A Case of Paraneoplastic Encephalitis Arising from Concurrent Thymoma and Stage I Adenocarcinoma of the Lung
Flaminio Pavesi MA1, Anna M. Cervantes-Arslanian MD2, Kei Suzuki MD3*
1Boston University School of Medicine, Boston, MA, United States
2Department of Neurology, Neurosurgery, and Medicine (Infectious Disease) Boston University School of Medicine, Boston, MA, United States
3Department of Surgery, Division of Thoracic Surgery, Boston University School of Medicine, Boston, MA, United States
*Corresponding Author: Dr. Kei Suzuki MD, Division of Thoracic Surgery, Boston Medical Center, 88 East Newton Street, Robinson 7308, Boston, MA 02118, USA
Received: 24 November 2020; Accepted: 10 December 2020; Published: 04 January 2021
Article Information
Citation: Flaminio Pavesi, Anna M. Cervantes-Arslanian, Kei Suzuki. A Case of Paraneoplastic Encephalitis Arising from Concurrent Thymoma and Stage I Adenocarcinoma of the Lung. Archives of Clinical and Medical Case Reports 5 (2021): 048-053.
View / Download Pdf Share at FacebookAbstract
Paraneoplastic Encephalitis (PE) is a rare syndrome that arises as a tumor-induced autoimmune response which can cause focal or diffuse nervous system dysfunction. Patients commonly present with a variety of neurologic symptoms, depending on the inflammatory distribution, and possibly with other symptoms stemming from the primary cancer. We herein present a never-smoker patient who presented with dysarthria, dysphagia and cognitive decline, and required numerous diagnostic examinations, sometimes invasive ones, for correct staging and diagnosing of the primary malignancy. Of note, despite the lack of clear evidence suggesting metastatic disease based on neuroimaging, a brain biopsy was nonetheless performed for diagnostic purposes. The patient was diagnosed with paraneoplastic encephalitis stemming from two independent primary cancers: an early stage lung adenocarcinoma and a concomitant type B3 thymoma. In addition to displaying a unique case of a paraneoplastic disorder associated with two simultaneous cancers, this report highlights the diagnostic challenge of paraneoplastic encephalitis and discusses important imaging findings with the goal of preventing invasive and evitable brain biopsies in similar future cases.
Keywords
<p>Paraneoplastic Neurologic Syndrome; Paraneoplastic Encephalitis; Stage I Thymoma; Stage I Adenocarcinoma of the lung</p>
Article Details
Abbreviations:
PE- Paraneoplastic encephalitis; CT- Computer tomography; MRI- Magnetic resonance imaging;
CSF- Cerebrospinal fluid; EBUS- Endobronchial ultrasound; AchR- Acetylcholine receptor; MG- Myasthenia gravis
1. Introduction
Paraneoplastic Neurologic Syndromes (PNS) are a rare, heterogenous group of disorders associated with cancer but caused by mechanisms other than direct metastatic infiltration in the nervous system [1-3]. Paraneoplastic Encephalitis (PE) is a subtype of PNS that may manifest as a widespread inflammation of the nervous system, in the case of encephalomyelitis, or, rather, as a localized encephalitis of the limbic system or of the brainstem, in the cases of limbic and brainstem encephalitis, respectively [4-7]. Patients present with a variety of neurologic symptoms, depending on the affected brain areas, that commonly arise with acute or subacute onset. Despite an unclear etiology, PE is now believed to be caused by an autoimmune reaction that arises when primary tumor cells synthesize neuronal peptides (onconeural antigens) normally found in the nervous system. Leukocytic infiltration into the brain parenchyma is then responsible for the manifestation of neurologic symptoms that commonly precedes the detection of the primary cancer [8-11]. We report a case of paraneoplastic encephalitis associated with both stage I lung adenocarcinoma and concomitant stage I thymoma that required an extensive diagnostic work up to identify and stage the malignancies. To our knowledge, this is the first case where two independent, primary tumors are both thought to be responsible for the onset of PLE.
2. Case Report
A 66-year-old, Haitian Creole, never smoker woman, with a medical history notable for well-controlled type 2 diabetes mellitus, hypertension and hysterectomy presented to the emergency department after noticing several weeks of symptoms including significant weight loss, left facial twitching, dysphagia, dysarthria, speech disturbance, hoarseness and word-finding difficulties. Over the next few weeks, the symptoms deteriorated. Her dysarthria and her slurring of speech, at first intermittent, became more consistent and she reported difficulties controlling her muscles of mastication resulting in inadvertent clenching and spasms of her jaw. Head computer tomography (CT) showed multiple hypodensities involving multiple lobes that appeared as T2/FLAIR hyperintense, T1 hypointense non-enhancing cortical and subcortical lesions on magnetic resonance imaging (MRI) (Figure 1A). While admitted to the hospital, she developed tonic-clonic seizures and progressive cognitive decline.
Further work-up included a chest CT, which revealed a part-solid 39mm mass in the right lower lobe, mediastinal lymphadenopathy, and an anterior mediastinal lesion (Figure 2). Lung biopsy confirmed adenocarcinoma with lepidic and acinar patterns. Total spine MRI did not note any evidence of metastasis. Electroencephalography (EEG) recorded frontotemporal focal cortical irritability and mild bilateral cerebral dysfunction. Cerebrospinal fluid (CSF) analysis and serum autoimmune encephalopathy panel were unremarkable. In light of these findings, both auto-antibody negative PE and metastatic disease were considered, and a brain biopsy was recommended for confirmation. Following a second evaluation at our hospital, the brain biopsy showed reactive astrogliosis with lymphocytic infiltrate and no evidence of metastatic disease. Upon follow up brain MRI (Figure 1B), some lesions were found to be enlarged, others had shrunk, and others were new as compared to the previous image.
A few weeks after discharge, the patient was again hospitalized for dizziness and confusion. Positron emission tomography CT was performed to stage her lung cancer, which showed avidity in the known lung cancer and her anterior mediastinal lesion. No activity was noted in her mediastinum. Laryngeal and thyroid cancer were excluded following nasolaryngoscopy and ultrasound exam. In order to complete staging of the primary lung cancer, flexible bronchoscopy and endobronchial ultrasound (EBUS) with lymph node biopsies was performed, and the mediastinum was confirmed to be negative for malignancy. The patient then underwent a left robot-assisted resection of the mediastinal mass, which showed a Masaoka stage I type B3 thymoma. Meanwhile, abnormally high levels of acetylcholine receptor antibodies were detected but clinical evaluation did not demonstrate findings consistent with symptomatic myasthenia gravis (MG). Having excluded all possibilities of metastatic disease, a right lower lobe lobectomy with en bloc right upper lobe wedge resection was performed with curative intent. Pathology revealed a pT1aN0 adenocarcinoma with lepidic-predominant morphology and invasive component of 9mm.
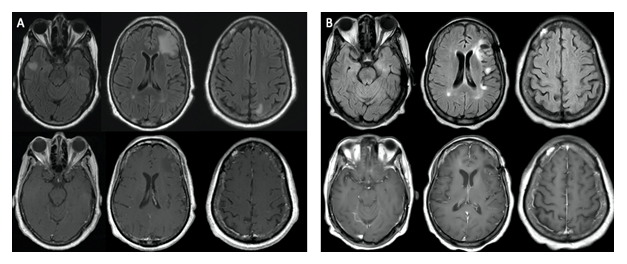
Figure 1: MRI images demonstrating (A) T2/FLAIR hyperintense, T1 hypointense non-enhancing cortical and subcortical lesions involving frontal lobes, insula, left parietal and right temporal lobes and (B) status post brain biopsy of the left frontal lobe lesion.
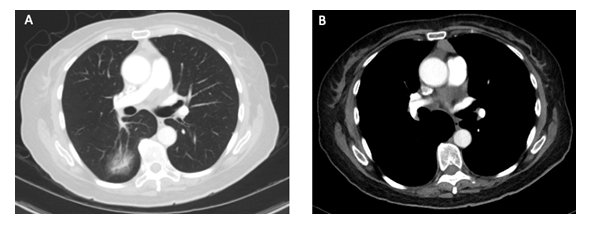
Figure 2: CT images demonstrating (A) 3.6 cm part-solid lung mass in the right lower lobe and (B) soft tissue prominence in the anterior mediastinum.
3. Discussion
PE is a rare disorder with a prevalence of less than 1% in cancer patients. It is most commonly observed in cases of small cell lung cancer (SCLC), neuroblastoma, thymoma and teratoma possibly because of their neuroendocrine origin and immunoregulatory involvement. Kidney, breast, testicular, non-small cell lung cancer (NSCL), and lymphoma have also been found in association with PE, though less commonly [4-6]. Although it is difficult to discern which primary malignancy contributed more to the development of PE, the current case depicts a double origin to the paraneoplastic disorder where both an early stage thymoma and an early stage lung adenocarcinoma are believed to be responsible for the onset of the paraneoplastic neurologic symptoms.
Due to its low prevalence, inconsistent semiology among patients and absence of highly selective clinical markers, numerous diagnostic procedures are often required in cases of PE [6, 7]. In this case, evidence gathered from numerous exams, including CSF analysis, neuroimaging, lung biopsy, EBUS with mediastinal lymph node biopsy, and nasolaryngoscopy, participated in the exclusion of nodal and distant metastatic disease of the two primary cancers. Additionally, despite the lack of onconeural antibodies, which are more commonly recorded in cases of SCLC [1-4, 12], the patient had abnormally high AchR autoantibodies without exhibiting a clinical picture compatible with MG. In addition to the common association between MG and thymomas, this finding may suggest the predisposition of patients with PNS to produce non-specific auto-antibodies due to its autoimmune etiology [13].
Importantly, although the neuroimaging evidence did not suggest typical appearance of brain metastases, the patient was recommended on two separate occasions to undergo an invasive brain biopsy in order to confidently exclude metastatic disease to the brain. Consistently with PE [14, 15], the lesions appeared as non-enhancing, not limited to the white matter, and free of diffusion abnormalities, thus not compatible with a classical presentation of metastatic disease, demyelinating disease, infection or vascular disease (Figure 1A). Nonetheless, due to the involvement of extratemporal brain areas that are not typically observed in PE cases and to the recurrence and increased size of lesions upon follow up MRI (Figure 2), the possibility of non-enhancing metastasis, albeit remote, was not ruled out. When approaching similar cases in the future, knowledge of paraneoplastic encephalitis and its imaging characteristics are essential for clinicians to frame their diagnostic approach and to avoid invasive procedures. This case report highlights how PE may present as multiple, scattered T2 hyperintense brain lesions involving various brain areas other than the more typically observed temporal-limbic lobe and brainstem. Moreover, due to its inflammatory nature, monitoring possible evolutions in the appearance of the noted brain lesions upon follow up neuroimaging is an additional tool towards the achievement of this goal.
4. Conclusion
We have presented a complicated case of PE secondary to both an early-stage lung adenocarcinoma and concurrent type B3 thymoma. This is the first documented case of PE associated with two simultaneous primary malignancies. In addition to its unique etiology, this case proposes itself to exhibit the extensive diagnostic work up that is needed in cases of paraneoplastic disorders and to offer an insight on relevant radiologic characteristics of PE with the hope of preventing invasive diagnostic procedures in future cases.
Acknowledgments
Dr. Suzuki (Corresponding author) is the guarantor of the content of this manuscript including both data collection and report.
Flaminio Pavesi (First Author) is responsible for writing the manuscript.
Dr. Cervantes-Arslanian (Second Author) contributed to the patient’s clinical management and to the editing of the presented work.
None of the authors have any financial statement to disclose, nor any conflict of interest in the merit of this work.
No external funding was needed for the completion of this work.
References
- Dalmau J, Rosenfeld MR. Paraneoplastic syndromes of the CNS. The Lancet Neurology 7 (2008): 327-340.
- Darnell RB, Posner JB. Paraneoplastic Syndromes Involving the Nervous System. New England Journal of Medicine 349 (2003): 1543-1554.
- Höftberger R, Rosenfeld MR, Dalmau J. Update on neurological paraneoplastic syndromes. Current Opinion in Oncology 27 (2015): 489-495.
- Rosenfeld MR, Titulaer MJ, Dalmau J. Paraneoplastic syndromes and autoimmune encephalitis: Five new things. Neurology: Clinical Practice 2 (2012): 215-223.
- Shen K, Xu Y, Guan H, et al. Paraneoplastic limbic encephalitis associated with lung cancer. Scientific Reports 8 (2018).
- Gultekin SH, Rosenfeld M, Voltz R, et al. Paraneoplastic limbic encephalitis: neurological symptoms, immunological findings and tumour association in 50 patients. Brain 123 (2000): 1481-1494.
- Graus FY, Delattre JC, Antoine Jundefined, et al. Recommended diagnostic criteria for paraneoplastic neurological syndromes. Journal of Neurology, Neurosurgery & Psychiatry 75 (2004): 1135-1140.
- Darnell RB. Onconeural antigens and the paraneoplastic neurologic disorders: at the intersection of cancer, immunity, and the brain. Proceedings of the National Academy of Sciences 93 (1996): 4529-4536.
- Alamowitch S, Graus F, Uchuya M, et al. Limbic encephalitis and small cell lung cancer. Clinical and immunological features. Brain 120 (1997): 923-928.
- Hurley K, Herron M, Mcdermott S, et al. Squamous Cell Lung Carcinoma Presenting With Refractory Status Epilepticus: A Case Report of Paraneoplastic Limbic Encephalitis. Respiratory Care 60 (2015).
- Bakheit AM, Kennedy PG, Behan PO. Paraneoplastic limbic encephalitis: clinico-pathological correlations. Journal of Neurology, Neurosurgery & Psychiatry 53 (1990): 1084-1088.
- Graus F, Dalmau J. Paraneoplastic neurological syndromes in the era of immune-checkpoint inhibitors. Nature Reviews Clinical Oncology 16 (2019): 535-548.
- Mckeon A. Paraneoplastic and Other Autoimmune Disorders of the Central Nervous System. The Neurohospitalist 3 (2013): 53-64.
- Tsunoda Y, Kiwamoto T, Homma S, et al. Paraneoplastic limbic encephalitis with late onset magnetic resonance imaging findings: A case report. Molecular and Clinical Oncology (2017).
- Deshmukh Y, Atre A, Vhora S, et al. A case report on paraneoplastic encephalitis associated with astrocytoma - An unknown entity. Indian Journal of Radiology and Imaging 26 (2016): 140.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
