Jejunum Hemolymphangioma Presenting as Chronic Anemia: A Case Report
Jiao Liu1, Meng Zhang1, Yesheng Li1, Yi Chen1, Yonggang Hong2*, Yangqing Huang1*
1Department of Hepatobiliary Surgery, Shanghai Public Health Clinical Center, Fudan University, Tongxin Road 921#, Hongkou, Shanghai, 200080, China
2Department of Anorectal Surgery, First Affiliated Hospital of Naval Military Medical University, Changhai Road 168#, Yangpu, Shanghai, 200433, China
*Corresponding Authors: Yonggang Hong, MD., Department of Anorectal Surgery, First Affiliated Hospital of Naval Military Medical University, Changhai Road 168#, Yangpu, Shanghai, 200433, China
Yangqing Huang, Ph.D., MD., Department of Hepatobiliary Surgery, Shanghai Public Health Clinical Center, Fudan University, Tongxin Road 921#, Hongkou, Shanghai, 200080, China
Received: 20 December 2021; Accepted: 12 January 2022; Published: 25 January 2022
Article Information
Citation: Jiao Liu, Meng Zhang, Yesheng Li, Yi Chen, Yonggang Hong, Yangqing Huang. Jejunum Hemolymphangioma Presenting as Chronic Anemia: A Case Report. Archives of Clinical and Medical Case Reports 6 (2022): 45-52.
View / Download Pdf Share at FacebookAbstract
Background: Hemolymphangioma is a rare disease with congenital malformation of both vascular and lymphatic vessels characterized by cystic dilation. It mostly occurs in cutaneous localizations like head, neck during childhood. Only few cases of hemolymphangioma occur in the small intestine, pancreas, esophagus have been reported. Hemolymphangioma is usually asymptomatic or lack of typical symptoms and the diagnosis is often made as an incidental finding during endoscopy. On rare cases the initial symptoms can be chronic anemia due to gastrointestinal bleeding. They constitute an unusual manifestation and there is a low incidence of this type of tumor.
Case Presentation: We report the case of a 42-year-old male, with a 7- month history of chronic anemia requiring blood transfusion. Hemoglobin and Hematocrit count were low, therefore further examinations were required to rule out bleeding sources or other causes of anemia. Double balloon enteroscopy showed a 20×20 mm lesion taking up 30% of the circumference in the proximal jejunum, with raised whitish edges, the center with a vascular appearance, and bleeding spots. Histological sections of the lesion revealed in the lamina propria and submucosal layer of the jejunum several markedly dilated thin-walled lymphatic spaces lined with single layers of flat endothelial cells. The final pathologic diagnosis was submucosal hemolymphangioma. Histological examination confirmed the diagnosis of submucosal hemolymphangioma.
Conclusion: The clinical symptoms of hemolymphangioma range from abdominal pain, anemia due to chronic blood loss in gastrointestinal tract, and obstruction to perforation. This case makes the point that hemolymphangioma should be concerned in the differential diagnosis of chronic anemia and other tumors in abdominal cavity with multiple diagnostic m
Keywords
<p>Hemolymphangioma; Hemangioma; Lymphangioma; Small intestine</p>
Article Details
1. Background
Hemolymphangioma includes hemangioma or vascular malformation and lymphangioma arising from congenital malformation of the vascular and lymphatic system, which is a rare benign primary neoplasm. Approximately 95% of hemolymphangioma are diagnosed in the head, neck and axilla in young person, especially in children before age 2, and the other 5% occur in the mediastinum and abdominal cavity, including the mesentery, small intestine, visceral organs, retroperitoneum and bones. The exact etiology of hemolymphangioma is still unclear, which is always considered to be congenital malformation of the vascular system and mesenchymal tissue [1]. The small intestine is the most common location and this type of lesion accounts for 10 % of the benign tumors in the duodenum, jejunum, and ileum, and hemolymphangioma represents 6% of small bowel tumors found in children and 1.4–2.4% in adults [2]. Intra- abdominal hemolymphangioma may produce some complications such as intestinal obstruction, volvulus or infarction, and gastrointestinal bleeding [3-5]. The male/female ratio is 1:2.5; however, when it comes to colon/rectum location, the ratio is 1:1 [5]. Even though there are several treatment options, the most effective is surgical resection [6].
1.1 Case presentation
A 42-year-old man was referred for a 7-month history of symptomatic persistent anemia and intermittent hematochezia requiring repeated blood transfusion. His medical history was remarkable for ankylosing spondylitis, acquired repairing of duodenal perforation and total hip arthroplasty. He denied any recent history of weight loss. He previously underwent esophagogastroduodenoscopy and colonoscopy, which revealed bleeding in the proximal jejunum.
Hemoglobin (Hb) and Hematocrit (Htc) count remained low, and thus examinations were conducted to explore possible bleeding sources as well as other causes of anemia, such as occult gastrointestinal bleeding. Laboratory test results at admission to our institution were consistent with microcytic hypochromic anemia (Hb 85g/L, Htc 28.1%), with iron 7.95μmol/L, total iron-binding capacity 47.5μmol/L, and transferrin saturation 16.7%. Fecal occult blood testing was positive and fecal transferrin was weakly positive. The coagulation profile was normal. Conditions such as autoimmune hemolytic anemia and paroxysmal nocturnal hemoglobinuria were ruled out as well. Tumor markers like CA19-9, CEA were normal. Upper gastrointestinal tract endoscopy showed chronic atrophic gastritis (Helicobacter pylori-negative) and a biopsy confirmed the diagnosis; no evidence of bleeding sites or scars of bleeding foci were found. Capsule endoscopy was performed next in order to study hidden causes of intestinal bleeding. Findings showed chronic gastritis and an ulcer lesion with bleeding stigmas in the first portion of jejunum. Colonoscopy showed scars of previous bleeding in the intestinal lumen, but no lesions suggesting colonic lesions as the source of anemia. The abdominal contrast-enhanced CT scan detected edematous striation of the root of the mesentery with reactive lymph nodes. There were no signs of ischemic or bowel obstruction or free liquid in the abdominal cavity (Figure 1).

Figure 1: The abdominal contrast-enhanced CT scan detected edematous striation of the root of the mesentery with reactive lymph nodes.
After these procedures, double balloon enteroscopy was carried out, reporting a 20×20 mm lesion taking up 30% of the circumference in the proximal jejunum, with raised whitish edges, a center with a vascular appearance, and bleeding spots (Figure 2).
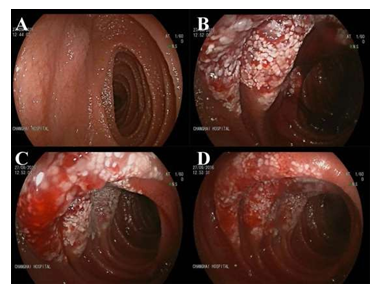
Figure 2: Double balloon enteroscopy reporting a lesion in the proximal jejunum.
Surgery confirmed the imaging findings. The affected part of intestine was easy to find (Figure 3). Considering the enterscopy findings and the multiple gastrointestinal bleeding episodes, the patient underwent surgical resection of the proximal jejunum lesion (first portion) and a lateral-lateral anastomosis of small intestine was performed. Surgery was well tolerated by patient, who received liquid diet 72 h in the postoperative period, patient had a regular evolution and no complications until discharge.
Macroscopic examination of the surgical specimen revealed a whitish, spongy lesion measuring 20 mm in diameter. Histological sections of the lesion revealed in the lamina propria and submucosal layer of the jejunum, several markedly dilated thin-walled lymphatic spaces lined with single layers of flat endothelial cells. The final pathologic diagnosis was submucosal hemolymphangioma. Histological examination confirmed the diagnosis of submucosal hemolymphangioma (Figure 4A (H&E staining) and B (D2-40 staining)). The postoperative course was uneventful. The patient remains well at regular outpatient follow-up with routine blood examination and there were no signs of recurrence at three years after surgery.
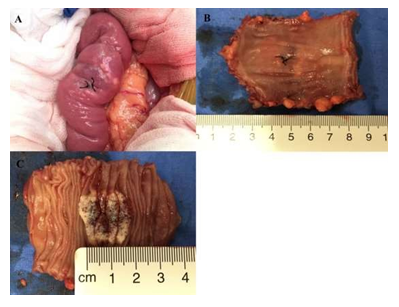
Figure 3: Surgery confirmed the imaging findings. Surgical specimen removed, showing a 2×2×0.5 cm mass with bleeding stigmata. A: Small bowel at laparotomy; B, C tumor after resection with edematous walls and clearly visible swollen, milky-white lacteals.
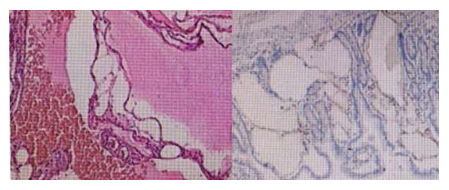
Figure 4: Hemangiolymphangioma with extension to mucosa and submucosa, free edges of injury. A: H&E staining; B: D2-40 staining.
2. Discussion
In this report, we have described an unusual case of jejunum hemolymphangioma with chronic anemia due to low grade gastrointestinal bleeding. Hemolymphangioma is a non-epithelial tumor with low incidence, being considered a benign disease with no invasive behavior. Approximately 95% of hemolymphangioma are diagnosed in the head, neck and axilla in young person, especially in children before age 2, and the other 5% occur in the mediastinum and abdominal cavity, including the mesentery, small intestine, visceral organs, retroperitoneum and bones [6]. The incidence of hemolymphangioma varies between 1.2 and 2.8 per every 1000 live births, and the male to female ratio is 1:2.5 [7].
These vascular tumors are endothelial neoplasias, characterized by an increase in cellular proliferation. On the other hand, vascular malformations are the result of the abnormal development of vascular structures during embryogenesis. Lymphatic malformations come in various forms, from a localized spongy lesion to a lesion compromising an anatomical region or multiple organs. Histologically demonstrated that they are characterized by micro or macrocysts, or both [8]. In this case presence, the patient received blood transfusion and medicine treatment with thalidomide before admission. Some researchs have been demonstrated that the thalidomide has shown the role in antiproliferative, immunomodulatory, and antiangiogenic properties [9,10]. Thalidomide has demonstrated potent antiangiogenic properties in numerous multicenter prospective trials over several years as therapy in the malignant B-lymphocyte plasma cell disorder, multiple myeloma [11]. Hemolymphangioma is a rare disease with congenital malformation of both vascular and lymphatic vessels characterized by cystic dilation. Hemolymphangioma includes hemangioma or vascular malformation and lymphangioma arising from congenital malformation of the vascular and lymphatic system. The abnormal venolymphatic communication between the dysembryoplastic vascular tissue and blood circulation could be the reason for the formation of this tumor, as well as the obstruction of lymph drainage or lymphatic damage [12]. These lymphatic lesions grow from a primordial sac which fails to connect with the rest of the lymphatic system during the embryonic development, and which tends to grow due to muscle contractions and increase intramural pressure. Intramural lymphatic obstruction, endothelial permeability damage, swelling, and congenital absence on lymph have been suggested as other possible causes for the development of intestinal lymphangiomas [13].
The differential diagnosis is essential by the immunohistochemical study of VIII factor antigen, D2-40, calretinina and human melanoma black-45 (HMB-45). Factor VIII and D2-40 in lymphatic malformations are positive, and in multicystic benign mesothelioma are negative. The HMB-45 is also positive in lymphangiomioma [14,15]. In our case, the immunohistochemical staining showed D2-40 positive (Figure 4B).
Although it is a benign proliferation of lymph or vascular vessels, it could transform into a lymphangiosarcoma especially after irradiation [16]. The surgical resection is the most effective treatment, especially when the lesion increases in size or applies pressure on the adjacent organs. Surgeons usually remove all tumor material from adjacent organs. It has been reported that local recurrences vary between 10 and 27 % for full resections and 50-100 % for partial resections in the next 2 years [17]. Many researches also did some partial resections because of the tumor usually associating with some complications such as infections, fistula and hemorrhage [6, 7, 13, 18].
The exact etiology of hemolymphangioma is still unclear. The clinical symptoms range from abdominal pain, anemia from blood loss, and obstruction to perforation. Because of the low incidence and vague symptoms of intra-abdominal hemolymphangioma, it is often not to diagnose. This case makes the point that hemolymphangioma should be concerned in the differential diagnosis of chronic anemia and other tumors in abdominal cavity with multiple diagnostic methods to confirm the presence of the condition.
3. Conclusion
Hemolymphangioma occurred in intestines are commonly asymptomatic, but sometimes present with gastrointestinal bleeding, abdominal pain, intussusception. Due to its rarity, very few reports with the characteristics described in this report have been published in the medical literature. With this report, we want to highlight including hemolymphangioma in the differential diagnosis of chronic anemia and gastrointestinal bleeding. The exact diagnosis is also difficult and may require multiple radiological investigations, capsule endoscopy and/or enteroscopy. Optimal treatment is radical surgical excision to avoid recurrence.
Authors Contribution
Yonggang Hong and Yangqing Huang contributed equally to this work as co-corresponding authors. Jiao Liu, Yi Chen, Yesheng Li, Meng Zhang, Yonggang Hong, Yangqing Huang designed the study and wrote the manuscript. All authors read and approved the final manuscript.
Conflict of Interest
The authors declare that they have no conflict of interest.
Reference
- Balderramo DC, Di Tada C, de Ditter AB, et al. Hemolymphangioma of the pancreas: case report and review of the literature. Pancreas 27 (2003): 197-199.
- Suthiwartnarueput W, Kiatipunsodsai S, Kwankua A, et al. Lymphangioma of thesmall bowel mesentery: a case report and review of the literature. World J Gastroenterol 18 (2012): 6328-6332.
- Levy AD, Cantisani V, Miettinen M. Abdominal lymphangiomas: imaging features with pathologic correlation. Am J Roentgenol 182 (2004): 1485-1491.
- Antoniou D, Soutis M, Loukas I, et al. Mesenteric cystic lymphangioma associated with intestinal malrotation in a newborn. Pediatr Med Chir 33 (2011): 141-142.
- Gomez-Galan, Mosquera-Paz, Ceballos, et al. Duodenal hemangiolymphangioma presenting as chronic anemia: a case report. BMC Res Notes 9 (2016): 426-430.
- Chen G, Cui W, Ji XQ, et al. Diffuse hemolynphangioma of the rectum: a report of a rare case. World J Gastroenterol 19 (2013):1494-1497.
- Fang YF, Qiu LF, Du Y, et al. Small intestinal hemolymphangioma with bleeding: a case report. World J Gastroenterol 18 (2012): 2145-2146.
- Marler JJ, Mulliken JB. Current management of hemangiomas and vascular malformations. Clin Plast Surg 32 (2005): 99-116.
- Ozeki M, Funato M, Kanda K, et al. Clinical improvement of diffuse lympangiomatosis with pegylated interferon alfa-2b therapy: case report and review of the literature. Pediatr Hematol Oncol 24 (2007): 513-524.
- Pauzner R, Mayan H, Waizman A, et al. Successful thalidomide treatment of persistent chylous pleural effusion. Ann Intern Med 146 (2007): 75.
- Hicks LK, Haynes AE, Reece DE, et al. A meta-analysis and systematic review of thalidomide for patients with previously untreated multiple myeloma. Cancer Treat Rev 34 (2008): 442-452.
- Figueroa RM, Lopez GJ, Servin TE, et al. Pancreatic hemolymphangioma. JOP 15 (2014): 399-402.
- Soontornmanokul T, Angsuwatcharakorn P, Viriyautsahakul V, et al. Jejunal Lymphangioma: rare case of GI bleeding. Gastrointest Endosc 76 (2012): 884-886.
- Campbell WJ, Irwin ST, Biggart JD. Benign lymphangioma of the jejunal mesentery: An unusual cause of small bowel obstruction. Gut 32 (1991):1568.
- Daniel S, Lazarevic B, Attia A. Lymphangioma of the mesentery of the jejunum: Report of a case and brief review of the literature. Am J Gastroenterol 78 (1983): 726-729.
- Rieker RJ, Quentmeier A, Weiss C, et al. Cystic lymphangioma of the small-bowel mesentery case report and a review of the literature. Pathol Oncol Res 6 (2000): 146-148.
- Monção CR. Approach to upper digestive hemorrhage with diagnosis of cavernous lymphangioma. Arq Bras Cir Dig 26 (2013): 69-70.
- Handra-Luca A, Montgomery E. Vascular malformations and hemangiolymphangiomas of the gastrointestinal tract: morphological features and clinical impact. Int J Clin Exp Pathol 4 (2011): 430-433.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
