Spontaneous Hemothorax of Tumoral Origin Complicating a Von Recklinghausen's Disease: Report of Two Cases in Cameroon and Litterature Review
LM Endale Mangamba*, 1, AD Balkissou2, V Tongue Essoh3, AD Batchom3, B Barche4, JP Engbang Ndamba3, BH Mbatchou Ngahane3
1Faculty of health sciences, University of Buea, Cameroon
2Faculty of Medicine and Biomedical Sciences of Garoua, University of Ngaoundéré, Cameroon
3Faculty of Medicine and Pharmaceutical Sciences, University of Douala, Cameroon
4Laquintinie Hospital of Douala, Cameroon
*Corresponding author: LM Endale Mangamba, Faculty of health sciences, University of Buea, Cameroon.
Received: 03 August 2022; Accepted: 12 December 2022; Published: 27 December 2022
Article Information
Citation: LM Endale Mangamba, AD Balkissou, V Tongue Essoh, AD Batchom, B Barche, JP Engbang Ndamba, BH Mbatchou Ngahane. Spontaneous Hemothorax of Tumoral Origin Complicating a Von Recklinghausen's Disease: Report of Two Cases in Cameroon and Litterature Review. Fortune Journal of Health Sciences 5 (2022): 610-616.
View / Download Pdf Share at FacebookAbstract
Von Recklinghausen's disease also known as neurofibromatosis type 1 (NF1), is one of the most common genetic diseases. It is responsible of the development of neurofibromas from nerve sheaths and very typical skin lesions. Chest damage according to the literature is rare. Spontaneous hemothorax is a subcategory of hemothorax that involves the accumulation of blood in the pleural space in the absence of trauma or other causes. This clinical entity secondary to vascular involvement is rarely described in the literature.
We present the cases of 2 young patients, with neurofibromatosis type 1 (NF1), without dermatological follow-up, or any contributive past medical history, with both massive spontaneous hemothorax. The first is a 26-year-old female nurse student, who consult for rapid worsening dyspnea over a week, associated with a dry and painful cough. She had no fever, no weight loss. Physical examination objectified NF1 lesions with coffee-milk stain type of skin neurofibromas, massive left fluid pleural effusion confirmed by imaging with a tumor of the posterior mediastinum. The second patient was 37 years old man, unemployed stable at the admission, also have a voluminous plexiform neurofibroma. Their evolution after pleural drainage was favorable. Patient with neurofibromatosis type1should be follow by dermatologist and refer in case of thoracic involvement. Spontaneous hemothorax are not exceptional, and can be associated with a tumor.
Keywords
<p>Recklinghausen's disease, Neurofibromatosis, phacomatosis, hemothorax, neurofibroma</p>
Article Details
Introduction
Neurofibromatosis is a group of three heterogeneous disorders that include neurofibromatosis type 1 (NF1), neurofibromatosis type 2 (NF2) and schwannomatosis. NF1 still known as Von Recklinghausen's disease is the most common phacomatoses [2] [3]. Its transmission occurs under the autosomal dominant mode. In 1988, the consensus conference of the National Institutes of Health in Bethesda, United States of America, clarified 7 cardinal criteria for the diagnosis of NF1. It is posed during the association of 2 criteria among the following: a first-degree relative with NF1, presence of 6 café-au-lait spots, 2 neurofibromas or 1 plexiform neurofibroma, axillary or inguinal lentigines, 2 Lisch nodules, characteristic bone lesions or glioma of the optic tract [4].
This condition is associated with multi-organ complications including damage of the nervous system, eyes, skin and bones. [5] People with NF1 have an increased risk of developing many pathologies, including cancers with cancers of the nervous system in the first place. Chest involment is however rare, 5-10% depending on the series. These are mainly diffuse interstitial lung diseases, lung cancers and pulmonary hypertension [6]. Fdil et al. in 2017, Ben Saad et al. in 2018, and Miura et al in 2003 reported cases of spontaneous pneumothorax in young subjects in Morocco. We did not find any studies on the subject in the sub-Saharan literature. [9]. We report 2 cases of massive spontaneous hemothorax in patients with NF1 since childhood who received conservative treatment.
2. Case Description
2.1 Case 1
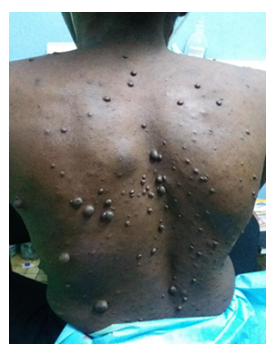
This is a 26-year-old patient, nursing student, with no contributory history, who came to consult for rapid worsening dyspnea for a week, associated with a dry and painful cough. She had no fever, no hyperperspiration, no weight loss. Physical examination objectified NF1 lesions with coffee-milk stain type of skin neurofibromas, (Figure 1), massive left fluid pleural effusion confirmed by imaging.
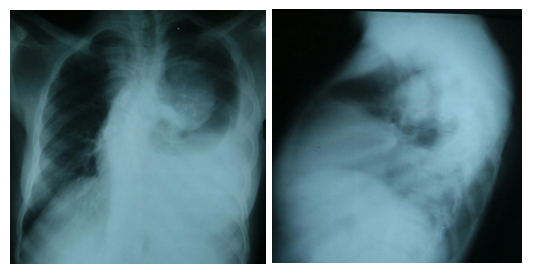
On the chest X-ray, there was a dense and homogeneous opacity erasing the left pleural sac with on the same side, a coarsely rounded, voluminous opacity, with clear limits postero-medium, mediastinal, which may be related to neurofibroma (Figure 2).
The patient also had moderate anemia at 7.5g/dL. Pleural fluid PCR by GeneXpert MTB/RIF was negative. The investigation for the presence of malignant cells within the pleural fluid was also negative. The patient received 2 pound of blood cell caps and a thoracocentesis has been done with drainage of 2800 ml of non-coagulable blood fluid. The evolution was simple clinically and radiologically with regression of dyspnea and cough and almost complete radiological cleaning of previous lesions until its discharge 5 days after removal of the drain.
2.2 Case 2
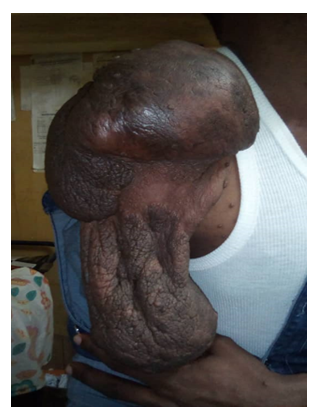
The second patient was 37 years old man, with NF1 diagnosed since the age of 5, without follow-up, nor any other contributory history. For several months, he had been experiencing progressive, painless swelling developed from one of the neurofibromas of the right shoulder (Figure 3).
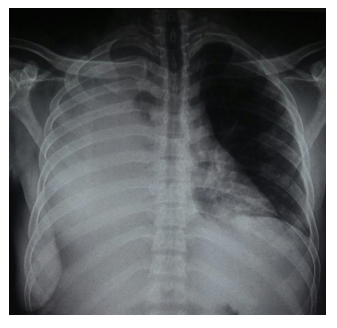
Faced with the rapid increase and the progressively disabling character with limitation of the function of the right shoulder, the patient consulted in a health facility near his home where an ablation of the largest portion of the tumor was done, without pathological examination. The evolution was marked a week later by the occurrence of rapidly worsening respiratory distress with dry cough that motivated an emergency consultation. On physical examination, NF1 was confirmed by the presence of disseminated neurofibromas of varying size and a giant plexiform neurofibroma of the right arm. The chest examination was in favor of a well-tolerated right massive fluid effusion confirmed by chest X-ray (Figure 4).
There were no malignant cells in the pleural fluid. The patient received local care for the surgical wound, chest drainage of 4300 ml of blood fluid. Clinical improvement and return to the lung wall motivated the patient's discharge on the 7th day of discharge 5 days later.
3. Discussion
NF1 is a rare genetic disease with an estimated prevalence of 1/3000 in the general population [10]. The diagnosis is easy with recognition of typical skin lesions associated or no with a family history of NF1. Although without family history of known cases of NF1, our two patients both had café-au-lait lesions on the body, particularly in the chest and the second had more advanced lesions including a large plexiform neurofibroma. Chest involvement during NF1 is rare and found in the literature as case-reports. [11] [12]. The most common causes of respiratory distress on NF1 usually include interstitial diseases and pulmonary hypertension. [6]. Spontaneous hemothorax complicating NF1 are rarely described. In the African literature, less than 10 cases have been described over more than 50 years [References]. NF1 lesions predispose to vascular complications including strictures, arterial ectasias and aneurysms [13]. In order to a better understanding of this clinical entity we reviewed the literature of less than 10 years on PubMed using the terms "Spontaneous hemothorax", "Neurofibromatosis type 1", and "Recklinghausen's disease". Of the 22 clinical cases reported, we did not notice a predominance over sex. The average age of patients ranged from 31 years in the work of Ben Saad et al., to 73 years in that of Gilvazquez et al. [14], [15]. The patients in our study were young. The diagnosis of hemothorax on NF1 is usually easy on clinical examination with the presence of a franc pleural effusion syndrome. The exploration pleural puncture highlight a blood fluid that does not coagulate, while a thorough examination makes it possible to find skin lesions typical of this phacomatosis. Management of pleural involment depends on the initial severity of the patient, the etiology involved when it is found and the technical platform. Our patients have benefited from conservative treatment with a good initial outcome. Fdil et al in Morocco in 2017 noted an evolution marked by the superinfection of the hemothorax and the patient benefited from a decortication with good secondary evolution. That of Degbelo et al was discharged from hospitalization after 8 days without immediate complication and a CT scan performed 1 month after the event, revealed a post-drainage pleural scar. [9] [16]. However, 18/22 patients (81.8%) received Video assisted thoracoscopic surgery video assistance (VATS) management for the most part. Two patients died prior to management. The basic treatment of spontaneous hemothorax is chest drainage combined with associated measures depending on the patient's condition at admission. After insertion of the chest tube, some patients may be potential candidates for VATS or thoracotomy [17]. In limited resources setting, some technical procedures are not available. An initial severity was rarely found, but is not exceptional. Both of our patients were stable at admission. D'Erriko et al in 2018 and Bidal et al in 2019 described severe cases from the onset with a fatal course, both on a ruptured aneurysm [18][13]. A neurofibroma-like tumor as found in our second patient or a mediastinal tumor whose histology must be specified for better management is most often at the origin of bleeding, more rarely vascular involvement of large or small arteries is responsible [3]. In our review, vascular causes were in the majority of the cases published. 20/22 cases (91%), in a patient the etiology could not be formally established despite the investigations made and a patient died before the diagnostic and therapeutic procedures.
4. Conclusion
Spontaneous hemothorax is not an exceptional complication of chest involvement during neurofibromatosis type I. Its occurrence in a patient can the patient's vital prognosis at stake. Management is usually surgical. But in hemodynamically stable patients, conservative treatment can be considered. Patients with this condition should be given special regular follow-up, including respiratory assessment.
Conflict’s D’interet
Authors do not declare any conflict of interest
Contribution Des Auteurs
All authors contributed to the conduct of this work.
All authors also declare that they have read and approved the final version of the manuscript.
endalem@yahoo.fr; jpauleng@gmail.com; barcheblaise@gmail.com, dbatchom@yahoo.fr; tonguevanessa@yahoo.com; mbatchou.ngahane@yahoo.com; dodobalkissou@gmail.com;
References
- D Patrini, N Panagiotopoulos, J Pararajasingham, L Gvinianidze, Y Iqbal, and DR Lawrence, “Etiology and management of spontaneous haemothorax,” J. Thorac 7 (2015): 520-526.
- PJ Cimino and DH Gutmann, “Neurofibromatosis type 1” (2018): 799-811.
- M Attia et al. “Spontaneous hemothorax and neurofibromatosis type 1: How to explain it, how to explore it and how to treat it?” Monaldi Arch. Chest Dis 89 (2019): 2.
- “National Institutes of Health Consensus Development Conference Statement: neurofibromatosis. Bethesda, Md., USA, July 13-15, 1987,” Neurofibromatosis 1 (1988): 172-8.
- RE Ferner and DH Gutmann, “Neurofibromatosis type 1 (NF1)” (2013): 939-955.
- L Reviron-Rabec et al. “Pulmonary complications of type 1 neurofibromatosis,” Rev. Mal. Respir 33 (2016): 460-473.
- E-M Jutant et al. “Pulmonary hypertension associated with neurofibromatosis type 1,” Eur. Respir. Rev 27 (2018): 180053.
- E-M Jutant et al. “Phenotype and Outcomes of Pulmonary Hypertension Associated with Neurofibromatosis Type 1,” Am. J. Respir. Crit. Care Med 202 (2020): 843-852.
- S Fdil, S Bouchikhi, and JE Bourkadi, “Hémothorax spontané: Complication rare de la neurofibromatose type 1,” Pan Afr. Med. J 28 (2017).
- DT Miller et al. “Health Supervision for Children with Neurofibromatosis Type 1,” Pediatrics 143 (2019): 5.
- G Camsari, A Gür, G Ozkan, ND Bakan, F Zengin, and A Külcü “[Thoracic findings in neurofibromatosis]”. Tuberk. Toraks 54 (2006): 267-72.
- AC Zamora, HR Collard, PJ Wolters, WR Webb, and TE King, “Neurofibromatosis-associated lung disease: a case series and literature review,” Eur. Respir. J 29 (2006): 210-214.
- R Bidad, C Hall, and E Blohm, “Fatal Tension Hemothorax Combined With Exanguination: A Rare Complication of Neurofibromatosis,” Clin. Pract. Cases Emerg. Med 3 (2019): 364-368.
- S Ben Saad, M Abdenadher, M Attia, H Daghfous, and F Tritar, “Un hémithorax blanc chez une jeune portant des tâches café au lait,” Rev. Pneumol. Clin 74 (2018): 100-103.
- PJ Gil Vázquez, ADA Arroyo Tristán, J Martínez Baños and J Torres Lanzas, “Massive spontaneous hemothorax in patients with neurofibromatosis type 1.,” Emergencias la Soc. Esp. Med. Emergencias 30 (2018): 190-193.
- FDA Degbelo, G Cito, B Guendil, M Christodoulou, and Z Abbassi. “Spontaneous Hemothorax in a Patient with von Recklinghausen’s Disease: A Case Report and Review of the Literature,” Am. J. Case Rep 20 (2019): 674-678.
- D Patrini, N Panagiotopoulos, J Pararajasingham, L Gvinianidze, Y Iqbal, and DR Lawrence. “Etiology and management of spontaneous haemothorax.,” J. Thorac. Dis 7 (2015): 520-6.
- S D’Errico, M Martelloni, FP Cafarelli, and G Guglielmi “Neurofibromatosis 1 and massive hemothorax: a fatal combination,” Forensic Sci. Med. Pathol 14 (2018): 377-380.


 Impact Factor: * 6.2
Impact Factor: * 6.2 Acceptance Rate: 76.33%
Acceptance Rate: 76.33%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 