Endoscopic Vacuum Therapy to Seal Recalcitrant Esophageal Leak After Esophageal Surgery in Children
Raja R Narayan MD MPH1, Megan A Foley MD2, Garrison M Carlos MD1, Matias Bruzoni
MD1, Gary Hartman MD1, Monique T Barakat MD PhD3, Roberto Gugig MD2*
1Division of Pediatric Surgery, Department of Surgery, Stanford University School of Medicine, Stanford, CA, USA
2Division of Pediatric Gastroenterology, Department of Pediatrics, Stanford University School of Medicine, Stanford, CA, USA
3Division of Gastroenterology and Hepatology, Department of Medicine, Stanford University School of Medicine, Stanford, CA, USA
*Corresponding author: Roberto Gugig, Division of Pediatric Gastroenterology, Department of Pediatrics, Stanford University School of Medicine, Stanford, CA, USA
Received: 25 August 2020; Accepted: 02 September 2020; Published: 03 September 2021
Article Information
Citation: Raja R Narayan, Megan A Foley, Garrison M Carlos, Matias Bruzoni, Gary Hartman, Monique T Barakat, Roberto Gugig. Endoscopic Vacuum Therapy to Seal Recalcitrant Esophageal Leak After Esophageal Surgery in Children. Journal of Surgery and Research 4 (2021): 459-464.
View / Download Pdf Share at FacebookAbstract
Postoperative esophageal leak is a serious adverse event that occurs in up to 20% following esophageal surgery. Increasingly, endoscopy is utilized to manage leaks refractory to surgical washout and drainage including endoscopic vacuum-assisted closure (EVAC) using negative pressure therapy to achieve source control and granulation. In this report, successful use of this endoscopic strategy to seal a persistent leak after esophageal diverticulectomy is described. A two-year-old male born with esophageal atresia and tracheoesophageal fistula underwent primary anastomosis on the first day of life. Two years later, he presented symptomatic from a diverticulum at the anastomosis and underwent diverticulectomy complicated by esophageal leak at the diverticulectomy site. Despite multiple surgical interventions to seal the leak and obtain source control, the esophageal leak persisted. A multidisciplinary team of surgeons, gastroenterologists, and radiologists determined in consensus with the family of the patient to proceed with EVAC beginning with intracavitary placement of a dressing sponge secured on a nasoesophageal tube. After six endoscopic sessions, the esophageal leak was determined to be closed on endoscopic and fluoroscopic imaging. The patient was ultimately discharged home nine days after removal of the endoscopic vacuum. The leak site remained closed on fluoroscopic esophagram studies performed 6 months later. Negative pressure therapy successfully closed a refractory esophageal leak in a child resulting from diverticulectomy. Future investigations should employ larger series to compare the efficacy, tolerance, and duration of therapy needed to close leaks from EVAC versus stenting.
Keywords
<p>Esophageal leak, Endoscopy, Endoscopic vacuum</p>
Article Details
1. Introduction
Postoperative leak is a serious adverse event associated with esophageal surgery, occurring in up to 20% of patients [1]. Esophageal leak management practices vary among institutions due to surgeon and endoscopist experiences. The optimal strategy has shifted from predominantly surgical to favoring endoscopic intervention first [2]. For refractory esophageal leaks, a new strategy has emerged using endoscopic vacuum-assisted closure (AC). This involves placing a surgical sponge on suction locally at the site of leak in an endocavitary or endoluminal position to aid in source control and to promote faster wound healing [3]. In this report, this novel endoscopic strategy resulted in successful resolution of a recalcitrant leak despite multiple surgical interventions in a child with tracheoesophageal fistula (TEF) and esophageal atresia (EA).
2. Materials and Methods
2.1 Patient History
The patient was born with VACTERL disorder including tetralogy of Fallot, ventricular septal defect, tracheomalacia, imperforate anus, and EA/TEF. He underwent an uneventful EA/TEF repair on day one of life. By 2 years old, he developed a proximal esophageal diverticulum at the repair site that resulted in choking and regurgitation. The patient then underwent right thoracotomy with diverticulectomy which was closed primarily in two layers. One week later, an esophageal leak was detected with an adjacent 2.8 cm gas and fluid collection directly communicating with the site of diverticulectomy. This prompted two redo right thoracotomies for chest washout and one attempt at esophageal repair with a bovine pericardial patch. The leak persisted thus the options of endoscopic stenting, EVAC, and diverting cervical esophagostomy were considered. A multidisciplinary team of surgeons and gastroenterologists determined in consensus with the family to proceed with EVAC.
2.2 Placement of the Endoscopic Vacuum-Assisted Closure Device
In the supine position, a neonatal Olympus XP190 gastroscope was advanced into the esophagus, where a diverticulum measuring 5.8 mm with suture material was visualized draining through two small fistula tracts into the right chest (Figure 1). Fluoroscopic views highlighted an irregular extraluminal tract extending toward the right lower lung base with pooling of contrast material. Based on these findings, suction tubing was advanced through the nares to the posterior pharynx and withdrawn from the mouth. The free end of this suction tubing was attached to a VAC GranuFoam dressing sponge (KCI USA, San Antonio, TX). The sponge size was matched to the size of the diverticulum cavity on fluoroscopic views and secured to the tubing with a silk suture. The sponge was advanced back through the mouth and down into the esophagus. A video laryngoscope and the Olympus pediatric gastroscope facilitated visualization. The pediatric gastroscope was introduced into the esophagus and rat tooth forceps were used to maneuver the foam sponge into the fistula cavity (Figure 2). Contrast was injected to confirm placement of the EVAC system. The suction tubing was connected to the vacuum unit (3M, KCI USA) to 125 mmHg negative pressure, and fluoroscopy visualized contrast evacuation from the diverticulectomy site.
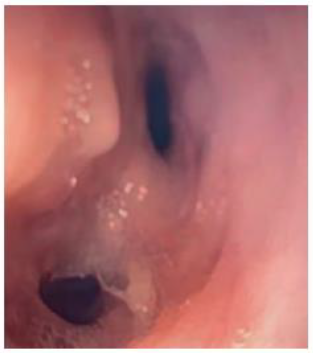
Figure 1: Endoscopic view of the esophageal diverticulum measuring larger than 5.8 mm containing two perforation sites. Day 0 – prior to placement of negative pressure therapy at initiation.
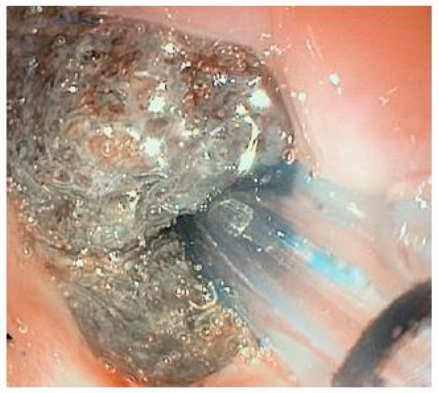
Figure 2: Endoscopic view of negative pressure sponge measuring 2.0 cm x 3.5 cm seated within the esophageal diverticulum. Day 0 – after placement of negative pressure therapy.
2.3 Serial Management of Endoscopic Vacuum-Assisted Closure Device
In subsequent procedures, the sponge was replaced with progressively smaller pieces as the fistula tracts granulated and diminished in size. Overall, five sponge exchange procedures were performed. With each exchange, the pediatric gastroscope visualized EVAC sponge removal. Contrast was introduced into the lumen to measure the size of the defect (Figure 3) and a new sponge was cut to fit the shrinking defect. Initially, exchanges were done within 3-5 days of each other, and then spaced out to 6-7 days as the healing process improved. Complete closure was achieved by 25 days following the initial procedure. Nine days after removal of the EVAC apparatus, the patient was discharged home without drains. Fluoroscopic esophagram studies obtained up to 6 months from discharge revealed no persistent leak or stricture. The patient receives the majority of their nutritional requirements through gastrojejunostomy tube bolus feeds with very little oral intake due to oral aversion.
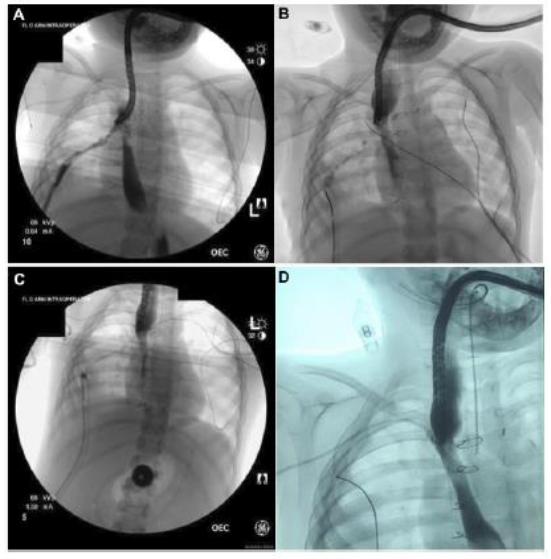
Figure 3: Serial fluoroscopic images illustrating closure of esophageal perforation over time. A: Day 8 – prior to placement of negative pressure therapy at the third endoscopic session. B: Day 14 – prior to placement of negative pressure therapy at the fourth endoscopic session. C: Day 17 – prior to placement of negative pressure therapy at the fifth endoscopic session. D: Day 25 – following removal of negative pressure therapy at sixth and final endoscopic session.
3. Discussion
This report details successful use of EVAC to seal a refractory leak from an esophageal diverticulum failing multiple operative interventions. Esophageal leak closure was successful after 25 days of EVAC with six endoscopic sessions. This device has not been approved by the FDA for intraluminal use and thus, this indication remains investigational. Several physiologic benefits arise from use of EVAC over stenting. First, the negative pressure system counteracts the physiologic low intrathoracic pressure created with breathing that pulls secretions through the perforation and into the chest [4]. Additionally, removal of necrotic material and fluid from the site of the perforation by EVAC promotes blood flow by activating cells that promote angiogenesis, granulation, and early closure [5]. Finally, EVAC generally requires a shorter duration of therapy than stenting, which is usually left in place for 6-8 weeks [6]. Relative to endoscopic stent placement, EVAC does carry the disadvantage of requiring multiple endoscopic sessions for exchange of the sponge as the fistula tract diminishes in size. However, the alternative option - endoscopic stent placement - is associated with complications including stent migration and erosion, which may also require repeated endoscopic interventions. Leaving a nasoesophageal tube in for EVAC additionally poses challenges related to displacement of the apparatus, particularly for the pediatric population. To address this issue, retrograde placement of the EVAC sponge inserted through a pre-existing gastrostomy site has been described [7] and may be better tolerated by children. Given the cylindrical shape of the sponge, if there is inadequate contact between the sponge and the esophageal wall, the VAC unit may fail to hold its seal. Serial endoscopic sessions are thus essential not only for sponge exchange, but also to ensure that the EVAC unit is properly functioning and not eroding at the site of the leak. Future investigations should seek to compare children treated with endoscopic negative pressure therapy with others managed with stenting or diverting cervical esophagostomy to compare morbidity, technical details, and success rates for each approach.
Acknowledgements
None
Declarations of Interest
The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article.
Funding Source
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- Whooley BP, Law S, Murthy SC, Alexandrou A, Wong J. Analysis of reduced death and complication rates after esophageal resection. Ann Surg 233 (2001): 338-344.
- Kassis ES, Kosinski AS, Ross P, Jr., Koppes KE, Donahue JM, Daniel VC. Predictors of anastomotic leak after esophagectomy: an analysis of the society of thoracic surgeons general thoracic database. Ann Thorac Surg 96 (2013): 1919-1926.
- Wedemeyer J, Schneider A, Manns MP, Jackobs S. Endoscopic vacuum-assisted closure of upper intestinal anastomotic leaks. Gastrointest Endosc 67 (2008): 708-711.
- de Moura DTH, de Moura B, Manfredi MA, et al. Role of endoscopic vacuum therapy in the management of gastrointestinal transmural defects. World J Gastrointest Endosc 11 (2019): 329-344.
- Huang C, Leavitt T, Bayer LR, Orgill DP. Effect of negative pressure wound therapy on wound healing. Curr Probl Surg 51 (2014): 301-331.
- Manfredi MA, Jennings RW, Anjum MW, Hamilton TE, Smithers CJ, Lightdale JR. Externally removable stents in the treatment of benign recalcitrant strictures and esophageal perforations in pediatric patients with esophageal atresia. Gastrointest Endosc 80 (2014): 246-252.
- Manfredi MA, Clark SJ, Staffa SJ, et al. Endoscopic Esophageal Vacuum Therapy: A Novel Therapy for Esophageal Perforations in Pediatric Patients. J Pediatr Gastroenterol Nutr 67 (2018): 706-712.


 Impact Factor: * 4.2
Impact Factor: * 4.2 Acceptance Rate: 72.62%
Acceptance Rate: 72.62%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 