Paradoxical Vocal Fold Motion in ALS Patient: A Case Report
Abdullah AlDaihani*, Salem AlDouseri, Mohammad AlDaihani
Division of Otolaryngology- Head & Neck, Department of Surgery, Dalhousie University, Canada
*Corresponding Author: Abdullah E. AlDaihani, Division of Otolaryngology- Head & Neck, Department of Surgery, Dalhousie University, Canada
Received: 03 March 2021; Accepted: 23 March 2021; Published: 05 April 2021
Article Information
Citation: Abdullah AlDaihani, Salem AlDouseri, Mohammad AlDaihani. Paradoxical Vocal Fold Motion in ALS Patient: a Case Report. Journal of Surgery and Research 4 (2021): 182-186.
View / Download Pdf Share at FacebookAbstract
Paradoxical vocal fold motion (PVFM) can occur at any stage of amyotrophic lateral sclerosis (ALS). PVFM results in glottic narrowing or complete blockage of airway in ALS patients, which eventually leads to respiratory insufficiency and sudden death. ALS typically presents initially with weakness of hands and bulbar musculature. Rarely, hoarseness is the initial symptom in ALS. This is a case report of how PVFM presents initially with hoarseness as a first symptom of ALS.
Keywords
<p>Paradoxical vocal fold motion, Amyotrophic lateral sclerosis, Vocal fold dysfunction, Laryngeal dysfunction, Laryngospasm, Bilateral vocal fold paresis</p>
Article Details
1. Introduction
Paradoxical vocal fold motion (PVFM), also known as vocal fold dysfunction;n (VFD), is glottic narrowing due to abnormal adduction of the true vocal folds, and often of the supraglottic structures, during respiration [1]. PVFM is known by many different terminologies in literature [2,3]. Historically, the exact etiology of PVFM has been poorly understood, in which it was treated as refractory asthma [4]. PVFM affects people of all ages but is predominant in young females [5,6].
PVFM causes a wide spectrum of symptoms, predominantly presenting as dyspnea, dysphonia, dysphagia, choking sensation, gastroesophageal reflux or cough [7,8]. It also presents with attacks of inspiratory stridor, a potentially life-threatening condition, where the vocal fold paradoxically adducts during inspiration, resulting in glottic narrowing or complete occlusion [9,10]. PVFM can be induced by multiple neurogenic causes that involve brainstem lesions such as amyotrophic lateral sclerosis (ALS) [10].
ALS is a neurodegenerative disorder characterized by progressive loss of both upper and lower motor neurons. Typically, involvement of weakness of hands and bulbar musculature, such as dysarthria and dysphagia, presents in initial manifestations. ALS progresses quickly, resulting in disability and respiratory failure that commonly leads to death 3–4 years after onset [11]. PVFM can occur at any stage of ALS progression [12]. Laryngospasm, another symptom of PVFM characterized by acute uncontrollable adduction of the vocal adduction, may account for sudden death of ALS patients [13].
PVFM and laryngospasm in ALS patients are mentioned less often in neurology literature than in otolaryngology literature [14,15]. Out of 441 ALS patients, only 3.9% reported hoarseness as the initial manifestation [15].
In this study, we discuss a case presented in our hospital with hoarseness as a first symptom of ALS. Three subtypes of ALS based on the symptomatology at disease onset. Bulbar subtypes are characterized by lower motor neuron damage in the brainstem, corticobulbar subtypes are characterized by damage in the brain stem and corticobulbar tract, and corticospinal subtypes are characterized by initial symptoms in the limbs secondary to anterior horn cell and possibly corticospinal tract involvement. Bulbar and corticobulbar subtypes often manifest early difficulties in speech and/or swallowing, and bulbar subtypes seem to have a more rapidly progressive disease pattern compared to corticospinal subgroups. Bulbar subtypes also manifest a faster rate of decline of orofacial and phonatory functions [6].
In regards to the phonatory system, perceptual features can include harshness, strain-strangled voice, breathiness, tremor, and pitch abnormalities [1]. In addition, features of spasmodic dysphonia (or focal laryngeal dystonia) have also been reported in speakers with ALS [7]. In cases of bulbar involvement, dysphonia can be the initial clinical symptom, and often results in a referral to the otolaryngologist before the diagnosis of ALS has been made [8]. Typically, laryngeal structure is normal in appearance. When corticobulbar involvement dominates, there is often a pattern of hyper-adduction of the vocal mechanism, and when bulbar involvement dominates, there is often a pattern of hypoadduction.
2. Case Report
A 42-year-old Indian female was initially admitted to the medical ward in Adan Hospital, Kuwait City as a case of Pneumonia in July 2018, confirmed by chest x-ray. Upon physical examination, she presented with hoarseness, short phonation time, hypophonia and dysphagia. She had not sought any medical advice pre-admission, although her symptoms had become progressively worse for the last few months, but she attributed it to her recent pneumonia and couple episodes of cold she had in February and April 2018.
One week post-admission, she developed weakness and hyperreflexia of her right arm. A neurology consultation was requested, and all blood work came back normal. Her CT head scan was normal as well, with no signs of cerebrovascular abnormality. Electromyography was requested, and ALS diagnosis was confirmed.
The patient’s speech evaluation, conducted by a speech therapist, indicated severe dysarthria characterized as flaccid, with compensatory hyperactivity in the laryngeal musculature. The respiratory and phonatory systems were noted as being more affected than articulation.
After the patient recovered from pneumonia, she was scheduled for an otolaryngology consultation. A Fiberoptic (Figure 1) and video laryngoscopic assessment were also performed by an ENT Surgeon, which adduction of the vocal cords with inspiration and abduction of the vocal cords with expiration. It wasn’t relived by distraction.
The patient was discharged against our medical advice and never showed up for any follow up afterwards. Nine months later, the patient came back to emergency department with an attack of acute dyspnea and stridor, most frequently at night. Spirometry was done and it showed severe impairment of respiratory flow through the larynx. Flexible laryngoscopy (Figure 2) revealed bilateral vocal cord abduction paresis; subsequently, tracheotomy was performed, she remained in the hospital for one week. One year after the tracheotomy, she experienced respiratory failure and passed away in the ICU after a failed attempt at resuscitation.
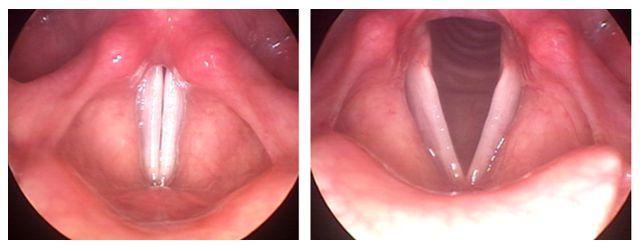
Figure 1: Flexible laryngoscopic view of the vocal cords from the patient, the right is during inspiration and left during expiration.
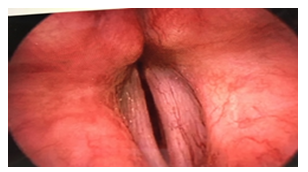
Figure 2: Flexible laryngoscopy 9 months later revealed bilateral vocal cord abduction paresis.
3. Conclusion
Hoarseness or stridor can be an initial symptom of ALS. Therefore, in cases of Paradoxical vocal fold motion of unknown aetiology, ALS should be taken into consideration in the context of differential diagnosis. Patients presenting with hoarseness, short phonation time, hypophonia and dysphagia should raise suspicion and justify prompt referral to an otolaryngologist. There is no enough data in this specific symptom. A case series would be a very good way to start.
Funding
None
Conflict of Interest
None declared
Ethical Approval
Not required
References
- Parsons JP, Benninger C, Hawley MP, Philips G, Forrest LA, Mastronarde JG. Vocal cord dysfunction: beyond severe asthma. Respiratory medicine 104 (2010): 504-509.
- Petrov AA. Vocal Cord Dysfunction: The spectrum across the ages. Immunology and Allergy Clinics 39 (2019): 547-560.
- Christensen PM, Heimdal JH, Christopher KL, Bucca C, Cantarella G, Friedrich G, et al. ERS/ELS/ACCP 2013 international consensus conference nomenclature on inducible laryngeal obstructions. European Respiratory Review 24 (2015): 445-450.
- Yelken K, Yilmaz A, Guven M, Eyibilen A, Aladag I. Paradoxical vocal fold motion dysfunction in asthma patients. Respirology 14 (2009): 729-733.
- Maturo S, Hill C, Bunting G, Baliff C, Ramakrishna J, Scirica C, et al. Pediatric paradoxical vocal-fold motion: presentation and natural history. Pediatrics 128 (2011): e1443-e1449.
- Forrest LA, Husein T, Husein O. Paradoxical vocal cord motion: classification and treatment. The Laryngoscope 122 (2012): 844-853.
- Koufman JA, Block C. Differential diagnosis of paradoxical vocal fold movement. American Journal of Speech-Language Pathology (2008).
- Marcinow AM, Thompson J, Chiang T, Forrest LA, deSilva BW. Paradoxical vocal fold motion disorder in the elite athlete: experience at a large division I university. The Laryngoscope 124 (2014): 1425-1430.
- Morris MJ, Christopher KL. Diagnostic criteria for the classification of vocal cord dysfunction. Chest 138 (2010): 1213-1223.
- Vander Graaff MM, Grolman W, Westermann EJ, Boogaardt HC, Koelman H, Vander Kooi AJ, et al. Vocal cord dysfunction in amyotrophic lateral sclerosis: four cases and a review of the literature. Archives of neurology. 66 (2009): 1329-1333.
- Van Es MA, Hardiman O, Chio A, Al-Chalabi A, Pasterkamp RJ, Veldink JH, et al. Amyotrophic lateral sclerosis. The Lancet 390 (2017): 2084-2098.
- Bigliardi G, Malaguti MC, Sola P, Georgoulopoulou E, Tondelli M, Barbi F, et al. Bilateral vocal cord paralysis: a rare onset of amyotrophic lateral sclerosis. Archives of neurology 67 (2010): 897-899.
- Gdynia HJ, Kassubek J, Sperfeld AD. Laryngospasm in neurological diseases. Neurocritical care 4 (2006): 163.
- Forshew D, Bromberg M. A survey of clinicians' practice in the symptomatic treatment of ALS. Amyotrophic Lateral Sclerosis and other motor neuron disorders 4 (2003): 258-263.
- Carpenter RJ, McDonald TJ, Howard Jr FM. The otolaryngologic presentation of amyotrophic lateral sclerosis. Otolaryngology 86 (1978): 479.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 