Visual Function Markers in Cardiac Surgery-Impact of Fundus Fluorescein Angiography in Assessing Ocular Circulation
Vasiliki Trantou1, Dimitrios V Moysidis2, Rodopi Stamatiou3, Andreas S Papazoglou2, Maria Kotoula4, Isaac Aidonidis3*, Ioannis Skoularigis5, Nikolaos Tsilimingas6, Tsironi Evangelia4
1Opthalmology Department, University Hospital of Larisa, University of Thessaly, Larisa, Greece
2AHEPA University Hospital of Thessaloniki, Aristotle University of Thessaloniki, Thessaloniki, Greece
3Department of Physiology, University of Thessaly Medical School, Larisa, Greece
4Opthalmology Department, University Hospital of Larisa, University of Thessaly, Larisa, Greece
5Cardiology Department, University Hospital of Larisa, University of Thessaly, Larisa, Greece
6Cardiothoracic Department, University Hospital of Larisa, University of Thessaly, Larisa, Greece
*Corresponding Author: Isaac Aidonidis, Associate Professor of Medical Physiology (Cardiovascular Section), University of Thessaly, Biopolis, Panepistimiou Street 3, Medical School of Larissa, Greece
Received: 29 April 2020; Accepted: 11 May 2020; Published: 14 May 2020
Article Information
Citation: Vasiliki Trantou, Dimitrios V Moysidis, Rodopi Stamatiou, Andreas S Papazoglou, Maria Kotoula, Isaac Aidonidis, Ioannis Skoularigis, Nikolaos Tsilimingas, Tsironi Evangelia. Visual Function Markers in Cardiac Surgery-Impact of Fundus Fluorescein Angiography in Assessing Ocular Circulation. Journal of Surgery and Research 3 (2020): 102-111.
View / Download Pdf Share at FacebookAbstract
Objectives: Cardiac surgery has been reported to be associated, albeit rarely, with postoperative vision alterations up to vision loss. In this study, we sought to examine if patients undergoing on-pump cardiac surgery are at higher risk for developing deranged vision based on analysis of standard visual parameters.
Methods: A total of 88 patients, who underwent coronary artery bypass graft (CABG, n=56) or aortic valve replacement (AVR, n=32) surgery were initially enrolled in our study in order to assess their visual function, right before and one month after surgery. The following examinations were performed: Visual Acquity (VA), Intraocular Pressure (IOP), Fundus Fluorescein Angiography Time (FFAT), and Optical Coherence Tomography (OCT). The collected data were statistically analysed by performing Mann Witney U Test in SPSS (CI=95%).
Results: One out of 88 patients (1.13%) showing complete vision loss after valvular surgery was excluded from study. From the remaining patients, 60 (69%) showed a decrease in the postoperative FFAT, while in 27 (31%) FFAT was increased. FFATs were improved in more patients with AVR (74%) than in those with CABG (66%) (p<0.05). Patients with extracorporeal circulation (ECC) time <90 min displayed improvement in FFAT, whereas those with ECC times >101 min had worser post- vs preoperative FFATs (38 vs 56%, respectively). No significant differences were observed in VA, IOP, OCT findings between the CABG and AVR group.
Conclusions: The vast majority of patients did not have any vision complications after surgery. They showed shorter FFATs post- vs preoperatively, implicating improved retinal flow. FFAT seemed to be dependent, at least partially, on the ECC time and possibly the type of operation.
Keywords
Open heart surgery, Postoperative vision disorders, Visual function markers, Fundus fluorescein angiography, Ocular microcirculation
Open heart surgery articles, Postoperative vision disorders articles, Visual function markers articles, Fundus fluorescein angiography articles, Ocular microcirculation articles
Open heart surgery articles Open heart surgery Research articles Open heart surgery review articles Open heart surgery PubMed articles Open heart surgery PubMed Central articles Open heart surgery 2023 articles Open heart surgery 2024 articles Open heart surgery Scopus articles Open heart surgery impact factor journals Open heart surgery Scopus journals Open heart surgery PubMed journals Open heart surgery medical journals Open heart surgery free journals Open heart surgery best journals Open heart surgery top journals Open heart surgery free medical journals Open heart surgery famous journals Open heart surgery Google Scholar indexed journals Postoperative vision disorders articles Postoperative vision disorders Research articles Postoperative vision disorders review articles Postoperative vision disorders PubMed articles Postoperative vision disorders PubMed Central articles Postoperative vision disorders 2023 articles Postoperative vision disorders 2024 articles Postoperative vision disorders Scopus articles Postoperative vision disorders impact factor journals Postoperative vision disorders Scopus journals Postoperative vision disorders PubMed journals Postoperative vision disorders medical journals Postoperative vision disorders free journals Postoperative vision disorders best journals Postoperative vision disorders top journals Postoperative vision disorders free medical journals Postoperative vision disorders famous journals Postoperative vision disorders Google Scholar indexed journals Visual function markers articles Visual function markers Research articles Visual function markers review articles Visual function markers PubMed articles Visual function markers PubMed Central articles Visual function markers 2023 articles Visual function markers 2024 articles Visual function markers Scopus articles Visual function markers impact factor journals Visual function markers Scopus journals Visual function markers PubMed journals Visual function markers medical journals Visual function markers free journals Visual function markers best journals Visual function markers top journals Visual function markers free medical journals Visual function markers famous journals Visual function markers Google Scholar indexed journals Fundus fluorescein angiography articles Fundus fluorescein angiography Research articles Fundus fluorescein angiography review articles Fundus fluorescein angiography PubMed articles Fundus fluorescein angiography PubMed Central articles Fundus fluorescein angiography 2023 articles Fundus fluorescein angiography 2024 articles Fundus fluorescein angiography Scopus articles Fundus fluorescein angiography impact factor journals Fundus fluorescein angiography Scopus journals Fundus fluorescein angiography PubMed journals Fundus fluorescein angiography medical journals Fundus fluorescein angiography free journals Fundus fluorescein angiography best journals Fundus fluorescein angiography top journals Fundus fluorescein angiography free medical journals Fundus fluorescein angiography famous journals Fundus fluorescein angiography Google Scholar indexed journals Ocular microcirculation articles Ocular microcirculation Research articles Ocular microcirculation review articles Ocular microcirculation PubMed articles Ocular microcirculation PubMed Central articles Ocular microcirculation 2023 articles Ocular microcirculation 2024 articles Ocular microcirculation Scopus articles Ocular microcirculation impact factor journals Ocular microcirculation Scopus journals Ocular microcirculation PubMed journals Ocular microcirculation medical journals Ocular microcirculation free journals Ocular microcirculation best journals Ocular microcirculation top journals Ocular microcirculation free medical journals Ocular microcirculation famous journals Ocular microcirculation Google Scholar indexed journals Ocular Perfusion Pressure articles Ocular Perfusion Pressure Research articles Ocular Perfusion Pressure review articles Ocular Perfusion Pressure PubMed articles Ocular Perfusion Pressure PubMed Central articles Ocular Perfusion Pressure 2023 articles Ocular Perfusion Pressure 2024 articles Ocular Perfusion Pressure Scopus articles Ocular Perfusion Pressure impact factor journals Ocular Perfusion Pressure Scopus journals Ocular Perfusion Pressure PubMed journals Ocular Perfusion Pressure medical journals Ocular Perfusion Pressure free journals Ocular Perfusion Pressure best journals Ocular Perfusion Pressure top journals Ocular Perfusion Pressure free medical journals Ocular Perfusion Pressure famous journals Ocular Perfusion Pressure Google Scholar indexed journals Intraocular Pressure articles Intraocular Pressure Research articles Intraocular Pressure review articles Intraocular Pressure PubMed articles Intraocular Pressure PubMed Central articles Intraocular Pressure 2023 articles Intraocular Pressure 2024 articles Intraocular Pressure Scopus articles Intraocular Pressure impact factor journals Intraocular Pressure Scopus journals Intraocular Pressure PubMed journals Intraocular Pressure medical journals Intraocular Pressure free journals Intraocular Pressure best journals Intraocular Pressure top journals Intraocular Pressure free medical journals Intraocular Pressure famous journals Intraocular Pressure Google Scholar indexed journals Optic Nerve Head articles Optic Nerve Head Research articles Optic Nerve Head review articles Optic Nerve Head PubMed articles Optic Nerve Head PubMed Central articles Optic Nerve Head 2023 articles Optic Nerve Head 2024 articles Optic Nerve Head Scopus articles Optic Nerve Head impact factor journals Optic Nerve Head Scopus journals Optic Nerve Head PubMed journals Optic Nerve Head medical journals Optic Nerve Head free journals Optic Nerve Head best journals Optic Nerve Head top journals Optic Nerve Head free medical journals Optic Nerve Head famous journals Optic Nerve Head Google Scholar indexed journals Extracorporeal Circulation articles Extracorporeal Circulation Research articles Extracorporeal Circulation review articles Extracorporeal Circulation PubMed articles Extracorporeal Circulation PubMed Central articles Extracorporeal Circulation 2023 articles Extracorporeal Circulation 2024 articles Extracorporeal Circulation Scopus articles Extracorporeal Circulation impact factor journals Extracorporeal Circulation Scopus journals Extracorporeal Circulation PubMed journals Extracorporeal Circulation medical journals Extracorporeal Circulation free journals Extracorporeal Circulation best journals Extracorporeal Circulation top journals Extracorporeal Circulation free medical journals Extracorporeal Circulation famous journals Extracorporeal Circulation Google Scholar indexed journals
Article Details
Abbreviations:
CABG-Coronary Artery Bypass Grafting; AVR-Aortic Valve Replacement; ECC-Extracorporeal Circulation; CPB-Cardiopulmonary Bypass; FFA-Fundus Fluorescein Angiography; VA-Visual Acuity; IOP-Intraocular Pressure; OCT-Optical coherence tomography; ACCT-Aortic Cross-Clamp Time; ONH-Optic Nerve Head; OPP-Ocular Perfusion Pressure
1. Introduction
Heart surgical operations rank among the most frequently performed operations worldwide. Their number has increased over the last decades due to the elevated frequency of metabolic syndrome causing factors, prolongation of life expectancy and technological improvements. In Greece and Germany, the approximate number of adult open heart surgery, which are conducted each year, approach 8.500 and 75.000 [1, 2], correspondingly, while coronary artery bypass grafting (CABG) and aortic valve replacement(AVR) consist the vast majority of them. One of the most significant factors contributing to the flowering of cardiac surgery and amelioration of its outcomes is the implication of extracorporeal circulation (ECC). Nevertheless, it has been blamed for potential complications, such as systematic inflammatory response, disseminated intravascular coagulation, neurological defects, multiple organ dysfunction, and especially, cerebral, renal, pulmonary and visual disorders [3]. The main responsible pathophysiological mechanisms leading to organ failure during and after cardiac surgery seem to be microembolisation, hypoperfusion and hypothermia [4]. This study has been designed to assess potential effects of cardiopulmonary bypass on visual function of patients utilizing markers enabling a comprehensive comparative assessment of visual alterations pre- versus postoperatively.
2. Material and Methods
Patients undergoing cardiac surgery in the General University Hospital of Larissa for the 5-year period between January 2015 and December 2019 were after informed consent prospectively enrolled in our database, so as to be examined for any differentiation in their visual function pre-and after surgery. Patients with diabetes mellitus and patients with severe vision problems were excluded from the study. More specifically, exclusion criteria included visual acquity less than 5/10, glaucoma, cataract, macular disorders, diabetic and hypertensive fundopathy. Our assessment was based on the following examinations: Visual Acuity Examination (VA), Intraocular Pressure Measurement (IOP), Fundus Fluorescein Angiography (FFA) and Optical coherence tomography (OCT). OCT detects defaults in the central macular area, the ganglion cell layer (GCL), the optic nerve head, the retina nerve fibrous layer (RNFL). FFA is a technique used to detect pathologies in the circulation of the retina and choroid. In order to assay the perfusion status in these parts of the fundus, we took photographs (2 photos/second) of the retina at different intervals by intravenously injecting the fluorescein (an orange water-soluble dye that when injected intravenously remains largely intravascular and circulates in the bloodstream).
The recorded phases of angiography are the following:
i. Arm-retina (Pre-arterial & arterial)
ii. Early venous
iii. Middle venous (arteriovenous)
iv. Late venous.
All the surgeries were performed by the same team of cardiothoracic surgeons and ECC tecnhicians. A total of 88 patients, 73 males and 15 females (age ranging from 30 to 74 years) having no significant eye disease comprised originally the study sample. One of them sustained vision loss post-operatively and hence was excluded from a further visual markers’ investigation. For the examination of the remaining 87 patients, we defined as improvement of their ocular microcirculation function the shortening of FFA phases duration (Group 1) and as deterioration its prolongation (Group 2). The improvement or deterioration of these 87 patients was assessed:
i. After any cardiac surgery operation (regardless of its type)
ii. In terms of the type of surgery (CABG or AVR)
iii. In terms of ECC time
iv. In terms of aortic cross-clump time (ACCT).
For the performance of the diagnostic examinations mydriasis was induced with the application of tropicamide 0.5%. The study was approved by the Deanship of University of Larisa, the hospital and the Healthcare Trust Ethics Committee. Every patient attested informed consent before being enrolled in our study database.
2.1 Statistics
Statistical analysis of the collected data was executed via Statistical Package for Social Analysis (SPSS) performing independent sample Mann Whitney U test. The tests were two-sided, while Confidence Interval was equal to 95% (statistical significance set at 0.05). In order to summarize the statistical analysis of studied variables, mean and Standard Deviation values were used for demonstration.
3. Results
3.1 Measurements independently on type of cardiac operation
In total of 87 included patients, 60 (69%) demonstrated postoperative improvement of their visual function (Group 1: decrease in FFA time) with greater decrease in Late venous phase, whereas the rest 27(21%) patients showed deterioration (Group 2: increase in FFA time). Both of these results satisfied the set level of statistical significance (p< 0.05). Preoperative (pre-op) FFA time (mean total duration of all phases) was statistically significantly longer in Group 1 (mean time: 120 seconds) than in Group 2 (mean time: 102 seconds), and thus could be used as a predictive marker of prognostic value, as depicted in Figure 2. In regard to VA, no statistically significant differentiation was found pre- and post-operatively in both groups (Group 1: mean VA= 0.90 ± 0.09 and Group 2: mean VA= 0.87 ± 0.12). It is noticeable that preoperative VA is greater in patients of Group 1 than in patients of Group 2. IOP was increased postoperatively in Group 1(pre-op:13.93 and post-op: 14.03 mmHg), whereas it was decreased in Group 2(pre-op:14.07 and post-op: 13.93 mmHg).
3.2 Assessment in terms of the type of surgery (CABG or AVR)
As illustrated in Figure 3 there was greater percentage of patients with FFA improvement in AVR (74%) than in CABG (66%). Moreover, in AVR surgeries we found higher percentage of decrease in FFA time (Late phase postoperative decrease: 17%) than in CABG (4% decrease). Besides the above mentioned results, another finding that is worth citing is the statistically significant deterioration in patients of Group 2 undergoing CABG, in contrast with the non-significant one (p>0.05) in Group 2 patients undergoing AVR. Additionally, in patients of Group 1 (improvement of retina and ONH microcirculation) we noticed that CABG resulted in a statistically significant difference between pre- and post-operative early-arm time values, while for patients undergoing AVR the statistically significant difference referred to middle-early phase, as shown in Figure 4. The preoperative values of IOP in patients of Group 1 who underwent CABG (14.11 ± 1.78 mmHg) were higher than those of Group 2 patients with CABG (13.58 ± 1.95 mmHg). On the contrary, preoperative values (13.65 ± 1.5 mmHg) of IOP in patients, who underwent AVR surgery and were visually ameliorated, were lower than the corresponding ones (15.25 ± 0.89 mmHg) of those who aggravated. Therefore high preoperative values of IOP in CABG patients and correspondingly low values in valvular disease patients could be used as positive prognostic indicator for assessing the overall postoperative vessel condition.
3.3 Assessment in terms of ECC time
We categorised ECC duration into 3 subgroups: a) <90 minutes: 30 patients, b) 91-100 minutes: 14 patients, and c) >101 minutes: 43 patients. Out of 60 patients of Group 1: 23 (38%) were <90 minutes in ECC, 10 (17%) 91-100 minutes and 27 (45%) >101 minutes. Out of 27 patients of Group 2: 7 (26%) were <90 minutes in ECC, 4 (15%) 91-100 minutes and 16 (59%) >101 minutes. The percentage of patients of Group 1 (38%) who have been in ECC for less than 90 minutes is significantly greater than the corresponding percentage of Group 2 patients (26%). On the other hand, 45% of Group 1 and 59% of Group 2 patients were in ECC for more than 101 minutes. The one male patient which lost his vision and excluded from further investigation, remained in ECC for more than 101 minutes.
Consequently, the duration of ECC application in cardiac operations seems to be an important factor influencing postoperative visual impairment. However, we found no statistically significant impact of ECC duration on postoperative FFA time in any of the 3 subgroups. There was no statistically significant differentiation in VA and IOP values pre- and post- operatively in regards with ECC time. However, patients with elevated VA and IOP preoperative values needed slightly prolonged application of ECC, even if there is no adequate statistical evidence to advocate that.
3.4 Assessment in terms of ACCT
In order to evaluate postoperative visual function in terms of ACCT, we separated our sample into 3 subgroups: 31 patients: <60 minutes, 44 patients: 61-90 minutes and 12 patients: >91 minutes. The statistical analysis proved that the number of improved patients was not statistically different than the number of those deteriorating depending on ACCT duration and thus ACCT should not be proposed as a prognostic marker. It is impressive, though, that statistically significant decrease in FFA time (improvement) was found only in patients with ischemia time >60 minutes and not in patients under ACCT for less than 60 minutes.
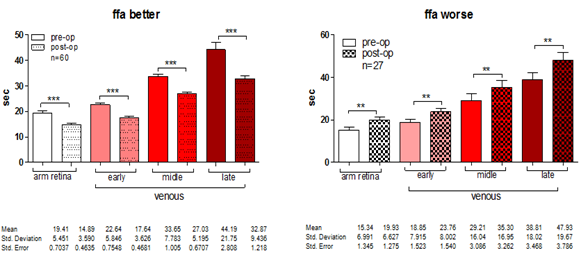
Figure 1: FFA of all patients included in the study.
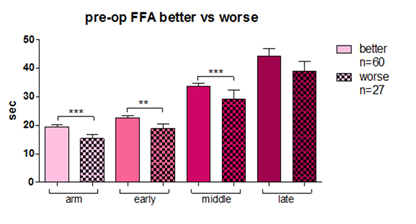
Figure 2: Comparison of preoperative FFA time in Group 1 and 2.
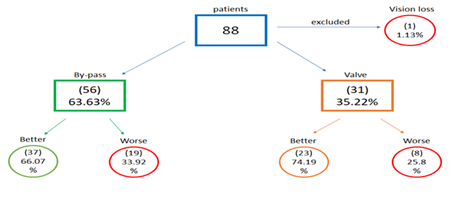
Figure 3: Improvement or deterioration in respect to the type of surgery.
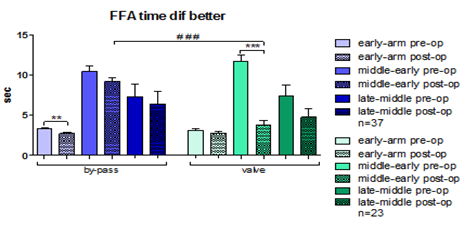
Figure 4: FFA time differences in relevance with the type of surgery.
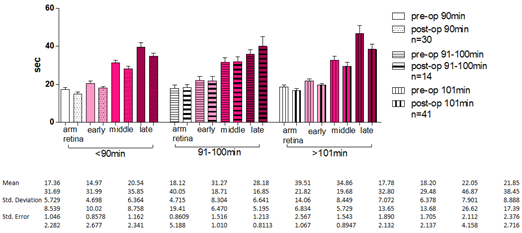
Figure 5: FFA time according to ECC time.
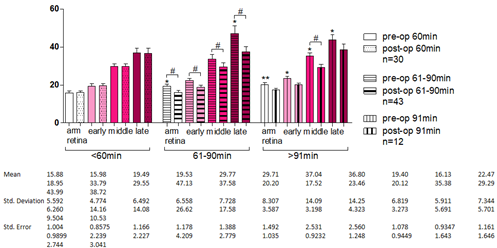
Figure 6: FFA time according to Aortic Cross-Clamp Time.
|
Preoperative Markers |
Prognostic value on improvement of ocular microcirculation and visual function |
|
Higher Preoperative FFA (total duration) |
+ |
|
Higher Preoperative VA |
+ |
|
CABG: Higher Preoperative IOP |
+ |
|
AVR: Lower Preoperative IOP |
+ |
Coronary Artery Bypass Grafting (CABG), Aortic Valve Replacement (AVR), Fundus Fluorescein Angiography (FFA), Visual Acuity (VA), Intraocular Pressure (IOP)
Table 1: Prognostic value of ophthalmologic indicators in predicting ocular vessels condition.
4. Discussion
Our study cannot explain a discrete mechanism responsible for visual complications associated with cardiac surgeries. However, pre- and postoperative ophthalmologic testing seems substantial to limit the occurrence of serious vision deficit in these patients. Only one from 88 patients (1.13%) experienced complete vision loss; the remaining patients showed no vision alterations compared to preoperative status. Postoperatively, FFATs were shorter in 69% of patients implicating a better retinal and choroidal circulatory response. Patients subjected to valvular surgery and having exposed to shorter ECC times yielded better FFATs than those undergoing CABG and longer ECC times. ΕCC consists a fundamental element of heart surgery giving the chance to the surgeon to safely perform the operation since ECC bypasses the cardiopulmonary system. However, it is admitted that ECC remains one of the main causes of surgical complications as it imposes hemodynamic alterations, which are triggered when autoregulatory mechanisms are abolished [5].
As far as microcirculation and ending arterial systems such as brain and retina are concerned, this autoregulatory mechanism is consisted of a static (myogenic, metabolic and neurogenic factors) and a dynamic (adaptation of local blood flow to perfusion pressure (PP) changes with alteration to vessels’ resistance) parameter [6]. More specifically, the retina and optic nerve head (ONH) are protected by their own autoregulation system under physiological circumstances [7]. ONH and retina’s static autoregulatory mechanism is based on the ability of their arteriolar vascular smooth muscle cells to respond to any differentiation in levels of systematic and local factors, such as PO2, PCO2 and metabolic products [8].
In parallel, their dynamic autoregulatory mechanism is responsible for maintaining local blood flow and vascular tone at specific levels, irrespectively of variations in the perfusion pressure, by special endothelial mechanotransduction [9]. But, since the level of constriction or dilation of arterioles or capillaries is limited, autoregulation is constricted to a certain critical range of perfusion pressure and is destroyed when ocular perfusion pressure (OPP) surpasses or falls below the critical range [5]. It has been shown that autoregulation is preserved functional with OPP (OPP= Mean Arterial Pressure (MAP)–IOP) levels: 50-150 mmHg [10]. Therefore, during ECC the autoregulatory mechanism is impaired as a result of: i) circulating microemboli that may cause vascular occlusion, ii) hypothermia provoking vasoconstriction, extravasation and consequent loss of vascular volume, iii) disordered autonomic neuronal tone and differentiation in levels of systemic substances such as PaO2 and PCO2, lactic acid and local metabolic factors released by the vascular endothelium. As a consequence, ECC seems to have significant impact on the microvasculature of the choroid-retina-optic nerve, since they influence their angiokinetic profile.
Nonetheless, this expected negative impact of ECC on ocular blood flow and function was not clearly imprinted in the results of our study. Our findings indicate that 69% of the operated patients had a decrease in their FFA time and thus improvement of their ocular vessels microcirculation. Moreover, the percentage of FFA improvement was greater in patients undergoing AVR (17%) than in CABG patients (4%) and thus valve replacement surgery seems to influence more positively than CABG the general vascular condition.
As far as the type of surgery is concerned, other findings, which have been also mentioned before, are: i) That the phase of FFA which should be postoperatively evaluated possibly differs in each type of surgery (CABG: Early-arm phase, AVR: Middle-Early phase), ii) That preoperative values of IOP (high in CABG patients and low in AVR patients) could be used as positive prognostic factors. In addition, preoperative VA and FFA total duration were higher in patients improved (Group 1) than in patients deteriorating (Group 2). Despite the prognostic importance of high preoperative VA, it is remarkable though that mean postoperative VA value did not significantly differ from the mean preoperative one, irrespective of the type of surgery, ECC and ACCT time. ECC associated hemodynamic complications are widely discussed in the scientific community. Although there is a tendency to blame ECC for optical complications, the results of our study do not confirm the existing evidence to the absolute level. This is extracted from the fact that in spite of our totally admitted finding that the duration of ECC application could be a possible, prognostic indicator of visual impairment, we also found that postoperative FFA time seems to be decreased not only in <90 minutes, but also in >101 minutes application of ECC. Even so, we encountered one case (out of 88) of postoperative vision loss, in which ECC was applied for over 101 minutes. This could be attributed to retinal ischemia and infarction owing to microemboli (platelet/ fibrin/ dictioerythrocytes) [11] or gas embolism [12], hypothermia, systematic inflammatory response, chorioretinal hypoperfusion or ischemic optic neuropathy induced by prolonged CPB [13].
Since the retina constitutes an embryological outgrowth of the brain and thus a potential pathway to assess changes in cerebral microvasculature, our finding of postoperative FFA time decrease in both ECC duration subgroups (ECC duration: <90 minutes and >101 minutes) could be of outmost importance (if it surpassed the traditional threshold for statistical significance) as it would demonstrate that ECC application does not play such a negative role in cerebral and ocular blood flow.
Furthermore, it could be of great interest to compare microvascular complications and especially the ocular and cerebral ones, between on-pump and off-pump CABG. As many studies, such as Bierbach et al. 2014 [14], indicate that off-pump technique minimizes ECC hemodynamic alterations in visceral microcirculation and end-organ damage, we confidently expect that the absence of CPB and its aforementioned complications, in conjunction with the avoidance of cardioplegia, could have a positive impact on the patient’s postoperative, visual and cerebral indicators, too. But, at the same time we are wondering if off-pump CABG would have similar outcomes, in terms of postoperative FFA time decrease, with the conventional method. Our surgical experience in off-pump technique is quite limited and thus no reliable conclusions could be extracted. However, a prospective randomized study [15] showed lower incidence of retinal microembolism and infarction after off-pump CABG, but further investigation is still needed.
Regarding ACCT time, it was evident that ACCT >60 minutes had significantly positive correlation with decreased postoperative FFA time and thus potential improvement of visual function. In consequence, the prolongation of ACCT does not have a definite, negative influence in ocular blood flow and microcirculation.
5. Limitations
The major limitation of the study is probably the relatively small sample size, which was obviously the result of the width of the exclusion criteria (cataract, glaucoma, visual acquity less than 5/10, diabetes mellitus, diabetic and hypertensive retinopathy and macular disease). From the potential 560 CABG and aortic valve patients in that were operated during the examination period, 320 patients were excluded due to the aforementioned criteria and 152 declined to participate in the study due to the complexity of the suggested examinations. The second limitation of the study is the fact that the improvement or deterioration was only determined by FFA time increase or decrease. Last, but not least, systematic and local hemodynamic changes are not solely the result of ECC application, but other covariates like haematocrit, excessive blood volume loss and surgical hypothermia could also have a negative impact on postoperative ocular hemodynamic condition [16, 17]. Therefore, in order to extract a more accurate etiological correlation between cardiac surgery-ECC and alterations in markers predicting visual disorders, major surgical complications and disorderation of intraoperative monitor parameters, such as excessive haematocrit-haemoglobin levels decrease, pulmonary gas exchange disorders and hemodynamic instability could have been taken into account and systematically presented.
6. Conclusion
Our data confirm reports of other investigators regarding the low incidence (≤ 1%) of serious vision problems in patients that underwent cardiac surgeries. Improvement of preoperative FFAT after the surgical procedure may be a sign of better ocular circulation possibly associated with increased cardiac output. Prolonged ECC times (>101 min) yielded longer postoperative FFAT translating into a worser circulatory response. We suggest that except of operative procedure related factors including adequate anticoagulation measures, preoperative ophthalmologic examination should be carried out in order to detect vulnerable patients, thereby further preventing vision loss after cardiac surgery.
Funding Statement
Not applicable
Conflict of Interest
Not applicable
References
- Beckmann A, Meyer R, Lewandowski J, et al. German Heart Surgery Report 2017: The Annual Updated Registry of the German Society for Thoracic and Cardiovascular Surgery. Thorac Cardiovasc Surg 66 (2018): 608-621.
- Beckmann A, Meyer R, Lewandowski J, et al. German Heart Surgery Report 2018: The Annual Updated Registry of the German Society for Thoracic and Cardiovascular Surgery. Thorac Cardiovasc Surg 67 (2019): 331-344.
- Ascione R, Ghosh A, Reeves BC, et al. Retinal and cerebral microembolization during coronary artery bypass surgery: a randomized, controlled trial. Circulation 112 (2005): 3833-3838.
- Yuruk K, Bezemer R, Euser M, et al. The effects of conventional extracorporeal circulation versus miniaturized extracorporeal circulation on microcirculation during cardiopulmonary bypass-assisted coronary artery bypass graft surgery. Interact Cardiovasc Thorac Surg 15 (2012): 364-370.
- Nenekidis I, Geiser M, Riva C, et al. Blood flow measurements within optic nerve head during on-pump cardiovascular operations. A window to the brain? Interact Cardiovasc Thorac Surg 12 (2011): 718-722.
- Luo X, Shen Y-M, Jiang M-N, et al. Ocular Blood Flow Autoregulation Mechanisms and Methods. J Ophthalmol (2015): 864-871.
- Prada D, Harris A, Guidoboni G, et al. Autoregulation and neurovascular coupling in the optic nerve head. Surv Ophthalmol 61 (2016): 164-186.
- Hill MA, Meininger GA. Arteriolar vascular smooth muscle cells: mechanotransducers in a complex environment. Int J Biochem Cell Biol 44 (2012): 1505-1510.
- Davies PF. Flow-mediated endothelial mechanotransduction. Physiol Rev 75 (1995): 519-560.
- Robinson F, Riva CE, Grunwald JE, et al. Retinal blood flow autoregulation in response to an acute increase in blood pressure. Invest Ophthalmol Vis Sci 27 (1986): 722-726.
- Tagarakis GI, Karangelis D, Tsolaki F, et al. Embolism as major cause of neurocognitive complications after heart surgery. Interact Cardiovasc Thorac Surg 12 (2011): 383.
- De Somer FMJJ, Vetrano MR, Van Beeck JPAJ, et al. Extracorporeal bubbles: a word of caution. Interact Cardiovasc Thorac Surg 10 (2010): 995-1001.
- Nenekidis I, Pournaras CJ, Tsironi E, et al. Vision impairment during cardiac surgery and extracorporeal circulation: current understanding and the need for further investigation. Acta Ophthalmol 90 (2012): 168-172.
- Bierbach B, Bomberg H, Pritzer H, et al. Off-pump coronary artery bypass prevents visceral organ damage. Interact Cardiovasc Thorac Surg 18 (2014): 717-726.
- Rainio A, Hautala N, Pelkonen O, et al. Risk of retinal microembolism after off-pump and on-pump coronary artery bypass surgery. J Cardiovasc Surg (Torino) 48 (2007): 773-779.
- Chamot SR, Petrig BL, Pournaras CJ, et al. Effect of isovolumic hemodilution on oxygen delivery to the optic nerve head. Klin Monbl Augenheilkd 219 (2002): 292-295.
- Roth S. The effects of isovolumic hemodilution on ocular blood flow. Exp Eye Res 55 (1992): 59-63.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 