Rare Keratopathy of Bilateral Anterior Amorphous Corneal Opacities with Spontaneous Corneal Stromalysis and Unilateral Descemetocele - Quantitative Framing with AS-OCT and Scheimpflug Imaging in inverse contrast
Shruthy Vaishali Ramesh1, Prasanna Venkatesh Ramesh2*, Aji K3, Meena Kumari Ramesh4, Ramesh Rajasekaran5
1Medical Officer, Department of Cataract and Refractive Surgery, Mahathma Eye Hospital Private Limited, Trichy, India
2Medical Officer, Department of Glaucoma and Research, Mahathma Eye Hospital Private Limited, Trichy, India
3Optometrist, Mahathma Eye Hospital Private Limited, Trichy, India
4Head of the Department of Cataract and Refractive Surgery, Mahathma Eye Hospital Private Limited, Trichy, India
5Chief Medical Officer, Mahathma Eye Hospital Private Limited, Trichy, India
*Corresponding author: Dr. Prasanna Venkatesh Ramesh, Department of Glaucoma and Research, Mahathma Eye Hospital Private Limited, Trichy, India
Received: 18 May 2021; Accepted: 25 May 2021; Published: 27 May 2021
Article Information
Citation: Shruthy Vaishali Ramesh, Prasanna Venkatesh Ramesh, Aji K, Meena Kumari Ramesh, Ramesh Rajasekaran. Rare Keratopathy of Bilateral Anterior Amorphous Corneal Opacities with Spontaneous Corneal Stromalysis and Unilateral Descemetocele - Quantitative Framing with AS-OCT and Scheimpflug Imaging in inverse contrast. Journal of Surgery and Research 4 (2021): 301-307.
View / Download Pdf Share at FacebookAbstract
Posterior amorphous corneal dystrophy (PACD) is classically characterised by bilaterally thin and flat central corneas with a deep, stromal sheet-like opacity. The epithelium, Bowman's layer, and anterior stroma are usually normal in PACD. Here, we report a case that had bilaterally anterior oval amorphous corneal opacities, which is different from the typical presentation of PACD. Corneal stromalysis occurred in them with progression to descemetocele unilaterally. To our knowledge, Scheimpflug images of anterior amorphous opacities especially with spontaneous corneal stromalysis and descemetocele are poorly reported in literature. Hence the objective of this manuscript was to throw light to the lacunae in literature, by describing the Scheimpflug images of this rare keratopathy in default and inverse contrast for better quantitative framing of the pathology.
Keywords
<p>Amorphous Keratopathy, Spontaneous Stromalysis, Descemetocele, Scheimpflug Imaging</p>
Article Details
1. Introduction
A flat and thin central cornea is a rare keratopathy, caused by long-term contact lens usage, dellen or posterior amorphous corneal dystrophy (PACD). PACD is characterised by bilaterally, thin and flat central corneas with deep, stromal sheet-like opacity where the epithelium, Bowman's layer, and anterior stroma are normal [1-3] .
2. Case Report
We report a case of anterior oval amorphous corneal opacities OU (Figure 1), which is different from its typical presentation. To add on; in this scenario, bilateral spontaneous corneal stromalysis had occurred with progression to descemetocele unilaterally OS. These anterior amorphous opacities were relatively transparent and were not associated with systemic or autoimmune diseases in this 66 year old female. Keratometry and corneal topography revealed a flat K (K1 8.00D@109°, K2 34.49@19° OD; K1 17.44@58°, K2 31.23@148°) in the central cornea. Anterior segment optical coherence tomography (AS-OCT) revealed (Figures 2 and 3) anterior hypo-reflective abnormalities in the amorphous opacities and some deep hyper-reflective opacities.
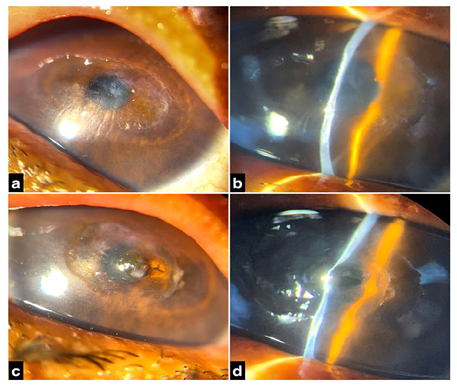
Figure 1a, b- Slit-lamp bio-microscopy in diffuse and slit examination respectively, showing an horizontally oval sheet-like opacity in the central, thin and flat cornea with Descemet folds associated with amorphous paracentral whitish deposits in OD.
c, d- Slit lamp bio-microscopy in diffuse and slit examination respectively, showing a central descemetocele with surrounding horizontally oval sheet-like opacity in the central, thin and flat cornea in OS.
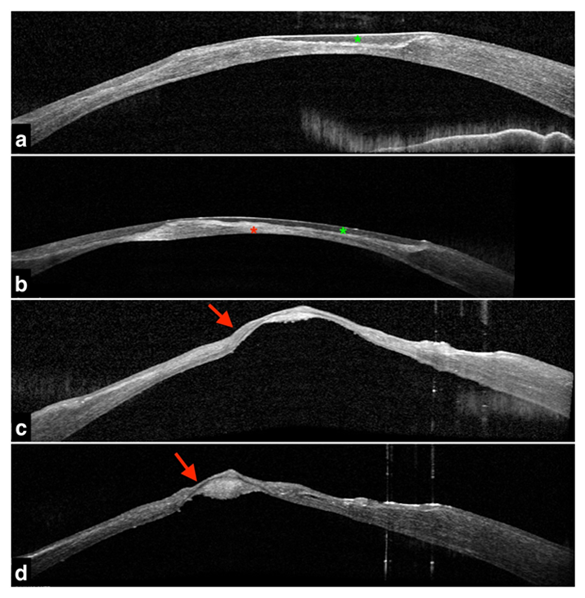
Figure 2: Anterior segment optical coherence topography.
a,b- Hypo-reflective abnormalities (green asterisk) are found in the anterior corneal stroma with some deep hyper-reflective opacities with an irregular thick Descemet's membrane (red asterisk) seen in OD.
c,d- Descemetocele (red arrows) seen in OS due complete loss of anterior stroma.
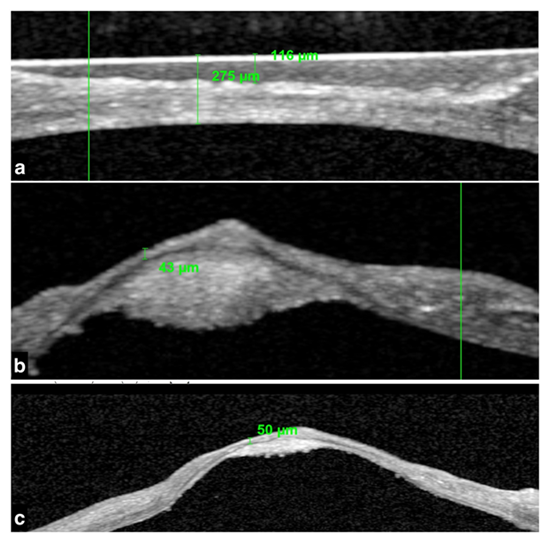
Figure 3a,b,c- Quantitative analysis of the central flat cornea in OD and descemetocele in OS done utilising anterior segment optical coherence topography.
4. Discussion
PACD is a rare, autosomal dominant disorder. The affected individuals classically demonstrate partial or complete posterior lamellar corneal opacification with decreased corneal thickness and flattening of the corneal curvature. Significant phenotypic variability exists; however, not only in the severity of these corneal findings, but also in the expression of a number of other associated abnormalities of the anterior ocular segment, including scleralization of the peripheral cornea, iris coloboma, corectopia, iris atrophy, and iridocorneal adhesions [4-12].
The anterior amorphous opacities in this case were relatively transparent and had vesicle-like appearance, which is different from the typical presentation. Despite multimodal imaging (Figure 4), we still cannot rule out the possibility of other variants of PACD. Additionally confocal imaging and histopathological examination are warranted [13] . Also, presumably if a corneal epithelium defect occurred, due to trivial trauma at the amorphous vesicle, these opacities melted easily and progressed to descemetocele within a short period [14]. To our knowledge, anterior amorphous opacities with spontaneous corneal stromalysis and descemetocele are poorly reported in literature. Hence this manuscript, aimed to throw light, by describing this rare keratopathy with Scheimpflug images.
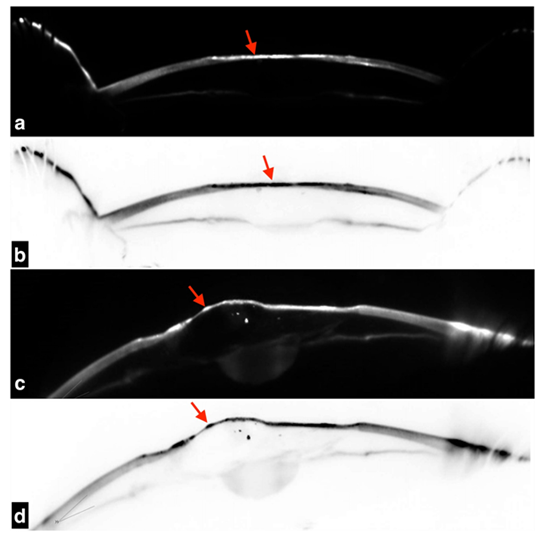
Figure 4a,b- Scheimpflug imaging of OD showing hyper-reflection in the anterior stromal region (red arrow) and a dark area (red arrow) in inverse colour respectively. c&d. Scheimpflug imaging of OS showing descemetocele with patchy hyper-reflection (red arrow) and dark patchy areas (red arrow) seen in inverse colour respectively.
4.1 Genetics
Genome-wide linkage analysis performed on the largest PACD family identified to date demonstrated a linkage to a 3.5 Mb region on chromosome 12q21.33. In total 26 genes were mapped to this region. Out of the 26, four of them namely, keratocan, lumican, decorin and epiphycan; encode small leucine-rich proteoglycans (SLRPs), a family of proteins involved in collagen fibrillogenesis and matrix assembly. In the cornea, these SLRPs are bound to glycosaminoglycans, thus forming the ground substance of the cornea [4]. They play an essential role in the maintenance of corneal transparency. Mutations in the SLRPs have been associated with corneal pathologies involving corneal clarity and curvature. Missense and nonsense mutations in keratocan are implicated in autosomal recessive cornea plana [15,16]. Nonsense mutations in decorin are implicated in congenital hereditary stromal dystrophy. PACD is however associated with a deletion of keratocan, lumican, decorin and epiphycan [17,18]. The implication of the deletion of the SLRP gene cluster forms the genetic basis of PACD. This provides additional evidence of the role of these genes in the development and maintenance of corneal clarity. PACD is associated with haploinsufficiency of SLRPs on chromosome 12, and is primarily caused by it.
4.2 Limitations
This case may be an atypical variant of PACD or other types of the rare amorphous corneal dystrophy/degeneration. Further genetic studies with genome-wide linkage analysis for more cases are needed to clarify this rare keratopathy.
5. Conclusion
To our knowledge, scheimpflug images of anterior amorphous corneal opacities, especially with spontaneous corneal stromalysis and descemetocele are poorly reported in literature. Hence the objective of this manuscript was to throw light to the lacunae in literature, by describing the Scheimpflug images of this rare keratopathy in default and inverse contrast for better quantitative framing of this pathologic process.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Yeniad B, Yigit B, Kozer BL et al. Effect of contact lenses on corneal thickness and corneal curvature during usage. Eye Contact Lens 29 (2003): 223-229.
- Insler MS, Tauber S, Packer A. Descemetocele formation in a patient with a postoperative corneal dellen. Cornea 8 (1989): 129-130.
- Carpel EF, Sigelman RJ, Doughman DJ. Posterior amorphous corneal dystrophy. Am J Ophthalmol 83 (1977): 629-632.
- Aldave AJ, Rosenwasser GO, Yellore VS, et al. Linkage of posterior amorphous corneal dystrophy to chromosome 12q21.33 and exclusion of coding region mutations in KERA, LUM, DCN, and EPYC. Invest Ophthalmol Vis Sci 51 (2010): 4006-4012.
- Castelo BB, Chalita MR, Casanova FH, et al. Posterior amorphous corneal dystrophy: ultrasound biomicroscopy findings in two cases. Cornea 21 (2002): 220-222.
- Dunn SP, Krachmer JH, Ching SS. New findings in posterior amorphous corneal dystrophy. Arch Ophthalmol 102 (1984): 236-239.
- Erdem U, Muftuoglu O, Hurmeric V. In vivo confocal microscopy findings in a patient with posterior amorphous corneal dystrophy. Clin Experiment Ophthalmol 35 (2007): 99-102.
- Grimm BB, Waring GO, Grimm SB. Posterior amorphous corneal dysgenesis. Am J Ophthalmol 120 (1995): 448-455.
- Johnson AT, Folberg R, Vrabec MP, et al. The pathology of posterior amorphous corneal dystrophy. Ophthalmology 97 (1990): 104-109.
- Moshegov CN, Hoe WK, Wiffen SJ, et al. Posterior amorphous corneal dystrophy. A new pedigree with phenotypic variation. Ophthalmology 103 (1996): 474-478.
- Oliveira LA, Vieira LA, Freitas D, et al. Posterior amorphous corneal dystrophy: case report. Arq Bras Oftalmol 69 (2006): 945-947.
- Roth SI, Mittelman D, Stock EL. Posterior amorphous corneal dystrophy. An ultrastructural study of a variant with histopathological features of an endothelial dystrophy. Cornea 11 (1992): 165-172.
- Waring GO, Malaty A, Grossniklaus H, et al. Climatic proteoglycan stromal keratopathy, a new corneal degeneration. Am J Ophthalmol 120 (1995): 330-341.
- Klintworth GK. Corneal dystrophies. Orphanet J Rare Dis 4 (2009): 7.
- Hassell JR, Birk DE The molecular basis of corneal transparency. Exp Eye Res 91 (2010): 326-335.
- Iozzo RV. Matrix proteoglycans: from molecular design to cellular function. Annu Rev Biochem 67 (1998): 609-652.
- Ebenezer ND, Patel CB, Hariprasad SM, et al. Clinical and molecular characterization of a family with autosomal recessive cornea plana. Arch Ophthalmol 123 (2005): 1248-1253.
- Bredrup C, Knappskog PM, Majewski J, et al. Congenital stromal dystrophy of the cornea caused by a mutation in the decorin gene. Invest Ophthalmol Vis Sci 46 (2005): 420-426.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 