Patient with Carcinoid Syndrome and Severe Cardiac Disease with Both Tricuspid an Pulmonary Lesions
Ileana Nitu¹,²*, Iulia Florentina Burcea¹,², Daniela Greere¹,², Adina Croitoru¹,², Catalina Poiana¹,²
1C.I. Parhon” National Institute of Endocrinology, Bucharest, Romania
2 Carol Davila” University of Medicine and Pharmacy, Bucharest, Romania
*Corresponding Author: Ileana Nitu, C.I. Parhon” National Institute of Endocrinology, Bucharest, Romania.
Received: 05 July 2022; Accepted: 20 July 2022; Published: 08 August 2022
Article Information
Citation: Ileana Nitu, Iulia Florentina Burcea, Daniela Greere, Adina Croitoru, Catalina Poiana. Patient with Carcinoid Syndrome and Severe Cardiac Disease with Both Tricuspid an Pulmonary Lesions. Archives of Clinical and Medical Case Reports 6 (2022): 562-567.
View / Download Pdf Share at FacebookAbstract
Introduction: Carcinoid Heart Disease (CHD) is a rare cardiac manifestation occurring in patients with advanced Neuroendocrine Tumours (NET) and the carcinoid syndrome, usually involving the right-sided heart valves and eventually leading to right heart failure. The additional burden of cardiac dysfunction heralds a steep decline in quality of life and survival, the prompt recognition of this disease being therefore of the utmost importance.
Case summary: We present the case of a 57 year old female patient diagnosed in 2016 with carcinoid tumor of the proxymal ileum (Grade 2 NET ki 67 8%) and liver metastasis. She underwent surgery with both tumor and secondary determinations resection, followed by chemotherapy in the same year. The patient comes to our clinic in February 2019, complaining in the last 5 months of abdominal pain associated with flushing of the face, simetric peripheral edema, progressive limitation in effort, appearance of dyspnea on moderate exertion and dry caugh. The transthoracic echocardiography detected severe dilation of the right chambers, systolic dysfunction of the right ventricle, tricuspid disease with severe regurgitation and mild stenosis. The echocardiography also revealed double pulmonary lesion with moderate regurgitation and mild stenosis. Endocrine evaluation revealed high levels of serum markers for Neuroendocrine Tumor (NET): serotonin (1543 ng/ml), cromogranin A (291 ng/ml), urinary level of 5-hydroxyindolacetic acid (84 mg/24 h).
Discussion: The carcinoid syndrome was considered to be the cause of the right-heart disease, regarding the fact that echocardiographic features suggested primary valvular lesions. Somatostatin analogue treatment was started with significant improvement in symptoms and NET markers, thus allowing cardiac surgery a month later with remission of symptom
Keywords
Neuroendocrine Tumours; Pulmonary; Carcinoid Syndrome; Valvular
Neuroendocrine Tumours articles; Pulmonary articles; Carcinoid Syndrome articles; Valvular articles
SARS-CoV-2 articles SARS-CoV-2 Research articles SARS-CoV-2 review articles SARS-CoV-2 PubMed articles SARS-CoV-2 PubMed Central articles SARS-CoV-2 2023 articles SARS-CoV-2 2024 articles SARS-CoV-2 Scopus articles SARS-CoV-2 impact factor journals SARS-CoV-2 Scopus journals SARS-CoV-2 PubMed journals SARS-CoV-2 medical journals SARS-CoV-2 free journals SARS-CoV-2 best journals SARS-CoV-2 top journals SARS-CoV-2 free medical journals SARS-CoV-2 famous journals SARS-CoV-2 Google Scholar indexed journals COVID-19 articles COVID-19 Research articles COVID-19 review articles COVID-19 PubMed articles COVID-19 PubMed Central articles COVID-19 2023 articles COVID-19 2024 articles COVID-19 Scopus articles COVID-19 impact factor journals COVID-19 Scopus journals COVID-19 PubMed journals COVID-19 medical journals COVID-19 free journals COVID-19 best journals COVID-19 top journals COVID-19 free medical journals COVID-19 famous journals COVID-19 Google Scholar indexed journals Neuroendocrine Tumours articles Neuroendocrine Tumours Research articles Neuroendocrine Tumours review articles Neuroendocrine Tumours PubMed articles Neuroendocrine Tumours PubMed Central articles Neuroendocrine Tumours 2023 articles Neuroendocrine Tumours 2024 articles Neuroendocrine Tumours Scopus articles Neuroendocrine Tumours impact factor journals Neuroendocrine Tumours Scopus journals Neuroendocrine Tumours PubMed journals Neuroendocrine Tumours medical journals Neuroendocrine Tumours free journals Neuroendocrine Tumours best journals Neuroendocrine Tumours top journals Neuroendocrine Tumours free medical journals Neuroendocrine Tumours famous journals Neuroendocrine Tumours Google Scholar indexed journals Pulmonary articles Pulmonary Research articles Pulmonary review articles Pulmonary PubMed articles Pulmonary PubMed Central articles Pulmonary 2023 articles Pulmonary 2024 articles Pulmonary Scopus articles Pulmonary impact factor journals Pulmonary Scopus journals Pulmonary PubMed journals Pulmonary medical journals Pulmonary free journals Pulmonary best journals Pulmonary top journals Pulmonary free medical journals Pulmonary famous journals Pulmonary Google Scholar indexed journals Chemotherapy articles Chemotherapy Research articles Chemotherapy review articles Chemotherapy PubMed articles Chemotherapy PubMed Central articles Chemotherapy 2023 articles Chemotherapy 2024 articles Chemotherapy Scopus articles Chemotherapy impact factor journals Chemotherapy Scopus journals Chemotherapy PubMed journals Chemotherapy medical journals Chemotherapy free journals Chemotherapy best journals Chemotherapy top journals Chemotherapy free medical journals Chemotherapy famous journals Chemotherapy Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals Leukemia articles Leukemia Research articles Leukemia review articles Leukemia PubMed articles Leukemia PubMed Central articles Leukemia 2023 articles Leukemia 2024 articles Leukemia Scopus articles Leukemia impact factor journals Leukemia Scopus journals Leukemia PubMed journals Leukemia medical journals Leukemia free journals Leukemia best journals Leukemia top journals Leukemia free medical journals Leukemia famous journals Leukemia Google Scholar indexed journals Carcinoid Syndrome articles Carcinoid Syndrome Research articles Carcinoid Syndrome review articles Carcinoid Syndrome PubMed articles Carcinoid Syndrome PubMed Central articles Carcinoid Syndrome 2023 articles Carcinoid Syndrome 2024 articles Carcinoid Syndrome Scopus articles Carcinoid Syndrome impact factor journals Carcinoid Syndrome Scopus journals Carcinoid Syndrome PubMed journals Carcinoid Syndrome medical journals Carcinoid Syndrome free journals Carcinoid Syndrome best journals Carcinoid Syndrome top journals Carcinoid Syndrome free medical journals Carcinoid Syndrome famous journals Carcinoid Syndrome Google Scholar indexed journals Valvular articles Valvular Research articles Valvular review articles Valvular PubMed articles Valvular PubMed Central articles Valvular 2023 articles Valvular 2024 articles Valvular Scopus articles Valvular impact factor journals Valvular Scopus journals Valvular PubMed journals Valvular medical journals Valvular free journals Valvular best journals Valvular top journals Valvular free medical journals Valvular famous journals Valvular Google Scholar indexed journals
Article Details
1. Introduction
Neuroendocrine Tumours (NETs) represent a rare class of neoplasms with an incidence between 2.5 and 5 cases per 100 000 people [1, 2]. It is also a rare cause of acquired valvular heart disease, although cardiac involvement has been described in up to 60% of patients with carcinoid syndrome. Without treatment, carcinoid heart disease has a poor prognosis with a 3 year survival rate as low as 31% (compared with 68% in patients without concomitant heart disease) [3]. Therefore, screening for heart involvement with transthoracic echocardiography is essential in patients with a newly diagnosed carcinoid tumour [4]. NETs of the small bowel, followed by the lung, large bowel, pancreas, appendix, and ovaries, have the highest incidence of heart disease [3]. Well-differentiated neuroendocrine tumors are rare. They are characterized by the secretion of a vasoactive substance (serotonin), responsible for the appearance of carcinoid syndrome symptoms (flushing, diarrhea, and bronchoconstriction) [2, 3]. The direct action of these vasoactive substances on the heart leads to carcinoid heart disease (CaHD; Hedinger’s syndrome), which is a frequent complication of carcinoid syndrome; CaHD can be the first presentation of carcinoid syndrome in up to 20% of patients. Right-sided heart failure is a prognostic factor of carcinoid syndrome [3]. Transthoracic echocardiography is indicated in all patients diagnosed with carcinoid syndrome, and it should be performed on a routine basis every 3-6 months depending on the clinical presentation of the patient and the severity of the disease [4]. Cardiac involvement can vary widely in patients. In fact, all heart valves may be affected, but the involvement of the tricuspid valve is most common [5]. Tricuspid leaflets show a specific involvement with thickened leaflets that have reduced mobility and are retracted. The concomitant regurgitation can vary from mild to severe [6-8]. Right ventricular volume overload can result in these two valves becoming dysfunctional and can lead to right heart failure [9].
As serotonin produced by the carcinoid tumour is inactivated in the lungs, right-sided carcinoid heart disease is more frequent than left-sided heart involvement (>90% of the cases) [5, 10]. Therefore, the presence or absence of an atrial septum defect or a Patent Foramen Ovale (PFO) needs to be thoroughly evaluated, because in the presence of PFO, left heart valve involvement is more frequent. Cardiac magnetic resonance imaging and Computed Tomogaphy (CT) can be helpful tools to assess damage of heart structures, as well as to evaluate cardiac metastases. Nuclear medicine imaging may be useful to detect the primary tumour; however, it may be of limited use when diagnosing carcinoid heart disease, except for depicting cardiac metastases [11]. In addition to transthoracic echocardiography, biomarkers such as N-terminal pro-brain natriuretic peptide (NT-proBNP), which is a well-established marker for heart failure, has been shown to also have diagnostic and prognostic relevance in patients with carcinoid tumours [12].
However, confounders may lead to false results. Foods and medications that may elevate the levels of 5-hydroxyindoleacetic acid (5-HIAA) should be avoided, and a 24 h urine sample should be taken [6]. Another biomarker is chromogranin A, which is a glycoprotein that is released from NET cells. Its levels are also associated with cardiac involvement [12]. The combination of NT-proBNP and chromogranin A has also been shown to determine survival probability, as patients with elevated levels of both parameters have only a survival probability of 16% compared with 81% in patients with normal levels of both parameters [12]. The management of CaHD is based on medical therapy (somatostatin analogues), resection of the primary tumor, and cardiac interventions. Surgical valve replacement is an effective treatment option for symptomatic patients, since it improves the symptoms and life quality [12].
2. Case report
In February 2019, a 57-year-old woman presented with heart failure with increasing dyspnea, lower limb edema, and cyanosis of her lips and extremities with refractory hypoxia. She was complaining in the last 5 months of abdominal pain associated with flushing of the face accompanied by night sweats, hepatalgia and asthenia, distension of the abdomen, simetric peripheral edema, progressive limitation in effort, appearance of dyspnea on moderate exertion and dry caugh. We noted that she was diagnosed in 2016 with carcinoid tumor of the proxymal ileum (Grade 2 NET ki 67 8%) and liver metastasis. She underwent surgery with both tumor and secondary determinations resection, followed by chemotherapy in the same year. Clinical examination revealed blood pressure of 120/70 mmHg, oxygen saturation at 85%, and a holosystolic murmur (grade 4/6) suggestive of tricuspid valve regurgitation. Lower limb edema up to her knee joint was also present. We also detected flushing, pale and dry mucous membranes, rales in pulmonary bases, hepatomegaly (liver palpable 4cm below the right costal margin), and positive fluid wave sign. A neurological examination was normal; in particular, there was no sensory or motor deficit and no language disorder. A 12-lead Electrocardiogram (ECG) showed sinus rhythm and a heart rate of 66 beats per minute, QRS axis +30 degrees, inverted T waves in D III and flattened T waves in aVF. She underwent transthoracic echocardiography, which showed dilated right heart cavities with Right Ventricular End Diastolic Diameter (RVEDD)/Left Ventricular End Diastolic Diameter (LVEDD) of 0.8, fractional area change of 30%, Right Atrial (RA) area of 25 cm2, and tricuspid ring of 39 mm; Tricuspid Annular Plane Systolic Excursion (TAPSE) was 12 mm and Right Ventricular (RV) velocity (S’) was 5 cm/second. Tricuspid valves appeared thickened and retracted with restricted mobility and poor coaptation responsible for a severe tricuspid regurgitation- vena contracta of 8 mm, proxymal radius of 7 mm and mild stenosis (Figure 1). Pulmonary valves were thickened with a moderate Pulmonary (IP) insufficiency (Figure 2). The left-sided valves were within normal limits. Our patient had a minimal mitral insufficiency. The function of the left ventricle was good with Ejection Fraction (EF) of 55%.
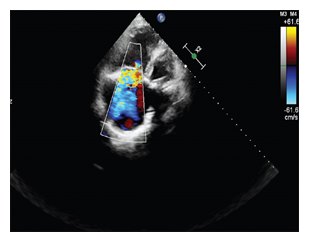
Figure 1: echocardiographic imaging- very severe tricuspid regurgitation with an enormous jet reaching the atrial roof.
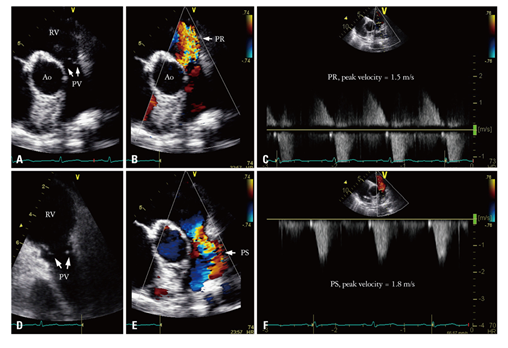
Figure 2: pulmonary valve regurgitation: 2D image- A, D; color Doppler interogation- B, E; continuous wave Doppler interogation with the measurement of peak systolic velocity- C, F.
Cardiac Magnetic Resonance (CMR) imaging revealed dilated right ventricle, with decreased systolic function, an ejection fraction of 44% (Figure 3), severe tricuspid regurgitation- regurgitant fraction of 51%, dilated tricuspid ring- area of 4,3cm2 and moderate pulmonary regurgitation- regurgitant fraction of 23%.
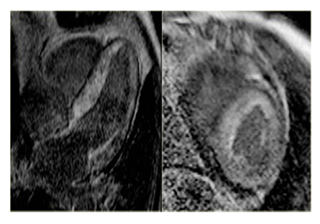
Figure 3: CMR imaging : dilated right ventricle (long and short axis examination); high degree of myocardial fibrosis Due to her former diagnosis of neuroendocrine tumour, a Computed Tomography (CT) of her thorax and abdomen was done and showed multiple hepatic and mesenteric nodules, as well as multiple bone secondary determinations.
Laboratory findings revealed increased serum NT-proBNP (2248 ng/ml), serotonin (1543 ng/ml), cromogranin A (291 ng/ml), urinary level of 5-hydroxyindolacetic acid (84 mg/24 hours, normal < 10 mg/24hours). An octreotide scan showed an intense radiotracer accumulation in her liver as well as normal distribution of the radiotracer in her kidneys and spleen (Figure 3). Paralleling her diagnostic workup by the patient’s cardiologist and endocrinologist, a comprehensive evaluation was undertaken in the structural heart valve clinic including discussions with oncology and cardiothoracic surgery as to the potential and feasibility of repair options. Medical therapy regarding right heart failure was initiated: loop diuretics, beta blocker, aldosterone receptor anthagonist-furosemide (80mg/day), carvedilolum (50mg/day) and spironolactone (50mg/day). Somatostatin analogue treatment was started with significant improvement in symptoms and NET markers, thus allowing cardiac surgery a month later with remission of symptoms related to heart failure, giving us time for deciding the adequate treatment for the NET. As valve repair was not feasible owing to severe fibrosis of the valve leaflets, valve replacement (Edwards St. Jude Epic, 33 mm bioprosthesis) was performed. Therefore, the patient received double bioprosthetic valves (tricuspid and pulmonary) in March 2019. She returns to our clinic six months later for endocrine evaluation which revealed a slight decrease in the level of serum markers for NET (serotonin 1393 ng/ml, urinary 5-hydroxyindolacetic acid 75,3 mg/24 h) and NT-proBNP (1543 ng/ml) compared to the first visit. The echocardiographic examination evidenced that both the tricuspid and pulmonary bioprosthesis were functional, with pressure gradients within the normal limits. Also, there was noticed an improvement in right ventricle function, with the increase of the ejection fraction, fractional area change (45%) and a decrease in the systolic pressure in the pulmonary artery compared to the first evaluation. She was also proposed for transesophageal echocardiography for a more accurate examination and there was no evidence regarding potential masses suggesting infective endocarditis at the level of the bioprosthetic valves. The patient is still under right-heart failure medical therapy with loop diuretics and beta blocker, but in lower doses and somatostatin analogues; her functional capacity improved significantly. The following PET–CT in February 2021 detected new neoplastic liver and bone enhancement. In the interdisciplinary tumour board, treatment with everolimus was discussed and administered. Unfortunately, our patient has poor prognosis despite initial successful cardiac surgery, due to the progression of carcinoid syndrome and multiple secondary determinations; we cannot exclude the possibility of bioprosthetic valves ‘stenosis as well as the worsening of her functional status. Furthermore, she needs multidisciplinary approach- combining biological therapy with right-heart failure medication and somatostatin analogues.
3. Discussion
Carcinoid syndrome was considered to be the cause of the right-heart disease, regarding the fact that echocardiographic features suggested primary valvular lesions and the initial diagnosis in 2016 didn’t reveal any hemodynamically significant valvulopathy. Carcinoid syndrome is a paraneoplastic syndrome mediated by humoral factors released by some carcinoid tumors. The majority of neuroendocrine tumors arise from the small intestine, particularly in the ileum, and they release into the systemic circulation a variety of vasoactive substances: serotonin, 5-hydroxytryptamine, 5-hydroxytryptophan, histamine, tachykinins, bradykinin, and prostaglandins, which explain the clinical manifestations of carcinoid syndrome [2]. Carcinoid syndrome is rare and once it is developed, more than 50% of the patients develop CaHD that can inaugurate this syndrome in 20% of cases. It is associated with a poor prognosis [3]. Symptoms of carcinoid syndrome (facial flushing, hypermotility of the gastrointestinal system, bronchoconstriction, and hypotension) usually occur in patients with hepatic metastatic lesions, due to the lack of hepatic inactivation of these released hormones [5]. These vasoactive substances act directly on the valvular endocardium and activate a fibrotic process. Histopathology reveals fibrous plaque-like and endocardial thickening leading to thickening and retraction of the valves. In this disease, the involvement of the right-sided heart valves is predominant and may be in the form of regurgitation and/or stenosis [6, 7]. Right-sided heart failure remains a major factor of morbidity in patients with CaHD [8]. Left-sided cardiac involvement is rarely observed (<10% of patients), particularly in the case of right-to-left shunt (a PFO) or bronchial carcinoids due to the bypassing of inactivation of serotonin within the lung [9].
The diagnosis is essentially based on biological examinations and echocardiography which remains the principal imaging modality in assessment of CaHD [6-10]. Biological examinations are helpful in the diagnosis of CaHD. High levels of N-terminal pro-brain natriuretic peptide (NT-proBNP), chromogranin-A (a neuroendocrine secretory protein), and urinary 5-HIAA (a metabolite of serotonin) are correlated with the progression of CaHD [11, 12]. Echocardiography shows typical valvular involvement: the tricuspid valve is constantly thickened, rigid, and retracted with restricted mobility and poor coaptation which lead to tricuspid regurgitation. Stenosing character is rarer (25% of cases) but may be associated. The pulmonary valve, which is more difficult to study, is reached in 30% of cases with regurgitation or pulmonary stenosis [10-13]. The right cavities and the inferior vena cava are dilated with or without right ventricular dysfunction [14]. Myocardial strain allows detection of an early right ventricular dysfunction in patients with CaHD independently of valvular involvement [15]. Of all patients with CaHD, ≤ 10% have lesions of the left-side valve. CaHD in the left side of the heart is less severe than in the right side of the heart. Left-sided heart involvement in carcinoid syndrome happens in patients with persistent foramen ovale or bronchial carcinoid [16]. Patients with CaHD have a decreased life expectancy compared with patients without cardiac lesions [17]. The treatment of patients with carcinoid syndrome is complex and involves multidisciplinary management. It is based on medical therapy, surgery of the tumor, and cardiac surgery [2]. Medical management consists of controlling the heart failure (diuretics agents and aldosterone antagonist) and symptoms of carcinoid syndrome. Therapy by somatostatin analogues allows improvement of symptoms and quality of life and decreases the incidence of CaHD from 50 to 20% [4]. New agents (telotristat and pasireotide) have shown promising results in patients with carcinoid syndrome refractory to somatostatin analogues [18]. Telotristat etiprate is a potent inhibitor of the synthesis of serotonin. The phase III TELESTAR clinical trial has shown that Telotristat may control the bowel movements in patients with carcinoid syndrome. It represents a new option for the treatment of patients with refractory carcinoid syndrome. However, other research is required to verify the safety and the benefit to control symptoms of that new drug [19]. Interferon alpha can be used as a complementary treatment for somatostatin analogues in refractory carcinoid syndrome. Because of its side effects, it should be initiated by 3 MU thrice weekly and then an individual titration [20]. Surgical resection of the primary tumor and resection of liver metastases appear to decrease the cardiac progression in CaHD and improve prognosis [16]. Since hepatic surgery exposes a patient to the risk of perioperative bleeding, hepatic intra-arterial treatment (transarterial chemoembolization, selective internal radiotherapy) is well suited to patients with hepatic metastases [21]. Valve replacement surgery or valvuloplasty is the only effective treatment for symptomatic CaHD; it improves the symptoms and increases the life expectancy of these patients [22]. The optimal moment for valve replacement surgery is not established. However, valvular surgery is proposed when patients become symptomatic or develop ventricular dysfunction provided they have a life expectancy of at least 1 year [20]. Bioprosthetic valves are generally preferred over a mechanical prosthesis, which requires anticoagulation for life and then exposes patients with liver metastases to the risk of bleeding in addition to the risk of prosthesis thrombosis in tricuspid position [23]. Percutaneous valve implantation is a novel option in high-risk patients with severe CaHD, who have poor performance status and comorbidities that do not allow open surgery [24].
4. Conclusion
The recognition, diagnosis, and treatment of neuroendocrine tumours and their associated syndromes are a rare but important subset of malignancies capable of affecting the cardiovascular system. Rapid diagnosis utilizing serum studies, CT scans, and echocardiography can help expedite the diagnosis and treatment of such rare conditions, and assist in the avoidance of complications due to the effects of circulating vasoactive substances. Despite its relatively well-recognized clinical symptoms, carcinoid syndrome and its associated heart disease still remains a challenging condition to manage and treat, oftens requiring the input of several subspecialties to treat the condition appropriately.
Declarations
Funding
No funding was received for presenting this case.
Conflicts of interest/Competing interests
The authors declare that there is no conflict of interest.
Ethics approval
Study approval was provided by the Ethics Committee of “Carol Davila” University of Medicine and Pharmacy, Bucharest.
Availability of data and material
All data generated or analyzed during this presentation are included in this published article.
Code of availability
Not applicable.
References
- Riedel M, Jou CJ, Lai S, et al. Functional and pharmacological analysis of cardiomyocytes differentiated from human peripheral blood mononuclear-derived pluripotent stem cells. Stem Cell Reports 3 (2014): 131-141.
- Liu J, Backx PH. Patch-clamp technique in ESC-derived cardiomyocytes. Methods Mol Biol 1181 (2014): 203-214.
- Farre C, Fertig N. New strategies in ion channel screening for drug discovery: are there ways to improve its productivity? Expert Opin Drug Discov 9 (2014): 1103-1107.
- Poulin H, Bruhova I, Timour Q, et al. Fluoxetine blocks Nav1.5 channels via a mechanism similar to that of class 1 antiarrhythmics. Mol Pharmacol 86 (2014): 378-389.
- Dobson R, Burgess MI, Pritchard DM, et al. The clinical presentation and management of carcinoid heart disease. Int J Cardiol 173 (2014): 29-32.
- Engelsman AF, van Duijvendijk P, Groenemeijer BE, et al. Tricuspid valve regurgitation as a presenting symptom of metastasized carcinoid tumor. Case Rep Gastroenterol 6 (2012): 643-649.
- Chowdhury MA, Taleb M, Kakroo MA, et al. Carcinoid heart disease with right to left shunt across a patent foramen ovale: a case report and review of literature. Echocardiography 32 (2015): 165-169.
- Dashwood A, Rahman A, Pavicic M. Carcinoid heart disease. Eur Heart J 36 (2015): 2326.
- Bradette S, Papas K, Pressacco J. Imaging features of carcinoid heart disease. Can Assoc Radiol J 65 (2014): 214-217.
- Patel C, Mathur M, Escarcega RO, et al. Carcinoid heart disease: current understanding and future directions. Am Heart J 167 (2014): 789-795.
- Simona Grozinsky-Glasberg ABG, Gross DJ. Carcinoid heart disease: from pathophysiology to treatment- ‘something in the way it moves’. Neuroendocrinology 101 (2015): 263-273.
- Catharina M, Korse BGT, de Groot CA, et al. Chromogranin-A and N-terminal pro-brain natriuretic peptide: an excellent pair of biomarkers for diagnostics in patients with neuroendocrine tumor. J Clin Oncol 27 (2009): 4293-4299.
- Lichtenauer M, Jirak P, Wernly B, et al. A comparative analysis of novel cardiovascular biomarkers in patients with chronic heart failure. Eur J Intern Med 44 (2017): 31-38.
- Schernthaner C, Lichtenauer M, Wernly B, et al. Multibiomarker analysis in patients with acute myocardial infarction. Eur J Clin Invest 47 (2017): 638-648.
- Jirak P, Fejzic D, Paar V, et al. Influences of Ivabradine treatment on serum levels of cardiac biomarkers sST2, GDF-15, suPAR and H-FABP in patients with chronic heart failure. Acta Pharmacol Sin 39 (2018): 1189-1196.
- Zurita AJ, Khajavi M, Wu HK, et al. Circulating cytokines and monocyte subpopulations as biomarkers of outcome and biological activity in sunitinib-treated patients with advanced neuroendocrine tumours. Br J Cancer 112 (2015): 1199-1205.
- Jirak P, Fejzic D, Paar V, et al. Influences of ivabradine treatment on serum levels of cardiac biomarkers sST2, GDF-15, suPAR and H-FABP in patients with chronic heart failure. Acta Pharmacol Sin 39 (2018): 1189-1196.
- Rachel P, Riechelmann AAP, Rego JFM, et al. Refractory carcinoid syndrome: a review of treatment options. Therapeutic Advances in Medical Oncology 9 (2017): 127-137.
- Castillo JG, Filsoufi F, Adams DH, et al. Management of patients undergoing multivalvular surgery for carcinoid heart disease: the role of the anaesthetist. Br J Anaesth 101 (2008): 618-626.
- Javier G, Castillo M, Federico Milla, et al. Surgical management of carcinoid heart valve disease. Semin Thoracic Surg 24 (2012): 254-260.
- IM Modlin, A Sandor. An analysis of 8305 cases of carcinoid tumors.Cancer 79 (1997): 813-829.
- M Kulke, R Mayer. Carcinoid tumors. N Engl J Med 340 (1999): 858-868.
- J Moller, P Pellikka, A Bernheim, et al. Prognosis of carcinoid heart disease: analysis of 200 cases over two decades.Circulation 112 (2005): 3320-3327.
- S Bhattacharyya, J Davar, G Dreyfus, et al. Carcinoid heart disease. Circulation 116 (2007): 2860-2865.


 Impact Factor: * 5.31
Impact Factor: * 5.31 Acceptance Rate: 75.63%
Acceptance Rate: 75.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
