Hereditary Nonpolyposis Colorectal Cancer (HNPCC): From Diagnosis to Surgical Management of the Main Hereditary Form of Colorectal Cancer: A Review
Guido Mantovani*, Michela De Angelis, Corrado Asteria, Francesco Di Lecce, Paolo Mazza, Luca Vicenzi, Luigi Boccia
Department of General Surgery, Ospedale Carlo Poma, Mantua, Italy
*Corresponding Author: Guido Mantovani, Department of General Surgery, Ospedale Carlo Poma, Mantua, Italy
Received: 22 January 2020; Accepted: 06 February 2020; Published: 12 February 2020
Article Information
Citation: Guido Mantovani, Michela De Angelis, Corrado Asteria, Francesco Di Lecce, Paolo Mazza, Luca Vicenzi, Luigi Boccia. Hereditary Nonpolyposis Colorectal Cancer (HNPCC): From Diagnosis to Surgical Management of the Main Hereditary Form of Colorectal Cancer: A Review. Journal of Surgery and Research 3 (2020): 020-030.
View / Download Pdf Share at FacebookAbstract
Colorectal cancer (CRC) is the third most common cancer worldwide, the 5% of CRC are hereditary. Among hereditary forms of CRC the most frequent is Hereditary Nonpolyposis Colorectal Cancer (HNPCC). HNPCC is an autosomal dominant genetic disease. Molecular basis of HNPCC are linked to genetic alterations, involving several mismatch repair (MMR) genes. Diagnosis of HNPCC is frequently difficult: HNPCC diagnosis process starts from family medical history and genetic counselling and continue with molecular diagnosis and genetic tests which often identify MMR genes mutation. In HNPCC the role of screening is irreplaceable for prevention and early diagnosis of CRC and extracolic cancer, such as endometrium, ovary, urothelium, stomach, small bowel, pancreas, brain, biliary ducts neoplasms. Surgical management of HNPCC is significantly different between colonic and rectal cancer. The main problem in surgical approach for HNPCC patients is related to the possibility of developing metachronous colorectal cancer. The choice is always between extensive and segmental resection. The aim of this study is to give some practical indications to surgeons for the management of HNPCC patients.
Keywords
<p>Hereditary Nonpolyposis Colorectal Cancer, Microsatellite Instability, Colorectal Cancer, Screening</p>
Article Details
1. Background
CRC is the third most common cancer worldwide, with more than 1 million of cases and 600.000 deaths per year [1]. About 85% of CRCs are sporadics, that is CRCs in which there is neither family history nor inherited mutation predisposing to CRC; approximately 10% of CRC are familial, this form may be due to the presence of gene mutations, predisposing to CRC or to environmental factors exposition, that may lead to an increased risk of developing CRC. The remaining 5% of CRC is hereditary. Among hereditary forms of CRC the most frequent is HNPCC, accounting for 3-4% of CRCs. HNPCC, as the name suggests, is not characterized by the appearance of polyps in the gastrointestinal tract, differently from Familial Adenomatous Polyposis (FAP), Peutz-Jeghers Syndrome and other polypoid forms of CRC. The distinction of HNPCC from sporadic CRC is not always obvious, rather sometimes it is very difficult and cannot prescind from a careful family medical history. The aim of this study is to describe clinical aspects, genetic and molecular features, diagnostic management, screening and surgical approach of HNPCC, giving some practical indications to surgeons for the management of this hereditary disease often difficult to recognize.
2. Introduction
HNPCC is an autosomal dominant genetic disease, with medium-high penetrance degree (30-70%) [2]. HNPCC is associated to increased risk of developing colorectal cancer (CRC) and other tumours (ovarian, gastric, urinary, cutaneous) in young age. The worldwide incidence of CRC related to HNPCC is 3-4 % of overall CRC, about 30.700 new cases every year [3], while the worldwide incidence of CRC is about 1.000.000 new cases every year. In HNPCC definition, it’s necessary to differentiate type I, the form associated only to development of colorectal cancer, from type II, in which there are extracolonic tumours associated to CRC, such as endometrial, ovarian, uterine, gastric, urinary and biliary.
3. Genetic Features
Molecular basis of HNPCC are linked to genetic alterations, involving several MMR genes. MMR is a multienzimatic complex, involved in the repair of DNA replication errors. These mutations regard the following genes: MLH1 [4], MSH2 [5], MSH6 [6], PMS2 [7], MLH3 [8] and EPCAM. EPCAM-gene is not implied in MMR, but it seems to inactivate his adjacent gene MSH2. Among all, MLH1 and MSH2 are the most frequently mutated, with an incidence of 50% and 40 %, respectively, whilst MSH6 mutations are present in approximately 10% of HNPCC families. PMS2, that has recently been identified, has an incidence from 2,2% to 5% [9]. The microsatellite sequences (Ms) are the DNA regions in which these errors accumulate more easily. Abnormalities of Ms are found in 85-95% of tumours associated with HNPCC and in 15-25% of sporadic colorectal cancer [10]. The most frequently observed genotype is mutation in genes MLH1 or MSH2, which is associated with "typical" tumour manifestations: early onset (mean age 44 years), colorectal cancer in proximal colon, poorly differentiated (G3), mucinous histology, presence of tumour ring-infiltrating lymphocytes and high MSI (MSI-H, with more than 30-40% microsatellite instability sequences) [11]. Germline mutations in MSH6, PMS2, MLH3, instead, manifest themselves by "atypical" tumour phenotypes, related to onset, penetrance and instability degree of MSI. Particularly, germline mutations in MSH6 seem to be associated with increased risk of developing endometrial cancer (lifetime risk is estimated to be 26% at age 70 years and 44% at age 80 years) [12], whilst germline mutations of PMS2 seems to be related to Turcot syndrome [13]. The lifetime risk of CRC according to HNPCC mutation carriers, in male and female, is summarized in Table 1.
4. Clinical Aspects and Classifications
Different criteria were developed to classify patients with HNPCC, Amsterdam criteria I (1991) [16] and Amsterdam criteria II (1998) [17], Revised Bethesda Guidelines (2004) [18] were developed to classify patients with HNPCC. Amsterdam Criteria II and Revised Bethesda Guidelines are reported in Table 2 and Table 3. Both Amsterdam and Bethesda criteria are often complex to apply and are burdened with estimated values of specificity (only 51% for Bethesda criteria) and sensibility (only 65% for Amsterdam criteria II).
|
Mutation Carrier |
Lifetime Risk of CRC (males) |
Lifetime risk of CRC (females) |
|
MLH1 and MSH2 [14] |
68.7% |
52% |
|
MSH6 [12] |
22% |
10% |
|
PMS2 [15] |
20% |
15% |
Table 1: Lifetime Risk of CRC in HNPCC-related Mutation Carrier.
|
Amsterdam II Criteria (all criteria must be met) |
|
Ø Three or more relatives with histologically confirmed CRC or cancer of the endometrium, small bowel, ureter, or renal pelvis, one affected relative being a first-degree relative of the other two; FAP should be excluded |
|
Ø Two or more successive generations are affected |
|
Ø At least one relative was diagnosed before the age of 50 years |
Table 2: Amsterdam II Criteria.
|
Revised Bethesda Guidelines (one or more of the following criteria must be met) |
|
Ø CRC before the age of 50 years |
|
Ø Synchronous or metachronous CRC or other HNPCC-related tumours*, regardless of age |
|
Ø CRC with MSI –H morphology** before the age of 60 years |
|
Ø CRC (regardless of age) and a first-degree relative with CRC or an HNPCC-related tumours before the age of 50 years |
|
Ø CRC (regardless of age) and two or more first or second-degree relatives diagnosed with CRC or an HNPCC-related tumours (regardless of age) |
*HNPCC-related tumours colorectal, endometrial, stomach, ovarian, pancreas, ureter and renal pelvis, biliary tract and brain (usually glioblastoma as seen in Turcot syndrome) tumours, sebaceous gland adenomas and keratoachantomas in Muir-Torre syndrome and carcinomas of the small bowel; **Presence of tumour- infiltrating lymphocytes, Crohn’s like lymphocytic reaction, mucinous/signet-ring cell differentiation, or medullary growth pattern.
Table 3: Revised Bethesda Guidelines.
5. Diagnosis
The HNPCC diagnosis process starts from a clinical suspicion, such as early onset of CRC (<50 years), familial history of CRC or the occurrence of multiple primary tumours, that fall into the category of cancer-related HNPCC type II, in at least two relatives (until the third generation), especially before the age of 50 years.
First step: family medical history and genetic counselling. Family history’s analysis requires a direct interview of patients or their closest relatives. The anamnesis should be able to reconstruct a comprehensive genealogical tree, which dates back at least to the third generation [19], to assess whether the disease is in their family or acquired and whether the patient matches the Amsterdam Criteria II or Revised Bethesda Guidelines (both or one of these criteria). Patients who meet these criteria will be submitted to specific screening, described below. If both Amsterdam and Bethesda criteria are not respected, patients will undergo classic CRC screening.
Second step: molecular diagnosis and genetic tests. If Amsterdam or Bethesda criteria are satisfied assessment of Ms of the tumour and immunohistochemical (IHC) investigations will be required. The order of molecular and genetic tests required is explained in Figure 1. The first step is the analysis of tumour phenotype, through the execution of IHC and Ms investigations on tumour DNA, searching for high instability (MSI-H), that interests at least 30% of analysed loci. If microsatellite sequences and IHC assessments are not diagnostic, i.e. microsatellite stability (MSS), or low MSI (MSI-L), or no loss of protein on IHC, patients will undergo classic CRC screening. If there is MSI-H and loss of expression on IHC, patients will be submitted to the second step, that consists in specific genetic tests, to identify germline mutations in MLH1, MSH2, MSH6, PMS2, summarized in Figure 1. These genetic tests are very expensive and, so it’s important a closely selection of the population to submit to these tests.
The presence of MSI in the tumour by itself is not enough to diagnose HNPCC because 10-15% of sporadic CRCs exhibit MSI. MSI in non-LS CRCs is usually caused by hypermethylation of the MLH1 gene. This acquired epigenetic inactivation of MLH1 is typically associated with mutations in the BRAF gene (specifically the V600E mutation), which has been described in about 35% of sporadic MSI-H CRCs. Therefore, identification of hypermethylation of MLH1 and/or BRAF V600E is an indication that a patient does not have the HNPCC germline mutation.
6. Screening
Patients with MMR genes mutations have more than 80% risk of developing colorectal cancer, compared to 5% of the general population, so this require close monitoring. The recent guidelines indicate to submit patients with MMR genes mutations to colonoscopy every 1-2 years, starting from 20-25 years of age, or 2-5 years before the youngest case in family. There is still no general consensus on the need to perform a colonoscopy every year or every two years, anyway it has been demonstrated its importance, in 2013, Vasen et al. [20] reviewed studies that reported the outcomes of colonoscopic surveillance in HNPCC before surgery, and although cancers did arise, the vast majority were early stage and death rates were very low. Practice guidelines for colon surveillance of HNPCC patients are summarized in Table 4. The risk of developing extracolic cancer (endometrium, ovary, urothelium, stomach, small bowel, pancreas, brain, biliary ducts) in patients with MMR gene mutations is about 37.5%, compared to 2% of general population [21]. This risk increase in patients with MSH2 mutations and in females. The district most affected is the endometrium (20-60%), while the ovary is the less one (9-12%). Gynaecological screening is standardized and consists of gynaecological examination, associated with trans-vaginal ultrasound with endometrial sampling every year from HNPCC is 1.6 to 19%, which is higher in Asian countries; screening programs provide EGDS every 1-3 years in patients with MMR genes mutations [22]. There is not general consensus about urothelial cancer (lifetime risk 28%), anyway the screening includes chemical and cytological urine examination, if necessary associated with abdominal ultrasound [23].
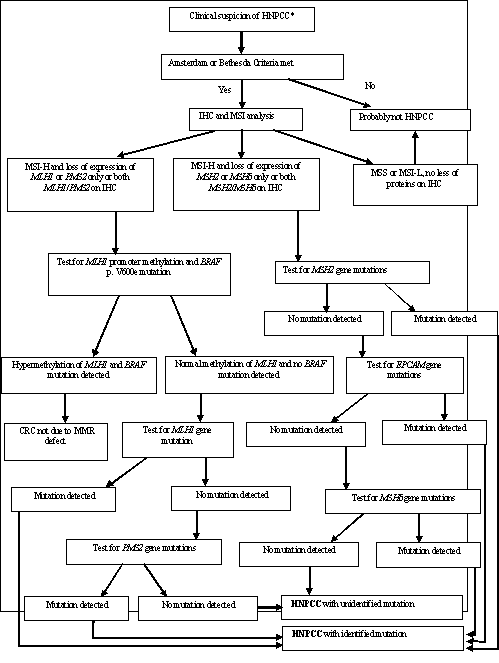
Figure 1: Suggested algorithm for HNPCC diagnosis.
*Early onset of CRC (<50 years), familial history of CRC or primary tumours, that fall into category of cancer
-related HNPCC type II (especially before 50 years or if more successive generations are affected).
|
Organization |
Age Screening Initiated |
Frequency |
Method |
|
American Cancer Society (2002) [24] |
21 years |
1-2 years until age of 40 years, then annually |
Colonoscopy |
|
GI Societies [25] (2003)a |
20-25 years |
1-2 years |
Colonoscopy |
|
Europe Mallorca Group (2007) [26] |
20-25 years |
1-2 years since 80 years |
Colonoscopy |
|
NCCN (2014) [27] |
20-25 years or 2-5 years before the youngest age at diagnosis in the family if it is before age of 25 years |
1-2 years |
Colonoscopy |
aGI Societies-American Academy of Family Practice, American College of Gastroenterology, American College of Physicians-American Society of Internal Medicine, American College of Radiology, American Gastroenterological Association, American Society of Colorectal Surgeons, and American Society for Gastrointestinal Endoscopy
Table 4: Practice Guidelines for Colon Surveillance of HNPCC patients.
7. Surgery
The main problem in surgical approach for HNPCC patients is related to the possibility (that they have) of developing metachronous colorectal cancer, estimated 16-40% at 10 years and 72% at 40 years [28]. The risk of metachronous CRC for MMR gene mutation carriers has been reported to be higher for MLH1 and MSH2 carriers and for subjects aged over 40 years [29].
8. Colonic Localization
The decision regarding which type of procedure to offer to patients with HNPCC and colon cancer is difficult. For patients and physicians, the task at hand is to measure the burden imposed by the larger initial procedure compared to benefits in the distant future. Abdominal colectomy with ileorectal anastomosis (IRA), has been recommended as procedure of choice in HNPCC patients with newly diagnosed colon cancer, as opposed to segmental resection. There are no reports of prospective and/or retrospective studies demonstrating survival benefit in patients undergoing IRA compared to those undergoing segmental resection. The disadvantage of abdominal colectomy with ileorectal anastomosis compared to segmental resection is mainly in the bowel frequency. Even though IRA is a more extensive procedure, it still has low morbidity and mortality [30]. It must be understood that this procedure does not prevent rectal cancer, which occurs between 3 and 20 % [31]. Thus, patients with extended resections will need endoscopic surveillance of the remainder rectum at risk.
The cumulative risk of metachronous CRC was 16% at 10 years, 41% at 20 years, and 62% at 30 years after segmental colectomy. Patients with HNPCC whose first colon cancer is treated with more extensive colonic resection have a lower risk of metachronous CRC than those receiving less extensive surgery. Other important issues to consider are the functional results of subtotal colectomy versus segmental resection (surveys suggest worse functional outcomes, but paradoxically not increased patient dissatisfaction), age (with not too much to gain in older patients than in those with many decades of expected life), and personal preference. Current recommendations in the USA suggest that people with HNPCC undergoing surgical resection of CRC should recieve an extensive resection rather than a segmental resection, even though this policy has not previously been proven to be superior to a policy of 1–2 yearly colonoscopic surveillance [32].
In Europe, current guidelines recommend the option of extensive resection be discussed with patients, particularly those under the age of 50 years [33]. Therefore in planning the extent of surgical resection for MMR gene mutation carriers presenting with colon cancer in the non-emergent setting, surgeons need to consider patient preference, patient age, bowel and sphincter function, as well as likely compliance with surveillance and the quality or otherwise of post-operative surveillance endoscopy. A heterogeneous group of 382 carriers of MMR gene mutations (172 MLH1, 167 MSH2, 23 MSH6, and 20 PMS2) with colorectal cancer who had surgery for their first colon cancer were analyzed using retrospective cohort analysis for age-dependent cumulative risks of metachronous CRC [28]. Detailed information about interval screening was unavailable, in patients who had segmental resections, 22% were diagnosed with metachronous CRC. On the other hand, none of 50 subjects who had extensive colectomy was diagnosed with metachronous CRC. Furthermore, the risk of metachronous CRC was reduced by 31% for every 10 cm of bowel removed.
In his study, Maeda [34] evaluated the outcomes in terms of quality of life achieved by two strategies of treatment: segmental resection versus total colectomy with ileo-rectal anastomosis for patients with HNPCC and CRC. He found that patients younger than 30 years old appeared to have better outcomes after segmental resection than IRA. This study differs from a similar study by Cappel et al [33] that reported only in terms of absolute survival and found that for young patients (age 27 years) with CRC and a MMR genetic defect, IRA conferred a survival benefit of 2.3 years relative to segmental resection. Extended procedure is in general favoured in the management of the HNPCC patients with newly diagnosed colorectal cancer. However, every case is different and treatment needs to be individualized. We can conclude that in HNPCC, extended resection are preferable in younger patients, while segmental resection is a good option in older individuals [33]. It is necessary to consider not only survival, but also quality of life when choosing between segmental resection and IRA. Approximately 25% of patients after IRA have five or more bowel movements per day [33]. Day-time and night-time incontinence occurs in 30% of patients [35].
8. Rectal Localization
In rectal surgery of HNPCC patients different surgical approaches are possible: local excision, segmental resection and extended resection. The first alternative is extremely rarely used and only for selected patients (important comorbidities and tumour amenable to local excision). Segmental resection includes anterior resection with primary anastomosis (LAR), proctectomy with coloanal anastomosis (CAA) or abdominoperineal resection (APR), if the sphincters are involved. Extended resection is represented by restorative proctocolectomy with ileal pouch anal anastomosis (RPC), if the sphincter can be saved or proctocolectomy with ileostomy if the sphincter cannot be saved; the experience with this type of surgery in HNPCC patients is still scarce.
The limit of segmental resection with primary anastomosis is the risk of metachronous cancer after proctectomy, with a median of 88-203 months post proctectomy [36] and a frequency from 15% to 51% [31]. In the report by Lee, 3 of 18 patients developed metachronous colon cancers at a median of 203 months post proctectomy [36]. RPC, instead, should be considered as a curative and at the same time prophylactic procedure, eliminating risk of metachronous colorectal cancers and in theory should be the procedure of choice for a patient with HNPCC and rectal cancer. However, in patients with RPC stool frequency is increased, with 6-8 daily bowel movements, at times inability to differentiate stool from gas and soiling in about 30 % of the cases [37]. Surveillance colonoscopies are mandatory after segmental rectal resection and pouchoscopies should be performed after RPC.
A survival advantage was not demonstrated in patients undergoing extended procedure. Stool frequency was less in segmental resection patients compared to extended resections. A study from the Dutch registry comparing segmental to extended resection in HNPCC patients, concluded that the functional outcome was worse after an extended procedure, but that there was no difference in generic quality of life between the two procedures [38]. In consideration of RPC as surgical option, we must explain and discuss approfonditely with patients about possible postoperative complications, such as female fecundity reduced by 50%, 1-2% of risk of erectile or ejaculatory dysfunction and the possibility of Pouch failure, that can lead to permanent ileostomy (10%) [39].
References
- Center MM, Jemal A, Smith RA, et al. Worldwide variations in colorectal cancer. CA Cancer J Clin 59 (2009): 366-378.
- Lynch HT, Lynch PM, Lanspa SJ, Snyder CL, Lynch JF and Boland CR. Review of the Lynch syndrome: history, molecular genetics, screening, differential diagnosis, and medicolegal ramifications. Clin Genet 76 (2009): 1-18.
- Hampel H, Frankel WL, Martin E, et al. Feasibility of screening for Lynch syndrome among patients with colorectal cancer. J Clin Oncol 26 (2008): 5783-5788.
- Bronner CE, Baker SM, Morrison PT, et al. Mutation in the DNA mismatch repair gene homologue hMLH1 is associated with hereditary nonpolyposis colon cancer. Nature 368 (1994): 258-261.
- Lindblom A, Tannergard P, Werelius B, et al. Genetic mapping of a second locus predisposing to hereditary nonpolyposis colorectal cancer. Nat Genet 5 (1993): 279-282.
- Hendriks YMC, Wagner A, Morreau H, et al. Cancer risk in hereditary nonpolyposis colorectal cancer due to MSH6 mutations: impact on counseling and surveillance. Gastroenterology 127 (2004): 17-25.
- Nicolaides NC, Papadopoulos N, Liu B, et al. Mutations of two PMS homologues in hereditary nonpolyposis colon cancer. Nature 371 (1994): 75-80.
- Hienonen T, Laiho P, Salovaara R, et al. Little evidence for involvement of MLH3 in colorectal cancer predisposition. Int J Cancer 106 (2003): 292-296.
- Nakagawa H, Lockman JC, Frankel WL, et al. Mismatch repair gene PMS2: disease-causing germline mutations are frequent in patients whose tumors stain negative for PMS2 protein, but paralogous genes obscure mutation detection and interpretation. Cancer Res 64 (2004): 4721-4727.
- Bellizzi AM, Frankel WL. Colorectal cancer due to deficiency in DNA mismatch repair function: a review. Adv Anat Pathol 16 (2009): 405-417.
- Laghi L, Bianchi P, Malesci A. Differences and evolution of the methods for the assessment of microsatellite instability. Oncogene 27 (2008): 6313-6321.
- Baglietto L, Lindor NM, Dowty JG, et al. Risks of Lynch syndrome cancers for MSH6 mutation carriers. J Natl Cancer Inst 102 (2010): 193-201.
- Peltomaki P, Vasen H. Mutations associated with HNPCC predisposition. Update of ICG-HNPCC/INSiGHT mutation database. Dis Markers 20 (2004): 269-276.
- Hampel H, Stephens JA, Pukkala E, et al. Cancer risk in hereditary nonpolyposis colorectal cancer syndrome: later age of onset. Gastroenterology 129 (2005): 415-421.
- Senter L, Clendenning M, Sotamaa K, et al. The clinical phenotype of Lynch syndrome due to germ-line PMS2 mutations. Gastroenterology 135 (2008): 419-428.
- Vasen HFA, Mecklin J-P, Meera Khan P, et al. The International Collaborative Group on Hereditary Nonpolyposis Colorectal Cancer (ICG-HNPCC). Dis Colon Rectum 34 (1991): 424-425.
- Vasen HFA, Watson P, Mecklin J-P. New clinical criteria for hereditary nonpolyposis colorectalcancer (HNPCC, Lynch syndrome) proposed by the International Collaborative Group on HNPCC. Gastroenterology 116 (1999): 1453-1456.
- Umar A, Boland CR, Terdiman JP, et al. Revised Bethesda Guidelines for hereditary nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite instability. J Natl Cancer Inst 96 (2004): 261-268.
- Rodriguez-Bigas MA, Moeslein G. Surgical treatment of hereditary nonpolyposis colorectal cancer (HNPCC, Lynch syndrome). Familial Cancer 12 (2013): 295-300.
- Vasen HF, Blanco I, Aktan-Collan K, et al. Revised guidelines for the clinical management of Lynch syndrome (HNPCC): recommendations by a group of European experts. Gut 62 (2013): 812-823.
- Barrow E, Robinson L, Alduaji W, et al. Cumulative lifetime incidence of exracolonic cancers in Lynch syndrome: a report of 121 families with proven mutation. Clin Genet 75 (2009): 141-149.
- National Comprehensive Cancer Network (NCCN) Practice Guidelines in Oncology-Lynch Syndrome, version 1 (2010).
- Koornstra JJ, Mourits MJE, Sijmons RH, et al. Agement of extracolonic tumours in patients with Lynch syndrome. Lancet Oncol 10 (2009): 400-408.
- Smith RA, Cokkinides V, von Eschenbach AC, et al. American Cancer Society guidelines for the early detection of cancer. CA Cancer J Clin 52 (2002): 8-22.
- Winawer S, Fletcher R, Rex D, et al. Colorectal cancer screening and surveillance: clinical guidelines and rationale-Update based on new evidence. Gastroenterology 124 (2003): 544-560.
- Vasen HF, Möslein G, Alonso A, et al. Guidelines for the clinical management of Lynch syndrome (hereditary non-polyposis cancer). J Med Genet 44 (2007): 353-362.
- National Comprehensive Cancer Network: NCCN Clinical Practice Guidelines in Oncology: Genetic/Familial High-Risk Assessment: Colorectal. Version 2 (2014).
- Parry S, Win AK, Parry B, et al. Metachronous colorectal cancer risk for mismatch repair gene mutation carriers: the advantage of more extensive colon surgery. Gut 60 (2011): 950-957.
- Vasen HFA, Abdirahman M, Brohet R, et al. One to 2-Year Surveillance Intervals Reduce Risk of Colorectal Cancer in Families With Lynch Syndrome. Gastroenterology 138 (2010): 2300-2306.
- Madden MV, Neale KF, Nicholls RJ, et al. Comparison of morbidity and function after colectomy with ileorectal anastomosis or restorative proctocolectomy for familial adenomatous polyposis. Br J Surg 78 (1991): 789-792.
- Kalady MF, Lipman J, McGannon E, et al. Risk of colonic neoplasia after proctectomy for rectal cancer in hereditary nonpolyposis colorectal cancer. Ann Surg 255 (2012): 1121-1125.
- Lindor NM, Petersen GM, Hadley DW, et al. Recommendations for the Care of Individuals With an Inherited Predisposition to Lynch Syndrome: A Systematic Review. JAMA 296 (2006): 1507-1517.
- De Vos tot Nederveen Cappel WH, Buskens E, van Duijvendijk P, et al. Decision analysis in the surgical treatment of colorectal cancer due to a mismatch repair gene defect. Gut 52 (2003): 1752-1755.
- Maeda T, Cannom RR, Beart RW Jr, et al. Decision model of segmental compared with total abdominal colectomy for colon cancer in hereditary nonpolyposis colorectal cancer. J Clin Oncol 28 (2010): 1175-1180.
- Aziz O, Athanasiou T, Fazio VW, et al. Metaanalysis of observational studies of ileorectal versus ileal pouch-anal anastomosis for familial adenomatous polyposis. Br J Surg 93 (2006): 407-417.
- Lee JS, Petrelli NJ, Rodriguez-Bigas MA. Rectal cancer in hereditary nonpolyposis colorectal cancer. Am J Surg 181 (2001): 207-210.
- Soravia C, Klein L, Berk T, et al. Comparison of ileal pouch-anal anastomosis and ileorectal anastomosis in patients with familial adenomatous polyposis. Dis Colon Rectum 42 (1999): 1028-1033.
- Haanstra JF, de Vos Tot Nederveen Cappel WH, Gopie JP, et al. Quality of life after surgery for colon cancer in patients with Lynch syndrome: partial versus subtotal colectomy. Dis Colon Rectum 55 (2012): 653-659.
- Vera N Tudyka and Susan K. Clark Surgical treatment in familial adenomatous polyposis. Annals of Gastroenterology 25 (2012): 201-206.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 