide the timing of drainage (class IIb level C) but without differencing SD and PCD [5, 6]. The main purpose of our study was to compare one-month and six-month mortality and recurrences according to the type of initial drainage procedure (PCD or SD) in a large reference tertiary center. Secondary objectives were to compare initial complications and duration of hospitalization between both groups.
2. Methods
2.1 Study design
This was an observational, prospective, monocentric study conducted in our Intensive Cardiac Care Unit (ICCU) (Rangueil University Hospital, Toulouse, France) from January 1, 2018 to December 31, 2018.
2.2 Population
All consecutive patients admitted for symptomatic PE during this period were included. Patients affected by iatrogenic PE secondary to endovascular procedure (such as radiofrequency arrhythmia ablation, coronary angioplasty…) were excluded for the analysis as they were directly managed in the catheterization laboratory. Patients admitted for pericardial effusion but without indication to drainage have not been included in the final analysis. This investigation conforms to the principles outlined in the Declaration of Helsinki. Our study was observational and did not modify existing diagnostic or therapeutic strategies. In accordance with French and European law (2016/679 settlement of the European parliament), all patients gave their informed non opposition and there was no longer a need to declare to the National Data Protection Commission. Patients were classified into two groups according to the type of drainage (SD or PCD). Type of drainage was decided by the team in charge of the patient at admission, taking into account the severity of the clinical picture but also the morphology of the patient. SD was preferentially used in case of severely overweight patients, post-cardiotomy PE, localization incompatible with PCD (for example cloistered PE) and lack of available medical expertise for percutaneous approach at the time of drainage.
2.3 Collected data
Information concerning baseline demographic and clinical characteristics, medical background, etiology of PE, biological parameters at admission, biological analysis of effusion and characteristics of drainage were collected. A procedure was defined as urgent in the presence of hemodynamic and/or respiratory instability and/or echocardiographic signs of tamponade (compressive effect on cardiac chambers or swinging heart). ESC triage score [6] was retrospectively used to highlight the severity of the population. In-hospital complications were prospectively collected and are defined by: cardiac, hepatic or peritoneal cavity puncture, pneumothorax, hemo or pneumo-mediastinum, cardiac arrest, malignant rhythm disorders, scar infection and/or anesthesia complication. Duration of hospitalization was defined as the number of days spent in the hospital.
2.4 Follow-up
Recurrences and vital status were collected at 1 and 6 months by a phone interview with the patient, his relatives or general practitioner.
2.5 Procedural description
All PCD were performed under local anesthesia by sub-xyphoid approach with ultrasound guidance. The needle is usually inserted between the xyphoid process and the left costal margin directed to the patient’s left shoulder with a 15° angle with a slowly aspirating way, until the pericardium is pierced and fluid appears in the syringe. Then, a guide wire is passed in order to facilitate the introduction of a drainage catheter [2]. The drain is then left in place for several days until it gives less than 50ml/day. SD was performed under general anesthesia generally by sub-xyphoid approach but sometimes by left thoracotomy. A 5 cm long vertical sub-xyphoid incision was made permitting the dissection of sub-cutaneous tissue to the pericardium which was opened permitting evacuation of fluid and, eventually, creation of a permanent fenestration (pericardio-pleural or pericardio- peritoneal window). Finally, a drain is left in the pericardium, fixed to the abdominal wall [15] for several days until it gives less than 50-75ml/day.
2.6 Statistical analysis
Quantitative data are reported using means ± standard deviations (sd). Qualitative data are presented as percentages. Comparative analyses were performed using Chi² and Fisher test for categorical variables and t-Student test for numerical variables. Binary logistic regression was used to determine predictors of mortality for all patients. A p value < 0.05 was considered significant. All statistical analyses were performed with XLSTAT software.
3. Results
3.1 Study population
One hundred and twenty nine patients were admitted for PE in our ICCU during the one-year inclusion period. Among them, 37 (29%) patients were not included in the final analysis (23 patients with iatrogenic tamponade managed in the catheterization laboratory and 14 patients who did not undergo drainage). A total of 92 patients were included in the final analysis: 54 (59%) in the SD group and 38 (41%) in the PCD group (Figure 1). Table 1 presents the characteristics of patients included according to the type of initial drainage. They were predominantly male (57%) with a mean age of 62 ± 15 years (from 20 to 100 years). Groups were not different for age, gender, comorbidities or current medication/therapy. Previous cancer reported for almost half of the population in both groups. Clinical presentations were the same between groups with frequent dyspnea, right heart failure signs and hypotension. The diagnosis of tamponade was most of the time clinic but electrical signs were common (low-voltage complexes in 26%). Initial TTE evaluation found the same effusion size estimation between groups. Fifty-four percent of PE patients were initially managed in another center, and were transferred in our ICCU for drainage. Patients were more severe in the PCD group according to the 2014 ESC triage score (8.7 (+/-2.4) vs 7.5 (+/- 2.7) for PCD and SD respectively; p = 0.029). The main PE etiology was cancer (n=44; 48%) especially lung (n=20; 22%) and breast cancer (n=8; 9%); and post-cardiotomy effusion were not infrequent concerning 7.2% of our population.
3.2 Procedural and effusion characteristics
The mean volume of effusion collected was 914 ± 641 ml for both techniques used for drainage. PCD was more frequently made in emergency (40 vs 20% for SD group; p = 0.045). Biological analysis found no significant difference between groups in terms of blood cultures, viral and bacterial serologies but neoplastic cells were more frequently found in the PCD group (47 vs 26%; p = 0.03) (Table 2). Whereas 43 effusions were related to neoplastic origin, neoplastic cells were found at the cytological analysis for only 32 (74%) patients. For 11 (26%) patients, the neoplastic origin was based on the context of evolutive cancer or neoplastic cells found in an associated pleural effusion. An adenocarcinoma TTF1+ was confirmed for 12 (28%) with neoplastic PE by cytological analysis. Neoplastic PEs were more frequently managed by PCD (61 for PCD vs 37% for SD group; p = 0.026) whereas SD was preferred for post-cardiotomy effusion (13% for SD vs 0% for PCD group; p = 0.037) (Table 2). An associated pleural drainage was frequent and more frequently made in case of SD (44 for SD vs 21% for PCD group; p = 0.02) (Table 1).
3.3 Surgical procedure
Sub-xyphoid approach was mainly used for SD (n= 25, 46%). Others approaches were left antero- lateral mini-thoracotomy (n=19, 35%), full-sternotomy (n=2; 4%), anterior thoracotomy (n=1; 1.9%) and left postero-lateral mini-thoracotomy (n=1; 2%). Fenestrations were frequently realized (n=29; 54%): 17 (59%) between pleura and pericardium spaces, and 12 (41%) between peritoneum and pericardium. One-month and 6-month recurrence were not different according to the presence of fenestration in case of SD (3 vs 3; p=1).
3.4 Complications
Seven patients initially managed by PCD (18%) required a conversion to SD due to the failure of the percutaneous procedure (n=3), right ventricular perforation (n=1) or persistent significant PE (n=3). One patient was in extremely hemodynamic instability at admission and presented a cardiac arrest before any drainage. There was a trends of more complications in the PCD group without reaching significance (7 in the SD group vs 15% in the PCD group; p = 0.39) (Table 3).
3.5 Outcomes according to the type of initial drainage
One-month and 6-months mortalities were not different between PCD and SD despite a trend to higher 6-months mortality in the PCD group as compared with the SD group (37 vs. 22%, respectively; p = 0.12) (Table 3). Neoplastic etiology was reported as the cause of deaths (n=22; 85%) for most of the patients, regardless of the group (79 vs 83% in PCD and SD groups, respectively; p = 1). Failure of PCD requiring SD, was associated with a trend to higher 6-month mortality (71 vs 29%; p = 0.08) compare to initial PCD success. Failure of a first SD needing a second surgical approach, was associated with a significant higher 6-months mortality (71 vs 22%; p = 0.01) compare to first SD success. No difference between groups was found in terms of 1-month or 6-months recurrences (Table 3). Neoplastic etiology reported for almost half of the recurrence (67 vs 17% in PCD and SD group, respectively, p = 0.06). In hospital length of stay was higher in the SD group compared to the PCD (8.2 vs 5.9 days; p = 0.02) (Table 3).
3.6 Factors associated with 6-months outcomes
In univariate analysis, a neoplastic etiology was highly associated with 6-months mortality (OR 11.8; CI 95% 3.6-38.5) as well as the presence of neoplastic cells in the cytological analysis of the PE (OR 6.4; CI 95% 2.4-17.3) (Table 4). Patients with adenocarcinoma TTF1+ PE had a higher mortality (58 vs 24% of death among patients with and without adenocarcinoma, respectively; p = 0.03). In univariate analysis, adenocarcinoma TTF1+ was significantly associated with 6-months mortality (OR 4.5; CI 95% 1.28-15.8). Interestingly previous medication (anticoagulant or antiplatelets), comorbidities, type of procedure, the emergency of the drainage or a recurrence were not associated with 6-months mortality. A previous pericardial effusion was associated with a higher risk of recurrence (OR 3.86; CI 95% 1.07-13.9) (Table 5).
|
All patients n= 92 |
Surgical drainage n= 54 (58.7%) |
Percutaneous drainage n = 38 (41.3%) |
p value |
|
|
Age (years) |
62 (15) |
60 (14) |
65 (15) |
0.13 |
|
Male, n (%) |
52 (57%) |
31 (57%) |
21 (55.3 %) |
0.84 |
|
BMI (Kg/m2) |
25.7 (6.2) |
26.3 (6.3) |
24.9 (5.9) |
0.33 |
|
Medical background, n (%) |
||||
|
Previous pericardial effusion |
16 (17.4 %) |
15 (27.8 %) |
1 (2.6 %) |
0.002 |
|
Previous heart disease |
29 (31.5 %) |
16 (29.6 %) |
13 (34.2 %) |
0.65 |
|
Previous cancer |
44 (47.8 %) |
23 (43.6 %) |
21 (55.3 %) |
0.23 |
|
Lung cancer |
20 (21.7 %) |
10 (18.5 %) |
10 (26.3 %) |
0.37 |
|
Breast cancer |
8 (8.7 %) |
4 (7.4 %) |
4 (10.5 %) |
0.7 |
|
Diagnosis of cancer with pericardial effusion, n (%) |
10 (10.9 %) |
4 (7.4 %) |
6 (15.8 %) |
0.31 |
|
Chronic renal failure (GFR < 50 ml/mn) |
11 (11.9 %) |
7 (13 %) |
4 (10.5 %) |
1 |
|
Obesity |
18 (20.7 %) |
15 (28.8 %) |
3 (8.1 %) |
0.017 |
|
Diabetes |
19 (20.9 %) |
12 (22.6 %) |
7 (18.9 %) |
0.7 |
|
High blood pressure |
40 (43.9 %) |
24 (45.3 %) |
16 (43.2 %) |
0.91 |
|
Current treatment, n (%) |
||||
|
Chemotherapy |
18 (19.6 %) |
9 (16.7 %) |
9 (23.7 %) |
0.4 |
|
Antiplatelet or anticoagulant |
56 (60.9 %) |
32 (59.3 %) |
24 (63.2 %) |
0.71 |
|
Urgent, n (%) ESC triage score ≥ 6 |
26 (28.3 %) 72 (78.3 %) |
11 (20.4 %) 38 (70.4 %) |
15 (39.5 %) 34 (89.5 %) |
0.045 0.039 |
Numerical variables: mean (standard deviation); categorical variables: number of patients (%) LVEF: Left Ventricular Ejection Fraction, BMI: Body Mass Index, GFR: Glomerular Filtration Rate
Table 1: Comparison of baseline characteristics according to the initial type of drainage.
|
All patients n= 92 |
Surgical drainage n= 54 (58.7%) |
Percutaneous drainage n = 38 (41.3%) |
p value |
|
|
Etiology, n (%) |
||||
|
Acute idiopathic pericarditis |
11 (11.9 %) |
9 (16.7 %) |
2 (5.3 %) |
0.12 |
|
Cancer |
43 (46.7 %) |
20 (37 %) |
23 (60.5 %) |
0.026 |
|
Infectious |
10 (10.9 %) |
4 (7.4 %) |
6 (15.8 %) |
0.31 |
|
Post-operative of cardiac surgery |
7 (7.6 %) |
7 (13 %) |
0 (0 %) |
0.037 |
|
Auto-immune disease |
2 (2.2 %) |
2 (3.6 %) |
0 (0 %) |
0.51 |
|
Unknown |
14 (15.2 %) |
8 (14.8 %) |
6 (15.8 %) |
1 |
|
Duration of drainage (days) |
2.7 (1.33) |
2.8 (1.3) |
2.5 (1.27) |
0.2 |
|
Pleural drainage, n (%) |
32 (34.8 %) |
24 (44.4 %) |
8 (21 %) |
0.02 |
|
Macroscopic aspect, n (%) |
||||
|
Hematic |
38 (41.3 %) |
19 (35.2 %) |
19 (50 %) |
0.15 |
|
Sero-hematic |
25 (27.2 %) |
14 (25.9 %) |
11 (28.9 %) |
0.74 |
|
Clear |
25 (27.2 %) |
19 (38.2 %) |
6 (15.8 %) |
0.04 |
|
Purulent |
2 (2.2 %) |
1 (1.8 %) |
1 (2.6 %) |
1 |
|
Clot |
2 (2.2 %) |
1 (1.8 %) |
1 (2.6 %) |
1 |
|
Biological analyses, n (%) |
||||
|
Bacteria a |
3 (3.3 %) |
1 (1.8 %) |
1 (2.6 %) |
1 |
|
Virus b |
17 (18.5 %) |
10 (18.5 %) |
7 (18.4 %) |
0.99 |
|
Neoplasic cells c |
32 (34.8 %) |
14 (25.9 %) |
18 (47.4 %) |
0.03 |
- Serratia Marcescens and Haemophilus parainfluenzae
- Ebstein Barr Virus by PCR analysis
- For 11 neoplastic effusions (25.6 %), the cytological analysis could not prove the malignant Numerical variables: mean (standard deviation); Categorical variables: number of patients (%)
Table 2: Comparison of pericardial effusion according to the initial type of drainage.
|
Surgical drainage n= 54 |
Percutaneous drainage n = 38 |
p value |
|
|
Mortality, n (%) |
|||
|
One month |
4 (7.4 %) |
3 (7.9 %) |
1 |
|
Six months |
12 (22.2 %) |
14 (36.9 %) |
0.12 |
|
Recurrences, n (%) |
|||
|
One month |
4 (7.4 %) |
4 (10.5 %) |
0.71 |
|
Six months |
6 (11.1 %) |
9 (23.7 %) |
0.11 |
|
Complications of drainage, n (%) |
4 (7.4 %) |
1 (5.3 %) |
0.39 |
|
Duration of hospitalization (days) |
8.2 (4.9) |
5.9 (4.4) |
0.02 |
Numerical variables: mean (standard deviaton); Categorical variables: number of patients (%)
Table 3: Comparison of outcomes according to the type of drainage.
|
Univariate analysis OR (95 % CI) |
p value |
|
|
Age |
1.01 (0.98-1,04) |
0.45 |
|
Percutaneous drainage |
1 (0.44-2.39) |
0.12 |
|
Surgical drainage |
0.94 (0.36-2.46) |
0.9 |
|
Male |
0.86 (0.34-2.14) |
0.74 |
|
Urgent |
0.89 (0.33-2.36) |
0.85 |
|
ESC triage score ≥ 6 |
0.66 (0.23-1.91) |
0.45 |
|
Obesity |
0.53 (0.14-2.07) |
0.23 |
|
Diabetes |
0.61 (0.18-2.04) |
0.41 |
|
Neoplasic effusion |
11.8 (3.6-38.5) |
< 0.0001 |
|
Infectious effusion |
0.1 (0.005-2.06) |
0.13 |
|
Acute idiopathic pericarditis |
0.06 (0.003-1.19) |
0.11 |
|
Recurrence |
0.31 (0.06-1.47) |
0.14 |
|
Antiplatelet or anticoagulant |
0.83 (0.33-2.09) |
0.69 |
|
Large (> 20 mm) effusion |
0.95 (0.34-2.7) |
0.92 |
|
Glycemia |
1.38 (0.92-2.1) |
0.11 |
|
Anemia |
1.16 (0.45-2.9) |
0.33 |
|
Neoplasic cells |
6.4 (2.4-17.3) |
< 0.0001 |
|
Adenocarcinoma TTF1 + |
4.5 (1.25-15.8) |
< 0.0001 |
|
Bacteria |
1.28 (0.11-14.7) |
0.84 |
|
Virus |
2.06 (0.69-6.2) |
0.19 |
Anemia: < 12 g/dl for women, < 13 g/dl for men, OR: Odds Ratio, CI: Confidence Interval
Table 4: Factors associated with six-month mortality in univariate analysis.
|
Univariate analysis OR (95 % CI) |
p value |
|
|
Age |
0.99 (0.95-1.03) |
0.52 |
|
Percutaneous drainage |
1 (0.44-2.3) |
0.32 |
|
Surgical drainage |
0.37 (0.11-1.23) |
0.1 |
|
Male |
0.61 (0.19-1.99) |
0.41 |
|
Urgent |
1.73 (0.51-5.87) |
0.38 |
|
ESC triage score ≥ 6 |
9.3 (0.49-175.2) |
0.14 |
|
Obesity |
0.29 (0.03-2.39) |
0.24 |
|
Diabetes |
1 (0.36-2.75) |
0.34 |
|
Neoplastic effusion |
1.39 (0.43-4.52) |
0.58 |
|
Acute idiopathic pericarditis |
0.57 (0.07-4.9) |
0.61 |
|
Previous pericardial effusion |
3.86 (1.07-13.9) |
0.039 |
|
Anti-platelet or anti-coagulant |
1 (0.43-2.31) |
0.57 |
|
Large (> 20 mm) effusion |
1.6 (0.4-6.35) |
0.5 |
|
Neoplastic cells |
1 (0.42-2.36) |
0.4 |
|
Duration of drainage (days) |
1 (0.71-1.4) |
0.56 |
Table 5: Factors associated with six-month recurrences
Anemia: < 12 g/dl for women, < 13 g/dl for men, OR: Odds Ratio, CI: Confidence Interval
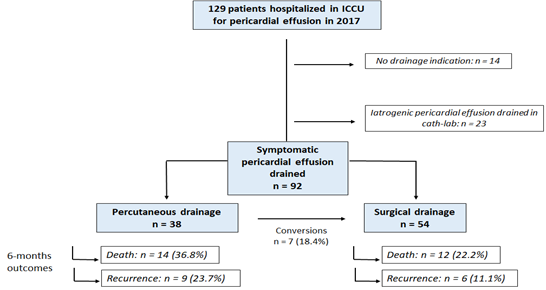
Figure 1: Flow Chart
4. Discussion
In this study, including 129 patients admitted in our ICCU for symptomatic non-iatrogenic PE of whom 92 required a drainage, there was no significant difference for mortality or recurrence at 1- month and 6-months between percutaneous and surgical management. Moreover, percutaneous drainage, performed under local anesthesia, was associated with a shorter length of stay that may justify its use as first line strategy. PE is not an infrequent pathology. The main etiology is represented by neoplasia, which is found in almost half of the case in our cohort, as previously reported [14]. In our cohort, only 12% of PE were idiopathic, which is slight below the 26% reported by Strobbe and al [14]. In past decades, post- cardiotomy effusions has become the main etiology representing about 15% of tamponade, nevertheless, this results is derived from surgical series [7, 15-16]. Our study shows that PE is associated with a significant mortality up to 8% at 1-month and 14% at six-month. PE mortality reported is extremely variable ranging from 2% [16], to 20% [12, 16]. These differences could be explained by differences of population characteristics and PE etiology, but also by different drainage techniques used. Deaths are secondary to neoplasia evolution in more than a half of our population, confirming that mortality is strongly linked to the etiology of the effusion [7, 11, 14, 16].
Limited data are available on large comparison between SD and PCD on the literature, justifying our study [6]. The disadvantages of the SD whatever the type is the need of (1) a specialized cardiac surgery center, (2) a general anaesthesia with its hypotension risk at induction especially in case of large effusions/cardiac tamponade, and (3) performing an abdominal (sub-xyphoid) or intercostal (thoracotomy) or vertical thoracic (sternotomy) incision, which is associated with post-operative pain and potential scarring. But the advantage of the SD, is the possibility to make large pericardial samples for patho-histological examination and potentially perform a pericardial wash. On the contrary, PCD could be applied in only few minutes, everywhere, under local anesthesia by a trained physician whatever his specialty. That explain, it is preferentially used in case of emergency with hemodynamic or respiratory instability, and/or large effusion in our cohort as in others centers [8, 19]. Moreover, the possible percutaneous use of larger drains, the possibility of ultrasound-guided alternative approaches and the possible pericardial/epicardial biopsy using flexible percutaneous pericardioscopy could favor its preferential use. Recurrences reach 7% of our population at 1-month and 14% at 6-months justifying a systematic follow-up in accordance with previous study [12, 13]. To date, several studies showed that PCD was more associated with recurrences [17] but it was not significant in our study. Beyond the type of drainage, neoplasia etiology is highly associated with more recurrence regardless of the technique of drainage. While one might think that SD with per-procedural fenestration could limit the risk of recurrence, we did not find any association between recurrence and initial pericardial fenestration realization in our series. A lack of power, or a rapid fenestration occlusion especially in case of neoplastic PE may explain this absence of difference. Moreover, data concerning the risk of neoplastic cells’ dissemination, particularly in the peritoneum when it is lung or breast cancer localized in the thorax, are missing. In our study, 2 patients affected by breast cancer presented bone metastasis few months after pericardo-peritoneal fenestration realized during the initial SD.
Data concerning complications of pericardial drainage are contradictory: old study reports higher complications with PCD [17] while a more recent series show higher complications with surgical procedure [7]. This should be explained by the improvement of percutaneous technique [12, 17], a better experience of physicians and the use of echocardiographic guidance [18, 19]. In our experience, PCD was safe with only one (2.6%) complication observed in our tertiary ICCU (a right ventricular perforation needed a surgical conversion). In 6 cases (15.8%), PCD by sub-xyphoid approach failed to drain PE and required a conversion into SD: in these cases, a trans-apical approach under ultrasound guidance would probably have limited the number of PCD failure but was not realized in our unit during this period. In our series, PCD was not associated with better outcomes in one-month and six-month mortality but it was associated with a shorter length of hospitalization (about 2 days less). Previous studies demonstrated a higher associated comfort and a shorter hospitalization [7, 20], explaining its preferentially use especially in case of neoplastic etiology. As previously described, obese patients and post-cardiotomy PE [7] were more frequently managed by SD due to technical consideration. Moreover, SD was the only technic use in case of failure of a first drainage, since it allows biopsies to improve diagnosis, and to perform a fenestration, in order to decrease recurrence even if it was not demonstrated in our cohort [13, 22].
Scores were proposed to help clinicians for the triage of cardiac tamponade patients without cardiogenic shock, where immediate pericardiocentesis must be mandatory and potentially lifesaving, but were only based on expert consensus [6, 12] with a class IIb C in the 2015 European guidelines [5]. It is interesting to note that the use of this score leads to consider 2 to 3 times more patients as needing emergency drainage compared to “clinical common sense” (Table 1), all the more justifying an easier and quicker technique to implement. Moreover, this score was not associated with 6-months outcomes as expected. But, to date, there is no recommendation to guide the choice between percutaneous or surgical drainage in case of PE due to the lack of large comparative and/or randomized study. Experts agree only on the fact that PE associated with type A aortic dissection, ventricular free wall rupture after myocardial infarction or severe recent chest trauma should be managed by SD as first line therapy [5, 6]. Our Figure 2 proposes a simple and easy to use algorithm for managing PE and choosing between SD and PCD. While SD remains the prerogative of specialized cardiac surgery centers, PCD should allow a wider diffusion of the technique limiting the systematic transfer of patients to tertiary centers. The absence of excess mortality, more frequent recurrences or more complications, no longer justify these tiring transfers for patients and costly for society when practitioners are trained [24].
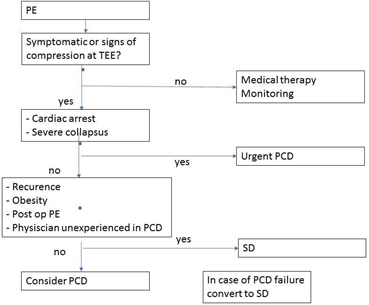
Figure 2: Proposed management algorithm of PE.
PE: pericardial effusion; PCD: percutaneous drainage; SD: surgical drainage; TEE: transthoracic echocardiography
4.1 Study limitations
This study has some inherent limitations related to its monocentric and observational design. Our limited number of patients did not permit definite conclusions in terms of mortality and recurrence especially at long term: larger series or randomized clinical trials are needed to conclude. However, a randomized study with parallel assignment seems not possible because of its frequency and the wide variety of clinical presentations. Our results highlight that clinical outcome of patients after PE management are closely linked to the etiology rather than the procedure management. In our cohort we did not find difference in terms of complications. However there is a trend with less favorable outcome with PCD. Generalization of our results should be made with caution since our teams applied PCD since more than 5 years, only by a sub-xyphoid approach, with a possible surgical backup if necessary. To date, PE drainage and type of procedure should stay a patient and team based decision, but with improvement in echo-guided pericardiocentesis and clinician’s experience, PCD is safe and appears as the first-line therapy for poorly tolerated and/or large PE. Percutaneous drainage should be more widely available in the future, subject to specific training of practitioners.
We did not perform a multivariate analysis given the small size of our series, especially since the univariate analysis found only one significant factor associated with the 6-month mortality (neoplastic nature of the effusion) and with the 6-month PE recurrences (previous PE). Quality of life after hospital discharge was not assessed in our study; neither were pain and analgesic consumption. Thus, we can only suggest that a less invasive procedure, be associated with a better experience and quality of life for patients especially in case of cancer. As PCD was preferentially used for cancer patients, it is possible that a trend towards more recurrences and higher mortality may be found with a longer follow-up. But in this specific population with metastatic cancer, prognosis is often poor at short and medium term, justifying our 6-months follow-up with no patient lost to follow-up.
5. Conclusion
In case of symptomatic non-iatrogenic pericardial effusions, percutaneous pericardial drainage is an efficient and reliable less invasive technique without significant difference in terms of mortality and recurrence at short and mid-term, with surgical drainage. It’s possible and wide availability, and its shorter length of hospitalization may justify its use as first line therapy in trained team especially for cancer patients.
Acknowledgements
The authors would like to thank O.Bouisset for her help in the revision of the manuscript.
References
- Shabetai R. Pericardial effusion: haemodynamic spectrum. Heart 90 (2004): 255-256.
- Spodick DH. Acute cardiac tamponade. N Engl J Med 349 (2003): 684-690.
- Larrey El. New surgical procedure to open the pericardium in the case of fluid in the cavity. Clin Chir 36 (1829): 303-337.
- Kopecky SL, Callahan JA, Tajik AJ, et al. Percutaneous pericardial catheter drainage: report of 42 consecutive cases. Am J Cardiol 58 (1986): 633-635.
- Adler Y, Charron P, Imazio M, et al. ESC Guidelines for the diagnosis and management of pericardial diseases 2015 ; Eur Heart Journal 36 (2015): 2921-2964.
- Ristic AD, Imazio M, Adler Y, et al. Triage strategy for urgent management of cardiac tamponade: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J 35 (2014): 2279-2284.
- Saltzman AJ, Paz YE, Rene AG, et al. Comparison of surgical pericardial drainage with percutaneous catheter drainage for pericardial effusion. J Invasive Cardiol 2012 November 24 (11): 590-593.
- Maggliolini S, De Carlini CC, Imazio M. Evolution of the pericardiocentesis technique. J Cardiovasc Med 19 (2018).
- Imazio M, Adler Y. Management of pericardial effusion. Eur Heart J 34 (2013): 1186-1197.
- Imazio M, Mayosi BM, Brucato A, et al. Triage and management of pericardial effusion. J Cardiovasc Med (Hagerstown) 11 (2010): 928-935.
- Levy PY, Corey R, Berger P, et al. Etiologic Diagnosis of 204 Pericardial Effusions. Medicine (Baltimore) 82 (2003): 385-391.
- Lekhakul A, Assawakawintip C, Fenstad ER, et al. Safety and outcome of percutaneous drainage of pericardial effusions in patients with cancer. Am J Cardiol 122 (2018): 1091-1094.
- Imazio M, Lazaros G, Valenti A, et al. Outcomes of idiopathic chronic large pericardial effusion. Heart 105 (2019): 477-481.
- Strobbe A, Adriaenssens T, Bennett J, et al. Etiology and long-term outcome of patients undergoing pericardiocentesis. J Am Heart Assoc 6 (2017): e007598.
- Peveri V, Pocard M, Martinod E. Sub-xyphoid surgical pericardial drainage for cardiac tamponnade. J Visc Surg 151 (2014): 457-460.
- Becit N, Unlü Y, Ceviz M, et al. Subxyphoid pericardiostomy in the management of pericardial effusions: case series analysis of 368 patients. Heart 91 (2005): 785-790.
- Rafique AM, Patel N, Biner S, et al. Frequency of recurrence of pericardial tamponade in patients with extended versus nonextended pericardial catheter drainage. Am J Cardiol 108 (2011): 1820-1825.
- Chandraratna PA, Mohar DS, Sidarous PF. Role of echocardiography in the treatment of cardiac tamponade. Echocardiography 31 (2014): 899-910.
- Maggioloni S, Gentile G, Farina A, et al. Safety, Efficacy, and Complications of Pericardiocentesis by Real-Time Echo-Monitored Procedure. Am J Cardiol 1 (2016): 43.
- Tsang TS, Enriquez-Sarano M, Freeman WK, et al. Consecutive 1127 therapeutic echocardiographically guided pericardiocenteses: clinical profile, practice patterns, and outcomes spanning 21 years. Mayo Clin Proc 77 (2002): 429-436.
- Lestuzzi C. Neoplastic pericardial disease: Old and current strategies for diagnosis and management. World J Cardiol 2 (2010): 270-279.
- Sagristà-Sauleda J, Mercé J, Permanyer-Miralda G, et al. Clinical clues to the causes of large pericardial effusions. Am J Med 109 (2000): 95-101.
- Halpern DG, Argulian E, Briasoulis A, et al. A novel pericardial effusion scoring index to guide decision for drainage. Crit Pathw Cardiol 11 (2012): 85-88.
- Imazio M, De Ferrari GM. Pericardiocentesis: No more a subspecialty technique. Trends Cardiovasc Med 29 (2019): 384-385.
Supplementary Information
|
Non-drained patients (n=14) |
|
|
Age (years) |
55 (19) |
|
Male, n (%) |
6 (42.8 %) |
|
BMI |
25.9 (6.8) |
|
Duration of hospitalization (days) |
4.8 (2.8) |
|
Medical background, n (%) |
|
|
Previous pericardial effusion |
2 (14.3 %) |
|
Previous cancer |
5 (35.7 %) |
|
Obesity |
3 (21.5 %) |
|
Etiology, n (%) |
|
|
Acute idiopathic pericarditis |
4 (28.6 %) |
|
Cancer |
3 (21.5 %) |
|
Post-operative of cardiac surgery |
1 (7.1 %) |
|
Unknown |
1 (7.1 %) |
|
Mortality, n (%) |
|
|
One month |
1 (7.1 %) |
|
Six month |
5 (37.5 %) |
|
Recurrences, n (%) |
|
|
One month |
1 (7.1 %) |
|
Six month |
2 (14.3 %) |
Numerical variables: mean (standard deviation) ; Categorical variables: number of patients (percentage) BMI: Body Mass Index
Supplementary Table 1: Description of non-drained patients


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 