Suboccipital-facial Anastomosis as a Proposition of the New Method of Facial Nerve Repair - Anatomic study
Kamil Krystkiewicz1*, Tomasz Wojciechowski2,3, Adrian Drozdz2,4, Tymon Skadorwa2,4, Bogdan Ciszek2,4, Marek Harat5,6, Marcin Tosik1
1Department of Neurosurgery and Neurooncology, Copernicus Memorial Hospital, Lódz, Poland
2Department of Descriptive and Clinical Anatomy, Medical University of Warsaw, Poland
3Department of Otorhinolaryngology, Head and Neck Surgery, Medical University of Warsaw, Poland
4Department of Pediatric Neurosurgery, Bogdanowicz Memorial Hospital for Children, Warsaw, Poland
5Department of Neurosurgery, 10th Military Research Hospital, Bydgoszcz, Poland
6Department of Neurosurgery and Neurology, Collegium Medicum in Bydgoszcz, Nicolaus Copernicus University in Torun, Torun, Poland
*Corresponding Author: Kamil Krystkiewicz, Department of Neurosurgery and Neurooncology, Copernicus Memorial Hospital, Lódz, Poland
Received: 15 January 2022; Accepted: 21 January 2022; Published: 27 January 2022
Article Information
Citation: Kamil Krystkiewicz, Tomasz Wojciechowski, Adrian Drożdż, Tymon Skadorwa, Bogdan Ciszek, Marek Harat, Marcin Tosik. Suboccipital-facial Anastomosis as a Proposition of the New Method of Facial Nerve Repair - Anatomic study. Journal of Surgery and Research 5 (2022): 51-63.
View / Download Pdf Share at FacebookAbstract
Background
A facial palsy is a cause of an aesthetical and functional problem. An operative technique used the most commonly in particular cases is a hypoglossal-facial anastomosis. However, a function of a tongue in these patients is usually not entirely preserved. Authors sought the solution for that neurological deficit, exploring the anatomical conditions to perform the anastomosis between the facial and the suboccipital nerve.
Materials and methods
The study was performed on the 5 cadavers (10 sides). In the first step, antromastoidectomy was performed. Next, incision was prolonged to the C2 level. The suboccipital nerve was microsurgically dissected. The facial nerve was transferred to the suboccipital nerve, where the anastomosis was performed.
Results
The anastomosis was possible in every case. The measurements were divided into 3 groups. The first group are three distances describing the extent of the antromastoidectomy: superior - 22±0.7 mm, posterior - 21±2.8 mm and anterior margin of procedure - 28±4.2 mm. The second group were the topographical distances crucial for identification of the neural structures: between the styloid process and the tip of mastoid process - 34±0.6 mm, from the stylomastoid foramen to the tip of mastoid process - 18±0.3 mm, from stylomastoid foramen to the suboccipital nerve - 34±0.6 mm. The last one were diameters of the nerves: suboccipital - 2.5±0.7 mm and facial nerve - 2.5±0.7 mm.
Conclusions
The study has revealed that it is possible to perform suboccipital-facial anastomosis. Nevertheless, the functional assessment h
Keywords
<p>Facial palsy surgery, Suboccipital-facial anastomosis, Crossover anastomosis, Vestibular schwannoma</p>
Article Details
Abbreviations
FN - facial nerve
HFA - hypoglossal-facial anastomosis
HHFA - hemihypoglossal-facial anastomosis
HN - hypoglossal nerve
SFA - suboccipital-facial anastomosis
SN - suboccipital-nerve
VS - vestibular schwannoma
1. Introduction
Surgical treatment of vestibular schwannomas (VS) could be followed by certain complications. One of them is a facial nerve (FN) paresis or palsy. In spite of application of a microsurgical techniques and an intraoperative neurophysiological monitoring, the functional outcome in the group with preserved nerve continuity is unsatisfactory in 19-50% [1,2]. Denervation of the area supplied with this nerve leads not only to movement impairment, like difficulties with eyelid closure and facial expression, but it is also a psychological discomfort [3]. There were proposed multiple ways to manage FN palsy after the VS surgery. Beginning with the static procedures, known widely in the plastic surgery, to dynamic reanimation surgeries: direct end-to-end suture, reconstruction with cable grafting and extratemporal crossover anastomosis. When the direct end-to-end anastomosis or use of interposition graft could not be performed, the crossover anastomosis should be employed [4,5]. There are multiple cranial and spinal nerves which were considered as axons donors. Among them, the hypoglossal-facial anastomosis (HFA) with its modifications is the most common choice in the treatment of these patients [2,6,7]. In classic procedure the completely transected hypoglossal nerve (HN) was used as a donor of motor fibers. However, the tongue atrophy and caused by it articulation and mastication disorders could severely interfere with normal day life activity [8]. Over past years many variations of the classic procedure were described in case to spare the tongue mobility. The most important seems to be the longitudinal division of HN [9], ‘side-to-end’ technique with transposition graft described by May [10], direct ‘end-to-side’ with transposition of the mastoid part of facial nerve (Darrouzet technique) [11] or described by Martins procedure hemihypoglossal-facial anastomosis (HHFA), where the intramastoid part of the FN is connected ‘end-to-end’ to the split HN [8]. Independently from the used technique, procedures are still related with risk of discrete or mild tongue atrophy which amounts in 30-57% [8,12]. Another disadvantage of HHFA is insufficient amount of fibers in the half of HN to provide the complete FN reinnervation [8]. Additionally, post-incision scar is placed on well visible region of head and neck. It also should be taken into consideration in the context of aesthetical results. All above mentioned complications related to HN injury may be avoided using other nerve. In searching for alternative nervous fibers donor in the region adjacent to the stylomastoid foramen, anastomosis with suboccipital nerve (SN) was proposed as potential solution. Mainly, because of the pure motor character of fibers and minor neurological complications caused by The anatomical relation and diameter of both nerves were taken under consideration, as well as the innervation area supplied by the SN. In our study we would like to explore anatomical conditions determining possibility to perform such procedure and to elaborate technique of suboccipital-facial anastomosis (SFA) as a treatment proposition in the proximal facial palsy.
2. Materials and methods
Study was performed on 5 cadavers, which were not supposed to have any middle ear pathology and surgery performed in the area of interest. Procedure was performed in both sides, which gives the number of 10 anastomosis. Typical three quarter prone position was used in every case. Microsurgical techniques of dissection were employed. All measurements were performed using standard caliper. Detailed description of procedure is presented in the Results section. Cadavers come from a voluntary donation program and are part of the existing collection of the Department of Clinical and Descriptive Anatomy, Medical University of Warsaw, Poland. The research was conducted according with the Polish Death and Funeral Act and with relevant guidelines and regulations. The Institutional Ethics Committee was informed about the ongoing study, and the Committee stated no need for its approval.
3. Results
Procedure could be divided into 4 major steps: 1) antromastoidectomy, 2) suboccipital triangle exposure, 3) anastomosis, 4) closure.
3.1 Antromastoidectomy
Incision starts in the retroauricular area, 0.5 cm inferior to the mastoid tip. It is prolonged to the suprameatal spine (spine of Henle). Incision is linear, lightly curved posteriorly (Figure 1). There is no need to perform the subcutaneous or pericranial flap in separate layer. Incision length of 5 cm is enough to evaluate the area of mastoid process. The tip of mastoid process and insertion of the sternocleidomastoid muscle should be visible in the inferior aspect of exposure, cartilage of external acoustic meatus in the anterior part, suprameatal spine superiorly to the external acoustic meatus. The mastoidectomy is performed in the triangle, which is limited by: the tip of mastoid process, the suprameatal spine and area where sigmoid sinus projects on the cranium (Figure 2). Average length of drilling necessary to expose the facial nerve properly is 28±4.2 mm in the anterior, 22±0.7 mm for the superior and 21±2.8 mm for posterior aspect of mastoidectomy. Compact bone is removed using the cutting burr and removal is performed until the antrum is visualized. From that moment procedure should be performed using the intraoperative microscope. During the drilling, there is no need to skeletonize the sigmoid sinus and the dura of middle fossa. There are several ways, which could be used for purposes of FN identification. The first one is depended on the location of the short crus of incus, which points the tympanic part of the canal. To visualize the incus, the drilling should be continued through the antrum, removing the cancellous bone covering the external acoustic meatus. The first structure after the opening of tympanic cavity will be the short crus of the incus pointing the prominence of FN canal. The second way to found the seventh cranial nerve is a relationship between its canal and prominence of the lateral semicircular canal. Beneath it, FN courses from the tympanic into the mastoid part of its canal. Third way is drilling through the tip of the mastoid process into anteromedial direction to found the nerve near the stylomastoid foramen. (Figure 3). It could be found approximately 18±0.3 mm from the tip of mastoid process. The diamond drill is used for skeletonization of the nerve canal. Thin layer of bone is left on its course, which is removed using the Penfield’s dissector. After that step, stylomastoid foramen is widened and tip of the mastoid process is removed. Thanks to such proceeding, the FN transposition will be much easier and less traumatizing for the nerve. After that, the second part of the procedure could be performed.
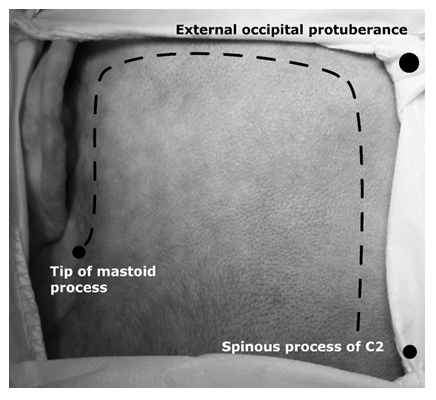
Figure 1: Skin incision, left side. Note the most important topographical cranial points, which should be palpated before surgery.

Figure 2: The area of antromastoidectomy
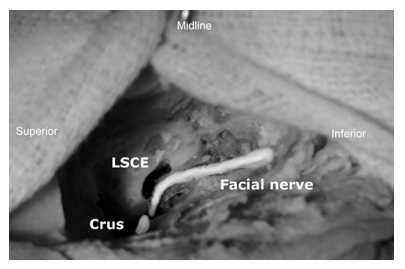
Figure 3: The course of left facial nerve. LSCE - eminence of lateral semicircular canal. SC - short crus of incus.
3.2 Suboccipital triangle exposure
The incision used for mastoidectomy ends on the level of external occipital protuberance (Figure 1). Incision is prolonged horizontally, parallel to superior nuchal line. The incision should be continued inferiorly in the paramedian line 2 cm laterally from the median line, parallel to the nuchal ligament, ending on the C2 level. The aim of the procedure is SN, which is found in the suboccipital triangle limited by obliquus capitis inferior and superior muscles and rectus capitis posterior major muscle. The superficial nuchal muscles - trapezius, splenius capitis and semispinalis capitis (Figure 4) are cut in one layer and suboccipital triangle is exposed. During separation of the last muscle - semispinalis, a great attention is required. This is caused by the fact, that SN innervates partially the semispinalis muscle. A small branch is arising from the main nerve trunk and penetrates the deeper surface of the muscle. That branch should be cut in the controlled fashion using the scissors, otherwise the main trunk could be ruptured. The suboccipital triangle is exposed (Figure 5). From that point, seeking for the SN in any case should be performed using the intraoperative microscope. Dissection in that area does not require vertebral artery mobilization and C1 laminectomy. However, vertebral artery should be separated from its sulcus on the posterior arch of atlas. From that point, course of the vertebral artery and venous plexus is evaluated. When SN is exposed, it is cut using the scalpel blade in the place, where it divides into muscular branches. The distance from that point to the stylomastoid foramen is in average 34±1.2 mm. Actually, it is a crucial measurement for the procedure, because this is the approximate length of the FN stump needed to be cut from the temporal bone. After that, the facial nerve should be located near its exit from the cranial base. The best way to achieve that, is a palpation of the styloid process, which should be searched behind the posterior belly of digastric muscle. That relevant in topography bony landmark is found 34±1.2 mm from the tip of mastoid process. It is important, because the stylomastoid foramen is located posteriorly from it and could be found 18±0.6 mm from the tip of mastoid process. That area is explored, facial nerve is dissected. After that, intramastoid part of nerve is cut in its canal in the length of 30±0.5 mm from the foramen. In the ending, the posterior belly of digastric muscle is cut and nerve is transposited near to the SN.
3.3 Anastomosis
The surfaces of the nerves stumps before suturing should be properly prepared. Transections should be performed using scalpel blade in both cases. Diameter of transected FN was 2.5±0.71 mm and 2.5 ±0.71 mm for SN. After that, anastomosis is performed (Figure 6). Fine 8-0 permanent monofilament suture is used. 2-3 sutures through the epineurium and nerves fascicles are performed. Anastomosis in the ending should not be performed under the tension.
3.4 Closure
The posterior belly of digastric muscle and superficial nuchal muscles should be sutured. The antromastoidectomy area should be closed typically, using the abdomen fat placed with the fibrin glue. Layers are sutured in the typical, multilayer fashion.
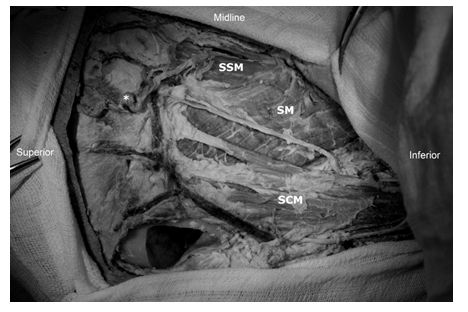
Figure 4: Left suboccipital area exposition after skin removal. SCM - sternocleidomastoid muscle, SM - splenius muscle, SSM - semispinalis muscle, * - suboccipital artery. Trapezius muscle is retracted medially.
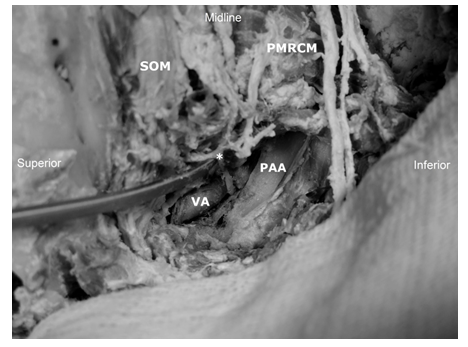
Figure 5: Left suboccipital triangle. Note the topographical relation of suboccipital nerve, which is found between vertebral artery and atlas. * - suboccipital nerve, VA - vertebral artery, PAA - posterior arch of atlas, SOM - obliquus superior muscle, PMRCM - rectus capitis posterior major muscle. Inferior oblique muscle is retracted.

Figure 6: Suboccipital-facial anastomosis. FN - facial nerve, SN - suboccipital nerve.
4. Discussion
Despite the modern microsurgical technique and intraoperative neuromonitoring, the preservation of the FN during the VS resection is a still challenging. Postoperative function of the facial muscles is evaluated using the House-Brackmann grading system [13]. Result is considered as satisfactory, when patient is able at least to close completely the eyelids (III grade). Basing on the large meta-analysis performed by Zou et al. [14], anatomical preservation of the FN was achievable in approximately 89% of cases. That results with 11% of patients, who from the assumption had poor functional outcome of the FN. Starnoni et al. provided an meta-analysis, where pooled satisfactory FN outcome after resection of large VS was 60,1% [15]. Large number of patients in whom any facial muscles reanimation procedure should be considered is significant. Mentioned articles were focused on a group of cases with the tumor larger than 3 cm, which is the risk factor of the postsurgical facial nerve palsy [15-17]. However, even in the groups with the smaller tumors, the risk of the FN damage is significant. Satisfactory function of the facial muscles is emphasized, because its weakness cause dangerous complications and decreased quality of life. The most important complication is weakened protection of a conjunctiva and cornea [18]. That explains the effort, which should be taken for proper management of the logophthalmos. Prophylaxis of the eye complications is based on constant moistening [19]. Although, conservative methods prevent from the ophthalmological complications, they do not influence the other important issue - decreased quality of life [18,20,21]. That is why the surgical procedure should be considered in the specific group of patients. One of the ways to manage the facial palsy is extratemporal crossover anastomosis [4]. There were proposed many nerves as donors for FN, but the most effective is the HN. HFA is known from the XX century [22]. Classic approach to that surgery required the complete transection of the HN. Cytomorphometric studies revealed that transected NH consists of 10 000 motor fibers [23]. Similar studies on the FN revealed that it is consisted from 7 000 axons [24]. Using the complete transection of the HN, there is a chance, that every single axon from the FN stump would have its continuation. However, use a part of the fibers forming the HN could not fulfill that aim. That assumptions explain the results presented by Martins et al. [8] in their comparison of the classic procedure with its variation - HHFA. Study revealed the slightly better outcome in House-Brackmann grading score in the classic procedure (III grade - 75%, IV grade - 25%) comparing to the HHFA (III grade - 71%, IV grade - 21%, V grade - 4%). However, the statistical analysis showed no differences in that aspect. Moreover, Martins described a scale of tongue function evaluation. (Table 1) [12]. What is important, basing on that scale the final outcome concerning the tongue function was significantly better for the hemihypoglossal variations. In the classic procedure all patients had transient problems with the speech and swallowing after surgery and 41% of them had that problem in the late follow-up. 75% of them was evaluated as grade III, 25% as grade IV in the Martins scale. On the other hand, in the hemihypoglossal group 75% of patients had no atrophy, 26% was in the grade II and 4% in the grade III. Dziedzic et al. [12] has a similar results. 84% of patients from that study had been evaluated as satisfactory in the House-Brackmann grading score. However, results of the tongue function were slightly better than Martins. 74% of cases were evaluated in the last control examination as grade I, 19% as grade II and 7% as grade III. These studies reveal, that there is always a probability of functional impairment and atrophy of the tongue, which could be not accepted by some patients. Other disadvantage of using the HN is a post incision scar in the submandibular area. That seems to be a marginal issue in the context of the face asymmetry, but the final outcome could be influenced by many factors. Concerning that disadvantages of the HFA, usage of the other nerve as a axons donor could be considered. The SN is formed by the dorsal rami of the first neuromere. Excluding the small number of proprioreceptive fibers to the atlanto-occipital joint, it is a pure motor nerve. The innervation area is limited to the suboccipital muscles: superior oblique, inferior oblique, rectus capitis posterior major and minor. Its fibers also innervates parts of the longissimus capitis and semispinalis. Function of the suboccipital muscles is mostly the control of complex rotation movement of the head. SN seems to be a good candidate as the donor, because has almost pure motor character and innervates the muscles, which function is not critical for the head movement. Moreover, there are no studies about the SFA in the literature. However, there is a lack of information about the number of fibers forming the SN. Cytomorphometric studies were focused mostly on other nerves, excluding the SN. Basing on the direct measurements, the diameter of the nerves is similar (2.5±0.7 mm). Of course, it provides only a macroscopic clue, that anastomosis is technically possible, but it gives no information about the number of fibers forming the SN. Further studies on the microstructure of that nerve should be performed. What is an advantage, usage of the SN gives an opportunity to assess all forming it fibers. During this procedure, the full diameter of SN is transected and connected with the FN. Moreover paralysis of the SN does not provide any significant complications [25]. During the procedures performed in the craniocervical junction, where there is a necessity to dissect the suboccipital triangle, SN is sacrificed. The model example of such procedure is the far lateral approach. During it, both muscles and nerves are cut for purposes of the vertebral artery mobilization from the transverse process of the atlas [26]. During the postoperative follow up, patients do not complain on a problem with the rotation of the head [25,27]. It seems to be a significant advantage, comparing to the HFA, where deficits are visible and could impact the quality of life. Moreover, anatomical topography of the SN favors the idea of its usage in the FN repair. The mean distance from the SN to the stylomastoid foramen, where facial nerve exits the cranial base, is 34±1.2 mm. There is no possibility to mobilize the proper length of extracranial part of FN for anastomosis purposes. The commonly used and accepted method for the solution of that problem is antromastoidectomy and facial canal exposure [11]. Thanks to the proper earlier dissection of the extratemporal part of the FN, the distance of 30±0.5 mm is a sufficient canal exposure for the tensionless anastomosis. The receiving of longest stump of the FN demands the exposure of tympanic part of canal. Localization of the FN does not cause an intraoperative problem. However, if there is a difficulty, the anatomy of that area should always be evaluated. Useful topographical point is always palpable styloid process. It could be found 34±1.2 mm medially and anteriorly to the mastoid process. The stylomastoid foramen is localized between them, approximately 18±0.6 mm medially and anteriorly from the mastoid process. Mentioned dimensions are rather constant. In classic HFA with the FN transposition, the incision from the retroauricular area is prolonged to the submandibular area. Unfortunately, that long linear incision lefts the scar in the visible place. In SFA the incision is planned in the suboccipital area, hided in the hairline. Flap used in the procedure is smaller, than typically used for the far lateral approach [25,26]. SFA solves some problems connected with the HFA or HHFA. However, because of the different anatomical relationship of the donor nerve, it creates a new ones. First of all, the suboccipital area is drained by the complex venous system. Vertebral artery is surrounded by the complex venous vessels. Arnautovic [27] defines it as a suboccipital cavernous sinus, because of the structural similarity to the internal carotid artery and cavernous sinus. Basing on the MRI and venography, Takahashi et al. [29] evaluated, that it is a constant structure and has complex draining area. What is more, during the nuchal muscles dissection, there is a risk of posterior jugular vein and deep cervical vein injury. However, intraoperative bleeding is manageable thanks to various hemostatic techniques and total mortality is low - 0-3% and caused by the other factors [25]. Second matter to discuss is unpredictable functional outcome. First of all, one of the possible complications could be facial synkinesis, visible during the neck movement. What is more, it is difficult to guess the function and rehabilitation outcome after the SFA. However, even the recovery of the facial muscles tonus, closure of the eyelids and achievement of the III grade in House-Brackmann grading is considered as a clinical success. This is why, this could be considered as a therapeutical option for the patients, who are not motivated for the long and difficult rehabilitation.
Limitations
Our study focus only on the anatomical conditions of such procedure. Basing on it, we couldn’t provide any results on the functional outcome. Further experimental studies, starting on the animal model, should be provided.
Conclusions
The study has revealed that anatomical conditions allows to perform SFA. That technique could provide motor supply for the facial nerve, at the same time sparing the tongue mobility. Nevertheless, the functional outcome in comparison to the HFA or HHFA has to be investigated in further studies.
References
- Falcioni M, Fois P, Taibah A, Sanna M. Facial nerve function after vestibular schwannoma surgery. J Neurosurg 115 (2011): 820-826.
- Samii M, Gerganov V, Samii A. Improved preservation of hearing and facial nerve function in vestibular schwannoma surgery via the retrosigmoid approach in a series of 200 patients. J Neurosurg 105 (2006): 527-35.
- Walker DT, Hallam MJ, Ni Mhurchadha S, et al. The psychosocial impact of facial palsy: our experience in one hundred and twenty six patients. Clin Otolaryngol 37 (2013): 474-477.
- Kunert P, Smolarek B, Marchel A. Facial nerve damage following surgery for cerebellopontine angle tumours. Prevention and comprehensive treatment. Neurol Neurochir Pol 45 (2011): 480-488.
- Yetiser S, Karapinar U. Hypoglossal-facial nerve anastomosis: a meta-analytic study. Ann Otol Rhinol Laryngol 116 (2007): 542-549
- González-Darder JM, Capilla-Guasch P, Escartín FP, et al. Side-to-End Hypoglossal-Facial neurorrhaphy for treatment of complete and irreversible facial paralysis after vestibular schwannoma removal by means of a retrosigmoid approach: A clinical and anatomic study. World Neurosurg 136 (2020): e262-e269.
- Dabiri S, Khorsandi AM, Moharreri M, et al. Results of end-to-side hypoglossal-facial nerve anastomosis in facial paralysis after skull base surgery. Iran J Otorhinolaryngol 32 (2020): 133-138.
- Martins RS, Socolovsky M, Siqueira MG, et al. Hemihypoglossal-facial neurorrhaphy after mastoid dissection of the facial nerve: results in 24 patients and comparison with the classic technique. Neurosurgery 63 (2008): 310-317.
- Arai H, Sato K, Yanai A. Hemihypoglossal-facial nerve anastomosis in treating unilateral facial palsy after acoustic neurinoma resection. J Neurosurg 82 (1995): 51-54.
- May M, Sobol SM, Mester SJ. Hypoglossal-facial nerve interpositional-jump graft for facial reanimation without tongue atrophy. Otolaryngol Head Neck Surg 104 (1991): 818-825.
- Darrouzet V, Guerin J, Bébéar JP. New technique of side-to-end hypoglossal-facial nerve attachment with translocation of the infratemporal facial nerve. J Neurosurg 90 (1999): 27-34.
- Dziedzic TA, Kunert P, Marchel A. Hemihypoglossal-Facial Nerve Anastomosis for Facial Nerve Reanimation: Case Series and Technical Note. World Neurosurg 118 (2018): e460-e467.
- House JW, Brackmann DE. Facial nerve grading system. Otolaryngol Head Neck Surg 93 (1985): 146-147.
- Zou P, Zhao L, Chen P, et al. Functional outcome and postoperative complications after the microsurgical removal of large vestibular schwannomas via the retrosigmoid approach: a meta-analysis. Neurosurg Rev 37 (2014): 15-21.
- Starnoni D, Giammattei L, Cossu G, et al. Surgical management for large vestibular schwannomas: a systematic review, meta-analysis, and consensus statement on behalf of the EANS skull base section. Acta Neurochir (Wien) 162 (2020): 2595-2617.
- Samii M, Gerganov VM, Samii A. Functional outcome after complete surgical removal of giant vestibular schwannomas. J Neurosurg 112 (2010): 860-867.
- Grinblat G, Dandinarasaiah M, Braverman I, et al. Large and giant vestibular schwannomas: overall outcomes and the factors influencing facial nerve function. Neurosurg Rev 44 (2020): 2119-2131.
- Nowak-Gospodarowicz I, Rózycki R, Rekas M. Quality of life in patients with unresolved facial nerve palsy and exposure keratopathy treated by upper eyelid gold weight loading. Clin Ophthalmol 14 (2020): 2211-2222.
- Cortese D, Capp L, McKinley S. Moisture chamber versus lubrication for the prevention of corneal epithelial breakdown. Am J Crit Care 4 (1995): 425-428.
- Ryzenman JM, Pensak ML, Tew JM Jr. Facial paralysis and surgical rehabilitation: a quality of life analysis in a cohort of 1,595 patients after acoustic neuroma surgery. Otol Neurotol 26 (2005): 516-521.
- Blom SSAH, Aarts H, Wever CC, et al. Quality of life, social function, emotion, and facial paresis in Dutch vestibular schwannoma patients. Laryngoscope Investig Otolaryngol 5 (2020): 477-484.
- Körte W. Ein Fall von Nervenpfropfung des N. facialis auf den N. hypoglossus. Dtsch Med Wschr 29 (1903): 293-295.
- Tiago RS, Faria FP, Pontes PA, et al. Morphometric aspects of the human hypoglossal nerve in adults and the elderly. Braz J Otorhinolaryngol 71 (2005): 554-558.
- Asaoka K, Sawamura Y, Nagashima M, et al. Surgical anatomy for direct hypoglossal-facial nerve side-to-end "anastomosis". J Neurosurg 91 (1999): 268-275.
- Flores BC, Boudreaux BP, Klinger DR, et al. The far-lateral approach for foramen magnum meningiomas. Neurosurg Focus 35 (2013): E12.
- Ladzinski P, Maliszewski M, Kaspera W, et al. Partial transcondylar approach - analysis of the surgical technique in cadaver simulation. Neurol Neurochir Pol 46 (2012): 245-256.
- Ladzinski P, Majchrzak H, Kaspera W, et al. Meningiomas of the upper and middle part of the clivus and surrounding structures: early and long-term outcome. Neurol Neurochir Pol 47 (2013): 332-344.
- Arnautovic KI, al-Mefty O, Pait TG, Krisht AF, et al. The suboccipital cavernous sinus. J Neurosurg 86 (1997): 252-262.
- Takahashi S, Sakuma I, Omachi K, et al. Craniocervical junction venous anatomy around the suboccipital cavernous sinus: evaluation by MR imaging. Eur Radiol 15 (2005):1694-1700.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 