Methylmalonic acidemia caused by Homozygous Missense variant c.1837C>T in Exon 11 of the MMUT gene which is Variant of Uncertain Significance: A Case Report
G Durga Phani Kumar1, Mashingbonio Dirinamai1, Bhutwala Senky Manishkumar1, Susheel kumar Saini*, 2, Ajay kumar Saini3, Seema kumari6
1MBBS, MD Pediatrics trainee, NIMS medical college, Jaipur, India
2MBBS, MD Pediatrics, FNB Pediatric Intensive care, Assistant Professor, NIMS medical college, Jaipur, India
3MBBS, DNB Pediatrics, SGMH, Delhi, India
4MBBS, MD Anesthesiology, Assistant Professor, NIMS medical college, Jaipur, India
*Corresponding author: Susheel kumar Saini. MBBS, MD Pediatrics, FNB Pediatric Intensive care, Assistant Professor, NIMS medical college, Jaipur, Rajasthan, India.
Received: 12 March 2023; Accepted: 21 March 2023; Published: 30 March 2023
Article Information
Citation: G Durga Phani Kumar, Mashingbonio Dirinamai, Bhutwala Senky Manishkumar, Susheel kumar Saini, Ajay kumar Saini, Seema kumari. Methylmalonic acidemia caused by Homozygous Missense variant c.1837C>T in Exon 11 of the MMUT gene which is Variant of Uncertain Significance: A Case Report. Fortune Journal of Health Sciences. 6 (2023): 115-118
View / Download Pdf Share at FacebookAbstract
Methylmalonic academia (MMA) is an autosomal recessive Inborn Error of Metabolism originating from a disorder of propionate metabolism. Although rare, it is one of the most frequent inborn errors of organic acid metabolism. The disease can manifest itself in the neonatal period or have late onset in childhood. The therapy is based on protein restriction and carnitine supplementation. The case report presents 10 months old male child who came with complaint of fever, loose stools and severe dehydration. On workup he was diagnosed as a case of isolated Methylmalonic acidemia. Clinical Exome sequencing based gene panel study showed a Homozygote Missense variant c.1837C>T in Exon 11 of the MMUT gene that resulted in the amino acid substitution p. Arg613Cys. Thus, the advanced genetic analysis along with clinical findings and typical imaging will help in appropriate diagnosis and management of the Inborn Error of Metabolism.
Keywords
<p>Methylmalonic aciduria, Rare, Organic academia, Pediatric, Variant</p>
Article Details
1. Objective
The methylmalonic acidemias (MMAs) are a heterogeneous group of autosomal recessive inborn errors of propionate metabolism. The term "methylmalonic acidemia" refers to a group of inborn errors of metabolism associated with elevated methylmalonic acid (MMA) concentration in the blood and urine that result from the failure to isomerize (convert) methylmalonyl-coenzyme A (CoA) into succinyl-CoA during propionyl-CoA metabolism in the mitochondrial matrix, without hyperhomocysteinemia or homocystinuria, hypomethioninemia, or variations in other metabolites, such as malonic acid.1In those individuals diagnosed by newborn screening and treated from an early age, there appears to be decreased early mortality, less severe symptoms at diagnosis, favorable short-term neurodevelopmental outcome, and lower incidence of movement disorders and irreversible cerebral damage.
However, secondary complications may still occur and can include intellectual disability, tubulointerstitial nephritis with progressive impairment of renal function, "metabolic stroke" (bilateral lacunar infarction of the basal ganglia during acute metabolic decompensation), pancreatitis, growth failure, functional immune impairment, bone marrow failure, optic nerve atrophy, arrhythmias and/or cardiomyopathy (dilated or hypertrophic), liver steatosis/fibrosis/cancer and renal cancer Precise genotype-phenotype correlations are difficult to determine since most affected individuals are compound heterozygotes and many pathogenic variants are not recurrent in the population. We are presenting a case of Isolated Methylmalonic academia due to Homozygous Missense variant c.1837C>T in Exon 11 of the MMUT gene (Variant of Uncertain Significance) who presented to us in state of metabolic stroke.
2. Case Presentation
A previously healthy 10 months -old boy presented to the pediatric emergency with mild grade fever for 2 days along with recurrent vomiting. He took IPD based treatment in local hospital where he had severe dehydration along with high anion gap severe metabolic acidosis. He received fluid bolus along with Sodium bicarbonate correction therapy along with other supportive treatment but metabolic acidosis did not improve. He took no medical or surgical treatment previously. His birth history was normal and perinatal period was uneventful. The parents denied consanguinity. His development was normal as per age till the beginning of illness. His elder sister had history similar illness at 9 months of age for which she was treated and survived.
His sister had global development delay after that illness but no metabolic workup was done. Upon presentation, during physical examination, he had severe dehydration along with acidotic breating. He had generalized hypotonia along with severe encephalopathy. The patient was admitted and laboratory investigations were carried out. His laboratory workup showed hemoglobin 8.3 g/dl, total leukocytes count 6670/mm3 with 72% neutrophils and platelets 339,000/mm3). His clotting parameters were within the normal limits. His transaminases were normal ALT 28 IU/L, AST 43 IU/L. His CRP was also normal 0.5 mg/dl (Normal Range: 0.01.0mg/dl) but blood ammonia level was elevated 142 micromol/L. His creatinine and electrolytes were normal. Blood gas analysis on admission showed pH 6.94, HCO3- 3.4 mmol/L, BE – 27.6 mmol/L, Lactate 1.2 mmol/L. Blood sugar levels were in normal limit. 2D ECHO and USG abdomen was done which also came normal.
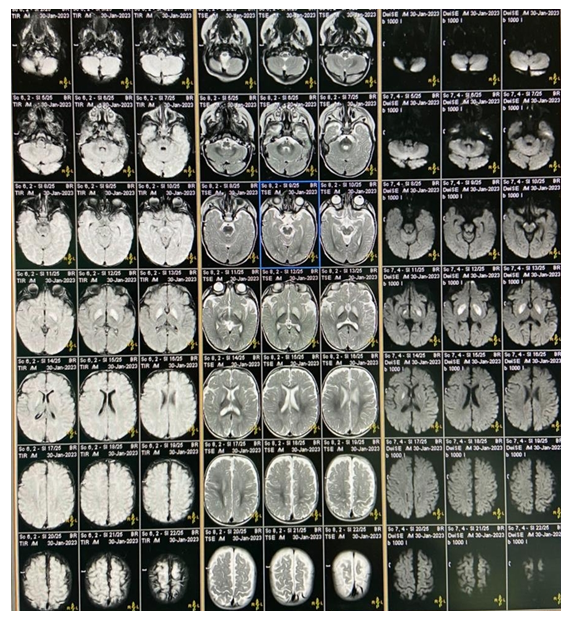
Magnetic resonance imaging (MRI) brain was done which showed hyperintensity in bilateral basal ganglia, predominantly in putamen with diffusion restriction involving bilateral cerebral peduncle; hyperintensity were also noted in subcortical location of bilateral parieto-occipital lobe. Findings were suggestive of metabolic / toxic encephalopathy. His cerebrospinal fluid examination was done which was reported normal. Biofire Meningitis Panel of cerebrospinal fluid was negative. Metabolic workup was done as history, family history, clinical examination and initial laboratory findings were suggestive of metabolic illness. Metabolic workup was in favour of Methylmalonic academia. GAS CHROMATOGRAPHY MASS SPECTROMETRY (GCMS) SCREENING showed increased excretion of Methylmalonic acid. Analyte result show Methylmalonic acid -2 level 2870.24 mmol/mol of creatinine with analyte reference value of 1216.56.
Vitamin B 12 level came normal while urine for ketones came 3 +. Whole genome sequencing showed homozygocity for a Uncertain Significance variant in the MMUT gene associated with Methylmalonic aciduria due to Methylmalonyl-CoA mutase deficiency. A Homozygote Missense variant c.1837C>T in Exon 11 of the MMUT gene that results in the amino acid substitution p. Arg613Cys was identified. He was intubated and mechanically ventilated in view of deterioration of sensorium and he was provided neuroprotective mesures. He was managed with L Carnitine along with diet specific for Methylmalonic aciduria. With time condition of the child improved and he was extubated. He also developed Central Diabetes Insipidus during the course of treatment which was managed with desmopressin. Child was discharged with the neurological Sequelae (Lost neck holding, no interest in surrounding, hypertonia in bilateral lower limbs) after 2 wks of hospitalization.
3. Discussion
Methylmalonic acidemia is caused by complete or partial deficiency of the enzyme methylmalonyl-CoA mutase (mut0 enzymatic subtype or mut– enzymatic subtype, respectively), a defect in the transport or synthesis of its cofactor, 5-deoxy-adenosyl-cobalamin (cblA, cblB, or cblD-MMA), or deficiencyof the enzyme methylmalonyl-CoA epimerase [1]. The diagnosis of MMA is established in a proband by identificationof biallelic pathogenic variants in MCEE, MMAA, MMAB, MMADHC, or MMUT or (in some instances) by significantly reduced activity of one of the following enzymes: methylmalonyl-CoA mutase, methylmalonyl-CoA mutase enzyme cofactor 5'-deoxyadenosylcobalamin, or methylmalonyl-CoA epimerase. Because of its relatively high sensitivity, easier accessibility, and noninvasive nature, molecular genetic testing can obviate the need for enzymatic testing in most instances. Methylmalonic acidemia can be classified into three general categories: vitamin B12-unresponsive apoenzyme defects, vitamin B12 coenzyme mutations, and the combined methylmalonic aciduria-homocystinuria remethylation variant. The vitamin B12-responsive form and the forms with homocystinuria are caused by defects in the metabolism of cobalamin (vitamin B12) [2]. The clinical course seen in patients can be very different depending on the type of methylmalonic acidemia. Methylmalonic acid is derived from hydrolysis of the acyl-coenzyme A substrate. Propionyl-coenzyme A and ketones accumulate in this disorder [3]. When propionyl-coenzyme A accumulates, it is toxic. All forms of disease are apparently inherited in the autosomal recessive fashion. Patients who are more severely affected are seen in early infancy with seizures, hypotonia, microcephaly, hyperammonemia, and ketoacidosis; they can have profound developmental and mental retardation as well.
Diagnosis of methylmalonic acidemia is made by the presence of large quantities of methylmaionic acid in the urine. Enzymes can be assayed to detect the specific enzymatic defect present. Treatment of the acute ketoacidotic episode of methylmaionic aciduria is similar to the treatment of propionic aciduria- decreasing (limiting) protein intake and treatment of the acidosis while maintaining adequate intake. The main principles of treatment are to provide supplemental vitamin B12 to those who are known to be vitamin B12 responsive; restrict natural protein, particularly of propiogenic amino acid precursors, while maintaining a high-calorie diet; address feeding difficulties, recurrent vomiting, and growth failure; provide supplemental carnitine to those with carnitine deficiency; reduce propionate production from gut flora; and provide emergency treatment during episodes of acute decompensation with the goal of averting catabolism and minimizing central nervous system injury.
In those with significant metabolic instability and/or renal failure, liver and/or renal transplantation may be considered.There are multiple pathogenic MMUT variants, that when in a homozygous state, lead to a specific predicted enzymatic phenotype. In our case a Homozygote Missense variant c.1837C>T in Exon 11 of the MMUT gene that resulted in the amino acid substitution p. Arg613Cys was identified. The observed variant has a minor allele frequency of 0.00012% in gnom AD exomes and genomes, respectively. The severity of the impact of this variant on the protein is medium, based on the effect of the protein and REVEL score. Rare Exome Variant Ensemble Learner (REVEL) is an ensembl method for predicting the pathogenicity of missense variants based on a combination of scores from 13 individual tools: MutPred, FATHMM v2.3, VEST 3.0, PolyPhen-2, SIFT, PROVEAN, Mutation Assessor, Mutation Taster, LRT, GERP++, SiPhy, phyloP, and phastCons. The REVEL score for an individual missense variant can range from 0 to 1, with higher scores reflecting greater likelihood that the variant is disease-causing. Based on the above evidence this variant classified as Uncertain Significance according to the ACMG guidelines.
4. Conclusion
Advanced genetic testing tools such as Whole genome sequencing, microarray etc has enabled the identification and characterization of several genes involved in various genetic syndromes at different pathophysiological levels. Thus, the report presents a rare case of Methymalonic acidemia based on advanced genetic diagnostics, which include multi gene panel study besides clinical history and MRI findings. These studies help in prenatal diagnosis and offer appropriate genetic counselling and decision making for future pregnancies
References
- Manoli I, Sloan JL, Venditti CP. Isolated Methylmalonic Acidemia. 2005 Aug 16. In: Adam MP, Everman DB, Mirzaa GM, et al., editors. Gene Reviews®. Seattle (WA): University of Washington, Seattle (1993-2023).
- Rosenberg LE, Fenton WA. Disorders of propionate and methylmalonate metabolism. In: Striver CR, Beaudet AL, Sly W, Valle D, eds. The metabolic basis of inherited disease. 6th ed. New York: McGraw-Hill (1989): 821-44.
- Scott CR. Disorders of amino acid metabolism. In: Emery EH, Rimoin DL, eds. Volume 2: principles and practices of medical genetics. New York: Churchill Livingston (1985): 1252-5.


 Impact Factor: * 6.2
Impact Factor: * 6.2 Acceptance Rate: 76.33%
Acceptance Rate: 76.33%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 