Reconstruction of Defects of the Lower Third of the Leg and Foot With a Reverse Sural Island Flap
Sokol Isaraj*, Nardi Kola, Ina Kola
Plastic Surgeon, University Hospital Center “Mother Theresa” of Tirana, Albania
*Corresponding Author: Sokol Isaraj, Plastic Surgeon, University Hospital Center “Mother Theresa” of Tirana, Albania
Received: 04 March 2020; Accepted: 16 March 2020; Published: 23 March 2020
Article Information
Citation: Sokol Isaraj, Nardi Kola, Ina Kola. Reconstruction of Defects of the Lower Third of the Leg and Foot With a Reverse Sural Island Flap. Journal of Surgery and Research 3 (2020): 031-042.
View / Download Pdf Share at FacebookAbstract
Background: Closure of lower limb defects asks for thorough knowledge of all the reconstructive options, starting with the use of local tissues. The scarce availability of such, makes this task difficult and demanding. Unique nature of blood supply to the leg, complicates this issue even more. Reverse sural island flap is what we found handy in solving the problem for defects of small to moderate size, on the lower leg and foot.
Objectives: The aim of this study is to report our experience with the use of the reverse sural island flap in the reconstruction of complex soft tissue defects of the lower leg and the foot.
Materials and methods: Following institutional review board approval, a database search was completed to identify all patients who underwent reconstruction of distal lower extremity and foot defects with a reverse sural island flap between 2011-2019. Data were collected from the medical records of patients, as regards: demographics, history of trauma or nature of the wound, personal medical history (accompanying diseases, smoking status), location of the defect, dimensions of the flap after being elevated, pedicle length etc.
Results: 15 cases received a reverse sural island flap, all the cases except one, being acute trauma. The remaining one was a chronic wound after a previous burn. In one of the cases the flap was delayed. In three of them flap it was used to cover defects of the dorsum of the foot. Flap dimension varied from 7 × 4 cm to 15 × 12 cm, length of the pedicle from 6 to 16 cm, two of the flaps were very close to the distal popliteal crease. In all of the cases, except one, donor area of the flap needed be skin grafted. Only two of the flaps were complicated: one by a partial necrosis, the other by a venous stasis, which in the end were both treated successfully. In none of the patients a
Keywords
<p>Foot and ankle defect, Soft tissue defects, Sural flap, Neurocutaneous, Fasciocutaneous flap, Distally based sural artery flap</p>
Article Details
1. Introduction
Defects of the lower third of the leg and foot pose an important challenge to the surgical team. Introduction of microsurgery changed the approach to reconstruction in general, and even to defects formerly considered inoperable. But, under circumstances when expertise is missing and the possibility of a free flap is not an option, we have to rely on regional solutions for each case. We have found the reverse sural flap a consistent and reliable solution for most of such cases. Fasciocutaneous flaps introduced first by Ponten in 1981, are still in use for the reconstruction of soft tissue defects of the lower 1/3 of the leg and foot [1]. In 1992 Masquelet referred to the sural neurocutaneous island flap, which was based on the blood supply offered by the vascular network accompanying the sural superficial nerve [2]. Masquelet noticed that arteries of such type, on their course, branch to several vessels towards the skin, and he gave anatomical and technical details in raising such flaps. Since then, this flap found wide use all over the world, received quick recognition and yielded promising clinical results. According to various classification criteria, but mostly determined by the surgical technique for harvesting and using this flap in various defects reconstruction, the sural flap has been referred to as reverse sural artery flap [3, 4], delayed sural flap [5], supercharged reverse sural flap [6], sural fasciomusculocutaneous flap [7], distally based sural flap [8, 9], cross-leg distally based sural flap [10], distally based sural neurocutaneous flap [11], distally based sural neuro-fasciomyocutaneous flap [12], distally based sural neuro-lesser saphenous veno-fasciocutaneous compound flap [13], nerve sparing distally based sural fasciocutaneous flap [14] etc. We have referred to it in our practice as the Reverse Sural Island Flap (RSIF) and will do so in this paper which is a review of the application of the flap at the Service of Burns and Plastic Surgery, University Hospital Center “Mother Theresa” of Tirana.
The reverse sural flap permits the soft tissue reconstruction without the need for microsurgery. A number of studies have established the utility of the RSIF in lower extremity and foot reconstruction over the past 30 years [15].
2. Materials and Methods
Following institutional review board approval, a database search was completed to identify all patients who underwent reconstruction of distal lower leg and foot defects with a RSIF between 2011-2019. Two cases were excluded from the material for lack of specific data.
Data were collected from the medical records of patients, as regards:
- demographics,
- history of trauma or nature of the wound,
- personal medical history (accompanying diseases, smoking status),
- location of the defect,
- dimension of the flap after being elevated,
- pedicle length,
- angle of rotation from the donor site to the defect,
- length of the surgical procedure,
- time lap from the moment of trauma to the moment of flap application,
- duration of hospital stay,
- number of operations per patient,
- final result.
Data were analyzed using simple mathematical averages, sample size didn’t allow us to make more detailed statistical processing.
Preoperatively, the design of the flap was done depending on the characteristics of the defect: pivot point of the flap was marked 5cm above the lateral malleolus. Pedicle length was designed almost equal with the distance from the proximal side of the defect to the distal side of the future flap. Flap design was approximated to the geometry of the defect to be reconstructed, adding to the size of the defect the intended extension of wound debridement. The more distal the defect, the closer to the popliteal crease was the proximal side of the designed flap. Flap was centered over the course of the lesser saphenous vein, which was generally easily identified after the application of a simple tourniquet, the same as the one that we use for i/v cannulation (Figure 1).
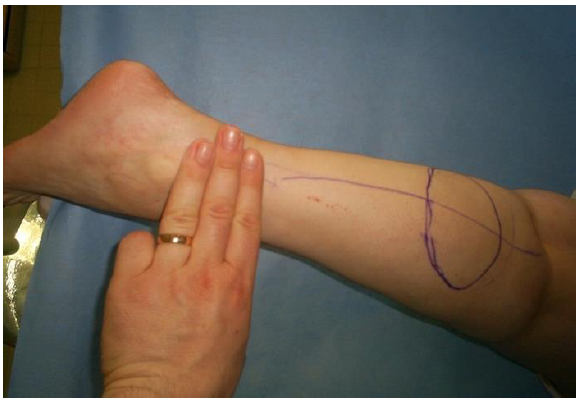
Figure 1: Design of the reverse sural island flap; pivot poing 5 cm above the lateral malleolus, or three finger breadth from it; pedicle designed over the course of the lesser saphenous vein, easily identified; shape and dimensions of the flap depends on the size and location of the defect.
Intervention was started after applying a tourniquet on the thigh, with the patient on the prone position or lateral position, in accordance with the location of the defect to be reconstructed. Debridement of the wound was done first and then incision on the course of the pedicle. Skin flaps were reflected on both sides, remaining over the superficial fascia on the intended pedicle and then flap was raised starting from the proximal edge, having always in its center the lesser saphenous vein. Elevation of the flap and pedicle was done under the deep fascia. Tourniquet was deflated, and viability of the flap was assessed carefully with visual assessment of the flap marginal capillary circulation. Then flap was rotated to the defect, through an incision that connects the edge of the defect with the pivot point. Flap is never tunneled, to avoid congestion and venous stasis, and finally sutured to the defect avoiding tension. Skin graft was used to cover the donor site and the exposed parts of the pedicle. The entire limb is dressed, so that a “window” can be created to expose the flap surface, for vitality monitoring.
3. Results
15 cases were treated by the authors between 2011-2019 with a reverse sural island flap for defects located on the lower third of the leg and foot. All the cases except three of them, were acute trauma. The remaining ones were postburn cases, after burns sustained years before. Table 1 shows data on the age, sex, anesthesia and location of the defect to be reconstructed. The average age of the patients was 34, 6 years: the youngest being 10 and the oldest 67 years old. Only 4 of our series were females (27% of them), the majority being males of active age. Most of the cases were done under general anesthesia. The defect was located on medial malleolus in 6 cases, dorsum of the foot in 3 cases, distal third of the leg in 2 cases, calcanear region in 3 cases, and anterior ankle in one.
Flap dimensions varied from 7 × 4 cm to 15 × 12 cm, length of the pedicle from 6 to 16 cm, two of the flaps were very close to the distal popliteal crease. In almost all of the cases donor area of the flap needed to be skin grafted, except one case where donor site was closed primarily and exposed part of the pedicle, covered with postage stamp size grafts, provided after debridement of the initial defect. In none of the patients was a detailed examination of vascular supply of the area deemed reasonable, all of them being nondiabetic and nonischemic legs. Average Time Lap from the moment of creation of the defect, to the application of the sural flap resulted 61 dyas. It was averaged excluding three cases (post burn cases) on which the initial trauma was sustained many years ago.
Mean hospital stay of the patients results 32 days, which in fact seems a lot. But this is mainly explained with the fact that most of the patients admitted needed considerable preoperative workup, and in most of the cases the wound at the moment of admission was not ready to receive the flap. This mandated delay of the operation until a favorable local condition was achieved. Only two of the flaps were complicated: one by a partial necrosis and the other by a venous stasis. Both of which in the end were treated successfully. In one patient more than two procedures were applied. The original defect was first attempted to be closed by a perforator flap from the posterior tibial perforators. Flap died because of venous congestion. After debridement, a delayed reverse sural flap was elevated, and the procedure was completed after 3 weeks with transfer of the delayed flap and skin graft. Healing of the flap was uneventful. Altogether four procedures were applied: elevation of the flap, two delay procedures in intervals of 10 days from each other, and three weeks later the flap was raised and final reconstruction completed (Figure 2).
There were only two cases where complications were encountered, but which both finally healed successfully. Flap suffering in one of the cases, was mainly due to the fact that the donor area of the flap was partially a granulated wound, so the flap was raised only from the 3/4th of the leg lateral side, to be transferred to a small area on the medial part of the ankle, on an exposed fractured medial malleolus. Flap necrosis was only marginal. No extra procedure was needed, and the small wound created after the necrosis healed secondarily (Figure 3).
On the other case, the flap was raised on a lipoedematous leg, with severe scarring, due to a trauma sustained 30-years ago. Patient had been suffering from a trophic ulcer on the medial malleolus for more than 4 years; bone was exposed on the wound bed. After considering all the reconstructive options, a reverse sural island flap was scheduled, in spite of the spread scarring on the leg. We did so, because the area where the anastomosis between median superficial sural artery and peroneal artery was scar-free. The dimensions of the flap were 12 × 8 cm, length of the pedicle 13.5 cm and the proximal border of the flap 7 cm from the popliteal crease. The flap was rotated 180°to the defect, covering it completely. Venous congestion was seen mainly on the lateral and medial part of the flap, and these areas were necrotised. The reason for this partial failure was the difficulty of the retrograde venous flow, on a leg, where venous circulation was problematic because of the lipoedema and spread scars (Figure 4). And the last case was done with direct closure of the donor area of the flap (Figure 5).
|
Cases |
Age |
Gender |
Smoking |
Anaesthesia |
Location of defect |
|
1 |
38 |
M |
No |
Spinal |
Medial Malleolus |
|
2 |
67 |
M |
No |
Spinal |
Medial Malleolus |
|
3 |
10 |
M |
No |
Endotracheal |
Distal third of tibia |
|
4 |
15 |
F |
No |
Endotracheal |
Medial malleolus |
|
5 |
22 |
M |
No |
Endotracheal |
Dorsum of foot |
|
6 |
53 |
F |
No |
Endotracheal |
Medial malleolus |
|
7 |
51 |
M |
No |
Endotracheal |
Lateral calcanear |
|
8 |
43 |
F |
No |
Endotracheal |
Calcanear region |
|
9 |
22 |
M |
No |
Endotracheal |
Distal third of leg |
|
10 |
23 |
M |
Yes |
Endotracheal |
Dorsum of foot |
|
11 |
34 |
M |
No |
Endotracheal |
Dorsum of foot |
|
12 |
40 |
M |
Yes |
Endotracheal |
Anteriolr ankle |
|
13 |
43 |
M |
Yes |
Endotracheal |
Medial malleolus |
|
14 |
29 |
M |
Yes |
Endotracheal |
Medial malleolus |
|
15 |
37 |
F |
No |
Endotracheal |
Calcanear region |
|
|
Avg=34,6 |
73.3% males |
80% nonsmokers |
87% endotracheal |
|
Table 1: General data on demographics, anaesthesia, location of the defect to be reconstructed.
|
1 |
Flap dimensions |
Flap rotation |
Pedicle length |
Duration of procedure |
Time lap |
Hospital stay |
More than 1 procedure |
|
8 × 5 cm |
170º |
14 cm |
1h35’ |
44 days |
44 |
N |
|
|
2 |
9 × 5 cm |
170º |
13 cm |
1h45’ |
1 year |
28 |
N |
|
3 |
5 × 5 cm |
110º |
10,5 cm |
1h20’ |
15 days |
40 |
N |
|
4 |
7 × 4 cm |
170º |
15 cm |
2h10’ |
14 days |
55 |
N |
|
5 |
15 × 12 cm |
170º |
16 cm |
2h15’ |
39 days |
15 |
N |
|
6 |
12 × 8 cm |
170º |
13,5 cm |
2h30’ |
30 years |
60 |
Y |
|
7 |
12 × 8 cm |
170º |
13 cm |
2h45’ |
5 years |
67 |
Y |
|
8 |
5 × 4 cm |
150º |
6 cm |
1h20 |
51 days |
26 |
N |
|
9 |
7 × 5 cm |
160º |
8 cm |
2h10’ |
60 days |
18 |
N |
|
10 |
15 × 8 cm |
180º |
15 cm |
2h45’ |
21 days |
28 |
Y |
|
11 |
7 × 4 cm |
180º |
14 cm |
2h30’ |
161 days |
19 |
N |
|
12 |
10 × 8 cm |
150º |
7 cm |
1h55’ |
57 days |
15 |
N |
|
13 |
6 × 3 cm |
170º |
12 cm |
2h10’ |
90 days |
60 |
Y |
|
14 |
7 × 4 cm |
160º |
13 cm |
1h45’ |
180 days |
8 |
N |
|
15 |
8 × 9 cm |
160º |
12,5 cm |
2h5’ |
2 days |
5 |
N |
|
Average |
58.4 cm2 |
170º |
11.75 cm |
2h4’ |
61 days |
32 days |
27% |
Mean surface area of the flap was 58.4 cm2
Table 2: Characteristics of the flaps.
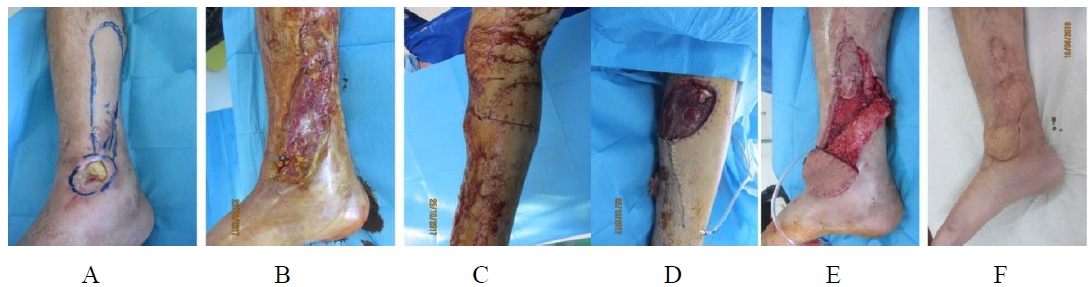
Figure 2: Exposed bone, medial malleolus (A), after failure of a perforator flap of the posterior tibial artery (B) where hardware is also exposed, planned to be covered with a delayed reverse sural island flap (C). Flap was raised after the delay (D), and rotated covering the exposed bone and hardware (E), and completely healed (F).
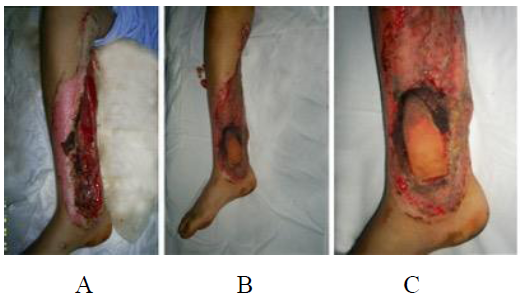
Figure 3: (A) Large wound on the right leg, exposed fractured medial malleolus, (B) Exposed malleolus is covered with a RSIF, the rest of the wound with a STG, (C) Marginal small necrosis of the flap is observed, which eventually healed spontaneously.
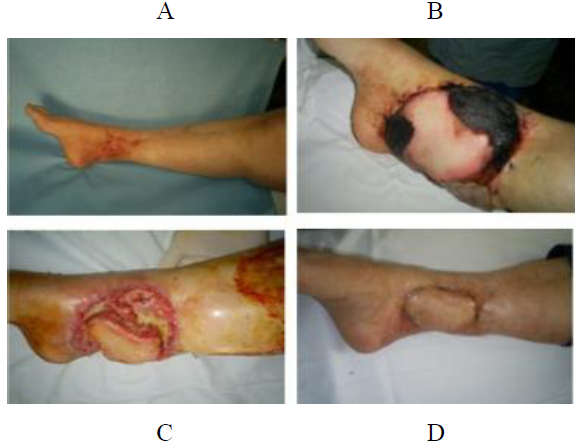
Figure 4: Chronic wound on the medial side of ankle with exposed medial malleolus (A), planned to be closed with a reverse sural island flap, resulted in partial necrosis of the flap because of the venous congestion (B). The resulting wound (C) was left to heal by secondary intention (D).
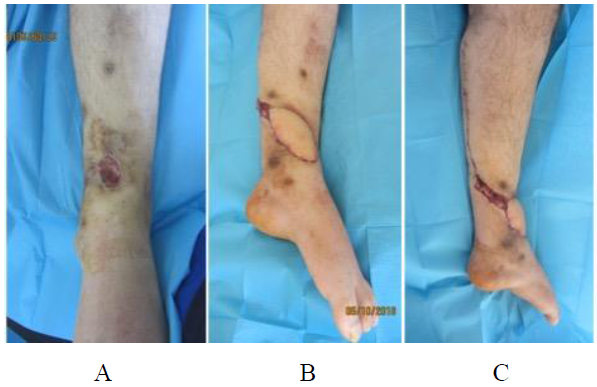
Figure 5: Chronic wound on the anteromedial part of the left angle (A) covered with a reverse sural island flap (B), whose donor site was closed primarily (C).
4. Discussion
Procedures described for the coverage of defects in distal third of the leg and foot, vary broadly, from the simple application of a split thickness skin graft [16, 17] to all kind of different flaps, from the muscle flaps [18, 19] septocutaneous flaps [20, 21], axial flaps [22, 23] and ending with the free flaps [24-26]. As a matter of fact, distal third of the leg is the domain of free flap in all the facilities where microsurgery can be performed. But if microsurgery is not possible, as is our case, such a rule is not valid. Generally, free flaps are superior to other methods because they allow reconstruction with well vascularized tissues. However, they required sophisticated infra-structure, well-trained surgical team and equipments. In any case, goals of the reconstruction are to provide stable soft-tissue coverage, preserve sensation, and allow for bipedal ambulation with normal weight bearing. RSIF is often said to have a favorable complication profile as evidenced by a recent meta-analysis that found 82% of flaps heal without any flap-related complications. The flap is strong enough even for heel coverage [27].
4.1 Anatomical basis of flap
Since the discovery of the distally based sural artery flap in 1992 by Masquelet et al. [2], the flap has been used to cover wound defects of the distal third of the leg. The distally based superficial sural artery flap is supplied by a superficial sural artery through a reverse flow, which takes contribution from the septocutaneous perforators of the peroneal artery in the distal part of the leg. As a matter of fact, in the calf midline, there is another structure that stays close to the sural nerve (SN), the lesser saphenous vein (LSV), supposedly included in all of the above-mentioned flaps. LSV somehow defines the course of the sural nerve, and plays an important role in draining the flap, but none of the above authors referred to any accompanying artery of the vein itself as contributing to the blood supply of the flap. In Pernkopf anatomy [28], LSV is depicted together with an unnamed artery. In Salmon Anatomy [29], the lesser saphenous artery is mentioned as a satellite of the LSV, but it is unclear if this is different from the artery accompanying the sural nerve.
On the posterior part of the lower leg, the accompanying artery of the sural nerve is named the sural superficial median artery (SSMA) and the accompanying artery of the lateral cutaneous sural nerve is named the sural superficial lateral artery (SSLA) [30]. Nakajima et al., studied in depth the anatomy of the accompanying artery of the LSV and its relations to the accompnanying artery of the SN [31]. They created theoretically a pedicled fasciocutaneous adipofascial flap, using the accompanying artery of the LSV and/or SN and named them respectively VAF (=venoadipofascial pedicled fasciocutanous flap of the lesser saphenous vein), NAF (=pedicled fasciocutaneous neuroadipofascial), VN-AF (pedicled fasciocutaneous venoneuroadipofascial of the LSV - SN). Of all these, VAF and VN-AF found clinical use.
Anatomical relations between LSV and SN are important for the topography of the flap. LSV originates in the rete venosum of the lateral malleolus and ascends to the calcanear tendon above the deep fascia. On the upper fourth of the calf, it enters the deep fascia and lies in the adipofascial tissues below it. Even here it is covered by a fascial sheath and lastly it drains into the popliteal vein. The SN, surrounded by a fascial sheath ascends in the adipofascial tissues above deep fascia, together with the LSV in the lower half of the leg. It enters the deep fascia at the middle point of the calf. In the proximal fourth of the calf, it ascends within the sheath that covers the LSV, usually dipped between the heads of the gastrocnemius muscle, and reaches the tibial nerve in the popliteal fossa. The peroneal communicating branch joins the SN above the deep fascia and in vicinity to the Calf Middle Point.
Masquelet flap can be considered a venoneurocutaneous flap more than a neurocutaneous flap. Nakajima advanced further the concept of the neurocutaneous flap to a venocutaneous, neurocutaneous and venoneurocutaneous and proposed a fasciocutaneous flap with an adipofascial pedicle depending on the accompanying arteries of the vein and/or nerve [32]. Naming these flaps as “adipofascial” doesn’t sound very familiar. However, this term is being used more frequently nowadays [33, 34, 35]. In all of our cases, the sural nerve has been always sacrificed, and most of patients complain of a slight paresthesia of the lateral aspect of the foot. This is one of the drawbacks of this kind of flap, although all of the patients deal very well with such a situation. Long-term follow-up of patients (11 of them) showed that none of them had sensation complaints of the above-mentioned type.
The exact name of the flap is still a matter of debate. Hasegawa considers the term “neurocutaneous flap” proposed by Masquelet as inappropriate and would rather call it the “distally based superficial sural artery flap [36]. Cavadas and Bonanand proposed the term “reverse flow sural island flap”, which sounds adequate to us, knowing the fact that the flap is not innervated and because the term “distally based” doesn’t say anything regarding the reverse flow of the flap [37]. Raising a reverse sural island flap is a much easier effort than raising the peroneal or anterior tibial flaps, the latters being supplied by deeper arteries and veins, hence less invasive because the major vessels are left undisturbed. Septocutaneous flaps, distally based, might show severe signs of congestive suffering, especially those based on the anterior tibial artery. But, 23 flaps of Nakajima et al., distally based, very rarely suffered of venous congestion. Furthermore, even in cases where such signs were present, the adipofascial layer of the flap survived very well, enabling covering of the remaining wound. So, these flaps can be considered safer than the peroneal and the anterior tibial artery flaps [31].
Torii et al. pretend that the retrograde flow occurs in each of the major arteries, even in the collateral branched of the posterior tibial artery [38]. Most of the authors agree that anastomosis occur mostly at a level 5cm above the lateral malleolus [2, 38, 39]. As for the venous drainage, the presence of a considerable number of valves on the lower leg, theoretically wouldn’t allow the retrograde flow. However, preliminary studies of more than one author, demonstrated the contrary [36, 40, 41]. However, the RSIF is often at risk for venous congestion, as it relies on communications between the venae comitantes of the sural nerve and the lesser saphenous vein, thus circumventing the valves of the deep venous system [42]. The explanation of this phenomenon can be found in several theoretical assumptions.
Lin emphasizes the existence of bridges that enable the connection beyond the valves in the venae commitante of radial artery [43]. According to Timmons denervation of the radial vein makes their valves incompetent and allowing so the retrograde flow [44]. Wee too, confirms the same for the venae comitantae of the anterior tibial artery, but emphasizes the importance of undermining beyond the pivot point. Undermining causes denervation of all the veins along their course [40]. Torii et al. found that the pressure gradient needed to overcome the valvular barrier can never be achieved inside the veins that drain the upper and lower limb. If such an assumption is true, all flaps of such type couldn’t survive, which in fact is not true [38]. According to Hasegawa how this type of flap drains, is still not clear [36].
Use of Duplex scan testifies to the fact that in all of the cases venous drainage occurs through the lesser saphenous vein. In patients where this examination was repeated, was seen that the slow, strained and continuous flow in the immediate postoperative period, became phasic and more physiologic in a later stage. These findings explain why the initial venous congestion encountered in some patients, was spontaneously and gradually improved [43]. Many techniques are described to decrease venous congestion including opening intervening skin, venous supercharging and exteriorizing pedicle [44].
In a paper from Almedia et al., 71 patients were treated with a reverse sural island flap. Only 3 of the cases (4, 2%) were considered unsuccessful, total necrosis of the flap. This is one of the largest series of patients and our results seem approximately the same with only a partial failure of the flap and two marginal necrosis [43]. There are studies to confirm that the selection of the cases must be very careful when applying a RSIF, to yield better results and less complication rate. Disadvantages of the flap are very few, positioning of the patients during the operation, and especially change of the position during operation in a number of cases; sensory changes on the lateral aspect of the foot (which was encountered in almost all cases); bulkiness of the flap when it comes to be applied on the foot, this was corrected in one of our cases by debulking and liposuction 6 months after the application of the flap.
5. Conclusion
Reverse sural island flap is, as a conclusion, a very useful tool for the reconstruction of small to moderate defects of the lower leg and foot, easy to be raised, one-stage rapid procedure with no special needs for complicated vascular preoperative workup, blood loss is minimal & preservation of the major vascular structure of the lower limbs is possible, and with a very low rate of failure and complications in carefully selected patients. Defects to be reconstructed vary from small to moderate size, and the flap variation from, skin, fascial, adipofascial with or without skin grafting the donor site.
References
- Ponten B. The fasciocutaneous flap. Its use in soft tissue defects of the lower leg. Br J Plast Surg 34 (1981): 215-220.
- Masquelet AC, Romana MC, Wolf G. Skin island flaps supplied by the vascular axis of the sensitive superficial nerves: Anatomic study and clinical experience in the leg. Plast Reconstr Surg 89 (1992): 1115-1121.
- Price MF, Capizzi PJ, Watterson PA, et al. Reverse sural artery flap: caveats for success. Ann Plast Surg 48 (2002): 496-504.
- Hong JP, Neligan PC, Song DH. Reconstructive surgery: lower extremity coverage. Plastic surgery 4 (2013): 127-150.
- Kneser U, Bach AD, Polykandriotis E, et al. Delayed reverse sural flap for staged reconstruction of the foot and lower leg. Plast Reconstr Surg 116 (2005): 1910-1917.
- Tan O, Atik B, Bekerecioglu M. Supercharged reverse-flow sural flap: a new modification increasing the reliability of the flap. Microsurgery 25 (2005): 36-43.
- Chen SL, Chen TM, Wang HJ. The distally based sural fasciomusculocutaneous flap for foot reconstruction. J Plast Reconstr Aesthet Surg 59 (2006): 846-855.
- Follmar KE, Baccarani A, Baumeister SP, et al. The distally based sural flap. Plast reconstr Surg 119 (2007): 138-148.
- Aoki S, Tanuma K, Iwakiri I, et al. Clinical and vascular anatomical study of distally based sural flap. Ann Plast Surg 61 (2008): 73-78.
- Eser C, Kesiktas E, Gencel E, et al. An alternative method to free flap for distal leg and foot defects due to electrical burn injury: distally based cross-leg sural flap. Ulus Travma Acil Cerrahi Derg 22 (2016): 46-51.
- Yilmaz M, Karatas O, Barutcu A. The distally based superifical sural artery flap. Plast Reconstr Surg 102 (1994): 1012-1020.
- Chang SM, Zhang K, Li HF, et al. Distally based sural fasciomyocutaneous flap: anatomic study and modified technique for complicated wounds of the lower third leg and weight bearing heel. Microsurgery 29 (2009): 205-213.
- Zhang F, Lin S, Song Y, et al. Distally based sural neuro-lesser saphenous venofasciocutaneous compound flap with a low rotation point: microdissection and clinical application. Ann Plast Surg 62 (2009): 395-404.
- Aydin OE, Tan O, Kuduban SD, et al. Nerve sparing-distally based sural flap. Microsurgery 31 (2011): 276-280.
- Costa-Ferreira A, Reis J, Pinho C, et al. The distally based island superficial sural artery flap: clinical experience with 36 flaps. Ann Plast Surg 46 (2001): 308-313.
- Lister GD. Use of an innervated skin graft to provide sensation to the reconstructed heel. Plast Reconstr Surg 62 (1978): 157-161.
- Maquieira NO. An innervated full-thickness skin graft to restore sensibility to finger tips and heels. Plast Reconstr Surg 53 (1974): 568-575.
- Hartrampf CR Jr, Scheflan M, Bostwick J. The flexor digitorum brevis muscle island fl aps. A new dimension in heel reconstruction. Plast Reconstr Surg 66 (1980): 264-270.
- Townsend PL. An inferiorly based soleus muscle flap. Brit J Plast Surg 31 (1978) 210-213.
- Yoshimura M, Imura S, Shimamura K, et al. Peroneal flap for reconstruction in the extremity. Plast Reconstr Surg 74 (1984): 402-409.
- Chang SM, Zhang F, Yu GR, et al. Modified distally based peroneal artery perforator flap for reconstruction of foot and ankle. Microsurg 24 (2000): 430-436.
- Lin SD, Chou CK, Lin TM, et al. The distally based lateral adipofacial flap. Brit J Plast Surg 51 (1998): 96-102.
- Voche P, Merle M, Stussi JD. Lateral supra-malleolar flap: Experience with 42 flaps. Ann Plast Surg 54 (2005): 49-54.
- Meland NB. Microsurgical reconstruction: The weight bearing surface of the foot. Microsurg 11 (1990) 54-58.
- Goldberg JA, Adkin P, Tsai TM. Microvascular reconstruction of the foot: weight-bearing pattern gait analysis and long-term follow up. Plast Reconstr Surg 92 (1993): 904-911.
- Chen SL, Chuang CJ, Chou TD, et al. Free medial sural artery perforator flap for ankle and foot reconstruction. Ann Plast Surg 54 (2005): 39-43.
- Ayyappan T, Chadha A. Super sural neurofasciocutaneous flaps in acute traumatic heel reconstructions. Plast Reconstr Surg 109 (2002): 2307-2313.
- Eduard Pernkopf. Atlas of Topographical and Applied Human Anatomy. Ed.: Ferner H. Philadelphia: Saunders (1964).
- Michel Salmon. Arteries of the Skin. Eds.: Taylor GI, Tempest MN. London: Churchil-Livingstone (1988): 61-62.
- Cormack GC, Lamberty BGH. The Arterial Anatomy of Skin Flaps. Edinburgh: Churchill Livingstone (1986): 380-382.
- Nakajima H, Imanishi N, Fukuzumi S, et al. Accompanying arteries of the lesser saphenous vein and sural nerve: anatomic study and its clinical applications. Plast Reconstr Surg 103 (1999): 104-120.
- Nakajima H, Imanishi N, Minabe T. Neuro and/or Venoadipofascial Pedicled Flap. Presented at the 36th Congress of Japan Society of Plastic and Reconstructive Surgery, Morioka (1993).
- El-Khatib H, Zeidan M. Island adipofascial flap based on distal perforators of the radial artery: An anatomic and clinical investigation. Plast. Reconstr. Surg 100 (1997): 1762-1766.
- Lai CS, Tsai CC, Lin SD. The reverse lateral arm adipofascial flap for elbow coverage. Ann Plast Surg 39 (1997): 196-200.
- Nakajima H, Fujino T, Adachi S. A new concept of vascular supply to the skin and classification of skin flaps according to their vascularization. Ann Plast Surg 16 (1986): 1-19.
- Hasegawa M, Torii S, Katoh H, et al. The distally based superficial sural artery flap. Plast Reconstr Surg 93 (1994): 1012-1020.
- Cavadas PC, Bonanand E. Reversed- flow sural island flap in the varicose leg . Plast Reconstr Surg 98 (1996): 901-902.
- Torii S, Namiki Y, Mori R. Reverse-flow island flap: Clinical report and venous drainage. Plast Reconstr Surg 79 (1987): 600-609.
- Fachinelli A, Masquelet A, Restrepo J, et al. The vascularized sural nerve: Anatomy and surgical approach. Int J Microsurg 3 (1981): 57.
- Wee JTK. Reconstruction of the lower leg and foot with the reverse pedicle anterior tibial flap: Preliminary report of a new fasciocutaneous flap. Br J Plast Surg 39 (1986): 327-337.
- Thatte RL. One-stage random pattern deepithelialized “turn over” flaps in the leg. Ann Acad Med Singapore 12 (1983): 441-445.
- Imanishi N, Nakajima H, Fukuzumi S, et al. Venous drainage of the distally based lesser saphenous-sural veno-neuroadipofascial pedicled fasciocutaneous flap: a radiographic perfusion study. Plast Reconstr Surg 103 (1999): 494-498.
- Almeida MF, da Costa PR, Okawa RY. Reverse-flow island sural flap. Plast. Reconstr. Surg 109 (2002): 583-591.
- Fujiwara M, Nagata T, Matsushita Y, et al. Delayed distally based sural flap with temporary venous supercharging. Microsurgery 33 (2013): 534-538.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 