Anti-Infective Properties of Anti-Cancer Cationic Peptides containing Survivin or Apolipoprotein E Sequences
Bruno Périchon1, Alphonse Garcia A2,3*
1Unit of Biology of Gram-positive Pathogenic Bacteria, Pasteur Institute, Paris, France
2E3 Phosphatase Laboratory-NMR Unit, Paris, France
3Department of Structural Biology and Chemistry and Dode-Design of Biology, Pasteur Institute, Paris, France
*Corresponding Author: Alphonse Garcia, E3 Phosphatase Laboratory-NMR Unit, Paris, France
Received: 20 September 2019; Accepted: 04 October 2019; Published: 10 October 2019
Article Information
Citation:
Bruno Périchon, Alphonse Garcia A. Anti-Infective Properties of Anti-Cancer Ationic Peptides containing Survivin or Apolipoprotein E Sequences. Journal of Biotechnology and Biomedicine 2 (2019): 161-168.
DOI: 10.26502/jbb.2642-91280019
View / Download Pdf Share at FacebookAbstract
Specific intracellular pathways regulated by Apolipoprotein E (ApoE), a PP2A activator or survivin, an inhibitor of Heat Shock Proteins (HSP), can impact human diseases including cancers. In this study, we have initially observed that anti-tumor Tat-survivin (shepherdin-Tat), and ApoE mimetics, Cog or Tat-Cog &Cog-Tat, peptides contained cationic sequences that share similar physical characteristics with LL17-32, a short active sequence of the anti-microbial human cathelecidin peptide. Furthermore, to investigate the potential host defense properties of these peptidic sequences we have comparatively analyzed anti-tumor and anti-bacterial properties of shepherdin-Tat and ApoE mimetic peptides with the human LL17-32 cathelecidin sequence using U87G cells, a relevant human
glioblastoma model, and a Group B Streptococcus agalactiae NEM316 ΔdltA that is highly sensitive to human LL17-32 cathelecidin. This study highligts two major insights indicating that similarly, to LL37 or LL17-32 cathelecidin sequences, shepherdin-Tat and ApoE mimetic peptides firstly impairs the growth of S. agalactiae NEM 316 dltA strain, and secondly inhibited the survival of human glioblastoma U87G cells. In conclusion, together, our results clearly indicate that cationic peptides with survivin and ApoE anti-tumor sequences, including scrambled shepherdinTat, a previously described biologically inactive anti-cancer molecule, behave as cathelecidin-like anti-infective host defense molecules. In addition, we identified hybrid Cog-Tat/Tat-Cog peptides as potential and highly potent therapeutic molecules with anti-glioblastoma and anti-bacterial effects.
Keywords
<p>Cationic sequences, PP2A, Survivin, Bacteria, Pathogen</p>
Article Details
Abbreviations:
S.agalactiae-Streptococcus agalactiae; PP2A-Protein Phosphatase 2A; HSP-Heat Shock Protein; ApoE-Apolipoprotein E; AA-amino acid
1. Introduction
Rationally designed anti-tumor peptides that can regulate specific intracellular targets, such as PP2A or HSP proteins, have been previously investigated. In this regard, firstly apolipoprotein E (ApoE), a plasma protein responsible for transporting lipid and cholesterol, and ApoE-mimetic peptides such as Cog 133, a peptide containing the amino acid residues 133-149 of the ApoE receptor binding region [1, 2], bind to the PP2A inhibitor SET to activate endogenous PP2A activity, a predictive anti-cancer target [3-5]. Interestingly, OP449, a Cog 133 bipartite mimetic peptide combining antennapedia cell penetrating and Cog133 sequences, displayed both in vitro and in vivo anti-tumor activity against B-cell chronic lymphocytic leukemia (CML) [6]. We have also more recently characterized ApoE Cog-mimetic peptides, named DPT-Cog, which combined the sequences of DPT-sh1 penetrating shuttle [7] with Cog133, and inhibited PI3K-dependent survival of U87G cells [8]. Secondly, the peptide sequence survivin79-83 is an antagonist of the interaction between Hsp90 and survivin. And Shepherdin-Tat, a bi-partite peptide resulting from the fusion of this sequence to Tat47-57 cell penetrating peptide (cpp), is a specific Hsp90 inhibitor that down regulated cell proliferation and cell viability in tumor cells and in tumor xenografted mice with no detectable toxic effects in control mice [9]. Anti-infective properties of cationic antimicrobial sequences have been previously documented and LL-37, the sole biologically active human antimicrobial peptide of the mammalian cathelecidin family, induced by the proteolytic cleavage of 18-kDa precursor HCT18 protein [10], is found in biological fluids. It has a broad spectrum of antimicrobial activity against Gram-positive and Gram-negative bacteria, fungi, and enveloped viruses [11, 12]. In addition, we have recently identified virally encoded cationic sequences with LL37-like anti-infective properties [13, 14]. In this study, following our initial observation that some survivin and ApoE mimetics Cog peptides contained potential anti-microbial cationic sequences, we have characterized three new host defense peptides, Shepherdin-Tat, Tat-Cog and Cog-Tat that inhibited the survival of human glioblastoma U87G cells and also the growth of S. agalactiae NEM 316 dltA strain.
2. Materials and Methods
2.1 Cell and peptide
We used human Glioblastoma U87G cells (kindly provided by Pr. Marie Dutreix, Curie Institute, Orsay) that have been previously approved by the Institut Pasteur, our institutional review board). Chemical solid-phase peptide synthesis of NH2-biotinylated peptides were commercially realized by the French Proteogenix company at >95% purity (for profile see web site https://www.proteogenix.science/company/). NH2-biotinylated peptides were, dissolved in DMSO and stored at -20°C pending use.
2.2 Cytotoxicity assays
A total of 3,000 cells were incubated for 24 hours with different concentrations of pharmacological agents. Cell cytotoxicity was analyzed by a colorimetric assay using 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (called MTT) for adherent cells as described by the manufacturer (Sigma). The assays were performed in triplicate and we used Gene 5 detection software (from BioTek-France) for data capture and export into Excel. And we used Microsoft Excel software 2016 for macOS for statistical analyses in histogram containing error bars indicating the standard deviation.
2.3 Bacterial strains and antibacterial susceptibility test
- agalactiae mutant NEM 316 dltA strain, which is characterized by a complete absence of D-alanine due to the insertional inactivation of dltA, was previously described [15]. The Minimum Inhibitory Concentrations (MICs) of each peptide were tested in Todd-Hewitt broth (THB) buffered with 50mM HEPES in 96-well Costar polypropylene microplates (Costar, Cambridge, USA) by a dilution method. Bacteria (10^6 CFU) were added in triplicates to wells containing increasing concentrations of the antimicrobial peptides. Plates were incubated 24h at 37°C and then read (OD600 nm) using microplate reader (Synergy 2, Biotek) for bacterial growth. The MICs90 was considered to be the peptide concentration that inhibited 90% growth.
3. Results
3.1 Survivin and cog-mimetics cationic sequences display similar physical characteristics with anti-microbial human LL17-32 cathelecidin peptide
As illustrated in Table 1 Shepherdin-Tat and ApoE mimetics Cog peptides, are hybrid bipartite sequences that contained the same protein transduction domain (PTD) sequence of the human immunodeficiency virus TAT protein (Tat47-57) combined with survivin79-83 (shepherdin) or with ApoE/Cog133-149 (Tat-Cog and Cog-Tat) sequences. Interestingly, these peptides are cationic and share common physical properties (details in Table 1 columns 2 and 3) with anti-microbial LL37/LL17-32 cathelecidin sequences. We therefore hypothesized that these hybrid peptides may also behave as new host defense molecules that, similarly to human LL37 cathelecidin, might be able to display toxicity against both U87G gliobastoma cells and S. agalactiae NEM 316 dltA strain, the gram-positive bacterial model. To test this hypothesis, we have comparatively analyzed the effect of LL17-32, a previously characterized efficient anti-bacterial and anti-tumoral sequence derived from LL37 human cathelecidin [12, 16], with hybrid bipartite peptides containing Tat47-57, respectively combined with LL17-32, a positive control, and with ApoE-cog133-149 and survivin79-87 sequences.
3.2 Effect of survivin and cog-mimetics cationic peptides on survival of U87G glioblastoma cells
Firstly, as shown in Figure 1 upper panel, using MTT assay, we found that both LL17-32 and Tat-LL17-32 inhibited the viability of U87G cells in a similar dose-dependent manner (estimated IC50=15μM). Furthermore concerning survivin mimetic peptides, we found that, in consistency with anti-tumor effects previously reported in different cancer cells [9], Shepherdin-tat, but not SCR, the hybrid control peptide combining Tat47-57 and survivin79-87 scrambled sequences [9], inhibited the viability of U87G cells in a dose-dependent manner (Figure 1, middle panel). Interestingly, similar effects were also observed in DPT-survivin after replacement of the Tat cpp by the cpp shuttle DPT-sh1 (Figure 1 middle panel). Secondly, concerning ApoE mimetic peptides, while Cog133-149 alone is not efficient, the presence of Tat penetrating sequence in N-ter (Tat-Cog) or in C-ter (Cog-Tat) of the ApoE mimetics hybrid sequences generates highly active hybrid peptides Cog (estimated IC50=5 μM) (Figure 1 bottom panel).
3.3 Effect of survivin and cog-mimetics cationic peptides on growth of S. agalactiae NEM 316 dltA strain
We have comparatively investigated the anti-microbial activities of LL17-32 and survivin or ApoE derivatives by monitoring bacterial growth of S. agalactiae NEM 316 ΔdltA strain. As shown in Table 1 (column 4), the MIC90 value of LL17-32, Tat-LL17-32, ShepherdinTat, SCR, DPT-survivin79-87 and Cog are in a restricted range (6.25-12.5 μM). Interestingly Tat-Cog or Cog-Tat are the more efficient peptides with a respective MIC90 value of 3.1 μM and 1.6 μM.
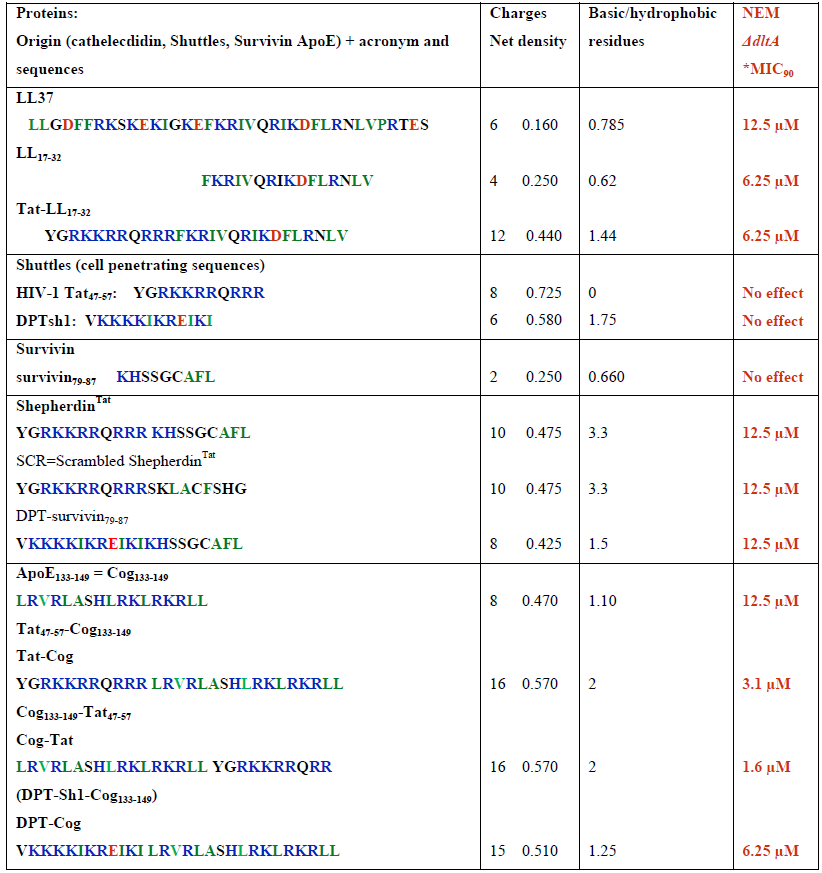
Table 1: Physical characteristics and anti-bacterial effects of cationic peptide sequences.
AA residues are expressed in one letter conventional code. Residues in red correspond to acidic residues, like D E, residues in blue correspond to basic residues, like R K H and in green correspond to hydrophobic uncharged residues, like F I L M V W A and P. °Charge density is calculated by dividing the net charge by the total number of amino acids, residues (for detailed calculation, see https:www.genescript.com/tools/peptide-properties-calculator. *The MIC (μM) of each peptide is an average of triplicate measurements performed by a dilution method in 96-well polypropylene microplate. The MICs90 was considered to be the peptide concentration that inhibited growth of 90% of the tested strains.
U87G cells were treated for 24 h with the different peptides and cell viability was assessed by MTT test (n=3). Upper panel: LL17-32, control penetrating Tat, and hybrid Tat-LL17-32 cationic sequences (0-25 μM). Middle panel: Shepherdin-Tat, SCR, DPT-survivin cationic peptides (0-150 μM), Lower panel: Cog, Tat-Cog, Cog-Tat cationic peptides (0-25 μM).
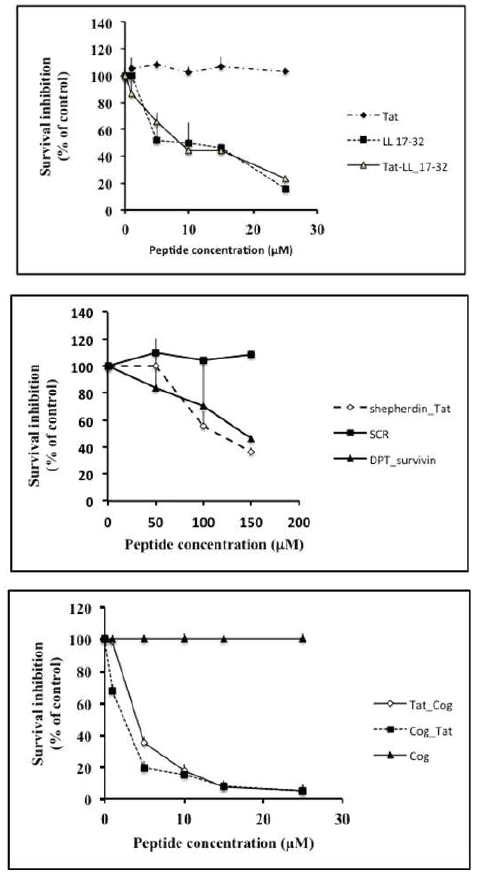
Figure 1: Effect of LL17-32, survivin and Cog mimetic peptides on viability of U87G glioblastoma cells.
4. Discussion
4.1 Host defense properties of anti-cancer peptides containing survivin and cog-mimetics sequences
In this study, we have hypothesized that shepherdin/survivin or ApoE/cog anti-tumor mimetics cationic peptides might be new host defense molecules with an additional potential antibiotic activity. The shepherdin family of Hsp90 inhibitors is derived from peptide sequence survivin79-83. This sequence or its scrambled peptide was previously made permeable to cells by adding the antennapedia helix III (shepherdin-ant) or HIV-1 Tat (shepherdin-Tat)/ scrambled shepherdin -Tat)/(SCR), cell penetrating sequences [9]. Interestingly the sherpherdin, but not the SCR molecules, can display anti-cancer activity through a dual mechanism involving the rapid disruption of mitochondrial function and decreased expression of Hsp90 client proteins, such as survivin, Akt, and CDK-6 [17]. Previous studies already indicated that treatment of glioblastoma cells with shepherdin caused the irreversible collapse of mitochondria, degradation of HSP90 client proteins in the cytosol, and tumor cell killing [18]. Hereby-in consistency with previous studies [9, 16, 17], we found that sherpherdin-Tat, but not the SCR, molecule inhibit survival of U87G cells. In contrast, and in addition to the anti-glioblastoma mediated by interaction of Sherpherdin-Tat with HSP we found an anti-Gram+ positive bacterial effect of both Sherpherdin-Tat/ SCR molecules, that share exactly the same physical cationic properties, against S. Agalactiae NEM 316 dltA strain.
ApoE, a protein initially involved the maintain of normal lipid homeostasis, modulates a variety of additional important biological functions including adaptive immune responses, host susceptibility to infection and anti-tumor [19]. The ApoE family of PP2A activators is derived from Cog133 peptide sequence and previous studies characterized anti-tumor effects of ApoE mimetics peptides [6]. Interestingly, we found, in contrast to fully inactive Cog133, that bipartite Cog-Tat or Tat-Cog penetrating peptides are potent and much more efficient inhibitors of U87G survival than shepherdin or LL17-32 peptides. However, surprisingly, while COG133 and LL17-32 peptides have similar anti-bacterial effects, Cog mimetic peptides are also more efficient inhibitors of the growth of S. agalactiae NEM 316 ΔdltA strain than Cog133 or LL17-32.
5. Conclusion
We have recently proposed that virally encoded cationic peptide sequences could penetrate human cells to deregulate important specific intracellular pathways and also could display LL37 cathelecidin-like antagonistic effects against multiple pathogens such as bacteria or viruses [13]. Similarly, in this study our results indicate that in addition to intracellular targeting of specific targets such as HSP or PP2A proteins, shepherdin or ApoE mimetics peptides may also display anti-bacterial effects based on cathelecidin like physical properties. In this regard future work involving viruses, microbes and parasites, will be necessary to study potential anti-infective effects of these new host defense molecules. In fine our data establish the rationale for the identification of a novel class host defense molecules that combined anti-tumor properties, through interfering with specific intracellular pathways, and anti-microbial effects, as a consequence of their cathelecidin-like physico-chemical properties.
Acknowledgements
The present study was supported by Institut Pasteur. The authors thank Patrick Trieu Cuot for providing S.agalactiae ΔdltA strain.
Financial Disclosure
The present study was supported by Institut Pasteur.
Conflict of Interest
The authors declare no conflict of interest.
References
- Lalazar A, Weisgraber KH, Rall SC, et al. Site-specific mutagenesis of human apolipoprotein E. Receptor binding activity of variants with single amino acid substitutions. J Biol Chem 263 (1998): 3542-3545.
- Mahley RW, Weisgraber KH, Huang Y. Apolipoprotein E: structure determines function, from atherosclerosis to Alzheimer’s disease to AIDS. J. Lipid Res 50 (2009): 183-188.
- Christensen DJ, Chen Y, Oddo J, et al. SET oncoprotein overexpression in B-cell chronic lymphocytic leukemia and non-Hodgkin lymphoma: a predictor of aggressive disease and a new treatment target. Blood 118 (2011): 4150-4158.
- Christensen DJ, Ohkubo N, Oddo J, et al. Apolipoprotein E and peptide mimetics modulate inflammation by binding the SET protein and activating protein phosphatase 2A. J Immunol 186 (2011): 2535-2542.
- Farrell AS, Allen-Petersen B, Daniel CJ, et al. The PP2A inhibitors SET and CIP2A are overexpressed in human pancreatic cancer and are important for pancreatic cancer cell growth and transformation; thus, antagonizing SET and/or CIP2A may be an innovative approach for the treatment of human pancreatic cancer. Mol Cancer Res 12 (2014): 924-939.
- Agarwal A, Mackenzie RJ, Pippa R, et al. Antagonism of SET using OP449 enhances the efficacy of tyrosine kinase inhibitors and overcomes drug resistance in myeloid leukemia. Clin Cancer Res 20 (2014): 2092-2103.
- Guergnon J, Dessauge F, Dominguez V, et al. Garcia Use of penetrating peptides interacting with PP1/PP2A proteins as a basis for a new Drug Phosphatase Technology. Mol Pharmacol 69 (2006): 1115-1124.
- Colle JH, Garcia A. The new APOE analog DPT-Cog inhibits PI3k/Akt-dependant survival of human radio-resistant U87G glioblastoma cells. Current Topics in Pharmaclogy 20 (2016): 33-37
- Plescia J, Salz W, Xia F, et al. Rational design of shepherdin, a novel anticancer agent. Cancer Cell. 7 (2005): 457-68.
- Dürr UH, Sudheendra US, Ramamoorthy A. LL-37, the only human member of the cathelicidin family of antimicrobial peptides. BBA 1758 (2006): 1408-1425.
- Ooi EH, Wormald PJ, Carney AS, et al. Fungal allergens induce cathelicidin LL-37 expression in chronic rhinosinusitis patients in a nasal explant model. Am J Rhinol 21 (2007): 367-372.
- Gwyer Findlay E, Currie SM, Davidson DJ. Cationic Host Defense Peptides: Potential as Antiviral Therapeutics. BioDrugs 27 (2013): 479-493.
- Garcia A. The Viral Quinta Columna Strategy: A new biological hypothesis to study infections in humans. Medical Hypotheses 113 (2018): 9-12.
- Colle JH, Périchon B, Garcia A. Anti-tumor and anti-bacterial properties of virally encoded cationic sequences Biologics: Target and Therapy 13 (2019): 117-126.
- Saar-Dover R, Bitler A, Nezer R, et al. D-alanylation of lipoteichoic acids confers resistance to cationic peptides in group B streptococcus by increasing the cell wall density. PLoS Pathog 8 (2012): e1002891.
- Wu WK, Wang G, Coffelt SB et al. Emerging roles of the host defense peptide LL-37 in human cancer and its potential therapeutic applications. Int J Cancer 127 (2010): 1741-1747.
- Gyurkocza B, Plescia J, Raskett CM et al. Antileukemic activity of shepherdin and molecular diversity of hsp90 inhibitors. J Natl Cancer Inst 98 (2006): 1068-1077.
- Siegelin MD, Plescia J, Raskett CM et al. Global targeting of subcellular heat shock protein-90 networks for therapy of glioblastoma. Mol Cancer Ther 9 (2010): 1638-1646.
- Vogar T, Guo N, Guly R et al. Apolipoprotein E: a potent inhibitor of endothelial and tumor cell proliferation. J Cell Biochem 54 (1994): 299-308.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 