Rheumatic Heart Disease: 30 years of Valve Surgery clarifying the Autoimmune Process
Pomerantzeff PMA*, Brandao CMA, Aiello VD, Demarchi LM, Sampaio RO, Spina G, Tanaka AC, Tarasoutchi F, Jatene FB, Kalil J, Guilherme L
Heart Institute University of São Paulo Medical School, São Paulo, Brazil
*Corresponding Author Pablo MA Pomerantzeff, Cardiovascular Surgery Division, Heart Institute University of São Paulo Medical School, São Paulo, Brazil
Received: 03 September 2019; Accepted: 16 September 2019; Published: 20 September 2019
Article Information
Citation: Pomerantzeff PMA, Brandao CMA, Aiello VD, Demarchi, LM, Sampaio RO, Spina G, Tanaka AC, Tarasoutchi F, Jatene FB, Kalil J, Guilherme L. Rheumatic Heart Disease: 30 years of Valve Surgery clarifying the Autoimmune Process. Journal of Surgery and Research 2 (2019): 198-204.
View / Download Pdf Share at FacebookAbstract
Rheumatic heart disease (RHD) is the major sequel of rheumatic fever (RF) and leads to valve damage and poor quality of life mainly in children and adolescents. This is due to the need for surgery to repair the lesions caused as a result of an autoimmune reaction due to S. pyogenes infections. In this review, we present the experience of 30 years of surgery for valve correction in both children/adolescents and adult people. The experiences acquired in surgical technical procedures as well as an overview of pathogenic mechanisms that permeate the autoimmune reactions leading to valve dysfunction are the focus of the present review.
Keywords
<p>Rheumatic fever, Rheumatic heart disease, Valve lesions, Histopathology, Autoimmune process, Valve heart disease surgery</p>
Article Details
1. Introduction
Rheumatic fever (RF) is the consequence of an inappropriate and delayed immune response to S. pyogenes infection in non-treated susceptible children and adolescents. Rheumatic heart disease (RHD) is the major sequel and is characterized by valvar lesions that can cause regurgitation and/or stenosis mainly in mitral and aortic valves. In the last 20 years, the mechanisms leading to valve damage have been better elucidated. Several genes are involved in the development of RF and RHD [1].
1.1 Inflammatory mediators
The inflammatory process that permeates heart lesions is responsible for the recruitment of leukocytes (neutrophils, monocytes and lymphocytes) into the heart tissue, through specific mediators such as chemokines, integrins and selectins that play an important role in the inflammatory process [2, 3]. This recruitment process leads to perivascular inflammation in the myocardium and subsequently valvulitis, with the mitral valve being the most frequently affected valve. It is of note that in the acute rheumatic episode, a large number of inflammatory cells such as neutrophils, monocytes/macrophages in addition to T and B lymphocytes are found [4, 5].
1.2 Autoimmune reactions
Some years ago it was identified the presence of cross reactive antibodies in the surface of valve endothelium [6] and infiltration of T cell clones into the valve tissue [7]. Several oligoclonal T cell expansions were found, indicating that some valve specific proteins were the targets of autoimmune reaction, that culminate into valvar dysfunction [8]. These T cells were able to recognize several epitopes of cardiac myosin and valve-derived proteins [9, 10]. The local and progressive inflammation mentioned above leads to tissue injury and cellular matrix disorganization that culminate in valvar dysfunction. Recently, we described a disorganized pattern of vimentin and reduced expression of collagen VI protein which was surrounded by lumican with limited co-localization [11] as a result of both the inflammatory and autoimmune processes. In Brazil, RF is still present today and is widely prevalent and is the leading cause of valvular heart disease and is thus responsible for significant morbimortality in children and young adults.
2. Surgical Procedures
During 30 years (1980-2010) of experience at the Heart Institute of University of Sao Paulo Medical School, we performed 14,601 valve operations, of which 11,311 (77.5%) were valve replacements and 3,290 (22.5%) were valve conservation procedures. Rheumatic patients represent 45.7% (5,169 patients) of the valve replacement group and 77.7% (2,556 patients) of the conservative group. Of those, 1,286 were valve repairs and 1,270 were valve commissurotomy procedures. It is interesting to note that in the 1990s, RF was the cause of 40% of the cardiac surgeries in children, and of over 30% of cardiac surgeries in young adults in the state of São Paulo [12]. The type of surgical procedure performed in the valves of rheumatic heart disease patients depends on the lesions resulting from this autoimmune attack to the heart valves. Different degrees of fibrosis, narrowing, retraction and even calcification of the valvar leaflets will determine the possibility of preservation or the need for valve replacement. Such decisions rely on the experience of the surgical team with the treatment of rheumatic valvar lesions. In the atrioventricular valves, evaluation of the annular dilation and the commitment of the subvalvular apparatus are also essential for decision making.
In young patients, valvar reconstruction is of particular importance, since bioprostheses, even the most recent ones with anti-calcification treatments, still have unsatisfactory outcomes, and the necessary anticoagulation treatment required for mechanical prostheses in this age group is not easy to institute. Mitral valve repair is technically more complex than mitral valve replacement and must be accompanied by careful intraoperative decision-making as pointed out earlier. Pitfalls are present, and probably more so than with mitral valve replacement. As the results of valve repair are superior to replacement, valve repair has become so desirable and popular that many patients come to the surgeon requesting the procedure. The goal of mitral valve repair is to re-establish valvar competence, anatomically preserving the left ventricular function, with as little prosthetic material as possible. The techniques of valve repair must be reproducible, have a low operative risk, provide long term durability and offer advantages over valve replacements. The appropriateness of mitral valve repair for rheumatic mitral valve disease is still an issue of discussion in the literature. Some authors support the idea of recommending valve repair in this group of patients. In our experience with rheumatic patients, we repair valves when we judge that the repair will satisfactorily restore the function to the valve, especially when the anterior leaflet is pliable. Efforts must be made to accomplish valve repair, since a large number of patients are young and might undergo more surgeries throughout life.
We have retrospectively analysed 330 patients who underwent a mitral valve repair during a 20-year period (1985-2005) [13]. The mean age was 26.9 ± 15.4 years, and 57.6% of patients were female. The most common techniques employed were bovine pericardial strip annuloplasty in 48.4% of patients and Carpentier ring annuloplasty in 22.6% of patients, associated with shortening of chordae (20%) and papillary muscle splitting (17.8%). Tricuspid valve repair (26.7%) and aortic valve replacement (27.2%) were the most common associated procedures. During this period, the hospital mortality rate was 0.9% (3 patients); 2 of the patients were children with active rheumatic fever. Linearized rates of freedom from thromboembolism, endocarditis, reoperation and late death were 0.2%, 0.2%, 3.5% and 0.5% of patients per year, respectively. The actuarial survival in 20 years rate was 86.4 ± 6.6% and the freedom from reoperation rate was 30.3 ± 11.1%. The issue arising from mitral valve repairs in this group is the need for reoperation. This is most frequently seen in younger patients (<16 years), as reported in previous published data [14], probably as the result of persistent or recurrent rheumatic carditis. Mitral valve repair, compared to replacement, is associated with higher survival rates and a lower risk of valve-related complications. Associated techniques of repair, such as chordae and papillary shortening, are very useful in rheumatic valves, mainly in younger patients. Regarding aortic rheumatic lesions, the usual finding in aortic insufficiency is leaflet thickening and retraction. Aortic valve repair is performed in specific situations. In these cases, we elongate the leaflets with bovine pericardial patches, mainly in young patients.
The experience of aortic valve repair at the Heart Institute consists of a small group of patients, similarly to other studies in the literature [15]. From 1980 to 2000, 39 patients underwent aortic valve repair for aortic insufficiency. The mean age was 24.7 ± 18.3 years. The aetiology was congenital in 21 patients (53.8%), rheumatic in 10 patients (25.6%), degenerative in 7 patients (17.9%) and infective endocarditis in 1 patient (2.6%). The techniques of valvuloplasty performed were suspension of the leaflets in 23 patients, partial annuloplasty in 2 patients, resection or plication of prolapsed cusp in 9 patients, leaflet extension with bovine pericardium patch in 3 patients and repair of leaflet perforation with bovine pericardium patch in 2 patients. No deaths were observed in this group during the hospitalization period. In the follow-up period, one patient was re-operated on (aortic valve replacement) one year after surgery due to residual aortic insufficiency. We observed one death in the follow-up period due to cardiac failure at nineteen months after surgery. Our results were satisfactory; however, we would like to reinforce that these techniques are feasible only in selected cases.
As early as 1969, Carpentier [16] introduced glutaraldehyde in low concentration for biological tissue fixation and used this technique to manufacture valves and proposed the term bioprosthesis. In 1977, Ionescu and co-workers [17] described the first incidence showing the clinical use of pericardial bioprostheses. In the Heart Institute of the University of Sao Paulo Medical School, bovine pericardial bioprostheses have been implanted since 1982. From 1982 to 1995, a total of 2607 bovine pericardial bioprostheses were performed in 2,259 patients [18]. The average age was 47.2 +/- 17.5 years and rheumatic fever was the main diagnosis in 1,031 patients (45.7%). There were associated procedures in 788 patients (32.9%), the most frequent being tricuspid valve repair (9.2%). The average hospital mortality rate was 8.6% (194 patients); 8.6% for the mitral group, 4.7% for the aortic group, and 12.8% for the double valve replacements. The linearized rates for calcification, thromboembolism, rupture, leak and endocarditis were, respectively, 1.1%, 0.2%, 0.9%, 0.1% and 0.5% of patients per year. The actuarial survival was 56.7+/- 5.4% in 15 years. Freedom rates from endocarditis, thromboembolism, rupture, leak, and calcification were 91.9 +/- 2%, 95 +/- 1.7%, 43.7 +/- 9.8%, 98.9 +/- 4.5%, and 48.8 +/- 7.9% in 15 years, respectively.
The hallmark of the acute rheumatic lesions in the heart is the presence of Aschoff’s bodies in myocardial and/or valvar tissue, characterized by nodular or granulomatous collections of cells arranged at the periphery of an area of fibrinoid necrosis. These nodular lesions occur in the interstitium of the myocardium, around small arterioles, or in the subendocardial region. The nodules are composed of T and B lymphocytes, macrophages, large mononuclear cells, and rarely polymorphonuclear leukocytes. The presence of activated macrophages in these lesions is consistent with an immune response of the CD4+ Th1 type [7]. In cardiac valves, the typical nodules are rare, and usually the acute lesion consists of a fibrin thrombus on the surface, underlined by a palisade of histiocytes. The acute valvar lesions are known to appear on the line of closure of the leaflets, as shown in Figure 1.
There is some controversy about the existence of a true myocarditis in acute rheumatic fever, since there is not direct lesion of cardiomyocytes, one of the injuries highlighted in the classical Dallas’ criteria [19]. In contrast, the inflammation lies in the interstitium around blood vessels as described above [20]. Moreover, laboratory markers of cell necrosis are usually absent in acute rheumatic fever. Regardless, the diffuse inflammation can disrupt the myocardial integrity, causing heart failure. Progression of the acute lesions over time results in modification of the Aschoff’s bodies, disappearance of the central fibrinoid necrosis and decrease in the number of cells. The healed nodule is fibrotic and located around myocardial blood vessels. In valves, the healing process leads to fibrous scarring which distorts the gross and histological morphology and is associated with neovascularization and sparse mononuclear inflammatory infiltrates. There also exists commissural and chordal fusion, retraction of leaflets and, in old lesions, deposits of calcium (Figure 2).
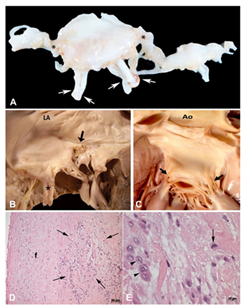
Figure 1: Typical gross lesions of chronic rheumatic mitral disease. (A) Surgical specimen of an excised mitral valve showing diffuse thickening of leaflets, commissural (asterisks) and chordal (arrows) fusion, characterizing chronic rheumatic commitment; (B) and (C) Typical gross lesions of chronic rheumatic mitral disease in necropsy hearts. (B) shows the atrial aspect of an opened mitral valve with diffuse leaflet thickening in addition to calcification and fusion of the posteromedial commissure (arrow); (LA=left atrium); (C) shows a close view of the ventricular aspect of a mitral valve with chronic rheumatic lesion, showing chordal shortening and fusion (arrows). Aortic valve (Ao) and ascending aorta are normal; (D) and (E) Hematoxylin and eosin-stained histological sections of an excised mitral valve; (D) The specific lesion of rheumatic activity, an Aschoff body (arrows), is seen in the valvular tissue with post-inflammatory fibrosis (f); x200; (E) Close view of Aschoff body showing Anitschkow cell (arrow) and syncytial Aschoff cells (arrow heads); Magnification x400.
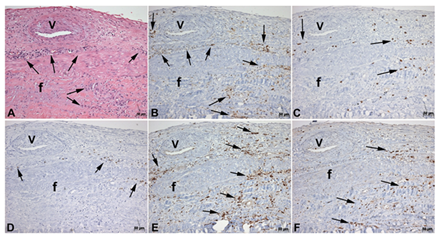
Figure 2: Rheumatic chronic valvulitis and immunohistochemical labeling of inflammatory cells. (A) Rheumatic chronic valvulitis with fibrosis (f), inflammatory cells (arrows) and thick-walled muscular vessel (V) (hematoxylin and eosin stain, x200). Immunohistochemical detection of inflammatory cells is brown color and indicated by arrows for (B)-CD4; (C)-CD8; (D)-CD20; (E)-CD68; (F)-myeloperoxidase. V- thick-walled muscular vessel and f- fibrosis.
New episodes of rheumatic activity, characterized by acute lesions occurring in valves with chronic rheumatic disease, are sometimes unexpected findings in surgically excised valves. This information is very useful for clinicians taking care of rheumatic patients since rheumatic activity may impact post-surgical evolution and may need specific therapeutic interventions.
3. Histopathology
Analysing all the valves and valvar fragments excises from patients with rheumatic fever over a period of 65 months at the Heart Institute (657 valves; 70% mitral), the team of cardiovascular pathologists found signs of acute rheumatic fever in 1.4% of the cases. All of these patients presented with superimposed signs of chronic rheumatic disease. There was a statistically significant difference between the age of the patients presenting signs of acute phase disease and those without (p<0.001, Mann-Whitney Rank Sum Test), and a significant association between the finding of acute-phase histological markers and having an age less than 26 years (chi-square test, p<0.001). The female gender was more common among the chronic (69.2%) and the acute rheumatic cases (66.7%).
It is of note that for those young and adult patients with severe lesions, surgical procedures combined with conventional measures will continue to be the choice treatment. The prospective of developing a vaccine to prevent S. pyogenes infections and consequently RF and RHD, will certainly modify this picture and bring a new quality of life for young people and children.
Conflicts of Interest
The authors declare no competing financial interests.
References
- Carapetis JR, Beaton A, Cunningham MW, et al. Acute rheumatic fever and rheumatic heart disease. Nat Rev Dis Primers 2 (2016): 15084.
- Faé KC, Palacios SA, Nogueira LG, et al. CXCL9/Mig mediates T cells recruitment to valvular tissue lesions of chronic rheumatic heart disease patients. Inflammation 36 (2013): 800-811.
- Tandon R, Sharma M, Chandrashekhar Y, et al. Revisiting the pathogenesis of rheumatic fever and carditis. Nat Rev Cardiol 10 (2013): 171-177.
- VeenaRaizada, Ralph C. Williams, Prem Chopraa, et al. Tissue distribution of lymphocytes in rheumatic heart valves as defined by monoclonal anti-T cell antibodies. Am J Med 74 (1983): 90-96.
- Stollerman GH. Rheumatogenic streptococci and autoimmunity. Clin Immunol Immunopathol 61 (1991): 131-142.
- Cunningham MW. Pathogenesis of group A streptococcal infections. Clin Microbiol Rev 13 (2000): 470-511.
- Guilherme L, Cunha-Neto E, Coelho V, et al. Human heart-infiltrating T-cell clones from rheumatic heart disease patients recognize both streptococcal and cardiac proteins. Circulation 92 (1995): 415-420.
- Luiza Guilherme, Nicolas Dulphy, Corinne Douay, et al. Molecular evidence for antigen-driven immune responses in cardiac lesions of rheumatic heart disease patients. Int Immunol 12 (2000): 1063-1074.
- Ellis NM, Li Y, Hildebrand W, et al. T cell mimicry and epitope specificity of cross-reactive T cell clones from rheumatic heart disease. J Immunol 175 (2005): 5448-5456.
- Faé KC, Da Silva DD, Oshiro SE, et al. Mimicry in recognition of cardiac myosin peptides by heart-intralesional T cell clones from rheumatic heart disease. J Immunol 176 (2006): 5662-5670.
- Martins CO, Demarchi L, Ferreira FM, et al. Rheumatic Heart Disease and Myxomatous Degeneration: Differences and Similarities of Valve Damage Resulting from Autoimmune Reactions and Matrix Disorganization. PLoS One 12 (2017): 0170191.
- Da Silva CH. Rheumatic fever: a multicenter study in the state of São Paulo. Pediatric Committee--São Paulo Pediatric Rheumatology Society. Rev Hosp Clin Fac Med Sao Paulo 54 (1999): 85-90.
- Pablo Maria Alberto Pomerantzeff, Carlos Manuel de Almeida Brandão, Osanan Amorim Leite Filho, et al. Mitral valve repair in rheumatic patients with mitral insuficiency: twenty years of techniques and results. Rev Bras Cir Cardiovasc 24 (2009): 485-489.
- Pomerantzeff PM, Brandão CM, Faber CM, et al. Mitral valve repair in rheumatic patients. Brazilian Journal of Cardiovascular Surgery 13 (1998): 211-215.
- Pomerantzeff PM, De Almeida Brandão CM, Stolf NA, et al. Valve reconstruction in the Heart Institute of São Paulo, Brazil. Semin Thorac Cardiovasc Surg 14 (2002): 324-327.
- Carpentier A, Lemaigre G, Robert L, et al. Biological factors affecting long-term results of valvular heterografts. J Thorac Cardiovasc Surg 58 (1969): 467-483.
- Ionescu MI, Tandon AP, Mary DA, et al. Heart valve replacement with the Ionescu-Shiley pericardial xenograft. J Thorac Cardiovasc Surg 73 (1977): 31-42.
- Pomerantzeff PM, Brandao CM, Cauduro P, et al. Fisics-Incor bovine pericardial bioprostheses: 15 year results. Heart Surg Forum 1 (1998): 130-135.
- Aretz HT. Myocarditis: the Dallas criteria. Hum Pathol 18 (1987): 619-624.
- Kamblock J, Payot L, Iung B, et al. Does rheumatic myocarditis really exists? Systematic study with echocardiography and cardiac troponin I blood levels. Eur Heart J 24 (2003): 855-862.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 