Mesenteric Root Abscess due to Perforated Diverticulum of the Third Part of the Duodenum with Concurrent Incidental Discovery of Sigmoid Colorectal Cancer
Panagiotis Patrikakos1*#, Nikiforos Rodis1#, Nikolaos Mpogiatzopoulos1, Konstantina Soukouli1, Aspasia Kapetanopoulou2, Konstantinos Syggounis2, Panagiota Xaplanteri3, Pavlos Athanasopoulos1
1Department of Surgery, General Hospital of Patras, Patras, Greece
2Department of Radiology, General Hospital of Patras, Patras, Greece
3Department of Microbiology, General Hospital of Eastern Achaia, Kalavrita, Greece
#Contributed equally.
*Corresponding Author: Panagiota Xaplanteri, Department of Microbiology, General Hospital of Eastern Achaia, Kalavrita, Greece
Received: 01 December 2022; Accepted: 12 December 2022; Published: 16 December 2022
Article Information
Citation: Panagiotis Patrikakos, Nikiforos Rodis, Nikolaos Mpogiatzopoulos, Konstantina Soukouli, Aspasia Kapetanopoulou, Konstantinos Syggounis, Panagiota Xaplanteri, Pavlos Athanasopoulos. Mesenteric Root Abscess due to Perforated Diverticulum of the Third Part of the Duodenum with Concurrent Incidental Discovery of Sigmoid Colorectal Cancer. Journal of Surgery and Research 5 (2022): 632-636.
View / Download Pdf Share at FacebookAbstract
Retroperitoneal abscesses originating from complicated duodenal diverticula are rare surgical entities. Herein we describe the case of an 81-year-old male patient who presented to the emergency department of our hospital, in a septic state. Computerized Tomography (CT) of the abdomen and retroperitoneal space revealed the presence of intraabdominal air. In the light of these findings the patient was submitted to emergency exploratory laparotomy that revealed a mesenteric root abscess with concurrent incidental discovery of sigmoid colorectal cancer. The abscess was drained, and Hartmann’s procedure was performed. Post-operative course was uneventful, and the patient was released on the tenth postoperative day. The patient returned 15 days later, due to fluid drainage from the site of the surgical drain. Minor leak from a diverticulum of the third duodenal part was documented by means of CT. Conservative treatment was chosen. The patient was discharged without any further complications and on follow up he is well three months afterwards. Conservative treatment was chosen due to the location of the diverticulum and the performance status of the patient. This was an intricate case of retroperitoneal, mesenteric root abscess, caused by a retroperitoneal duodenal diverticulum perforation of the third part.
Keywords
<p><em>Streptococcus anginosus</em>, <em>Candida albicans</em>, Duodenal diverticulum, Mesenteric root abscess, Retroperitoneal, Perforation, Surgery</p>
Article Details
Introduction
Primary mesenteric abscesses are a rare event [1]. Hematogenic spread by means of bacteremia originating from a distant focus causing such an entity, is also rare [2]. Other possible causes may include suppuration of a mesenteric lymph node, perforated duodenal ulcer and duodenal diverticulum perforation [3]. In our case, an senior male patient was diagnosed with a mesenteric root abscess, possibly due to a ruptured duodenal diverticulum, along with incidental discovery of colorectal cancer.
Case Presentation
A male patient of 81 years old presented to the emergency department of surgery complaining of worsening deep epigastric pain radiating to the back. His vitals were: core temperature of 39oC, heart rate 110 beats/min, blood pressure of 90/60mmHg. He reports fever, since about five days. His condition had reportedly been treated as a urinary tract infection with per os ciprofloxacin and metronidazole for the forementioned interval. The patient was sufficiently resuscitated with crystalloid intravenous fluids. Broad spectrum IV antibiotic coverage with meropenem, metronidazole and caspofungin was promptly initiated. A complete blood count and basic metabolic panel and urine analysis were obtained, along with abdominal/chest films. An abdominal computerized tomography (CT) examination with IV contrast was also ordered. The initial laboratory workup results were the following: White blood cells count: 14.400/mm3, neutrophil percentage: 86.70%, C reactive protein :30.9 mg/dl (normal lab. reference range: 0-0.5 mg/dl). Urine analysis was negative. On initial assessment by the radiologist, the abdominal CT scan revealed the presence of intra-abdominal air (figure 1) with elements of entrenchment near the jejunum (figure 2) with retroperitoneal extension along the course of the superior mesentery artery (figure 3). A possible inflammatory mass of the sigmoid colon and several para-aortic lymph nodes were also mentioned (figure 4). Due to the patient’s toxic state in conjunction with the CT findings, an emergency exploratory laparotomy was promptly decided, and the patient was brought to the operating table. Intraoperative findings: Upon investigation of the abdomen during the operation, a sizeable, fluctuant mass was discovered at the mesenteric root, affecting the mesentery of the first meter of jejunum, at a relative distance from the jejunal lumen, below the transverse colon, medial to the ligament of Treitz (figure 5). No viscera perforation was detected. The lesser sac was explored, with no remarkable findings, and Kocher maneuver was performed, exposing the first and second part of the duodenum. The duodenum was inspected, and the fourth part mobilized, without any significant findings and without obvious pancreatic pathology. A worrisome, noteworthy intraoperative finding was a sigmoid hard mass with macroscopic features compatible with possible neoplasm, was also found incidentally (figure 6). Upon incision of the mesenteric mass there was significant flow of purulent, foul-smelling liquid (figure 5). The pus was sufficiently drained and there was no obvious communication of the abscess cavity with adjacent viscera. Samples of fluid were collected and sent for culture. Sigmoidectomy with Hartmann’s pouch and end-colostomy was also performed, as well as appendicectomy, for the purpose of elimination of possible contamination source. Finally, a multilumen drainage tube was placed in the abscessed cavity and the abdomen was closed.
Culture results of the abscess contents proved positive for Streptococcus anginosus and Candida albicans.
The patient’s postoperative course was uneventful. The drain was removed, and he was discharged on the tenth postoperative day, hemodynamically normal, afebrile, with per os levofloxacin, cefixime and fluconazole.
After 15 days, the patient was readmitted to the clinic complaining of small amount of liquid exudate from the removed drainage tube fistulous tract. A Foley catheter was inserted into the tract, which produced traces of purulent discharge (about 10 ml) and the resulting fluid was sent to the laboratory for analysis. The fluid had elevated amylase content. Patient was treated conservatively. An abdominal CT scan was ordered, which revealed a minor leak originating from what appeared to be a duodenal diverticulum of the third part (figure 7). Flow from the fistula was significantly reduced at the first 48 hours (less than 10ml /24h).
Abdominal CT scan after a week, with per os contrast medium was obtained but showed no leak into other viscera or the peritoneum. The catheter was removed 10 days later without complications (figure 8).
Pathology report of the removed sigmoid specimen revealed an adenocarcinoma of intermediate differentiation, 2 cm in diameter with metastatic infiltration of one of the excised lymph nodes. TNM staging was T3 N1a M0.
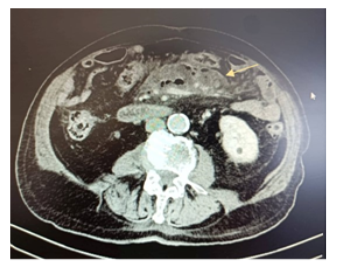
Figure 1: Free intra-abdominal air
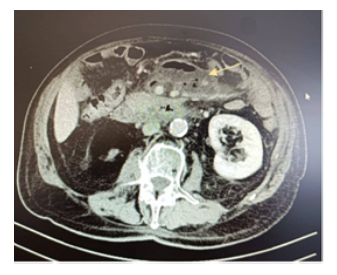
Figure 2: Entrenchment near the jejunum
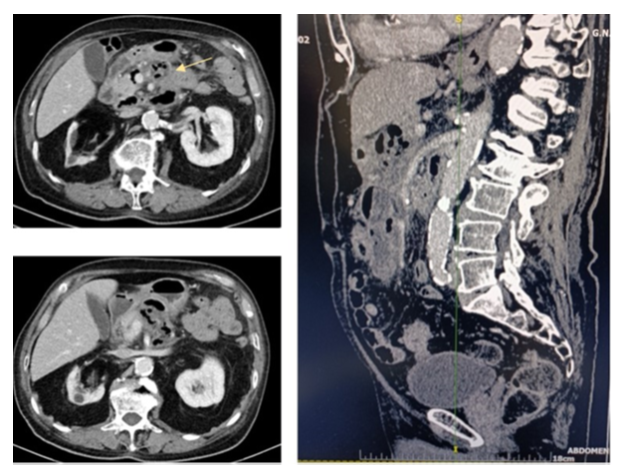
Figure 3: Retroperitoneal extension along the superior mesentery artery
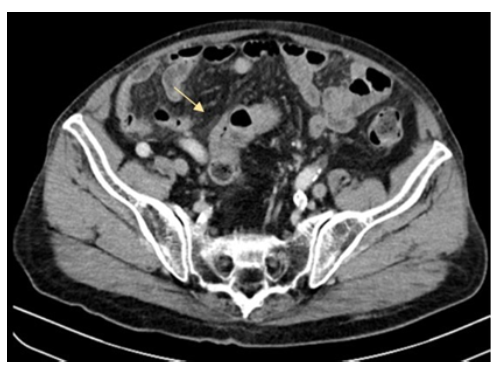
Figure 4: Sigmoid mass
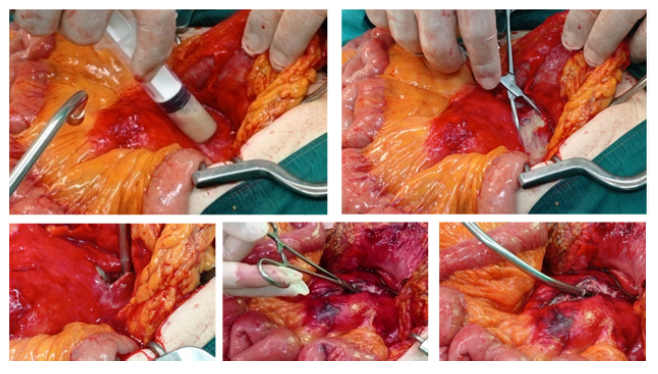
Figure 5: Intra-operative pictures. Abscess aspiration, incision, and drainage
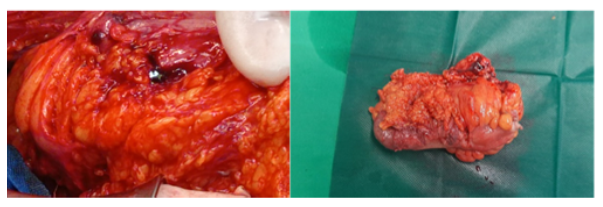
Figure 6: Sigmoid neoplasm specimen
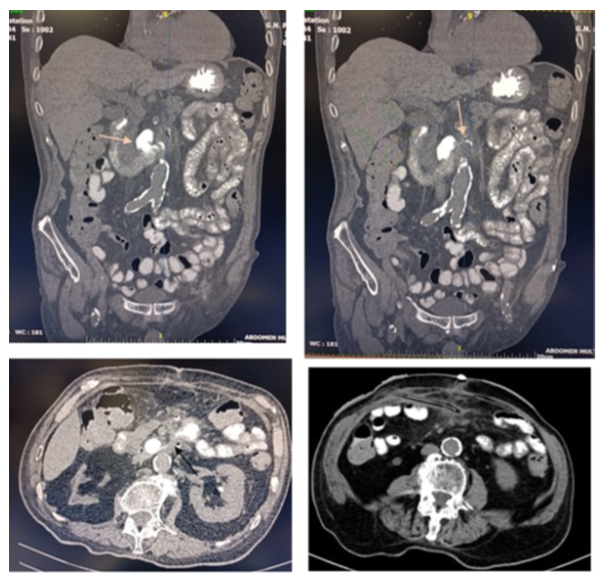
Figure 7: Duodenal diverticulum and leak. Foley catheter insertion
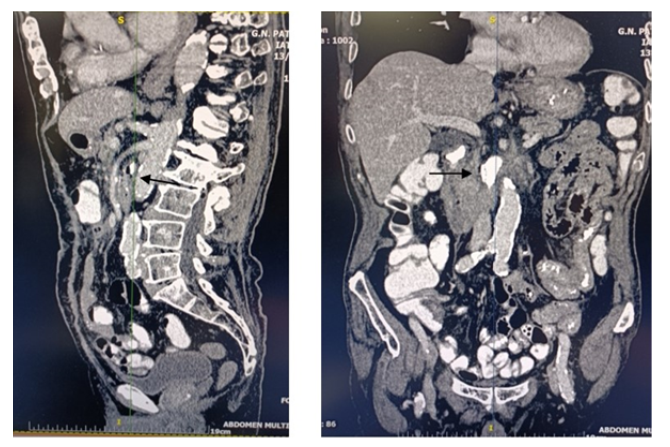
Figure 8: No abdominal leak after catheter removal
Discussion
Questions that arise from the study of this case include the true origin of the abscess, interpretation of culture results, possible communication between the abscess cavity and the gastrointestinal system or pancreas and probable association with a ruptured duodenal diverticulum.
The Streptococcus anginosus group (SAG), or otherwise described as Streptococcus milleri group, include the species: Streptococcus anginosus, Streptococcus intermedius, and Streptococcus constellatus. Those strains are opportunistic pathogens that immerge in recent studies as significant pathogens causing pyogenic infections. Of those, S. intermedius and S. constellatus provoke dental plaque and are related to periodontal diseases, while S. anginosus colonizes the oral cavity, gastrointestinal, and genitourinary tract. SAG can spread via transient bacteremia in people with bad oral hygiene, gingivitis, and tooth abscesses [2,4].
In literature SAG is the causative agents of intra-abdominal abscesses, peritonitis, soft tissue abscesses, and arthritis, osteomyelitis, and lung abscesses. S. anginosus is related to gastrointestinal and genitourinary tract infections. SAG is also related to the formation of pyogenic liver abscesses. The mechanism seems to be hematogenous spread due to diverticulitis of the sigmoid colon. Predisposing factors are the presence of solid tumors (like colorectal carcinoma or rectal adenocarcinoma), hematologic malignancies, type 2 diabetes mellitus, high blood pressure, AIDS, unhealthy diet, alcohol, drug consumption, and smoking, myasthenia gravis, Parkinson’s disease, chronic kidney failure, chronic respiratory disease, viral hepatitis, and connective tissue disease. The mechanisms of infection where neoplasms are concerned is that the cancerous lesions enable those microorganisms to enter blood circulation. Patients with SAG infection over the age of 65 and immunocompromised have significantly higher mortality rates. In a study in Spain the majority of intra-abdominal S. anginosus infections were of abdominal origin (20/33), with the most common diagnoses being cholangitis/cholecystitis (18%) and appendicitis (12%). The source of infection could not be determined in three cases. Nine cases of bacteremia (27%) were polymicrobial. Six patients (18%) had septic shock. In four the infection was polymicrobial and in two the infection was of abdominal origin. Eighteen of 33 patients (54%) required surgery. Five patients died [4,5].
Candida species are the main funguses of gastrointestinal and genitourinary tracts flora in humans yet are capable of invasive infections when imbalance occurs in those environments [6,7]. Intra-abdominal candidiasis in the form of peritonitis, purulent or necrotic infection, or abscess formation is usually related to patients who underwent abdominal surgery, suffer from Crohn’s disease, gastrointestinal perforation, or anastomotic leak, or acute necrotizing pancreatitis [6-8]. It is related to increased morbidity [7].
The duodenum is the second most common place of occurrence for diverticula after the colon. The incidence ranges from 1% to 22% in various studies [3,9-11]. Most of diverticula are acquired and are extraluminal pseudodiverticula in nature. The most frequent site of occurrence is the second part of the duodenum, in the vicinity of the ampulla of Vater (about 2cm), due to possible weakness of papilla predisposing protrusion of mucosa and submucosa [3,9,11,12].
Duodenal diverticula are asymptomatic in 90% of cases. Prevalence of complications can reach 0,03% per year [11], the most common ones being cholopancreatic stasis, lumen obstruction, ulceration, diverticulitis, hemorrhage, and perforation. Most frequent causes for the later include diverticulitis, ischemia and distention owing to enterolithiasis or food retention and bacterial overgrowth inside the diverticulum. Furthermore, foreign body ingestion, trauma, and iatrogenic injury during ERCP are also common culprits. Perforation symptoms usually include epigastric pain, nausea, and vomiting. However, retroperitoneal perforation often becomes entrenched, and the symptoms mentioned above might be absent in these patients [9,11].
When it comes to treatment, surgery has been classically preferred to treat perforation, although conservative treatment could also be adequate, especially in hemodynamically normal patients [9,11].
Surgical intervention includes diverticulectomy with single/ double layer closure or duodenal diversion, gastroenteric anastomosis, duodenostomy, duodenal segmental resection and pylorus-preserving Whipple in severe cases. Identification of the papilla is of uttermost importance in diverticula with proximity to the ampulla of Vater. However, rate of post-operative complications is high, including duodenal leak, fistula, common bile duct injury, pancreatitis peritonitis and sepsis, thus surgery is usually reserved for selected cases.
Non-operative treatment consists of bowel rest, nasogastric decompression, broad antibiotic coverage, intravenous fluids and/or parenteral nutrition, possibly percutaneous/endoscopic catheter drainage and endoscopic cleaning of diverticulum pouch. It is a safe alternative in non-septic, mildly affected patients without peritonitis, as the initial treatment [3,9,11]. Physicians should always co-evaluate the clinical condition of every patient with comorbidities for treatment pathway related decision-making, in order to avoid surgical complications [9,11,13,14].
Considering the performance status of our patient and the location of the duodenal diverticulum (third part), between the aorta and the superior mesenteric artery, which was inaccessible without major surgical procedures (gastroenterological evaluation for intraluminal treatment was not a favorable option), we decided not to intervene any further. In the three-month follow-up the patient is well and without any complications.
Conclusion
This was an intricate case of retroperitoneal, mesenteric root abscess, caused by a retroperitoneal duodenal diverticulum perforation of the third part. We strongly suggest careful evaluation of radiologic findings in cases with a similar presentation consisting of intra-abdominal air in the vicinity of the duodenum, mesentery, and superior mesenteric artery, which can be atypical, in conjunction with careful examination and assessment of patients in septic condition presenting with acute abdomen.
Conflicts of interest
We declare no conflict of interest.
Funding
None.
Ethical approval
Institutional Review Board Statement: The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of GENERAL HOSPITAL OF PATRAS (protocol code 46884, date 29 November 2022).
References
- Dudley HAF and Maclaren IF. Primary mesenteric abscess. Lancet 12 (1956): 1182-1184.
- Terzi HA. Intra-abdominal abscess and primary peritonitis caused by streptococcus anginosus. Jundishapur J. Microbiol 9 (2016): 33-63.
- Thorson CM, Paz Ruiz PS, Roeder RA, et al. The perforated duodenal diverticulum. Arch. Surg 147 (2012): 81-88.
- Pilarczyk-Zurek M, Sitkiewicz I, Koziel J. The Clinical View on Streptococcus anginosus Group- Opportunistic Pathogens Coming Out of Hiding. Front. Microbiol 13 (2022): 677.
- Salavert M, Gómez L, Rodriguez CM, et al. Seven-year review of bacteremia caused by Streptococcus milleri and other viridans streptococci. Eur. J. Clin. Microbiol. Infect. Dis. 15 (1996): 365-371.
- Kauffman C. Candida infections of the abdomen and thorax 8 (2018): 669.
- V. Rodolico et al. Intra-abdominal Candida spp infection in acute abdomen in a quality assurance (QA)-certified academic setting. J. Clin. Pathol 70 (2017): 579-583.
- John RK. Intra-abdominal candidiasis. Global Alliance for Infections in Surgery 5 (2019): 569.
- Sahned J, Hung FS, Mohammed SD, et al. Duodenal Diverticulitis: To Operate or Not To Operate?. Cureus 11 (2019) 1-5.
- Barakat M, Bansal R., Chun S, et al. A Case of Unresolved and Worsening Retroperitoneal Abscess. Am. J. Gastroenterol 112 (2017): S1127-S1128.
- Costa SV, Santos B, Magalhães S, et al. Perforated duodenal diverticulum: Surgical treatment and literature review. Int. J. Surg. Case Rep 5 (2014): 547-550.
- Shimada A. Perforated duodenal diverticulum successfully treated with a combination of surgical drainage and endoscopic nasobiliary and nasopancreatic drainage: a case report. Surg. Case Reports 6 (2020): 4-9.
- Martínez-Cecilia D. Conservative management of perforated duodenal diverticulum: A case report and review of the literature. World J. Gastroenterol 14 (2008): 1949-1951.
- Sampsel J and Zaugg P. Perforation and Abscess of a Duodenal Diverticulum. Arch Surg 960 (1960):542-544.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 