Human Papillomavirus 16 E6/E7 Oncoproteins Transfection: Identification of Acquired Robertsonian Translocations in Human Keratinocytes Involving Chromosomes 13, 14, and 15
Eva McGhee1,2*, Sameeran Das3, Adin Handler1, Julian Handler1, Joel Palefsky2
1Charles R. Drew University of Medicine and Science, Department of Internal Medicine, 1731 East 120th Street, Los Angeles CA 90059, USA
2University of California San Francisco, Department of Medicine, Box 0126, San Francisco, CA 94143, USA
3Yale University, New Haven, Connecticut 48097, USA
*Corresponding Authors: Dr. Eva McGhee, Charles R. Drew University of Medicine and Science, Department of Internal Medicine, 1731 East 120th Street, Los Angeles CA 90059, USA
Received: 24 January 2020; Accepted: 10 February 2020; Published: 09 March 2020
Article Information
Citation:
Eva McGhee, Sameeran Das, Adin Handler, Julian Handler, Joel Palefsky. Human Papillomavirus 16 E6/E7 Oncoproteins Transfection: Identification of Acquired Robertsonian Translocations in Human Keratinocytes Involving Chromosomes 13, 14, and 15. Archives of Clinical and Medical Case Reports 4 (2020): 206-217.
View / Download Pdf Share at FacebookAbstract
Human papillomavirus16 (HPV) E6/E7 oncoproteins are associated with increased chromosomal instability through Robertsonian aberrations. Robertsonian translocations are common constitutional abnormalities in humans but are considered rare occurrences in cancer cells. Research is sparse surrounding Robertsonian translocations in viral infected cells; therefore, we examined whether HPV-E6/E7 transfection could facilitate the formation of Robertsonian aberrations. We used spectral karyotyping (SKY) and array-comparative genomic hybridization (array-CGH) to score Robertsonian aberrations in 16-MT cells, which were derived from human keratinocytes that were transfected with HPV16 E6/E7 oncoproteins. We analyzed 30 metaphases to determine the types of Robertsonian translocations present in the 16-MT cells. The results showed non-homologous Robertsonian translocations in 100% of metaphases scored and homologous acrocentric rearrangements-isochromosome types in 75% of metaphases scored. We studied two types Robertsonian translocations: der(13;14), the non-homologous type, and i(13), the homologous isochromosomes type. The results showed that HPV E6/E7 oncoproteins induce genomic instability, causing Robertsonian translocations on chromosomes 13 and 14. This analysis provides a reliable method for future research in the mechanisms of Robertsonian translocations as a result of centromeric fusion in HPV-transformed cells.
Keywords
<p>Human papillomavirus; Robertsonian translocations; Chromosomes</p>
Article Details
1. Introduction
Robertsonian translocations, typically inherited de novo, are the most common constitutional chromosomal rearrangements[1]. Moreover, the most prevalent constitutional Robertsonian translocations are der(13;14) and der(14;21), while all other reported combinations are rare [2]. The distribution of acquired Robertsonian translocations differs from that of the constitutional type [3]. All forms of Robertsonian rearrangements have been reported in hematologic malignancies, with isochromosomes accounting for over 50% of cases [1, 2]. The majority of acquired cases involved other chromosomal rearrangements in addition to the Robertsonian translocations [2]. The presence of these additional rearrangements suggests genomic instability within affected cells [4].
Robertsonian translocations are connected with a number of cancers, where 90% of cases were reported as constitutional in origin and approximately 3% as acquired [3, 5-11]. Constitutional translocations, specifically der(13;14), may predispose certain individuals to T- or B- cell malignancies, depending on the location of the break sites on chromosome 14 [12, 13]. If the breakpoint occurs at the T-cell a-receptor locus on chromosome 14, then the affected individual may be predisposed to T-cell malignancy. If the break occurs in the short arm of chromosome 14, it does not affect the T-cell a-receptor; however, the individual is at risk to develop a lymphocytic malignancy, such as lymphoma [13, 14]. Furthermore, a tandem translocation between chromosomes 13 and 14, with the breakpoint at 14q, may lead to B-cell malignancy [12]. Previous reports show that pre-malignant and malignant hematological disorders have been reported in individuals with constitutional rearrangements involving chromosomes 13 and 14 [15] and der(15;22) [16, 17].
Little is known about the connection between Robertsonian translocations involving virus-associated transformation and genomic instability. The heterozygous balanced translocation between chromosomes 13 and 17 correlates with several Robertsonian chromosomes in Moloney murine leukemia virus-transformed cell lines [18-21]. In this study we analyzed cells transfected by HPV16 E6/E7, known as 16-MT, using spectral karyotyping (SKY) and array-comparative genomic hybridization (array-CGH) techniques. We show that Robertsonian translocations can, in fact, be acquired in cells that are transfected with HPV16 E6/E7 oncogenes, an important observation given that Robertsonian translocations are not often presented as a common acquired event.
2. Materials and Methods
2.1 Cell growth-16-MT cells
The 16-MT cell line was generated from primary keratinocytes, and stable transfection was confirmed through dot blot analysis. The 16-MT cell line was grown in complete keratinocyte growth media (KGM CC-4131), (Clonetics, San Diego, CA) and in complete Dulbecco’s modified Eagle’s medium, containing 10% FCS (DMEM) (3:1). The cell solution was then incubated at 37ºC in a 5% CO2 incubator. The standard protocol for tissue culture growth of 16-MT cells is described in Molecular cytogenetic characterization of human papillomavirus16-transformed foreskin keratinocyte cell line 16-MT [22]. Subculturing of the 16-MT cell was halted at passage 16 for study. Slides were aged for 3 days to prepare them for spectral karyotyping.
2.2 Spectral karyotyping (SKY)
SKY analysis was performed on 16-MT cells on unstained slides using aged metaphases. The SKY probes were pre-annealed, denatured, hybridized, covered with a glass cover-slip, and sealed with rubber cement. The slides hybridized at 37°C for 40 to 48 hours in a moisture chamber. The slides were aged in 2x saline sodium citrate (SSC) at 37ºC for 30 minutes. Cells were pretreated with pepsin (30 m g/mL pepsin in 0.01 mol/L HCl) at 37ºC for 5 min and 1x phosphate buffered saline (PBS) at room temperature for 5 minutes. The cells were then postfixed in 1% formaldehyde in 1x PBS/MgCl2 at room temperature for 5 minutes and washed in PBS. Finally, the slides were dehydrated in 70, 80, and 100% ethanol for 2 minutes. The SKY probes (Vial 1, SKY kit; Spectral Imaging, Carlsbad, CA) were denatured at 76ºC for 7 minutes and then allowed to preanneal at 37ºC for 60 minutes. The slides were denatured at 76ºC for 5 minutes in 70% formamide–2 -SSC at a pH of 7.0 and were then dehydrated in 70, 80, and 100% ethanol for 2 minutes and allowed to air dry. The hybridization mixture was applied to the slides, covered with a glass coverslip, and sealed with rubber cement. The slides were hybridized at 37ºC for 40 to 48 hours in a moisture chamber. Following hybridization, the slides were washed with 50% formamide–2x SSC for 10 minutes each at 43ºC thrice, followed by another two washes in 2x SSC for 10 minutes, one wash in 0.4x SSC for 5 minutes, and a final wash in 0.1% Tween 20–4x SSC for 2 minutes at room temperature. Next, 80 mL of blocking reagent (Vial 2, SKY kit; Applied Spectral Imaging) was applied to each slide, which were then covered with a plastic coverslip and incubated at 37ºC for 45 minutes.
The SD200 SpectraCubeTM spectral imaging system was used to acquire spectral images (Applied Spectral Imaging, Migdal Ha’emek, Israel) [23]. The SD200 imaging system, attached to a Nikon E600 microscope, consisted of an optical head (a Sagnac interferometer) coupled with a multiline charge-coupled device CCD camera (Hamamatsu, Bridgewater, NJ) to capture images at discrete interferometric steps. The images were stored as a stack in a Pentium 586/300 MHz computer.
2.3 Array-comparative genomic hybridization (array-CGH)
Array-CGH analysis was performed to further analyze the 16-MT cell line, as previously described [23, 24]. The array consisted of at least 2,464 genomic clones printed in triplicate with an average resolution of 1.4 Mb across the genome. The data were analyzed using the UCSF SPOT computer software [25]. A second computer program, SPROC, was used to calculate the average ratios of the triplicate spots for each clone. 16-MT cell DNA and normal reference DNA from whole blood were labeled by random priming with Cy3- or Cy5-labeled nucleotides and hybridized for 2 days to the array. Sixteen-bit, 1024-pixel images, through 4’,6-diamidino-2-phenylindole (DAPI) Cy3 and Cy5 staining, were collected using a custom CCD camera system [26]. The UCSF SPOT computer software was used [25] to automatically segment the array spots and calculate the log2 ratios of the total Cy3 and Cy5 intensities for each spot after background subtraction. SPROC was used again to calculate the average ratios of the triplicate spots, standard deviations of the triplicates, and plotting positions for each clone on the array for the July 2003 freeze of the human genome sequence draft (http://genome.ucsf.edu). SPROC also implements a filtering procedure to reject data based on a number of criteria, including low reference-to-DAPI signal intensity and low correlation of the Cy3 and Cy5 intensities with a spot. The data files were edited to remove ratios of clones, for which only one of the triplicates remained after SPROC analysis and the standard deviation of the log2 ratios of the triplicates was O 0.2 [24].
3. Results
3.1 Spectral karyotyping analysis (SKY) of the 16-MT cell line
In Figure 1, SKY analysis confirmed the presence of the der(13;14) Robertsonian translocation and the isochromosome-Robertsonian translocation i [13] rearrangement. Several other numerical and structural aberrations were noted in the 16-MT cells involving duplications and deletions of chromosomal material [22]. Additional painting with chromosome 13- and 14-specific probes was performed to accurately observe the frequency of the Robertsonian aberrations, which are depicted by the arrow in figure 1A. Figures 1B and 1C present DAPI and classified images, respectively. Figure 1D shows dicentric chromosomes involving der(13;14). Figure 1E –H, focusing on clone number 2, depicts specific probes 13 and 14, that indicate i (13) and der(13;14), respectively. Figure 1F is a DAPI image, and figure 1G and 1H demonstrate SKY analysis of Robertsonian translocations.
3.2 Array-CGH of robertsonian imbalances
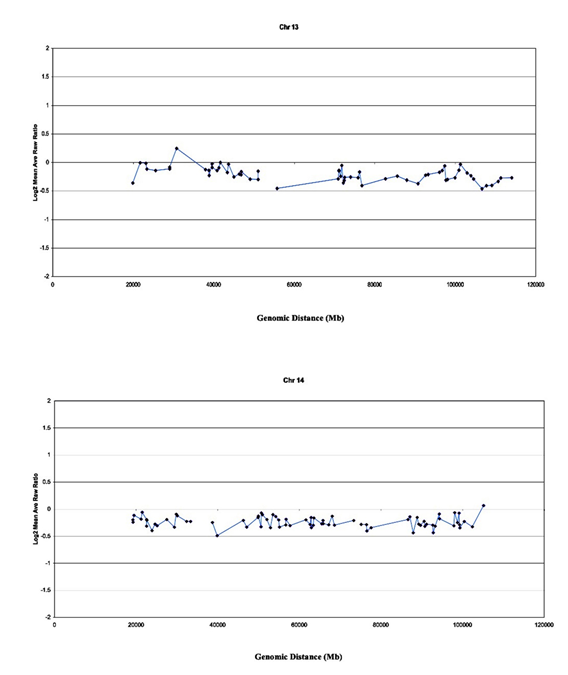
Array-CGH was performed to determine and map chromosomal imbalances to confirm unbalanced Robertsonian translocations. Figure 2 provides precise measurements (standard deviation of log2 ratios = 0.05-0.01) of numerical chromosomal changes that involve imbalances in Robertsonian chromosomes 13 and 14. Other observations through array-CGH revealed chromosomal gains and losses in 16-MT cells [22].
3.3 16MT and cervical cancer cells
In Table 1, 16MT cells showed Robertsonian types for chromosomes 13,14, der(13;14) (q10; q10)the 13;14 event occurred 30 times out of 30. The homologous acrocentric genetic event i(13) occurred 18 times out of 30. These genomic instability events in the 16MT cells were compared to the cervical cancer cells genetic events. HeLa cell showed non-homologous events der(14;15) (q10;q10) Siha showed no non–homologous events. CaSki cells showed non-homologous resulting in der(14;14) (q10;q10), der(14;14) (q10;q10), and der(15;21) (q10;q10).
3.4 Comprehensive summary of robertsonian translocations
The comprehensive summary for cervical cancer cells showed hypotriploid for HeLa and SiHa cells, and Hypertriploid for CaSki cells, as all outlined in Table 2.
Figure 1: Cytogenetic characterization-representative karyotype from SKY analysis composite of the 16-MT keratinocyte cells. (A) Red-green-magenta image showing several Robertsonian nonhomologue der(13;14) translocations, (B) DAPI image, (C) classified image, (D) red-green-blue metaphase image indicating structural and numerical changes in chromosomes, 13,14 and 15, (E) Red-green-magenta image showing a Robertsonian non-homologue and an isochromosome i(13) derivatives, (F) DAPI image, (G) der(13;14), (H) i(13).
Figure 2: Array-CGH image-profile of the 16-MT keratinocyte cell line. Array-CGH analysis was used to detect and map chromosome gains and losses in the 16-MT cells by hybridizing target of genomic DNA from a test and a reference sample to sequences immobilized on prepared slides. These probes are genomic DNA sequences (i.e., artificial chromosome, or BAC, that are mapped on the genome). The array CGH data show specific chromosomes of interest that indicate Robertsonian derivatives, der(13;14) and i(13).
|
Cell type |
Robertsonian type |
Chromosome |
N-number |
|
16-MT |
Non-homologous |
der(13;14) (q10;q10) |
30/30 |
|
Homologous acrocentric |
i(13) |
18/30 |
|
|
isochromosomes |
|||
|
HeLa |
Non-homologous |
der(14;15) (q10;q10) |
|
|
SiHa |
Non-homologous |
0 |
|
|
CaSki |
Non-homologous |
der(13;13) (q10;q10) x2 |
|
|
der(14;14) (q10;q10) |
|||
|
der(15;21) (q10;q10) x2 |
Table 1: Robertsonian chromosomes that are involved in structural and numerical aberrations for 16-MT cells in comparison to those in HeLa, SiHa, and CaSki cells. The chromosomes involved in the translocations and the Robertsonian type are listed.
|
Structural/numerical imbalances |
Number of appearances |
Robertsonian chromosome involved |
|
|
Hela Cells: Hypotriploid |
|||
|
der(13;19;3) |
3 |
13 |
|
|
der(13;19) |
9 |
13 |
|
|
der(13;10) |
3 |
13 |
|
|
der(14;15) |
1 |
14 |
|
|
der(13;19;15) |
1 |
13 |
|
|
-14 Chromosome |
1 |
14 |
|
|
-15 Chromosome |
1 |
15 |
|
|
- 21Chromosome |
4 |
21 |
|
|
-22 Chromosome |
4 |
22 |
|
|
+14 Chromosome |
3 |
14 |
|
|
+15 Chromosome |
14 |
15 |
|
|
+22 Chromosome |
4 |
22 |
|
|
SiHa: Hypotriploid |
|||
|
+13 Chromosome |
3 |
13 |
|
|
-13 Chromosome |
1 |
13 |
|
|
+14 Chromosome |
33 |
14 |
|
|
+15 Chromosome |
12 |
15 |
|
|
+21 Chromosome |
21 |
21 |
|
|
-21 Chromosome |
2 |
22 |
|
|
-22 Chromosome |
22 |
22 |
|
|
der(15;19) |
8 |
15 |
|
|
der(21;2) |
3 |
21 |
|
|
CaSki 78-82 Hypertriploid |
|||
|
+13 Chromosome |
5 |
13 |
|
|
+14 Chromosome |
10 |
14 |
|
|
-15 Chromosome |
3 |
15 |
|
|
-21 Chromosome |
7 |
21 |
|
|
-22 Chromosome |
3 |
22 |
|
|
der(13;19) |
2 |
13 |
|
|
der(14;11) |
2 |
14 |
|
|
der(15;6) |
4 |
15 |
|
|
der(22;13;1) |
1 |
22;13 |
|
|
der(13;13) |
1 |
13 |
|
|
der(15;8) |
1 |
15 |
|
|
der(22;8) |
1 |
22 |
|
|
der(15;13;7) |
1 |
15 |
|
|
der(13;9;1) |
1 |
13 |
|
|
der(14;14) |
2 |
14 |
|
|
der(21;9) |
1 |
21 |
|
|
der(21;10) |
1 |
21 |
|
|
der(15;5) |
1 |
15 |
|
|
der(15;10) |
2 |
21 |
|
|
der(15;21) |
1 |
15 |
|
|
der(21;16) |
1 |
21 |
|
|
der(22;10) |
1 |
22 |
|
|
der(22;6;13) |
1 |
22;13 |
|
|
der(13;3) |
1 |
13 |
|
|
der(22;13;17) |
1 |
22;13 |
|
|
der(13;19) |
1 |
13 |
|
|
der(11;14) |
1 |
14 |
|
|
der(15;6) |
1 |
15 |
|
Table 2: Comprehensive summary of reported Robertsonian translocations in HeLa, SiHa, and CaSki cells. Genomic analysis, using SKY, was conducted to generate the results displayed in the table. The table presents various numerical imbalances in each cell line, along with the number of appearances and the chromosome involved in the Robertsonian translocation.
4. Discussion
This study was conducted to examine the molecular and genetic mechanisms by which high-risk HPV16 E6/E7 genes contribute to Robertsonian translocations, genomic instability, and, eventually, cancer. HPV-associated genomic instability is a property that contributes to cancer and is associated with the malignant transformation of human epithelial cells in vitro through the deregulation of HPV oncoproteins [22]. We utilized 16-MT cells as a research model to study acquired Robertsonian translocations. We show, through SKY, that der(13;14) Robertsonian translocations were in 100% of metaphases scored. The derivative chromosome breakpoints, identified by SKY, appeared to have broken in the centromeric region, with the two chromosome pieces (q arms) fusing together after. This occurrence is linked with decondensed pericentromeric heterochromatin, which allows breakage and recombination at the same site [2].
We suggest that Robertsonian translocations may form through a distinct mechanism involving homologous recombination in chromosomes 13 and 14. SKY analyses of the 16-MT cells show a hypertriploid karyotype involving Robertsonian translocations and other isochromosomes. Robertsonian translocations are unique in their relationship with acrocentric chromosomes. As it was once believed that Robertsonian translocations have no direct clinical phenotypes, it is now accepted that Robertsonian translocations have long-term consequences [2, 27]. The acrocentric short arms contain repetitive DNA within the nucleolar-organizing region (NOR). NOR activity and expression of rRNA genes are increased in proliferating tumor tissues and reflect transcription activity in cells [2]. Cancerous cells infected with HPV exhibit both non-homologous translocations and homologous isochromosomes. The 16-MT cells showed several acquired Robertsonian translocations throughout metaphase spreads. These findings surrounding Robertsonian translocations suggest that induced chromosomal instability is caused by HPV16 integration into fragile sites of the DNA that produce specific chromosomal changes.
In addition to Robertsonian translocation der(13;14), the rare homologous acrocentric rearrangement i(13) is of particular interest. The homologous acrocentric rearrangement of the q-arms of chromosome 13 resulted in one chromosome 13; therefore, there are still two copies of chromosome 13, one rearranged with two q-arms of chromosome 13 and one normal copy. SKY analysis showed der(13;14) translocations in 100% of all metaphases analyzed. Also, i(13) aberrations were in 75% of metaphases scored, indicating that the 16-MT cell line may be a strong model for studying acquired genomic instability damage. Analysis of array-CGH data showed that Robertsonian chromosomes 13 and 14 have distinct regions of focal gain and loss. The array-CGH results revealed cryptic deletions and gains involving chromosomes where localization is cytogenetically compatible with the aberrant regions identified by SKY. The high level of separate chromosome aberrations might also reflect early or late events that could be induced by HPV16 E6/E7 transfection, thus exhibiting focal amplifications and deletions superimposed upon the background of whole chromosome gains.
In this analysis, we describe two types of Robertsonian rearrangements in the 16-MT cells, isochromosomes and Robertsonian translocations. Our findings show 100% of Robertsonian translocations occurred between non-homologous acrocentric chromosomes. Moreover, they are dicentric, suggesting that the translocations most likely result from both short arm recombination and centric fusion, as initially explained by Robertson in 1916 and Fryns in 1982 [27]. Additionally, we analyzed an HPV16-transfected cell line that contained acquired Robertsonian aberrations. Any whole arm translocation between two acrocentric chromosomes with a breakage and reunion site within the vicinity of the centromeres is a Robertsonian translocation. The der(13;14) form through homologous recombination of repetitive DNA sequences on chromosomes 13 and 14; this explains the high frequency of der(13;14) in the population of cells examined and the uniform breakpoint at the molecular level. The occurrences of der(13;14) may reflect the presence of a homologous repetitive sequence on other Robertsonian chromosomes that could be an inverted event in chromosome 14, and it could lead to preferential pairing and exchange between chromosome 13, 14, and 21.
Robertsonian translocations are very common in humans. The molecular mechanisms of chromosomal translocations are complex and are not fully understood. Studies show organization of the human genome is a hierarchy in the nucleus and every chromosome/chromatin structure has its preferential position and territory. Our findings suggest that the arrangements of chromosomes and gene loci in cells are responsible for non-random chromosomal translocations in human cancer. The results further suggest that such exchanges are more selective for chromosomes 13 and 14 than for chromosomes 15, 21and 22. The possible role of centromeric alpha satellite DNA in the etiology of 13q and 14q Robertsonian translocation involving the common and non-random association of chromosomes 13 and 14. Is hypothesized.
5. Summary
We found two specific types of Robertsonian translocations in the 16-MT cells: the non-homologous type der(13;14) and the homologous acrocentric type i(13). Additionally, we found other chromosomal gains and losses from the 16-MT composite karyotype associated with E6/E7 transformation and induced genomic instability. The HPV16 E6/E7 oncoproteins may predispose cells to additional genomic instability, such as sister chromatid exchanges, micronuclei, double strand breaks and bridge fusion breakage. These Robertsonian rearrangements may also facilitate progression to metastasis as it causes a number of genetic changes to accumulate. The data presented here are consistent with previous reports describing chromosomal instability in HPV-infected cells. Most importantly, this is the first detailed report on Robertsonian translocations appearing in HPV-transformed cells. The identification of Robertsonian-acquired changes in 16-MT cells, using advanced molecular cytogenetic techniques such as SKY and array-CGH, supports the usefulness of the 16-MT cell line as an in vitro model for studying important steps during HPV16 induced genomic instability in human keratinocytes.
Acknowledgments
The authors would like to thank Philip Cotter, Jingly Weier and Ha Nam Nguyen at UCSF for their technical contributions to these studies and the UCSF Comprehensive Cancer Center staff members for their technical support on the array-CGH work. Supported by NIH-NCI GRANTS: 3R01CA054053-10S1; U54 CA143931-04S1
References
- Herring DB, Harbert KA, Wenger SL, et al. Acquired Robertsonian translocations in leukemia: two more cases. Cancer Genet Cytogenet 158 (2005): 196-197.
- Welborn J. Acquired Robertsonian translocations are not rare events in acute leukemia and lymphoma. Cancer Genet Cytogenet 151 (2004): 14-35.
- Morgan R, Sandberg AA, Jarzabek V, et al. An acquired Robertsonian translocation in prolymphocytic leukemia: a case presentation and review. Cancer Genet Cytogenet 25 (1987): 293-301.
- Brunelli M, Russo C, Costantini S, et al. Association of the cytogenetic pattern and latero-cervical metastasis of laryngeal cancer. Acta Otorhinolaryngol Ital 14 (1994): 553-560.
- Fan YS, Baer MR, Sait SN, et al. An acquired Robertsonian translocation dic(14;14)(p11;p11) in a patient with a myelodysplastic syndrome following treatment of multiple myeloma. Cancer genetics and cytogenetics 30 (1988): 133-137.
- Ma SK, Chow EY, Wan TS, et al. Robertsonian translocation as an acquired karyotypic abnormality in leukaemia. Br J Haematol 98 (1997): 213-215.
- Ino T, Okamoto M, Tsuzuki M, et al. An acquired Robertsonian 13;15 translocation in acute myelogenous leukemia (M5b). Int J Hematol 59 (1994): 131-135.
- Agag R, Sacks J, Silver L. Congenital midline cervical cleft. Cleft Palate Craniofac J 44 (2007): 98-101.
- Qian J, Xue Y, Sun J, et al. Constitutional Robertsonian translocations in (9;22)-positive chronic myelogenous leukemia. Cancer Genet Cytogenet 132 (2002): 79-80.
- Chinnappan D, Philip A, Wu X, et al. Acquired Robertsonian translocations in two leukemia patients. Cancer Genet Cytogenet 131 (2001): 104-108.
- Multani AS, Kacker RK, Pathak S. Are Robertsonian translocations rare in cancers? Cancer Genet Cytogenet 93 (1997): 179-180.
- Kapanadze B, Makeeva N, Corcoran M, et al. Comparative sequence analysis of a region on human chromosome 13q14, frequently deleted in B-cell chronic lymphocytic leukemia, and its homologous region on mouse chromosome 14. Genomics 70 (2000): 327-334.
- White VA, Gascoyne RD, Paton KE. Use of the polymerase chain reaction to detect B- and T-cell gene rearrangements in vitreous specimens from patients with intraocular lymphoma. Archives of ophthalmology 117 (1999): 761-765.
- Caccia N, Bruns GA, Kirsch IR, et al. T cell receptor alpha chain genes are located on chromosome 14 at 14q11-14q12 in humans. J Exp Med 161 (1985): 1255-1260.
- Satge D, Moore SW, Stiller CA, et al. Abnormal constitutional karyotypes in patients with neuroblastoma: a report of four new cases and review of 47 others in the literature. Cancer Genet Cytogenet 147 (2003): 89-98.
- Fryns JP. The cardio-facio-cutaneous (CFC) syndrome and Robertsonian 15/22 translocation. Ann Genet, 35 (1992): 186-188.
- Becher R, Wendt F, Kuhn D. Constitutional Robertsonian T(15;22) in Ph-positive CML. Hum Genet 76 (1987): 399.
- Karst C, Trifonov V, Romanenko SA, et al. Molecular cytogenetic characterization of the mouse cell line WMP2 by spectral karyotyping and multicolor banding applying murine probes. Int J Mol Med 17 (2006): 209-213.
- Spira J, Wiener F, Klein G. Robertsonian translocation studies on the significance of trisomy 15 in murine T-cell leukemia. Cancer Genet Cytogenet 9 (1983): 45-49.
- Spira J, Wiener F, Ohno S. Is trisomy cause or consequence of murine T cell leukemia development? Studies on Robertsonian translocation mice. Proc Natl Acad Sci USA 76 (1979): 6619-6621.
- Zornig M, Klett C, Lovec H, et al. Establishment of permanent wild-mouse cell lines with readily identifiable marker chromosomes. Cytogenet Cell Genet 71 (1995): 37-40.
- McGhee EM, Cotter PD, Weier JF, et al. Molecular cytogenetic characterization of human papillomavirus16-transformed foreskin keratinocyte cell line 16-MT. Cancer Genet Cytogenet 168 (2006): 36-43.
- Rauen KA, Albertson DG, Pinkel D. Additional patient with del(12)(q21.2q22): further evidence for a candidate region for cardio-facio-cutaneous syndrome? American journal of medical genetics 110 (2002): 51-56.
- Snijders AM, Nowak N, Segraves R, et al. Assembly of microarrays for genome-wide measurement of DNA copy number. Nature genetics 29 (2001): 263-264.
- Jain AN, Tokuyasu TA, Snijders AM, et al. Fully automatic quantification of microarray image data. Genome research 12 (2002): 325-332.
- Pinkel D, Segraves R, Sudar D, et al. High resolution analysis of DNA copy number variation using comparative genomic hybridization to microarrays. Nature genetics 20 (1998): 207-211.
- Fryns JP, Kleczkowska A, Van den Bergue. Robertsonian t(Dq;Dq) translocations in man. J Genet Hum 30 (1982): 101-117.



 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
