A Comparative Study Between Fine Needle Aspiration Cytology and Core Needle Biopsy in the Diagnosis of Palpable Breast Lumps
Sharang S Kulkarni*, Sheetal Murchite, Aniket Patil
Department of General Surgery, Dr. D. Y. Patil Hospital and Research Institute, Kolhapur, Maharashtra, India
*Corresponding Author: Sharang S Kulkarni, Department of General Surgery, Dr. D. Y. Patil Hospital and Research Institute, Kolhapur, Maharashtra, India
Received: 02 April 2022; Accepted: 15 April 2022; Published: 21 April 2022
Article Information
Citation: Sharang S Kulkarni, Sheetal Murchite, Aniket Patil. A Comparative Study Between Fine Needle Aspiration Cytology and Core Needle Biopsy in the Diagnosis of Palpable Breast Lumps. Journal of Surgery and Research 5 (2022): 221-228.
View / Download Pdf Share at FacebookAbstract
Background
Breast carcinoma is a frequently diagnosed cancer in women worldwide. An effective diagnosis is necessary to manage this cancer in its preliminary stages. After a radiological examination of the palpable breast lumps, a core needle biopsy (CNB) or fine needle aspiration cytology (FNAC), is performed.
Objectives
The study focuses on comparing the accuracy and efficacy of FNAC and CNB in diagnosing palpable breast lumps.
Design
A randomized, open-labeled, comparative research.
Setting
A tertiary care center from October 2018 to 2020.
Patients and Method
66 female patients who presented to the out-patient department with a palpable breast lump were selected after informed consent. The randomization technique was used to divide patients into two groups, i.e., undergoing FNAC or CNB. The results thus obtained were compared with the post-operative histopathological examination statistically. Sample size 66 female patients with palpable breast lumps
Main Outcome Measures
Biostatistical parameters such as sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy.
Results
The sensitivity of CNB and FNAC was 92.8% and 86.6% respectively, whereas the specificity and positive predictive value of both the tests were 100%. NPV of CNB was 95%, and that of FNAC was 90%. The accuracy of CNB was 96.96% and that of FNAC was 93.90%.
Conclusion
Better cellularity, the possibility of immunohistochemical analysis and better characterization of specimens make core needle biopsy more efficacious than FNAC. It is more sensitive and accurate than FNAC. Core needle biopsy provides a more dependable preoperative diagnosis which can help in creating a pru
Keywords
<p>Breast neoplasms, Core needle biopsy, Fine needle aspiration cytology</p>
Article Details
1. Introduction
Breast cancer is one of the most prevalent cancers in the U.S. in women with an estimated number of new cases diagnosed in the year 2020 being 2,76,480 and estimated deaths being 42,170 (7%) [1-3] According to 2012 global statistics, nearly 1.7 million women were identified as suffering from breast carcinoma, with 5,22,000 deaths [4]. In Asian countries, the prevalence of palpable breast lumps is increasing in countries namely China, Taiwan, India, etc., between the age range of 40 to 50 years [5]. In India, there are 1,392,179 cancer cases as of 2020 and 57.0% of these cases are breast cancer [6]. It is crucial to recognize a breast lump in the early stages. The early evaluation and prompt diagnosis are reliant upon a detailed history of the lesion, and the ‘Triple Assessment’ which includes detailed clinical breast examination, radio-imaging and tissue examination [7]. The standard radio-diagnostic modalities used in diagnosis of breast lumps are mammography, ultrasonography, and magnetic resonance imaging (MRI) in accordance with BI-RADS (Breast Imaging, Reporting, and Data System), a quality control, numerical coding system developed by multiple healthcare groups, globally, but a trademark of American College of Radiology [8]. The gold standard modality for diagnosis of a breast lesion is histopathological examination of the specimen retrieved post-operatively. However, a routine excision biopsy of such specimen would not be prudent and necessary, as majority of the palpable breast lesions are often benign. Use of minimally invasive, cost-conscious and accurate modalities of pathological examination is prudent. Such methods of diagnosis with easy reproducibility, short learning curve and less preparation in clinical practice have taken over the now obsolete excision biopsies. Core needle biopsy (CNB) and fine needle aspiration cytology (FNAC) are the techniques applied to diagnose and characterize breast tumors [9]. FNAC is a easily applicable, dependable, quick and reproducible investigation. It is also a patient-friendly and usually requires minimal or no anaesthesia, giving frequently true positive results, thereby making it a tool of choice for the diagnosis of breast lesions. Despite all these advantages, a few limitations like less cellularity and the inability to characterize the lesions (e.g., Ductal carcinoma in-situ (DCIS) or Atypical ductal hyperplasia from invasive neoplasm) put FNAC on a weighing scale against Core needle biopsy. On the other hand, Core needle biopsy (CNB) provides adequate tissue for definitive histological diagnosis, distinguishes between invasive cancer and carcinoma-in-situ in patients having suspicious radiological findings. The accuracy of CNB increases with multiple passes through the lesion. Also, recent advances like the use of a 14-gauge core needle, automated large core biopsy guns and vacuum assistance have increased the efficacy as well as ease of the procedure substantially. Besides the samples provided by CNB can also be subjected to hormonal assays, i. e., ER/PR and HER2/neu status and it can also comment on the grade of the tumor as well as lymphovascular invasion. Both the procedure has their merits and demerits [10]. Current study mainly focuses on the comparison of FNAC with CNB. The comparison is done between accuracy, sensitivity, specificity as well as positive predictive value (PPV), and negative predictive value (NPV) of FNAC and CNB regarding palpable breast lumps.
2. Patients and Method
The trial was conducted from October 2018 to 2020 in a tertiary care teaching hospital. The trial was approved by the institutional review board. Women presenting with clinical features of palpable breast lumps were included. Women recognized with an already diagnosed breast lump, not consenting for CNB or FNAC and with contraindications to the procedures were excluded. Informed consent was taken before the onset of the study. Patients were selected and divided into two groups equally after randomization. Based on their clinical evaluation, patients were advised to take up ultrasonography to characterize a breast carcinoma as solid or complex cystic. Every alternate patient was advised to undergo either CNB or FNAC. Appropriate surgical interventions were planned and performed as per the test results and the specimens obtained during the surgical procedure were investigated with the histopathological examination (HPE). Estrogen and Progesterone Receptor (ER/PR) testing of the retrieved tissue was performed wherever found necessary. In cases where surgical intervention was not required or when patients did not consent to surgery, the patients were still recruited for the research, but the reports were analyzed separately. CNB and FNAC outcomes were compared with postoperative histopathological reports (HPR) for validation and biostatistical analysis.
2.1 Statistical analysis
The sample size was derived using the following formula by Rajeev Kumar Malhotra and A Indrayan [11].
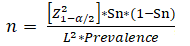
n = required sample size, Sn = anticipated sensitivity (82%), = size of the critical region (1- is the confidence level) (5%), = Standard normal deviate corresponding to the specified size of a critical region ( Absolute precision (5%) desired on either side (half-width of the confidence interval) of specificity and sensitivity, Prevalence = 27%. The power of this research was set based on the values obtained from the Suvradeep Mitra et al. 2016, the values were = 5%, Sn = 82%, L=5%, and prevalence = 27% [12]. Data were analyzed using the statistical software R (version 3.6.3). Categorical variables are presented as percentage and frequency. Continuous variables are depicted as mean and standard deviation (SD). Cohens-Kappa was employed to assess the agreement of CNB and FNAC with the histopathological reports specificity, sensitivity, PPV, NPV and the accuracy of the two tests were compared.
3. Results
The study recruited 66 women who were recognized with palpable breast lumps. Among 66 patients, 56% (n=37) were diagnosed with a benign tumor and 44% (n=29) had malignant lesions. The benign lesions were categorized in accordance to the type of tumor i.e., fibroadenoma (67.5%, n=25), antibioma (8.1%, n=3), fibrocystic disease (5.4%, n=2), Phyllodes tumor (5.4%, n=2), lipoma (2.7%, n=1), benign simple cyst (2.7%, n=1), galactocele (2.7%, n=1), duct ectasia (2.7%, n=1), and pseudoangiomatous hyperplasia (2.7%, n=1). The malignant lesions were categorized as: infiltrating duct carcinoma (72.4%, n=21), ductal carcinoma in-situ (20.6%, n=6), infiltrating micropapillary carcinoma (3.4%, n=1), and inflammatory carcinoma (3.4%, n=1). The agreement between CNB and HPR was 0.937 (p<0.0001) and that between FNAC and HPR was 0.876 (p<0.0001). CNB diagnosed 19 malignant carcinomas with 100% accuracy. CNB reported 14 subjects with benign tumors and 1 false-negative report was received, i.e., an invasive ductal carcinoma was misidentified as a phyllodes tumor. For one lesion with invasive ductal carcinoma, CNB could not ascertain if the carcinoma were in-situ or invasive. Three invasive ductal carcinomas were reported as ‘atypical ductal hyperplasia’ (Table 1). Two FNAC reports were false negatives. It was noticed that both these lesions were invasive ductal carcinomas, however, were identified as benign cystic lesions and fat necrosis, respectively. Though FNAC was capable to assess a true positive malignant report in 13 cases, it could not differentiate an in-situ lesion from an invasive lesion (Table 2). The sensitivity of CNB to detect a malignant lesion was 92.8% and that of FNAC was 86.6%. Both the tests demonstrated 100% specificity and PPV. NPV of CNB and FNAC were 95% and 90% respectively. Accuracy with CNB was 96.96%, and that of FNAC was 93.90% (Table 3).
|
HPR: MALIGNANT |
HPR: BENIGN |
|
|
CNB: MALIGNANT |
13 |
0 |
|
CNB: BENIGN |
1 |
19 |
HPR: Histopathology report
Table 1: Comparison of CNB results with the histopathology report.
|
HPR: MALIGNANT |
HPR: BENIGN |
|
|
FNAC: MALIGNANT |
13 |
0 |
|
FNAC: BENIGN |
2 |
18 |
HPR: Histopathology report
Table 2: Comparison of FNAC results with the histopathology report.
|
FNAC |
CNB |
|
|
Sensitivity |
86.60% |
92.80% |
|
Specificity |
100% |
100% |
|
Positive predictive value (PPV) |
100% |
100% |
|
Negative predictive value(NPV) |
90% |
95% |
|
Accuracy |
93.90% |
96.96% |
Table 3: Comparison of sensitivity, specificity, positive predictive value, negative predictive value and accuracy of FNAC with CNB.
4. Discussion
Breast carcinoma is one of the commonest cancers diagnosed in women worldwide. A palpable lump in breast is the most frequent complaint of females coming to the outpatient department. Pain, nipple discharge, skin changes are some other frequent complaints. Triple assessment includes clinical breast assessment, ultrasonography, and FNAC. FNAC is a primary diagnostic tool in the rural setup where CNB is not affordable [13]. Axillary lymph node metastasis and metastatic lesions were identified by FNAC [14]. A survey performed on the usage of CNB and FNAC concluded that there is growth in the usage of CNB in the last 5 years [15]. Salim Tahir et al, in a study conducted on fifty patients, concluded that FNAC has 85% sensitivity and 98.6% specificity while CNB has 92% sensitivity and 100% specificity in classifying breast tumor lesions [16]. Another study by Shashirekha et al reported values that were in agreement with our study i.e., specificity was observed to be 84.3% and 97%, while sensitivity and PPV were 100%, NPV was 53.3% and 74% with FNAC and CNB respectively [17]. Saha et al, reported that values of FNAC and CNB sensitivity were 69%, and 88.3%, NPV was 38.1%, and 53.3%, accuracy was 74%, and 86% while specificity and PPV were 100%, respectively [18]. Shaila et al, expressed the values for specificity as 84.6% and 89.7%, sensitivity as 72.4% and 96.5%, accuracy as 79.4% and 92.6% of FNAC and CNB, respectively [19]. All the studies reported CNB to possess greater accuracy when compared to FNAC in diagnosing and characterizing a palpable breast mass.
Takhellambam et al described the utilization of FNAC as a tool to categorize breast carcinoma in 62 cases, the test identified 42 as benign, 19 with malignancy, and 1 as indeterminate. In this research, 2 cases were not classified correctly when validated using the respective HPR. In low-grade malignancy, FNAC is not accurate in reporting if a malignant lesion is in-situ or infiltrative. Therefore, it becomes prudent that a careful approach is adopted while deciding the surgical extent of clearance of lymph nodes and cancerous tissue based on an FNAC alone [20] Brancato et.al, compared FNAC and CNB when coupled with the ultrasound reports. CNB was better in diagnosing palpable breast lumps [21].
A review done by Mitra et al. observe that there is a general bias toward CNB, but both methods have highlights and challenges. In the instance of FNAC, accuracy, knowledge about the procedure, and difficulty in interpreting FNAC smears are a few challenges of this investigation. Differentiation of in-situ carcinomas and fibroadenomas cannot be performed by FNAC [12]. On the other hand, there are some barriers to decreased sensitivity and the diagnostic accuracy of FNAC which has been explained by Nassar 2011. According to Nassar, 2011, FNAC is replaced by CNB as there are some limitations in diagnostic accuracy, decreased sensitivity [14]. Metastatic lesions, axillary lymph node metastasis and their progression were still evaluated with the FNAC procedure. FNAC can also utilize the new techniques to enhance its power in breast carcinoma diagnosis. The techniques like proteomic pattern expression, methylation profiling. FNAC associated with the image obtaining techniques also has higher sensitivity and specificity. CNB has to be utilized in most cases as a major diagnostic tool after clinical examination and radiological imaging techniques. To increase the effectiveness in the precession of CNB, modern techniques like vacuum assistance, mammography, and MRI guidance should be advocated [14].
Based on the existing literature and the current research observations, two techniques i.e., FNAC and CNB are accurate, sensitive, specific and have good PPV and NPV values in classifying breast tumors. CNB is more accurate as well as efficacious in diagnosing and characterizing palpable breast lumps. Despite being a more invasive, technically difficult and expensive modality, the results obtained by core needle biopsy more efficiently point towards appropriate surgical management as well as chemotherapy. The ability to characterize a lesion as in-situ or infiltrating, commenting on micro-architecture of low-grade tumors, lymphovascular involvement and to provide enough tissue samples for tumor marker and hormonal assays (ER/PR and HER2/neu status) makes core needle biopsy a more prudent tool than FNAC for diagnosis of a breast lump.
FNAC is a less invasive, affordable, easily reproducible modality for a quick diagnosis, especially during screening. Although it shows comparable biostatistics, the need for a thorough enough diagnosis to decide a treatment plan is often not met. Hence, in cases with ambiguous or suspicious reports by FNAC, a core needle biopsy is warranted. Clinical as well as radiological investigations can raise suspicion of a malignant lesion. In such cases, core needle biopsy should be the first line of tissue diagnosis. FNAC can be reserved as a diagnostic tool for recurrent or metastatic diseases.
To summarize, core needle biopsy is a superior diagnostic tool than fine needle aspiration cytology for diagnosis of a palpable breast lump. Therefore a wider and more frequent use of core needle biopsy should be advocated and implemented. With the advent of less radical surgical methods coupled with neoadjuvant chemotherapy, it is possible to attain complete clearance from the carcinoma in a single surgical procedure. Therefore, a thorough diagnosis procured after performing a core needle biopsy helps in chalking out an algorithm of management. Although fairly dependable, it is advisable to keep the limitations of core needle biopsy in mind.
A comparatively small sample size of the current study can be a limitation. A comparative study involving two diagnostic modalities for breast lumps is of paramount importance in a developing nation like India, where awareness about breast cancer is needed to be augmented. Loss in follow-up pertaining to the current coronavirus pandemic during the course of the study is also a causative factor for a small sample size. We, however, want to establish that the data obtained was compared with similar studies having comparable sample size, therefore have statistically overcome this limitation. Inherent discrepancy in the sample sizes, of aspiration cytology and biopsy can be a limitation. Taking this into account, the objective of the study is to advocate the use of core needle biopsy over FANC in the diagnosis of palpable breast lumps.
Conflict of interest
None
References
- National Cancer Institute, Surveillance, Epidemiology, and End Results Program. (SEER). Cancer stat facts: female breast cancer (2019).
- Siegel R, Miller K, Jemal A. Cancer statistics 2019. CA: A Cancer Journal for Clinicians 69 (2019): 7-34.
- DeSantis C, Ma J, Gaudet M, et al. Breast cancer statistics 2019. CA: A Cancer Journal for Clinicians 69 (2019): 438-451.
- Tao Z, Shi A, Lu C, et al. Breast Cancer: Epidemiology and Etiology. Cell Biochemistry and Biophysics 72 (2014): 333-338.
- Leong S, Shen Z, Liu T, et al. Is Breast Cancer the Same Disease in Asian and Western Countries?. World Journal of Surgery 34 (2010): 2308-2324.
- Mathur P, Sathishkumar K, Chaturvedi M, et al. Cancer Statistics, 2020: Report From National Cancer Registry Programme, India. JCO Global Oncology 6 (2020): 1063-1075.
- Saslow D, Hannan J, Osuch J, et al. Clinical Breast Examination: Practical Recommendations for Optimizing Performance and Reporting. CA: A Cancer Journal for Clinicians 54 (2004): 327-344.
- Gokhale S. Ultrasound characterization of breast masses. Indian Journal of Radiology and Imaging 19 (2009): 242-247.
- Tikku G, Umap P. Comparative Study of Core Needle Biopsy and Fine Needle Aspiration Cytology in Palpable Breast Lumps: Scenario in Developing Nations. Turk Patoloji Derg 32 (2016): 1-7.
- Sustova P, Klijanienko J. Value of combined use of fine?needle aspiration and core needle biopsy in palpable breast tumors performed by pathologist: Institut Curie experience. Diagnostic Cytopathology 48 (2019): 71-77.
- Malhotra R, Indrayan A. A simple nomogram for sample size for estimating sensitivity and specificity of medical tests. Indian Journal of Ophthalmology 58 (2010): 5-19.
- Mitra S, Dey P. Fine-needle aspiration and core biopsy in the diagnosis of breast lesions: A comparison and review of the literature. CytoJournal 13 (2016): 18.
- Kharkwal S, Sameer, Mukherjee A. Triple test in carcinoma breast. J Clin Diagn Res 8 (2014): NC09-11.
- Nassar A. Core needle biopsy versus fine needle aspiration biopsy in breast--a historical perspective and opportunities in the modern era. Diagn Cytopathol 39 (2011): 380-388.
- Tabbara SO, Frost AR, Stoler MH, et al. Changing trends in breast fine-needle aspiration: results of the Papanicolaou Society of Cytopathology Survey. Diagn Cytopathol 22 (2000): 126-130.
- Tahir S, Ahmad S. Clinicopathological and Radiological Evaluation of Benign Breast Diseases- A Comparison Between Fnac And Core Biopsy. Journal of Evolution of Medical and Dental Sciences 5 (2016): 6936-6939.
- Shashirekha CA, Singh R, Ravikiran HR, et al. Fine needle aspiration cytology versus trucut biopsy in the diagnosis of breast cancer: a comparative study. International Surgery Journal 4 (2017): 3718.
- Saha A, Mukhopadhyay M, Das C, et al. FNAC Versus Core Needle Biopsy: A Comparative Study in Evaluation of Palpable Breast Lump. J Clin Diagn Res 10 (2016): EC05-08.
- Mitra S, Rai R, Mitra R, et al. Comparative evaluation of FNAC, core needle biopsy and excisional biopsy in subtyping of breast lesions. Tropical Journal of Pathology and Microbiology 2 (2016): 9-15.
- Takhellambam YS, Lourembam SS, Sapam OS, et al. Comparison of ultrasonography and fine needle aspiration cytology in the diagnosis of malignant breast lesions. J Clin Diagn Res 7 (2013): 2847-2850.
- Brancato B, Crocetti E, Bianchi S, et al. Accuracy of needle biopsy of breast lesions visible on ultrasound: audit of fine needle versus core needle biopsy in 3233 consecutive samplings with ascertained outcomes. Breast 21 (2012): 449-54.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 